Rapid Progress in the Use of Immunomodulatory Drugs and Cereblon E3 Ligase Modulators in the Treatment of Multiple Myeloma
Abstract
Simple Summary
Abstract
1. Introduction
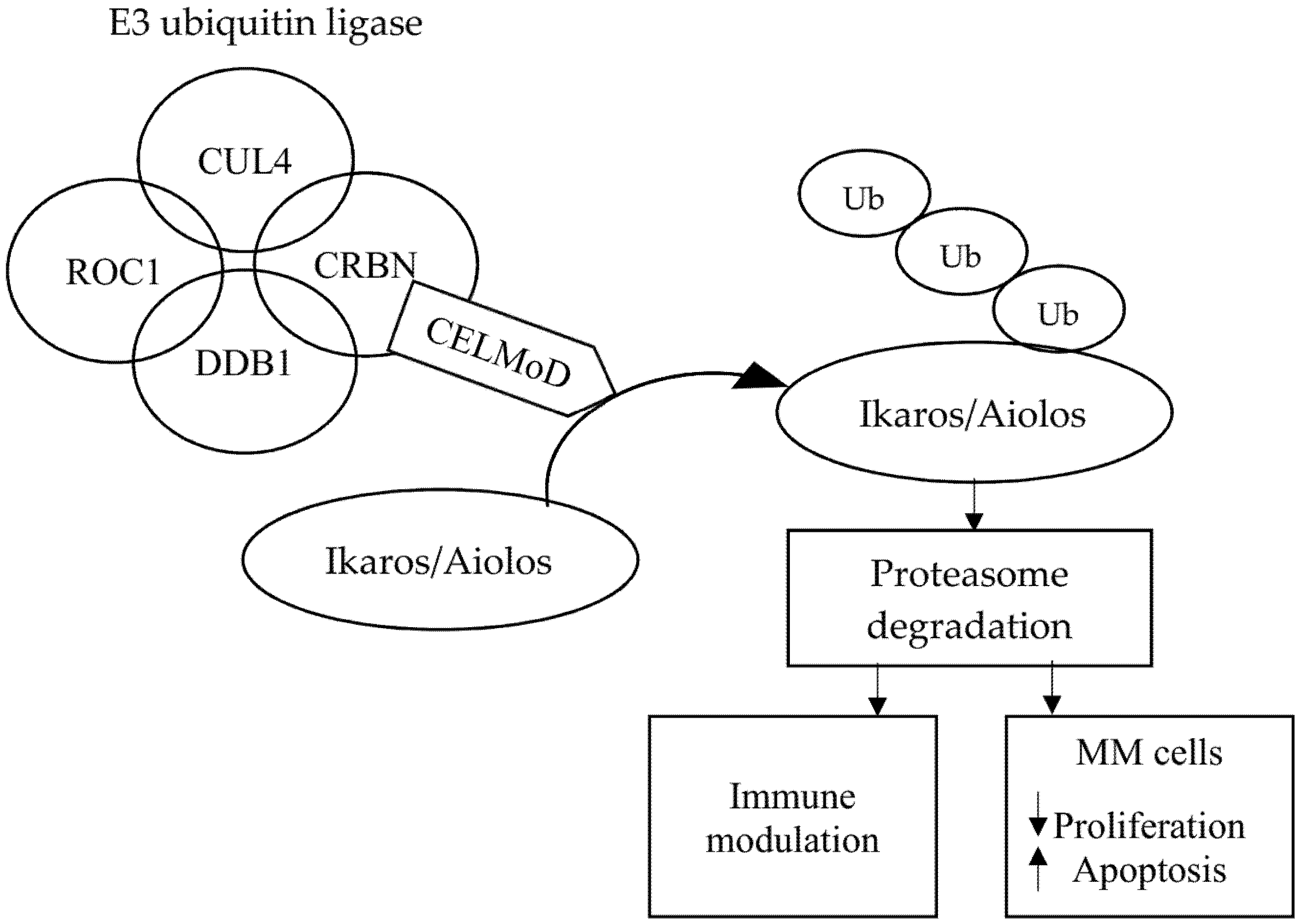
2. Mechanism of Action of Immunomodulatory Drugs and Cereblon E3 Ligase Modulators in the Treatment of Multiple Myeloma
3. Immunomodulatory Drugs in Newly Diagnosed Multiple Myeloma
3.1. Thalidomide
3.1.1. Thalidomide for the Treatment of Newly Diagnosed Multiple Myeloma in Patients Eligible for ASCT
3.1.2. Thalidomide for the Treatment of Newly Diagnosed Multiple Myeloma in Patients Ineligible for ASCT
3.2. Lenalidomide
3.2.1. Lenalidomide for the Treatment of Newly Diagnosed Multiple Myeloma in Patients Eligible for ASCT
3.2.2. Lenalidomide in the Treatment of Newly Diagnosed Multiple Myeloma in Patients Ineligible for ASCT
4. Immunomodulatory Drugs in Maintenance Therapy after ASCT
4.1. Thalidomide
4.2. Lenalidomide
5. Immunomodulatory Drugs in the Treatment of Relapsed/Refractory Multiple Myeloma
5.1. Thalidomide
5.2. Lenalidomide
5.3. Pomalidomide
6. The New Generation of Immunomodulatory Drugs—Cereblon E3 Ligase Modulators
6.1. Iberdomide (CC-220)
6.2. Avadomide (CC-122)
6.3. CC-92480
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Usmani, S.Z.; Hoering, A.; Cavo, M.; San Miguel, J.; Goldschmidt, H.; Hajek, R.; Turesson, I.; Lahuerta, J.J.; Attal, M.; Barlogie, B.; et al. Clinical predictors of long-term survival in newly diagnosed transplant eligible multiple myeloma—An IMWG Research Project. Blood Cancer J. 2018, 8, 123. [Google Scholar] [CrossRef] [PubMed]
- Holstein, S.A.; McCarthy, P.L. Immunomodulatory drugs in multiple myeloma: Mechanisms of action and clinical experience. Drugs 2017, 77, 505–520. [Google Scholar] [CrossRef]
- Liu, J.; Liu, W.; Mi, L.; Zeng, X.; Cai, C.; Ma, J.; Wang, L. Incidence and mortality of multiple myeloma in China, 2006–2016: An analysis of the Global Burden of Disease Study 2016. J. Hematol. Oncol. 2019, 12, 136. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, S.V. Multiple myeloma: Every year a new standard? Hematol. Oncol. 2019, 37 (Suppl. 1), 62–65. [Google Scholar] [CrossRef] [PubMed]
- Aragon-Ching, J.B.; Li, H.; Gardner, E.R.; Figg, W.D. Thalidomide analogues as anticancer drugs. Recent Pat. Anticancer Drug Discov. 2007, 2, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Raza, S.; Safyan, R.A.; Lentzsch, S. Immunomodulatory frugs (IMiDs) in multiple myeloma. Curr. Cancer Drug Targets 2017, 17, 846–857. [Google Scholar] [CrossRef]
- McBride, W.G. Thalidomide and congenital abnormalities (letter). Lancet 1961, 2, 1358. [Google Scholar] [CrossRef]
- Vargesson, N. The teratogenic effects of thalidomide on limbs. J. Hand Surg. Eur. 2019, 44, 88–95. [Google Scholar] [CrossRef]
- Ito, T.; Ando, H.; Suzuki, T.; Ogura, T.; Hotta, K.; Imamura, Y.; Yamaguchi, Y.; Handa, H. Identification of a primary target of thalidomide teratogenicity. Science 2010, 327, 1345–1350. [Google Scholar] [CrossRef]
- Rajkumar, S.V.; Witzig, T.E. A review of angiogenesis and antiangiogenic therapy with thalidomide in multiple myeloma. Cancer Treat. Rev. 2000, 26, 351–362. [Google Scholar] [CrossRef]
- Singhal, S.; Mehta, J.; Desikan, R.; Ayers, D.; Roberson, P.; Eddlemon, P.; Munshi, N.; Anaissie, E.; Wilson, C.; Dhodapkar, M.; et al. Antitumor activity of thalidomide in refractory multiple myeloma. N. Engl. J. Med. 1999, 341, 1565–1571. [Google Scholar] [CrossRef]
- Richardson, P.G.; Delforge, M.; Beksac, M.; Wen, P.; Jongen, J.L.; Sezer, O.; Terpos, E.; Munshi, N.; Palumbo, A.; Rajkumar, S.V.; et al. Management of treatment-emergent peripheral neuropathy in multiple myeloma. Leukemia 2012, 26, 595–608. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, O. Immunomodulatory drugs and their therapeutic effect in hematological malignancies through cereblon. Hematol. Med. Oncol. 2017, 2, 1–8. [Google Scholar] [CrossRef][Green Version]
- Dalla Torre, C.; Zambello, R.; Cacciavillani, M.; Campagnolo, M.; Berno, T.; Salvalaggio, A.; De March, E.; Barilà, G.; Lico, A.; Lucchetta, M.; et al. Lenalidomide long-term neurotoxicity: Clinical and neurophysiologic prospective study. Neurology 2016, 87, 1161–1166. [Google Scholar] [CrossRef]
- Lacy, M.Q.; McCurdy, A.R. Pomalidomide. Blood 2013, 122, 2305–2309. [Google Scholar] [CrossRef]
- Derudas, D.; Capraro, F.; Martinelli, G.; Cerchione, C. Old and new generation immunomodulatory drugs in multiple myeloma. Panminerva Med. 2020, 62, 207–219. [Google Scholar] [PubMed]
- Ruchelman, A.L.; Man, H.W.; Zhang, W.; Chen, R.; Capone, L.; Kang, J.; Parton, A.; Corral, L.; Schafer, P.H.; Babusis, D.; et al. Isosteric analogs of lenalidomide and pomalidomide: Synthesis and biological activity. Bioorg. Med. Chem. Lett. 2013, 23, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, P.P.; Lopez-Girona, A.; Miller, K.; Carmel, G.; Pagarigan, B.; Chie-Leon, B.; Rychak, E.; Corral, L.G.; Ren, Y.J.; Wang, M.; et al. Structure of the human Cereblon-DDB1-lenalidomide complex reveals basis for responsiveness to thalidomide analogs. Nat. Struct. Mol. Biol. 2014, 21, 803–809. [Google Scholar] [CrossRef]
- D’Amato, R.J.; Loughnan, M.S.; Flynn, E.; Folkman, J. Thalidomide is an inhibitor of angiogenesis. Proc. Natl. Acad. Sci. USA 1994, 91, 4082–4085. [Google Scholar] [CrossRef]
- Davies, F.; Baz, R. Lenalidomide mode of action: Linking bench and clinical findings. Blood Rev. 2010, 24 (Suppl. 1), S13–S19. [Google Scholar] [CrossRef]
- Ríos-Tamayo, R.; Martín-García, A.; Alarcón-Payer, C.; Sanchez-Rodriguez, D.; Del Valle Diaz de la Guardia, A.M.; Collado, C.G.G.; Morales, A.J.; Chacon, M.J.; Barrera, J.C. Pomalidomide in the treatment of multiple myeloma: Design, development and place in therapy. Drug Des. Dev. Ther. 2017, 11, 2399–2408. [Google Scholar] [CrossRef]
- Quach, H.; Ritchie, D.; Stewart, A.K.; Nesson, P.; Harrison, S.; Smyth, M.J.; Prince, H.M. Mechanism of action of immunomodulatory drugs (IMiDS) in multiple myeloma. Leukemia 2010, 24, 22–32. [Google Scholar] [CrossRef]
- Lopez-Girona, A.; Mendy, D.; Ito, T.A.; Miller, K.H.; Gandhi, A.K.; Kang, J.; Karasawa, S.; Carmel, G.; Jackson, P.E.; Abbasian, M.; et al. Cereblon is a direct protein target for immunomodulatory and antiproliferative activities of lenalidomide and pomalidomide. Leukemia 2012, 26, 2326–2335. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Handa, H. Cereblon and its downstream substrates as molecular targets of immunomodulatory drugs. Int. J. Hematol. 2016, 104, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Sievers, Q.L.; Petzold, G.; Bunker, R.D.; Renneville, A.; Słabicki, M.; Liddicoat, B.J.; Abdulrahman, W.; Mikkelsen, T.; Ebert, B.L.; Thomä, N.H. Defining the human C2H2 zinc finger degrome targeted by thalidomide analogs through CRBN. Science 2018, 362, eaat0572. [Google Scholar] [CrossRef]
- Krönke, J.; Udeshi, N.D.; Narla, A.; Grauman, P.; Hurst, S.N.; McConkey, M.; Svinkina, T.; Heckl, D.; Comer, E.; Li, X.; et al. Lenalidomide Causes Selective Degradation of IKZF1 and IKZF3 in Multiple Myeloma Cells. Science 2014, 343, 301–305. [Google Scholar] [CrossRef]
- Lu, G.; Middleton, R.E.; Sun, H.; Naniong, M.; Ott, C.J.; Mitsiades, C.S.; Wong, K.-K.; Bradner, J.E.; Kaelin, W.G., Jr. The Myeloma Drug Lenalidomide Promotes the Cereblon-Dependent Destruction of Ikaros Proteins. Science 2014, 343, 305–309. [Google Scholar] [CrossRef]
- John, L.B.; Ward, A.C. The Ikaros gene family: Transcriptional regulators of hematopoiesis and immunity. Mol. Immunol. 2011, 48, 1272–1278. [Google Scholar] [CrossRef]
- Bjorklund, C.C.; Lu, L.; Kang, J.; Hagner, P.; Havens, C.G.; Amatangelo, M.; Wang, M.; Ren, Y.; Couto, S.S.; Breider, M.; et al. Rate of CRL4CRBN substrate Ikaros and Aiolos degradation underlies differential activity of lenalidomide and pomalidomide in multiple myeloma cells by regulation of c-Myc and IRF4. Blood Cancer J. 2015, 5, e354. [Google Scholar] [CrossRef] [PubMed]
- Chanan-Khan, A.A.; Swaika, A.; Paulus, A.; Kumar, S.K.; Mikhael, J.R.; Rajkumar, S.V.; Dispenzieri, A.; Lacy, M.Q. Pomalidomide: The new immunomodulatory agent for the treatment of multiple myeloma. Blood Cancer J. 2013, 3, e143–e148. [Google Scholar] [CrossRef] [PubMed]
- Castelli, R.; Cassin, R.; Cannavo, A.; Cugno, M. Immunomodulatory drugs: New options for treatment of myelodysplastic syndromes. Clin. Lymphoma Myeloma Leuk. 2013, 13, 1–7. [Google Scholar] [CrossRef]
- Corral, L.G.; Haslett, P.A.; Muller, G.W.; Chen, R.; Wong, L.M.; Ocampo, C.J.; Patterson, R.T.; Stirling, D.I.; Kaplan, G. Differential cytokine modulation and T cell activation by two distinct classes of thalidomide analogues that are potent inhibitors of TNF-alpha. J. Immunol. 1999, 163, 380–386. [Google Scholar] [PubMed]
- Davies, F.E.; Raje, N.; Hideshima, T.; Lentzsch, S.; Young, G.; Tai, Y.T.; Lin, B.; Podar, K.; Gupta, D.; Chauhan, D.; et al. Thalidomide and immunomodulatory derivatives augment natural killer cell cytotoxicity in multiple myeloma. Blood 2001, 98, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Hideshima, T.; Akiyama, M.; Podar, K.; Yasui, H.; Raje, N.; Kumar, S.; Chauhan, C.; Treon, S.P.; Richardson, P.; et al. Molecular mechanisms whereby immunomodulatory drugs activate natural killer cells: Clinical application. Br. J. Haematol. 2005, 128, 192–203. [Google Scholar] [CrossRef]
- Noonan, K.; Rudraraju, L.; Ferguson, A.; Emerling, A.; Pasetti, M.F.; Huff, C.A.; Bornello, I. Lenalidomide-induced immunomodulation in multiple myeloma: Impact on vaccines and antitumor responses. Clin. Cancer Res. 2012, 18, 1426–1434. [Google Scholar] [CrossRef] [PubMed]
- Matyskiela, M.E.; Zhang, W.; Man, H.W.; Muller, G.; Khambatta, G.; Baculi, F.; Hickman, M.; LeBrun, L.; Pagarigan, B.; Carmel, G.; et al. A cereblon modulator (CC-220) with improved degradation of Ikaros and Aiolos. J. Med. Chem. 2018, 61, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.D.; Correa, M.; Nagy, M.A.; Alexander, M.; Plantevin, V.; Grant, V.; Whitefield, B.; Huang, D.; Kercher, T.; Harris, R.; et al. Discovery of CRBN E3 ligase modulator CC-92480 for the treatment of relapsed and refractory multiple myeloma. J. Med. Chem. 2020, 63, 6648–6676. [Google Scholar] [CrossRef]
- Moreau, P.; San Miguel, J.; Sonneveld, P.; Mateos, M.V.; Zamagni, E.; Avet-Loiseau, H.; Hajek, R.; Dimopoulos, M.A.; Ludwig, H.; Einsele, H.; et al. Multiple myeloma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28, iv52–iv61. [Google Scholar] [CrossRef]
- Cavo, M.; Pantani, L.; Petrucci, M.T.; Patriarca, F.; Zamagni, E.; Donnarumma, D.; Crippa, C.; Boccadoro, M.; Perrone, G.; Falcone, A.; et al. Bortezomib-thalidomide- dexamethasone is superior to thalidomide-dexamethasone as consolidation therapy after autologous hematopoietic stem cell transplantation in patients with newly diagnosed multiple myeloma. Blood 2012, 120, 9–19. [Google Scholar] [CrossRef]
- Moreau, P.; Hulin, C.; Macro, M.; Caillot, D.; Chaleteix, C.; Roussel, M.; Garderet, L.; Royer, B.; Brechignac, S.; Tiab, M.; et al. VTD is superior to VCD prior to intensive therapy in multiple myeloma: Results of the prospective IFM2013-04 trial. Blood 2016, 127, 2569–2574. [Google Scholar] [CrossRef]
- Barlogie, B.; Annaissie, E.; van Rhee, F.; Haessler, J.; Hollmig, K.; Pineda-Roman, M.; Cottler-Fox, M.; Mohiuddin, A.; Alsayed, Y.; Tricot, G.; et al. Incorporating bortezomib into upfront treatmentfor multiple myeloma: Early results of total therapy 3. Br. J. Haematol. 2007, 138, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Moreau, P.; Attal, M.; Hulin, C.; Arnulf, B.; Belhadj, K.; Benboubker, L.; Bene, M.C.; Broijl, A.; Caillon, H.; Caillot, D.; et al. Bortezomib, thalidomide, and dexamethasone with or without daratumumab before and after autologous stem-cell transplantation for newly diagnosed multiple myeloma (CASSIOPEIA): A randomised, open-label, phase 3 study. Lancet 2019, 394, 29–38. [Google Scholar] [CrossRef]
- Dimopoulos, M.A.; Moreau, P.; Terpos, E.; Mateos, M.V.; Zweegman, S.; Cook, G.; Delforge, M.; Hájek, R.; Schjesvold, F.; Cavo, M.; et al. EHA-ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2021, 32, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Jacobus, S.J.; Cohen, A.D.; Weiss, M.; Callander, N.; Singh, A.K.; Parker, T.L.; Menter, A.; Yang, X.; Parsons, B.; et al. Carfilzomib or bortezomib in combination with lenalidomide and dexamethasone for patients with newly diagnosed multiple myeloma without intention for immediate autologous stem-cell transplantation (ENDURANCE): A multicentre, open-label, phase 3, randomised, controlled trial. Lancet Oncol. 2020, 21, 1317–1330. [Google Scholar]
- Palumbo, A.; Bringhen, S.; Liberati, A.M.; Caravita, T.; Falcone, A.; Callea, V.; Montanaro, M.; Ria, R.; Capaldi, A.; Zambello, R.; et al. Oral melphalan, prednisone, and thalidomide in elderly patients with multiple myeloma: Updated results of a randomized controlled trial. Blood 2008, 112, 3107–3114. [Google Scholar] [CrossRef]
- Waage, A.; Gimsing, P.; Fayers, P.; Abildgaard, N.; Ahlberg, L.; Bjorkstand, B.; Carlson, K.; Dahl, I.M.; Forsberg, K.; Gulbrandsen, N.; et al. Melphalan and prednisone plus thalidomide or placebo in elderly patients with multiple myeloma. Blood 2010, 116, 1405–1412. [Google Scholar] [CrossRef]
- Rajkumar, S.V.; Jacobus, S.; Callander, N.S.; Fonseca, R.; Vesole, D.H.; Williams, M.E.; Abonour, R.; Siegel, D.S.; Katz, M.; Greipp, P.R.; et al. Lenalidomide plus high-dose dexamethasone versus lenalidomide plus low-dose dexamethasone as initial therapy for newly diagnosed multiple myeloma: An open-label randomised controlled trial. Lancet Oncol. 2010, 11, 29–37. [Google Scholar] [CrossRef]
- Durie, B.M.G.; Hoering, A.; Sexton, R.; Abidi, M.H.; Epstein, J.; Rajkumar, S.V.; Dispenzieri, A.; Kahanic, S.P.; Thakuri, M.C.; Reu, F.J.; et al. Longer term follow-up of the randomized phase III trial SWOG S0777: Bortezomib, lenalidomide and dexamethasone vs. lenalidomide and dexamethasone in patients (Pts) with previously untreated multiple myeloma without an intent for immediate autologous stem cell transplant (ASCT). Blood Cancer J. 2020, 10, e53. [Google Scholar]
- Facon, T.; Dimopoulos, M.A.; Dispenzieri, A.; Catalano, J.V.; Belch, A.; Cavo, M.; Pinto, A.; Weisel, K.; Ludwig, H.; Bahlis, N.J.; et al. Final analysis of survival outcomes in the phase 3 FIRST trial of up-front treatment for multiple myeloma. Blood 2018, 131, 301–310. [Google Scholar] [CrossRef]
- Palumbo, A.; Hajek, R.; Delforge, M.; Kropff, M.; Petrucci, M.T.; Catalano, J.; Gisslinger, H.; Wiktor-Jędrzejczak, J.; Zodelava, M.; Weisel, K.; et al. Continuous lenalidomide treatment for newly diagnosed multiple myeloma. N. Engl. J. Med. 2012, 366, 1759–1769. [Google Scholar] [CrossRef]
- Stewart, A.K.; Jacobus, S.; Fonseca, R.; Weiss, M.; Callander, N.S.; Chanan-Khan, A.A.; Rajkumar, S.V. Melphalan, prednisone, and thalidomide vs melphalan, prednisone, and lenalidomide (ECOG E1A06) in untreated multiple myeloma. Blood 2015, 126, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Facon, T.; Usmani, S.Z.; Plesner, T.; Orlowski, R.Z.; Touzeau, C.; Basu, S.; Bahlis, N.; Goldschmidt, H.; O’Dwyer, M.; et al. Updated Analysis of Daratumumab Plus Lenalidomide and Dexamethasone (D-Rd) Versus Lenalidomide and Dexamethasone (Rd) in Patients with Transplant-Ineligible Newly Diagnosed Multiple Myeloma (NDMM): The Phase 3 Maia Study. Blood 2020, 136, 24–26. [Google Scholar] [CrossRef]
- Wijermans, P.; Schaafsma, M.; Termorshuizen, F.; Ammerlaan, R.; Wittebol, S.; Sinnige, H.; Zweegman, S.; van Marwijk Kooy, M.; van der Griend, R.; Lokhorst, H.; et al. Phase III study of the value of thalidomide added to melphalan plus prednisone in elderly patients with newly diagnosed multiple myeloma: The HOVON 49 Study. J. Clin. Oncol. 2010, 28, 3160–3166. [Google Scholar] [CrossRef]
- Hulin, C.; Facon, T.; Rodon, P.; Pegourie, B.; Benboubker, L.; Doyen, C.; Dib, M.; Guillerm, G.; Salles, B.; Eschard, J.P.; et al. Efficacy of melphalan and prednisone plus thalidomide in patients older than 75 years with newly diagnosed multiple myeloma: IFM 01/01 trial. J. Clin. Oncol. 2009, 27, 3664–3670. [Google Scholar] [CrossRef]
- Facon, T.; Mary, J.Y.; Hulin, C.; Benboubker, L.; Attal, M.; Pegourie, B.; Renaud, M.; Harousseau, J.L.; Guillerm, G.; Chaleteix, C.; et al. Melphalan and prednisone plus thalidomide versus melphalan and prednisone alone or reduced-intensity autologous stem cell transplantation in elderly patients with multiple myeloma (IFM 99-06): A randomised trial. Lancet 2007, 370, 1209–1218. [Google Scholar] [CrossRef]
- Beksac, M.; Haznedar, R.; Firatli-Tuglular, T.; Ozdogu, H.; Aydogdu, I.; Konuk, N.; Sucak, G.; Kaygusuz, I.; Karakus, S.; Kaya, E.; et al. Addition of thalidomide to oral melphalan/prednisone in patients with multiple myeloma not eligible for transplantation: Results of a randomized trial from the Turkish Myeloma Study Group. Eur. J. Haematol. 2011, 86, 16–22. [Google Scholar] [CrossRef]
- Fayers, P.M.; Palumbo, A.; Hulin, C.; Waage, A.; Wijermans, P.; Beksac, M.; Bringhen, S.; Mary, J.Y.; Gimsing, P.; Termorshuizen, F.; et al. Thalidomide for previously untreated elderly patients with multiple myeloma: Meta-analysis of 1685 individual patient data from 6 randomized clinical trials. Blood 2011, 118, 1239–1247. [Google Scholar] [CrossRef]
- Palumbo, A.; Bringhen, S.; Larocca, A.; Rossi, D.; Di Raimondo, F.; Magarotto, V.; Patriarca, F.; Levi, A.; Benevolo, G.; Vincelli, I.D.; et al. Bortezomib-melphalan- prednisone-thalidomide followed by maintenance with bortezomib- thalidomide compared with bortezomib-melphalan-prednisone for initial treatment of multiple myeloma: Updated follow-up and improved survival. J. Clin. Oncol. 2014, 32, 634–640. [Google Scholar] [CrossRef]
- Rajkumar, S.V.; Hayman, S.R.; Lacy, M.Q.; Dispenzieri, A.; Geyer, S.M.; Kabat, B.; Zeldenrust, S.R.; Kumar, S.; Greipp, P.R.; Fonseca, R.; et al. Combination therapy with lenalidomide plus dexamethasone (Rev/Dex) for newly diagnosed myeloma. Blood 2005, 106, 4050–4053. [Google Scholar] [CrossRef]
- Roussel, M.; Lauwers-Cances, V.; Robillard, N.; Hulin, C.; Leleu, X.; Benboubker, L.; Marit, G.; Moreau, P.; Pegourie, B.; Caillot, D.; et al. Front-line transplantation program with lenalidomide, bortezomib, and dexamethasone combination as induction and consolidation followed by lenalidomide maintenance in patients with multiple myeloma: A phase II study by the Intergroupe Francophone du Myélome. J. Clin. Oncol. 2014, 32, 2712–2717. [Google Scholar]
- Rosiñol, L.; Oriol, A.; Rios, R.; Sureda, A.; Blanchard, M.J.; Hermandez, M.T.; Martinez-Martinez, R.; Moraleda, J.M.; Jarque, I.; Bargay, J.; et al. Bortezomib, lenalidomide, and dexamethasone as induction therapy prior to autologous transplant in multiple myeloma. Blood 2019, 134, 1337–1345. [Google Scholar] [CrossRef] [PubMed]
- Cavo, M.; Gay, F.; Beksac, M.; Pantani, L.; Petrucci, M.T.; Dimopoulos, M.A.; Dozza, L.; van der Holt, B.; Zweegman, S.; Oliva, S.; et al. Autologous haematopoietic stem-cell transplantation versus bortezomib-melphalan-prednisone, with or without bortezomib-lenalidomide-dexamethasone consolidation therapy, and lenalidomide maintenance for newly diagnosed multiple myeloma (EMN02/HO95): A multicentre, randomised, open-label, phase 3 study. Lancet Haematol. 2020, 7, e456–e468. [Google Scholar]
- Richardson, P.G.; Weller, E.; Lonial, S.; Jakubowiak, A.J.; Jagannath, S.; Raje, N.S.; Avigan, D.E.; Xie, W.; Ghobrial, I.M.; Schlossman, R.L.; et al. Lenalidomide, bortezomib, and dexamethasone combination therapy in patients with newly diagnosed multiple myeloma. Blood 2010, 116, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Flinn, I.; Richardson, P.G.; Hari, P.; Callander, N.; Noga, S.J.; Stewart, A.K.; Turturro, F.; Rifkin, R.; Wolf, J.; et al. Randomized, multicenter, phase 2 study (EVOLUTION) of combinations of bortezomib, dexamethasone, cyclophosphamide, and lenalidomide in previously untreated multiple myeloma. Blood 2012, 119, 4375–4382. [Google Scholar] [CrossRef] [PubMed]
- Alsina, M.; Landgren, O.; Raje, N.; Niesvizky, R.; Bensinger, W.I.; Berdeja, J.G.; Kovacsovics, T.; Vesole, D.H.; Fang, B.; Kimball, A.S.; et al. A phase 1b study of once-weekly carfilzomib combined with lenalidomide and dexamethasone in patients with newly diagnosed multiple myeloma. Am. J. Hematol. 2021, 96, 226–233. [Google Scholar] [CrossRef]
- Jasielec, J.; Kubicki, T.; Raje, N.; Vij, R.; Reece, D.; Berdeja, J.; Derman, B.A.; Rosenbaum, C.A.; Richardson, P.; Gurbuxani, S.; et al. Carfilzomib, lenalidomide, and dexamethasone plus transplant in newly diagnosed multiple myeloma. Blood 2020, 136, 2513–2523. [Google Scholar] [CrossRef] [PubMed]
- Voorhees, P.M.; Kaufman, J.L.; Laubach, J.; Sborov, D.W.; Reeves, B.; Rodriguez, C.; Chari, A.; Silbermann, R.; Costa, L.J.; Anderson, L.D., Jr.; et al. Daratumumab, lenalidomide, bortezomib, and dexamethasone for transplant-eligible newly diagnosed multiple myeloma: The GRIFFIN trial. Blood 2020, 136, 936–945. [Google Scholar] [CrossRef]
- Landgren, O.; Hultcrantz, M.; Diamond, B.; Lesokhin, A.M.; Mailankody, S.; Hassoun, H.; Tan, C.; Shah, U.A.; Lu, S.X.; Salcedo, M.; et al. Safety and effectiveness of weekly carfilzomib, lenalidomide, dexamethasone, and daratumumab combination therapy for patients with newly diagnosed multiple myeloma: The MANHATTAN nonrandomized clinical trial. JAMA Oncol. 2021, 7, 862–868. [Google Scholar] [CrossRef]
- Gay, F.; Musto, P.; Scalabrini, D.R.; Galli, M.; Belotti, A.; Zamagni, E.; Bertamini, L.; Zambello, R.; Quaresima, M.; De Sabbata, G.; et al. Survival analysis of newly diagnosed transplant-eligible multiple myeloma patients in the randomized Forte trial. Blood 2020, 136, 35–37. [Google Scholar] [CrossRef]
- Benboubker, L.; Dimopoulos, M.A.; Dispenzieri, A.; Catalano, J.; Belch, A.R.; Cavo, M.; Pinto, A.; Weisel, K.; Ludwig, H.; Bahlis, N.; et al. Lenalidomide and dexamethasone in transplant-ineligible patients with myeloma. N. Engl. J. Med. 2014, 371, 906–917. [Google Scholar] [CrossRef] [PubMed]
- Zweegman, S.; van der Holt, B.; Mellqvist, U.H.; Salomon, M.; Bos, G.M.; Levin, M.D.; Visser-Wisselaar, H.; Hansson, M.; van der Velden, A.W.G.; Deenik, W.; et al. Melphalan, prednisone, and lenalidomide versus melphalan, prednisone, and thalidomide in untreated multiple myeloma. Blood 2016, 127, 1109–1116. [Google Scholar] [CrossRef]
- Facon, T.; Kumar, S.; Plesner, R.Z.; Orlowski, R.Z.; Moreau, P.; Bahlis, N.; Basu, S.; Nahi, H.; Hulin, C.; Quach, H.; et al. Daratumumab plus Lenalidomide and Dexamethasone for Untreated Myeloma. N. Engl. J. Med. 2019, 380, 2104–2115. [Google Scholar] [CrossRef] [PubMed]
- Lokhorst, H.M.; van der Holt, B.; Zweegman, S.; Vellenga, E.; Crockewit, S.; van Oers, M.H.; von dem Borne, P.; Wijermans, P.; Schaafsma, R.; de Weerdt, O.; et al. A randomized phase 3 study on the effect of thalidomide combined with adriamycin, dexamethasone, and high-dose melphalan, followed by thalidomide maintenance in patients with multiple myeloma. Blood 2010, 115, 1113–1120. [Google Scholar] [CrossRef]
- Stewart, A.K.; Trudel, S.; Bahlis, N.J.; White, D.; Sabry, W.; Belch, A.; Reiman, T.; Roy, J.; Shustik, C.; Kovacs, M.J. A randomized phase 3 trial of thalidomide and prednisone as maintenance therapy after ASCT in patients with MM with a quality-of-life assessment: The National Cancer Institute of Canada Clinicals Trials Group Myeloma 10 Trial. Blood 2013, 121, 1517–1523. [Google Scholar] [CrossRef]
- Van de Donk, N.W.; van der Holt, B.; Minnema, M.C.; Vellenga, E.; Crookewit, S.; Kersten, M.J.; von dem Borne, P.A.; Ypma, P.; Schaafsma, R.; de Weerdt, O.; et al. Thalidomide before and after autologous stem cell transplantation in recently diagnosed multiple myeloma (HOVON-50): Long-term results from the phase 3, randomised controlled trial. Lancet Haematol. 2018, 5, e479–e492. [Google Scholar] [CrossRef]
- Morgan, G.J.; Gregory, W.M.; Davies, F.E.; Bell, S.E.; Szubert, A.J.; Brown, J.M.; Coy, N.N.; Cook, G.; Russell, N.H.; Rudin, C.; et al. The role of maintenance thalidomide therapy in multiple myeloma: MRC Myeloma IX results and meta-analysis. Blood 2012, 119, 7–15. [Google Scholar] [CrossRef]
- Ludwig, H.; Durie, B.G.M.; McCarthy, P.; Palumbo, A.; San Miguel, J.; Barlogie, B.; Morgan, G.; Sonneveld, P.; Spencer, A.; Anderson, K.C.; et al. IMWG consensus on maintenance therapy in multiple myeloma. Blood 2012, 119, 3003–3015. [Google Scholar] [CrossRef] [PubMed]
- Morgan, G.J.; Davies, F.E.; Gregory, W.M.; Bell, S.E.; Cook, G.; Drayson, M.T.; Owen, R.O.; Ross, F.M.; Jackson, G.H.; Child, J.A. Long-term follow-up of MRC Myeloma IX trial: Survival outcomes with bisphosphonate and thalidomide treatment. Clin. Cancer Res. 2013, 19, 6030–6038. [Google Scholar] [CrossRef]
- Jackson, G.; Davies, F.E.; Pawlyn, C.; Cairns, D.; Striha, A.; Hockaday, A.; Sakauskiene, I.; Jones, J.R.; Kishore, B.; Garg, M.; et al. Lenalidomide maintenance significantly improves outcomes compared to observation irrespective of cytogenetic risk: Results of the myeloma XI trial. Blood 2017, 130 (Suppl. 1), 436. [Google Scholar]
- Gay, F.; Oliva, S.; Petrucci, M.T.; Conticello, C.; Catalano, L.; Corradini, P.; Sinscalchi, A.; Magarotto, V.; Pour, L.; Carella, A.; et al. Chemotherapy plus lenalidomide versus autologous transplantation, followed by lenalidomide plus prednisone versus lenalidomide maintenance, in patients with multiple myeloma: A randomised, multicentre, phase 3 trial. Lancet Oncol. 2015, 16, 1617–1629. [Google Scholar] [CrossRef]
- Attal, M.; Lauwers-Cances, V.; Hulin, C.; Leleu, X.; Caillot, D.; Escoffre, M.; Arnulf, B.; Macro, M.; Balhadj, K.; Garderet, L.; et al. Lenalidomide, bortezomib, and dexamethasone with trans- plantation for myeloma. N. Engl. J. Med. 2017, 376, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.K.; Shah, J.J.; Morphey, A.N.; Lee, H.C.; Manasanch, E.E.; Patel, K.K.; Miller, C.P.; Phillips, S.; Crumpton, B.N.; Johnson, R.J.; et al. Updated results of a phase II study of lenalidomide- elotuzumab as maintenance therapy post-autologous stem cell transplant (AuSCT) in patients (Pts) with multiple myeloma (MM). Blood 2018, 132 (Suppl. 1), 1982. [Google Scholar] [CrossRef]
- Gambella, M.; Omede, P.; Spada, S.; Muccio, V.E.; Gilestro, M.; Saraci, E.; Grammatico, S.; Larocca, A.; Conticello, C.; Bernardini, A.; et al. Minimal residual disease by flow cytometry and allelic-specific oligonucleotide real-time quantitative polymerase chain reaction in patients with myeloma receiving lenalidomide maintenance: A pooled analysis. Cancer 2019, 125, 750–760. [Google Scholar] [CrossRef] [PubMed]
- de Tute, R.M.; Cairns, D.; Rawstron, A.; Pawlyn, C.; Davies, F.E.; Jones, J.R.; Kaiser, M.F.; Hockaday, A.; Striha, A.; Henderson, R.; et al. Minimal residual disease in the maintenance setting in myeloma: Prognostic significance and impact of lenalidomide. Blood 2017, 130 (Suppl. 1), 904. [Google Scholar] [CrossRef]
- Oliva, S.; Hofste op Bruinink, D.; Rihova, L.; D’Agostino, M.; Pantani, L.; Capra, A.; van der Holt, B.; Troia, R.; Petrucci, M.T.; Villanova, T.; et al. Minimal residual disease (MRD) assessment by multiparameter flow cytometry in transplant eligible myeloma in the EMN02/HOVON95 MM trial. Blood Cancer J. 2021, 11, 106. [Google Scholar] [CrossRef] [PubMed]
- Glasmacher, A.; Hahn, C.; Hoffmann, F.; Naumann, R.; Goldschmidt, H.; von Lilienfeld-Toal, M.; Orlopp, K.; Schmidt-Wolf, I.; Gorschluter, M. A systematic review of phase-II trials of thalidomide monotherapy in patients with relapsed or refractory multiple myeloma. Br. J. Haematol. 2006, 132, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Kropff, M.; Baylon, H.G.; Hillengass, J.; Robak, T.; Hajek, R.; Liebisch, P.; Goranov, S.; Hulin, C.; Blade, J.; Caravita, T.; et al. Thalidomide versus dexamethasone for the treatment of relapsed and/or refractory multiple myeloma: Results from OPTIMUM, a randomized trial. Haematologica 2012, 97, 784–791. [Google Scholar] [CrossRef][Green Version]
- Palumbo, A.; Facon, T.; Sonneveld, P.; Blade, J.; Offidani, M.; Gay, F.; Moreau, P.; Waage, A.; Spencer, A.; Ludwig, H.; et al. Thalidomide for treatment of multiple myeloma: 10 years later. Blood 2008, 111, 3968–3977. [Google Scholar] [CrossRef]
- Palumbo, A.; Rajkumar, S.V.; Dimopoulos, M.A.; Richardson, P.G.; San Miguel, J.; Barlogie, B.; Harousseau, J.; Zonder, J.A.; Cavo, M.; Zangari, M.; et al. Prevention of thalidomide- and lenalidomide-associated thrombosis in myeloma. Leukemia 2008, 22, 414–423. [Google Scholar] [CrossRef]
- Stewart, A.K.; Siegel, D.; Ludwig, H.; Facon, T.; Goldschmidt, H.; Jakubowiak, A.; San Miguel, J.F.; Obreja, M.; Blaedel, J.; Dimopoulos, M.A. Overall survival (OS) of patients with relapsed/refractory multiple myeloma (RRMM) treated with carfilzomib, lenalidomide, and dexamethasone (KRd) versus lenalidomide and dexamethasone (Rd): Final analysis from the randomized phase 3 ASPIRE trial. Blood 2017, 130 (Suppl. 1), 743. [Google Scholar] [CrossRef]
- Richardson, P.G.; Kumar, S.K.; Masszi, T.; Grzasko, N.; Bahlis, N.J.; Hansson, M.; Pour, L.; Sandhu, I.; Ganly, P.; Baker, B.W.; et al. Final overall survival analysis of the TOURMALINE-MM1 phase III trial of ixazomib, lenalidomide, and dexamethasone in patients with relapsed or refractory multiple myeloma. J. Clin. Oncol. 2021, 39, 2430–2442. [Google Scholar] [CrossRef]
- Bahlis, H.J.; Dimopoulos, M.A.; White, D.J.; Benboubker, L.; Cook, G.; Leiba, M.; Ho, P.J.; Kim, K.; Takezako, N.; Moreau, P.; et al. Daratumumab plus lenalidomide, and dexamethasone in relapsed/refractory multiple myeloma: Extended follow-up of POLLUX, a randomized, open-label phase 3 study. Leukemia 2020, 34, 1875–1884. [Google Scholar] [CrossRef]
- Dimopoulos, M.A.; Lonial, S.; White, D.; Moreau, P.; Palumbo, A.; San Miguel, J.; Shpilberg, O.; Anderson, K.; Grosicki, S.; Spicka, I.; et al. Elotuzumab plus lenalidomide/dexamethasone for relapsed or refractory multiple myeloma: ELOQUENT-2 follow-up and post-hoc analyses on progression-free survival and tumour growth. Br. J. Haematol. 2017, 178, 896–905. [Google Scholar] [CrossRef] [PubMed]
- San Miguel, J.; Weisel, K.; Moreau, P.; Lacy, M.; Song, K.; Delforge, M.; Karlin, L.; Goldschmidt, H.; Banos, A.; Oriol, A.; et al. Pomalidomide plus low-dose dexamethasone versus high-dose dexamethasone alone for patients with relapsed and refractory multiple myeloma (MM-003): A randomised, open-label, phase 3 trial. Lancet Oncol. 2013, 14, 1055–1066. [Google Scholar] [CrossRef]
- Baz, R.C.; Martin, T.G., 3rd; Lin, H.Y.; Zhao, X.; Shain, K.H.; Cho, H.J.; Wolf, J.L.; Mahindra, A.; Chari, A.; Sullivan, D.M.; et al. Randomized multicenter phase 2 study of pomalidomide, cyclophosphamide, and dexamethasone in relapsed refractory myeloma. Blood 2016, 127, 2561–2568. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.G.; Oriol, A.; Beksac, M.; Liberati, A.M.; Galli, M.; Schjesvold, F.; Lindsay, J.; Weisel, K.; White, D.; Facon, T. Pomalidomide, bortezomib, and dexamethasone for patients with relapsed or refractory multiple myeloma previously treated with lenalidomide (OPTIMISMM): A randomised, open-label, phase 3 trial. Lancet Oncol. 2019, 20, 781–794. [Google Scholar] [CrossRef]
- Dimopoulos, M.A.; Terpos, E.; Boccadoro, M.; Delimpasi, S.; Beksac, M.; Katodritou, E.; Moreau, P.; Baldini, L.; Symeonidis, A.; Bila, J.; et al. Daratumumab plus pomalidomide and dexamethasone versus pomalidomide and dexamethasone alone in previously treated multiple myeloma (APOLLO): An open-label, randomised, phase 3 trial. Lancet Oncol. 2021, 22, 801–812. [Google Scholar] [CrossRef]
- Attal, M.; Richardson, P.G.; Rajkumar, S.V.; San-Miguel, J.; Beksac, M.; Spicka, I.; Leleu, I.; Schjesvold, F.; Moreau, P.; Dimopoulos, M.A.; et al. Isatuximab plus poma- lidomide and low-dose dexamethasone versus pomalidomide and low-dose dexamethasone in patients with relapsed and refractory multiple myeloma (ICARIA-MM): A randomised, multicentre, open-label, phase 3 study. Lancet 2019, 394, 2096–2107. [Google Scholar] [CrossRef]
- Meletios, A.; Dimopoulos, M.A.; Dytfeld, D.; Grosicki, S.; Moreau, P.; Takezako, N.; Hori, M.; Leleu, X.; LeBlanc, R.; Suzuki, K.; et al. Elotuzumab plus Pomalidomide and Dexamethasone for Multiple Myeloma. N. Engl. J. Med. 2018, 379, 1811–1822. [Google Scholar]
- Dimopoulos, M.A.; Weisel, K.C.; Song, K.W.; Delforge, M.; Karlin, L.; Goldschmidt, H.; Moreau, P.; Banos, A.; Oriol, A.; Garderet, L.; et al. Cytogenetics and long-term survival of patients with refractory or relapsed and refractory multiple myeloma treated with pomalidomide and low-dose dexamethasone. Haematologica 2015, 100, 1327–1333. [Google Scholar] [CrossRef]
- Dimopoulos, M.A.; Palumbo, A.; Corradini, P.; Cavo, M.; Delforge, M.; Di Raimondo, F.; Weisel, K.C.; Oriol, A.; Hansson, M.; Vacca, A.; et al. Safety and efficacy of pomalidomide plus low-dose dexamethasone in STRATUS (MM-010): A phase 3b study in refractory multiple myeloma. Blood 2016, 128, 497–503. [Google Scholar] [CrossRef]
- Larocca, A.; Montefusco, V.; Bringhen, S.; Rossi, D.; Crippa, C.; Mina, R.; Galli, M.; Marcatti, M.; La Verde, G.; Giuliani, N.; et al. Pomalidomide, cyclophosphamide, and prednisone for relapsed/refractory multiple myeloma: A multicenter phase 1/2 open-label study. Blood 2013, 122, 2799–2806. [Google Scholar] [CrossRef]
- Garderet, L.; Kuhnowski, F.; Berge, B.; Roussel, M.; Escoffre-Barbe, M.; Lafon, I.; Facon, T.; Leleu, X.; Karlin, L.; Perrot, A.; et al. Pomalidomide, cyclophosphamide, and dexamethasone for relapsed multiple myeloma. Blood 2018, 132, 2555–2563. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, M.A.; Weisel, K.; Moreau, P.; Anderson, L.D., Jr.; White, D.; San-Miguel, J.; Sonneveld, P.; Engelhardt, M.; Jenner, M.; Corso, A.; et al. Pomalidomide, bortezomib, and dexamethasone for multiple myeloma previously treated with lenalidomide (OPTIMISMM): Outcomes by prior treatment at first relapse. Leukemia 2021, 35, 1722–1731. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.J.; Stadtmauer, E.A.; Abonour, R.; Cohen, A.D.; Bensinger, W.I.; Gasparetto, C.; Kaufman, J.L.; Lentzsch, S.; Vogl, D.T.; Gomes, C.L.; et al. Carfilzomib, pomalidomide, and dexamethasone for relapsed or refractory myeloma. Blood 2015, 126, 2284–2290. [Google Scholar] [CrossRef]
- Krishnan, A.; Kapoor, P.; Palmer, J.M.; Tsai, N.C.; Kumar, S.; Lonial, S.; Htut, M.; Karanes, C.; Nathwani, N.; Rosenzweig, M.; et al. Phase I/II trial of the oral regimen ixazomib, pomalidomide, and dexamethasone in relapsed/refractory multiple myeloma. Leukemia 2018, 32, 1567–1574. [Google Scholar] [CrossRef]
- Amatangelo, M.; Bjorklund, C.C.; Kang, J.; Polonskaia, A.; Viswanatha, S.; Thakurta, A. Iberdomide (CC-220) Has Synergistic Anti-Tumor and Immunostimulatory Activity Against Multiple Myeloma in Combination with Both Bortezomib and Dexamethasone, or in Combination with Daratumumab in Vitro. Blood 2018, 132 (Suppl. 1), 1935. [Google Scholar] [CrossRef]
- Schafer, P.H.; Ye, Y.; Wu, L.; Kosek, J.; Ringheim, G.; Yang, Z.; Liu, L.; Thomas, M.; Palmisano, M.; Chopra, R. Cereblon modulator iberdomide induces degradation of the transcription factors Ikaros and Aiolos: Immunomodulation in healthy volunteers and relevance to systemic lupus erythematosus. Ann. Rheum. Dis. 2018, 77, 1516–1523. [Google Scholar] [CrossRef]
- Lonial, S.; Amatangelo, M.; Popat, R.; Minnema, M.C.; Zonder, J.A.; Larsen, J.; Oriol Rocafiguera, A.; Campagnaro, E.L.; Rodriguez Otero, P.; Badros, A.Z.; et al. Translational and Clinical Evidence of a Differentiated Profile for the Novel CELMoD, Iberdomide (CC-220). Blood 2019, 134 (Suppl. 1), 3119. [Google Scholar] [CrossRef]
- Van de Donk, N.W.C.J.; Popat, R.; Larsen, J.; Minnema, M.C.; Jagannath, S.; Oriol, A.; Zonder, J.; Richardson, P.G.; Rodriguez-Otero, P.; Badros, A.Z.; et al. First results of Iberdomide (IBER.; CC-220) in combination with dexamethasone (DEX) and daratumumab (DARA) or bortezomib (BORT) in patients with relapsed/refractory multiple myeloma (RRMM). Blood 2020, 136 (Suppl. 1), 16–17. [Google Scholar] [CrossRef]
- Lonial, S.; Richardson, P.G.; Popat, R.; Stadtmauer, E.; Larsen, J.; Oriol, A.; Knop, S.; Jagannath, S.; Cook, G.; Badros, A.Z.; et al. Iberdomide (IBER) in Combination with Dexamethasone (DEX) and Daratumumab (DARA), Bortezomib (BORT), or Carfilzomib (CFZ) in Patients (PTS) with Relapsed/Refractory Multiple Myeloma (RRMM). In Proceedings of the EHA Virtual Congress Platform, Online, 9–17 June 2021; p. S187. [Google Scholar]
- Rasco, D.W.; Papadopoulos, K.P.; Pourdehnad, M.; Gandhi, A.K.; Hagner, P.R.; Li, Y.; Wei, X.; Chopra, R.; Hege, K.; DiMartino, J.; et al. A first-in-human study of novel Cereblon modulator Avadomide (CC-122) in advanced malignancies. Clin. Cancer Res. 2019, 25, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.G.; Vangsted, A.J.; Ramasamy, K.; Trudel, S.; Martinez, J.; Mateos, M.V.; Rodriguez-Otero, P.; Lonial, S.; Popat, R.; Oriol, A.; et al. First-in-human phase 1 study of the novel CELMoD agent CC-92480 combined with dexamethasone in patients with relapsed/refractory multiple myeloma. J. Clin. Oncol. 2020, 38, S208. [Google Scholar] [CrossRef]
- Chamberlain, P.P.; Cathers, B.E. Cereblon modulators: Low molecular weight inducers of protein degradation. Drug Discov. Today Technol. 2019, 31, 29–34. [Google Scholar] [CrossRef] [PubMed]


| Trial/Author | Regimen | Patients, n | ORR, % | ≥VGPR, % | Median PFS, mo or % | Median OS, mo or % |
|---|---|---|---|---|---|---|
| Transplant eligible | ||||||
| Thalidomide-based | ||||||
| Cavo et al. [39] | TD | 238 | 87 | 31 | NR; 60% at 36 mo | NR; 88% at 36 mo |
| VTD | 236 | 96 | 62 | NR; 48% at 36 mo | NR; 90% at 36 mo | |
| Moreau et al. [40] | VCD | 169 | 83 | 56 | NR | NR |
| VTD | 169 | 92 | 66 | NR | NR | |
| CASSIOPEIA [42] | Dara-VTD | 543 | 92 | 83 | NR; 93% at 18 mo | NR |
| VTD | 542 | 90 | 78 | NR; 85% at 18 mo | NR | |
| Lenalidomide-based | ||||||
| ENDURANCE [44] | VRd | 542 | 84 | 65 | 34.4 | NR; 84% at 36 mo |
| KRd | 545 | 87 | 74 | 34.6 | NR; 86% at 36 mo | |
| Transplant ineligible | ||||||
| Thalidomide-based | ||||||
| Palumbo et al. [45] | MPT | 167 | 69 | 45 | 21.8 | 45.0 |
| MP | 164 | 48 | 15 | 14.5 | 47.6 | |
| Palumbo et al. [46] | VMPT-VT | 254 | 89 | 59 | 35.3 | NR; 61% at 60 mo |
| VMP | 257 | 81 | 50 | 24.8 | NR; 51% at 60 mo | |
| Lenalidomide-based | ||||||
| Rajkumar et al. [47] | RD | 223 | 79 | NR | NR | NR; 75% at 24 mo |
| Rd | 225 | 68 | NR | NR | NR; 87% at 24 mo | |
| SWOG S0777 [48] | Rd | 261 | 79 | 53 | 29 | 69 |
| VRd | 264 | 90 | 75 | 41 | NR; 69% at 60 mo | |
| FIRST [49] | Rd (cont) | 535 | 81 | 48 | 26 | 59 |
| Rd18 | 541 | 79 | 47 | 21 | 62 | |
| MPT | 547 | 67 | 30 | 22 | 49 | |
| MM-015 [50] | MPR-R | 152 | 77 | 77 | 31 | NR; 70% at 36 mo |
| MPR | 153 | 68 | 68 | 14 | NR; 62% at 36 mo | |
| MP | 154 | 50 | 50 | 13 | NR; 66% at 36 mo | |
| ECOG E1A06 [51] | MPT-T | 154 | 75 | 25 | 21 | 52.6 |
| MPR-R | 152 | 70,4 | 32 | 19 | 47.7 | |
| MAIA [52] | Rd | 369 | 82 | 57 | 34 | NR |
| Dara-Rd | 368 | 93 | 81 | NR; 60% at 48 mo | NR | |
| Trial/Author | Regimen | Patients, n | ORR, % | ≥VGPR, % | Median PFS, mo | Median OS, mo or % |
|---|---|---|---|---|---|---|
| Lenalidomide-based | ||||||
| ASPIRE [90] | KRd | 396 | 87.1 | 70 | 26 | 48.3 |
| Rd | 396 | 66.7 | 40 | 17,6 | 40.4 | |
| TOURMALINE-MM1 [91] | Ixa-Rd | 360 | 78 | 48 | 20.6 | N/A |
| Rd | 362 | 72 | 39 | 14.7 | N/A | |
| POLLUX [92] | Dara-Rd | 286 | 93 | 80.4 | 44.5 | NR |
| Rd | 283 | 76.4 | 49.3 | 17.5 | NR | |
| ELOQUENT-2 [93] | Elo-Rd | 321 | 79 | 36 | 18.5 | 43.7 |
| Rd | 325 | 66 | 30 | 15 | 39.6 | |
| Pomalidomide-based | ||||||
| NIMBUS [94] | Pd | 302 | 31 | 7 | 4 | 12.7 |
| Dex | 153 | 10 | 1 | 1.9 | 8.1 | |
| Baz et al. [95] | PCD | 34 | 65 | 12 | 9.5 | N/A |
| Pd | 36 | 39 | 14 | 4.4 | 16.8 | |
| OPTIMISMM [96] | PVd | 281 | 82 | 52.7 | 11.2 | NR |
| Vd | 278 | 50 | 18.3 | 7.1 | NR | |
| APOLLO [97] | Dara-Pd | 151 | 69 | 51 | 12.4 | NR |
| Pd | 153 | 46 | 20 | 7 | NR | |
| ICARIA-MM [98] | Isa-Pd | 154 | 60 | 32 | 11.5 | NR; 72% at 12 mo |
| Pd | 153 | 35 | 9 | 6.5 | NR; 63% at 12 mo | |
| ELOQUENT-3 [99] | Elo-Pd | 60 | 53 | 20 | 10.3 | NR |
| Pd | 57 | 26 | 9 | 4.7 | NR | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Charliński, G.; Vesole, D.H.; Jurczyszyn, A. Rapid Progress in the Use of Immunomodulatory Drugs and Cereblon E3 Ligase Modulators in the Treatment of Multiple Myeloma. Cancers 2021, 13, 4666. https://doi.org/10.3390/cancers13184666
Charliński G, Vesole DH, Jurczyszyn A. Rapid Progress in the Use of Immunomodulatory Drugs and Cereblon E3 Ligase Modulators in the Treatment of Multiple Myeloma. Cancers. 2021; 13(18):4666. https://doi.org/10.3390/cancers13184666
Chicago/Turabian StyleCharliński, Grzegorz, David H. Vesole, and Artur Jurczyszyn. 2021. "Rapid Progress in the Use of Immunomodulatory Drugs and Cereblon E3 Ligase Modulators in the Treatment of Multiple Myeloma" Cancers 13, no. 18: 4666. https://doi.org/10.3390/cancers13184666
APA StyleCharliński, G., Vesole, D. H., & Jurczyszyn, A. (2021). Rapid Progress in the Use of Immunomodulatory Drugs and Cereblon E3 Ligase Modulators in the Treatment of Multiple Myeloma. Cancers, 13(18), 4666. https://doi.org/10.3390/cancers13184666







