Interplay between IL-10, IFN-γ, IL-17A and PD-1 Expressing EBNA1-Specific CD4+ and CD8+ T Cell Responses in the Etiologic Pathway to Endemic Burkitt Lymphoma
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Populations
2.2. Characteristics of Study Populations
2.3. T Cell Immunophenotyping by Flow Cytometry
2.4. Statistical Analysis
3. Results
3.1. Heterogeneity of EBNA1 and PfSEA-1A Specific T Cell Responses
3.2. Magnitude of Cytokine Responses to EBNA1 and PfSEA-1A
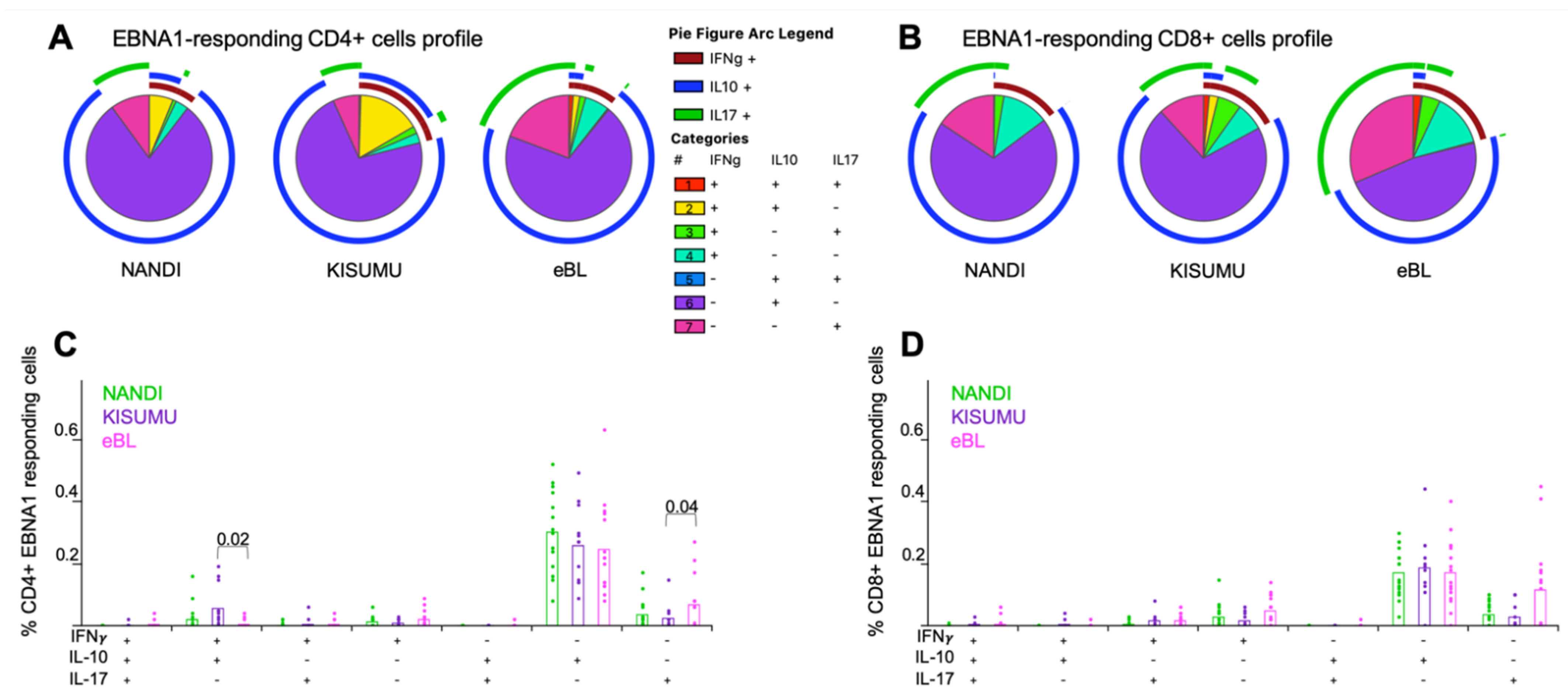
3.3. Mono- and Poly-Functional IFN-γ, IL-10 and IL-17A T Cell Responses to EBNA1 and PfSEA-1A
3.4. EBNA-1 and PfSEA-1A Specific T Cell Subsets
3.5. PD-1 Expression, as a Marker of Activation or Exhaustion
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Parkin, D.M.; Bray, F.; Ferlay, J.; Jemal, A. Cancer in Africa 2012. Cancer Epidemiol. Biomarkers Prev. 2014, 23, 953–966. [Google Scholar] [CrossRef] [Green Version]
- Stefan, C.; Bray, F.; Ferlay, J.; Liu, B.; Maxwell Parkin, D. Cancer of Childhood in Sub-Saharan Africa. Ecancermedicalscience 2017, 11. [Google Scholar] [CrossRef]
- Moormann, A.M.; Bailey, J.A. Malaria—How This Parasitic Infection Aids and Abets EBV-Associated Burkitt Lymphomagenesis. Curr. Opin. Virol. 2016, 20, 78–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burkitt, D. A Tumour Safari in East and Central Africa. Br. J. Cancer 1962, 16, 379–386. [Google Scholar] [CrossRef] [Green Version]
- De-Thé, G. The Epidemiology of Burkitt’s Lymphoma: Evidence for a Causal Association with Epstein-Barr Virus. Epidemiol. Rev. 1979, 1, 32–54. [Google Scholar] [CrossRef]
- Moormann, A.M.; Chelimo, K.; Sumba, O.P.; Lutzke, M.L.; Ploutz-Snyder, R.; Newton, D.; Kazura, J.; Rochford, R. Exposure to Holoendemic Malaria Results in Elevated Epstein-Barr Virus Loads in Children. J. Infect. Dis. 2005, 191, 1233–1238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piriou, E.; Asito, A.S.; Sumba, P.O.; Fiore, N.; Middeldorp, J.M.; Moormann, A.M.; Ploutz-Snyder, R.; Rochford, R. Early Age at Time of Primary Epstein-Barr Virus Infection Results in Poorly Controlled Viral Infection in Infants from Western Kenya: Clues to the Etiology of Endemic Burkitt Lymphoma. J. Infect. Dis. 2012, 205, 906–913. [Google Scholar] [CrossRef]
- Reynaldi, A.; Schlub, T.E.; Chelimo, K.; Sumba, P.O.; Piriou, E.; Ogolla, S.; Moormann, A.M.; Rochford, R.; Davenport, M.P. Impact of Plasmodium Falciparum Coinfection on Longitudinal Epstein-Barr Virus Kinetics in Kenyan Children. J. Infect. Dis. 2016, 213, 985–991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Snider, C.J.; Cole, S.R.; Chelimo, K.; Sumba, P.O.; Macdonald, P.D.M.; John, C.C.; Meshnick, S.R.; Moormann, A.M. Recurrent Plasmodium Falciparum Malaria Infections in Kenyan Children Diminish T-Cell Immunity to Epstein Barr Virus Lytic but Not Latent Antigens. PLoS ONE 2012, 7, e31753. [Google Scholar] [CrossRef]
- Moormann, A.M.; Chelimo, K.; Sumba, P.O.; Tisch, D.J.; Rochford, R.; Kazura, J.W. Exposure to Holoendemic Malaria Results in Suppression of Epstein-Barr Virus-Specific T Cell Immunosurveillance in Kenyan Children. J. Infect. Dis. 2007, 195, 799–808. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaymaz, Y.; Oduor, C.I.; Yu, H.; Otieno, J.A.; Ong’echa, J.M.; Moormann, A.M.; Bailey, J.A. Comprehensive Transcriptome and Mutational Profiling of Endemic Burkitt Lymphoma Reveals EBV Type-Specific Differences. Mol. Cancer Res. 2017, 15, 563–576. [Google Scholar] [CrossRef] [PubMed]
- Grande, B.M.; Gerhard, D.S.; Jiang, A.; Griner, N.B.; Abramson, J.S.; Alexander, T.B.; Allen, H.; Ayers, L.W.; Bethony, J.M.; Bhatia, K.; et al. Genome-Wide Discovery of Somatic Coding and Noncoding Mutations in Pediatric Endemic and Sporadic Burkitt Lymphoma. Blood 2019, 133, 1313–1324. [Google Scholar] [CrossRef] [PubMed]
- Panea, R.I.; Love, C.L.; Shingleton, J.R.; Reddy, A.; Bailey, J.A.; Moormann, A.M.; Otieno, J.A.; Ong’echa, J.M.; Oduor, C.I.; Schroeder, K.M.S.; et al. The Whole-Genome Landscape of Burkitt Lymphoma Subtypes. Blood 2019, 134, 1598–1607. [Google Scholar] [CrossRef]
- Roca-Feltrer, A.; Carneiro, I.; Armstrong, J.R. Estimates of the Burden of Malaria Morbidity in Africa in Children under the Age of 5 Years. Trop. Med. Int. Health 2008, 13, 771–783. [Google Scholar] [CrossRef]
- Chattopadhyay, P.K.; Chelimo, K.; Embury, P.B.; Mulama, D.H.; Sumba, P.O.; Gostick, E.; Ladell, K.; Brodie, T.M.; Vulule, J.; Roederer, M.; et al. Holoendemic Malaria Exposure Is Associated with Altered Epstein-Barr Virus-Specific CD8(+) T-Cell Differentiation. J. Virol. 2013, 87, 1779–1788. [Google Scholar] [CrossRef] [Green Version]
- Moss, D.J.; Burrows, S.R.; Castelino, D.J.; Kane, R.G.; Pope, J.H.; Rickinson, A.B.; Alpers, M.P.; Heywood, P.F. A Comparison of Epstein-Barr Virus-Specific T-Cell Immunity in Malaria-Endemic and -Nonendemic Regions of Papua New Guinea. Int. J. Cancer 1983, 31, 727–732. [Google Scholar] [CrossRef]
- Whittle, H.C.; Brown, J.; Marsh, K.; Greenwood, B.M.; Seidelin, P.; Tighe, H.; Wedderburn, L. T-Cell Control of Epstein-Barr Virus-Infected B Cells Is Lost during P. Falciparum Malaria. Nature 1984, 312, 449–450. [Google Scholar] [CrossRef]
- Moormann, A.M.; Heller, K.N.; Chelimo, K.; Embury, P.; Ploutz-Snyder, R.; Otieno, J.A.; Oduor, M.; Münz, C.; Rochford, R. Children with Endemic Burkitt Lymphoma Are Deficient in EBNA1-Specific IFN-Gamma T Cell Responses. Int. J. Cancer 2009, 124, 1721–1726. [Google Scholar] [CrossRef] [Green Version]
- Long, H.M.; Taylor, G.S.; Rickinson, A.B. Immune Defence against EBV and EBV-Associated Disease. Curr. Opin. Immunol. 2011, 23, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Hislop, A.D.; Taylor, G.S. T-Cell Responses to EBV. In Current Topics in Microbiology and Immunology; Springer: Heidelberg/Berlin, Germany, 2015; pp. 325–353. [Google Scholar]
- Shannon-Lowe, C.; Rickinson, A. The Global Landscape of EBV-Associated Tumors. Front. Oncol. 2019, 9, 713. [Google Scholar] [CrossRef] [Green Version]
- Kutok, J.L.; Wang, F. Spectrum of epstein-barr virus–associated diseases. Annu. Rev. Pathol. Mech. Dis. 2006, 1, 375–404. [Google Scholar] [CrossRef]
- Kang, M.-S.; Kieff, E. Epstein-Barr Virus Latent Genes. Exp. Mol. Med. 2015, 47, e131. [Google Scholar] [CrossRef] [Green Version]
- Paludan, C.; Schmid, D.; Landthaler, M.; Vockerodt, M.; Kube, D.; Tuschl, T.; Münz, C. Endogenous MHC Class II Processing of a Viral Nuclear Antigen after Autophagy. Science 2005, 307, 593–596. [Google Scholar] [CrossRef] [PubMed]
- Paludan, C.; Bickham, K.; Nikiforow, S.; Tsang, M.L.; Goodman, K.; Hanekom, W.A.; Fonteneau, J.-F.; Stevanovic, S.; Munz, C. Epstein-Barr Nuclear Antigen 1-Specific CD4 Th1 Cells Kill Burkitt’s Lymphoma Cells. J. Immunol. 2002, 169, 1593–1603. [Google Scholar] [CrossRef] [Green Version]
- Khanna, R.; Burrows, S.R.; Steigerwald-Mullen, P.M.; Moss, D.J.; Kurilla, M.G.; Cooper, L. Targeting Epstein-Barr Virus Nuclear Antigen 1 (EBNA1) through the Class II Pathway Restores Immune Recognition by EBNA1-Specific Cytotoxic T Lymphocytes: Evidence for HLA-DM-Independent Processing. Int. Immunol. 1997, 9, 1537–1543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.P.; Brooks, J.M.; Al-Jarrah, H.; Thomas, W.A.; Haigh, T.A.; Taylor, G.S.; Humme, S.; Schepers, A.; Hammerschmidt, W.; Yates, J.L.; et al. CD8 T Cell Recognition of Endogenously Expressed Epstein-Barr Virus Nuclear Antigen 1. J. Exp. Med. 2004, 199, 1409–1420. [Google Scholar] [CrossRef]
- Voo, K.S.; Fu, T.; Wang, H.Y.; Tellam, J.; Heslop, H.E.; Brenner, M.K.; Rooney, C.M.; Wang, R.-F. Evidence for the Presentation of Major Histocompatibility Complex Class I-Restricted Epstein-Barr Virus Nuclear Antigen 1 Peptides to CD8+ T Lymphocytes. J. Exp. Med. 2004, 199, 459–470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tellam, J.; Connolly, G.; Green, K.J.; Miles, J.J.; Moss, D.J.; Burrows, S.R.; Khanna, R. Endogenous Presentation of CD8+ T Cell Epitopes from Epstein-Barr Virus-Encoded Nuclear Antigen 1. J. Exp. Med. 2004, 199, 1421–1431. [Google Scholar] [CrossRef] [Green Version]
- O’Garra, A.; Vieira, P. T(H)1 Cells Control Themselves by Producing Interleukin-10. Nat. Rev. Immunol. 2007, 7, 425–428. [Google Scholar] [CrossRef]
- Muranski, P.; Restifo, N.P. Essentials of Th17 Cell Commitment and Plasticity. Blood 2013, 121, 2402–2414. [Google Scholar] [CrossRef]
- Chelimo, K.; Embury, P.B.; Sumba, P.O.; Vulule, J.; Ofulla, A.V.; Long, C.; Kazura, J.W.; Moormann, A.M. Age-Related Differences in Naturally Acquired T Cell Memory to Plasmodium Falciparum Merozoite Surface Protein 1. PLoS ONE 2011, 6, e24852. [Google Scholar] [CrossRef]
- Sallusto, F.; Langenkamp, A.; Geginat, J.; Lanzavecchia, A. Functional Subsets of Memory T Cells Identified by CCR7 Expression. In Current Topics in Microbiology and Immunology; Springer: Heidelberg/Berlin, Germany, 2000; pp. 167–171. [Google Scholar]
- Salisch, N.C.; Kaufmann, D.E.; Awad, A.S.; Reeves, R.K.; Tighe, D.P.; Li, Y.; Piatak, M., Jr.; Lifson, J.D.; Evans, D.T.; Pereyra, F.; et al. Inhibitory TCR Coreceptor PD-1 Is a Sensitive Indicator of Low-Level Replication of SIV and HIV-1. J. Immunol. 2010, 184, 476–487. [Google Scholar] [CrossRef] [Green Version]
- Butler, N.S.; Moebius, J.; Pewe, L.L.; Traore, B.; Doumbo, O.K.; Tygrett, L.T.; Waldschmidt, T.J.; Crompton, P.D.; Harty, J.T. Therapeutic Blockade of PD-L1 and LAG-3 Rapidly Clears Established Blood-Stage Plasmodium Infection. Nat. Immunol. 2011, 13, 188–195. [Google Scholar] [CrossRef] [Green Version]
- Gutierrez, F.R.S.; Mariano, F.S.; Oliveira, C.J.F.; Pavanelli, W.R.; Guedes, P.M.M.; Silva, G.K.; Campanelli, A.P.; Milanezi, C.M.; Azuma, M.; Honjo, T.; et al. Regulation of Trypanosoma Cruzi-Induced Myocarditis by Programmed Death Cell Receptor 1. Infect. Immun. 2011, 79, 1873–1881. [Google Scholar] [CrossRef] [Green Version]
- Bhadra, R.; Gigley, J.P.; Weiss, L.M.; Khan, I.A. Control of Toxoplasma Reactivation by Rescue of Dysfunctional CD8+ T-Cell Response via PD-1-PDL-1 Blockade. Proc. Natl. Acad. Sci. USA 2011, 108, 9196–9201. [Google Scholar] [CrossRef] [Green Version]
- Raj, D.K.; Nixon, C.P.; Nixon, C.E.; Dvorin, J.D.; DiPetrillo, C.G.; Pond-Tor, S.; Wu, H.-W.; Jolly, G.; Pischel, L.; Lu, A.; et al. Antibodies to PfSEA-1 Block Parasite Egress from RBCs and Protect against Malaria Infection. Science 2014, 344, 871–877. [Google Scholar] [CrossRef] [Green Version]
- Kaymaz, Y.; Oduor, C.I.; Aydemir, O.; Luftig, M.A.; Otieno, J.A.; Ong’echa, J.M.; Bailey, J.A.; Moormann, A.M. Epstein-Barr Virus Genomes Reveal Population Structure and Type 1 Association with Endemic Burkitt Lymphoma. J. Virol. 2020, 94, e02007–e02019. [Google Scholar] [CrossRef] [PubMed]
- Rainey, J.J.; Mwanda, W.O.; Wairiumu, P.; Moormann, A.M.; Wilson, M.L.; Rochford, R. Spatial Distribution of Burkitt’s Lymphoma in Kenya and Association with Malaria Risk. Trop. Med. Int. Health 2007, 12, 936–943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ernst, K.C.; Adoka, S.O.; Kowuor, D.O.; Wilson, M.L.; John, C.C. Malaria Hotspot Areas in a Highland Kenya Site Are Consistent in Epidemic and Non-Epidemic Years and Are Associated with Ecological Factors. Malar. J. 2006, 5, 78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forconi, C.S.; Cosgrove, C.P.; Saikumar-Lakshmi, P.; Nixon, C.E.; Foley, J.; Ong’echa, J.M.; Otieno, J.A.; Alter, G.; Münz, C.; Moormann, A.M. Poorly Cytotoxic Terminally Differentiated CD56negCD16pos NK Cells Accumulate in Kenyan Children with Burkitt Lymphomas. Blood Adv. 2018, 2, 1101–1114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heller, K.N.; Upshaw, J.; Seyoum, B.; Zebroski, H.; Münz, C. Distinct Memory CD4+ T-Cell Subsets Mediate Immune Recognition of Epstein Barr Virus Nuclear Antigen 1 in Healthy Virus Carriers. Blood 2007, 109, 1138–1146. [Google Scholar] [CrossRef]
- Sallusto, F.; Geginat, J.; Lanzavecchia, A. Central Memory and Effector Memory T Cell Subsets: Function, Generation, and Maintenance. Annu. Rev. Immunol. 2004, 22, 745–763. [Google Scholar] [CrossRef]
- Roederer, M.; Nozzi, J.L.; Nason, M.C. SPICE: Exploration and Analysis of Post-Cytometric Complex Multivariate Datasets. Cytometry A 2011, 79, 167–174. [Google Scholar] [CrossRef] [Green Version]
- Seder, R.A.; Darrah, P.A.; Roederer, M. T-Cell Quality in Memory and Protection: Implications for Vaccine Design. Nat. Rev. Immunol. 2008, 8, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Wilson, E.B.; Brooks, D.G. The Role of IL-10 in Regulating Immunity to Persistent Viral Infections. Curr. Top. Microbiol. Immunol. 2011, 350, 39–65. [Google Scholar] [PubMed] [Green Version]
- Speiser, D.E.; Utzschneider, D.T.; Oberle, S.G.; Münz, C.; Romero, P.; Zehn, D. T Cell Differentiation in Chronic Infection and Cancer: Functional Adaptation or Exhaustion? Nat. Rev. Immunol. 2014, 14, 768–774. [Google Scholar] [CrossRef]
- Long, H.M.; Chagoury, O.L.; Leese, A.M.; Ryan, G.B.; James, E.; Morton, L.T.; Abbott, R.J.M.; Sabbah, S.; Kwok, W.; Rickinson, A.B. MHC II Tetramers Visualize Human CD4+ T Cell Responses to Epstein-Barr Virus Infection and Demonstrate Atypical Kinetics of the Nuclear Antigen EBNA1 Response. J. Exp. Med. 2013, 210, 933–949. [Google Scholar] [CrossRef]
- Meckiff, B.J.; Ladell, K.; McLaren, J.E.; Ryan, G.B.; Leese, A.M.; James, E.A.; Price, D.A.; Long, H.M. Primary EBV Infection Induces an Acute Wave of Activated Antigen-Specific Cytotoxic CD4+ T Cells. J. Immunol. 2019, 203, 1276–1287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Münz, C. Cytotoxicity in Epstein Barr Virus Specific Immune Control. Curr. Opin. Virol. 2021, 46, 1–8. [Google Scholar] [CrossRef]
- Münz, C. Latency and Lytic Replication in Epstein-Barr Virus-Associated Oncogenesis. Nat. Rev. Microbiol. 2019, 17, 691–700. [Google Scholar] [CrossRef] [Green Version]
- Hviid, L.; Kurtzhals, J.A.; Adabayeri, V.; Loizon, S.; Kemp, K.; Goka, B.Q.; Lim, A.; Mercereau-Puijalon, O.; Akanmori, B.D.; Behr, C. Perturbation and Proinflammatory Type Activation of V Delta 1(+) Gamma Delta T Cells in African Children with Plasmodium Falciparum Malaria. Infect. Immun. 2001, 69, 3190–3196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hviid, L.; Kurtzhals, J.A.; Dodoo, D.; Rodrigues, O.; Rønn, A.; Commey, J.O.; Nkrumah, F.K.; Theander, T.G. The Gamma/delta T-Cell Response to Plasmodium Falciparum Malaria in a Population in Which Malaria Is Endemic. Infect. Immun. 1996, 64, 4359–4362. [Google Scholar] [CrossRef] [Green Version]
- Futagbi, G.; Welbeck, J.E.; Tetteh, J.K.A.; Hviid, L.; Akanmori, B.D. Selective Activation of TCR-Gammadelta+ Cells in Endemic Burkitt’s Lymphoma. Malar. J. 2007, 6, 69. [Google Scholar] [CrossRef] [Green Version]
- Price, A.M.; Luftig, M.A. To Be or Not IIb: A Multi-Step Process for Epstein-Barr Virus Latency Establishment and Consequences for B Cell Tumorigenesis. PLoS Pathog. 2015, 11, e1004656. [Google Scholar] [CrossRef]
- Artavanis-Tsakonas, K.; Tongren, J.E.; Riley, E.M. The War between the Malaria Parasite and the Immune System: Immunity, Immunoregulation and Immunopathology. Clin. Exp. Immunol. 2003, 133, 145–152. [Google Scholar] [CrossRef]
- Parsons, E.; Otieno, J.A.; Ong’echa, J.M.; Nixon, C.E.; Vulule, J.; Münz, C.; Stewart, V.A.; Moormann, A.M. Regulatory T Cells in Endemic Burkitt Lymphoma Patients Are Associated with Poor Outcomes: A Prospective, Longitudinal Study. PLoS ONE 2016, 11, e0167841. [Google Scholar] [CrossRef] [Green Version]
- Morales, O.; Mrizak, D.; François, V.; Mustapha, R.; Miroux, C.; Depil, S.; Decouvelaere, A.-V.; Lionne-Huyghe, P.; Auriault, C.; de Launoit, Y.; et al. Epstein-Barr Virus Infection Induces an Increase of T Regulatory Type 1 Cells in Hodgkin Lymphoma Patients. Br. J. Haematol. 2014, 166, 875–890. [Google Scholar] [CrossRef]
- Roncarolo, M.G.; Gregori, S.; Bacchetta, R.; Battaglia, M.; Gagliani, N. The Biology of T Regulatory Type 1 Cells and Their Therapeutic Application in Immune-Mediated Diseases. Immunity 2018, 49, 1004–1019. [Google Scholar] [CrossRef] [Green Version]
- McLane, L.M.; Abdel-Hakeem, M.S.; Wherry, E.J. CD8 T Cell Exhaustion During Chronic Viral Infection and Cancer. Annu. Rev. Immunol. 2019, 37, 457–495. [Google Scholar] [CrossRef] [Green Version]
- Buckle, G.; Maranda, L.; Skiles, J.; Ong’echa, J.M.; Foley, J.; Epstein, M.; Vik, T.A.; Schroeder, A.; Lemberger, J.; Rosmarin, A.; et al. Factors influencing survival among Kenyan children diagnosed with endemic Burkitt lymphoma between 2003 and 2011: A historical cohort study. Int. J. Cancer 2016, 139, 1231–1240. [Google Scholar] [CrossRef] [PubMed]







| Cytokine | Antigen Stimulation | T Cell Type | Nandi | Kisumu n (%) | eBL n (%) | Nandi vs. Kisumu (p Value) | Nandi vs. eBL (p Value) | Kisumu vs. eBL (p Value) |
|---|---|---|---|---|---|---|---|---|
| IFN-γ responders | EBNA1 | CD4 | 10/15 (66%) | 11/13 (84%) | 6/15 (40%) | 0.005 | 0.0004 | <0.0001 |
| CD8 | 10/17 (58%) | 7/11 (63%) | 8/16 (50%) | 0.56 | 0.32 | 0.008 | ||
| PfSEA-1A | CD4 | 7/12 (58%) | 10/11 (90%) | 5/12 (41%) | <0.0001 | 0.02 | <0.0001 | |
| CD8 | 4/12 (33%) | 8/10 (80%) | 5/11 (45%) | <0.0001 | 0.11 | <0.0001 | ||
| IL-10 responders | EBNA1 | CD4 | 5/15 (33%) | 5/13 (38%) | 11/15 (73%) | 0.55 | <0.0001 | <0.0001 |
| CD8 | 9/17 (52%) | 6/11 (54%) | 9/16 (56%) | 0.88 | 0.67 | 0.88 | ||
| PfSEA-1A | CD4 | 10/12 (83%) | 5/11 (45%) | 9/12 (75%) | <0.0001 | 0.22 | <0.0001 | |
| CD8 | 12/12 (100%) | 3/10 (30%) | 9/11 (81%) | <0.0001 | <0.0001 | <0.0001 | ||
| IL-17 responders | EBNA1 | CD4 | 11/15 (73%) | 6/13 (46%) | 10/15 (66%) | 0.0002 | 0.35 | 0.006 |
| CD8 | 9/17 (52%) | 2/11 (18%) | 11/16 (68%) | <0.0001 | 0.03 | <0.0001 | ||
| PfSEA-1A | CD4 | 6/12 (50%) | 6/11 (54%) | 5/12 (41%) | 0.67 | 0.25 | 0.08 | |
| CD8 | 6/12 (50%) | 5/10 (50%) | 3/11 (27%) | >0.99 | 0.001 | 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forconi, C.S.; Mulama, D.H.; Saikumar Lakshmi, P.; Foley, J.; Otieno, J.A.; Kurtis, J.D.; Berg, L.J.; Ong’echa, J.M.; Münz, C.; Moormann, A.M. Interplay between IL-10, IFN-γ, IL-17A and PD-1 Expressing EBNA1-Specific CD4+ and CD8+ T Cell Responses in the Etiologic Pathway to Endemic Burkitt Lymphoma. Cancers 2021, 13, 5375. https://doi.org/10.3390/cancers13215375
Forconi CS, Mulama DH, Saikumar Lakshmi P, Foley J, Otieno JA, Kurtis JD, Berg LJ, Ong’echa JM, Münz C, Moormann AM. Interplay between IL-10, IFN-γ, IL-17A and PD-1 Expressing EBNA1-Specific CD4+ and CD8+ T Cell Responses in the Etiologic Pathway to Endemic Burkitt Lymphoma. Cancers. 2021; 13(21):5375. https://doi.org/10.3390/cancers13215375
Chicago/Turabian StyleForconi, Catherine S., David H. Mulama, Priya Saikumar Lakshmi, Joslyn Foley, Juliana A. Otieno, Jonathan D. Kurtis, Leslie J. Berg, John M. Ong’echa, Christian Münz, and Ann M. Moormann. 2021. "Interplay between IL-10, IFN-γ, IL-17A and PD-1 Expressing EBNA1-Specific CD4+ and CD8+ T Cell Responses in the Etiologic Pathway to Endemic Burkitt Lymphoma" Cancers 13, no. 21: 5375. https://doi.org/10.3390/cancers13215375







