Opioids and Breast Cancer Recurrence: A Systematic Review
Abstract
:Simple Summary
Abstract
1. Introduction
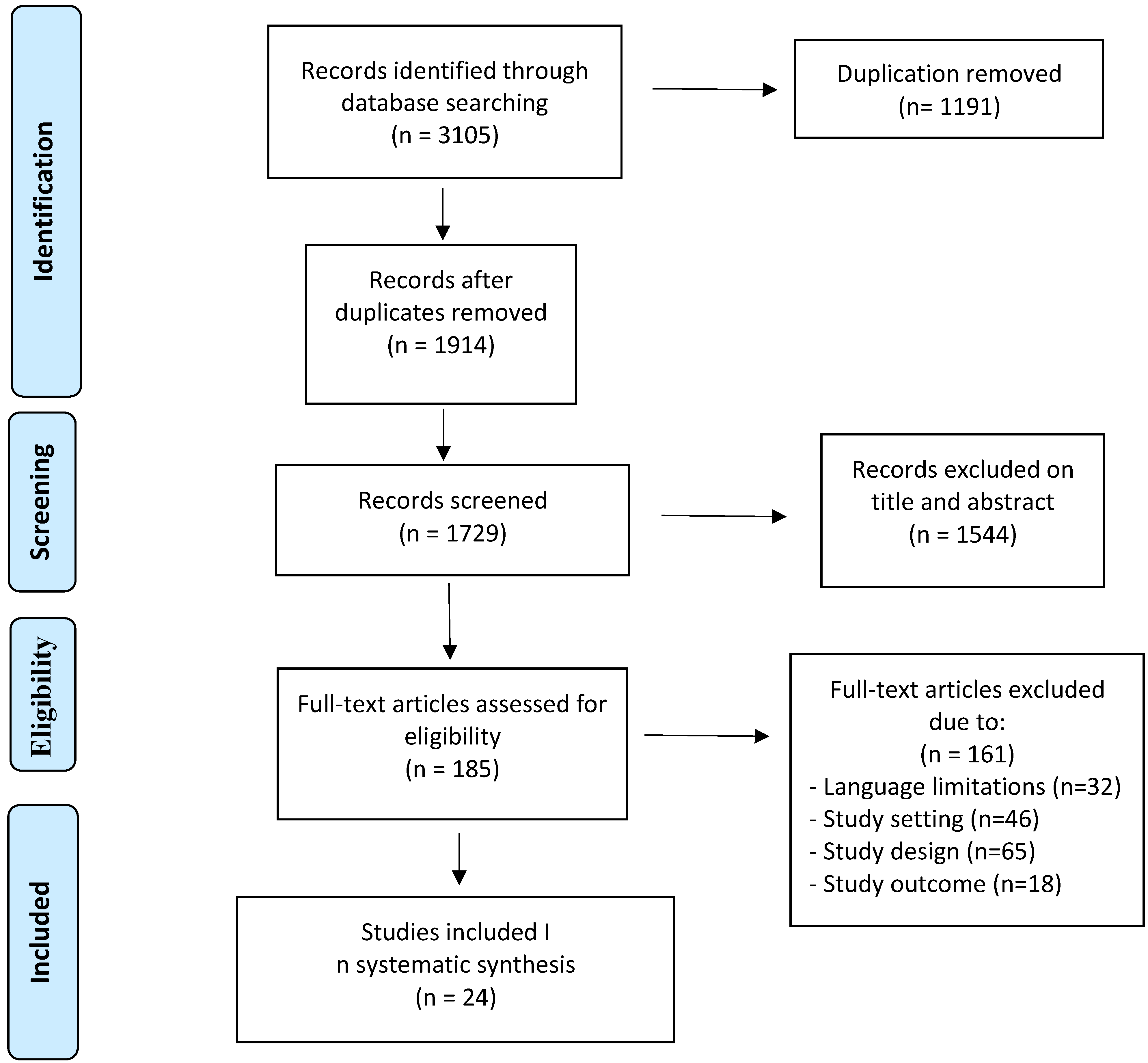
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Study Collection and Selection
3. Discussion
3.1. Opioids and Immune System
3.2. Opioids and Angiogenesis
3.3. Opioids and Tumor Growth
3.4. Chronic Use of Opioids
3.5. Clinical Data: Retrospective and Prospective Studies
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Breast Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/breast-cancer (accessed on 15 September 2021).
- Shakhar, G.; Ben-Eliyahu, S. Potential prophylactic measures against postoperative immunosuppression: Could they reduce recurrence rates in oncological patients? Ann. Surg. Oncol. 2003, 10, 972–992. [Google Scholar] [CrossRef] [PubMed]
- Kim, R. Anesthetic technique and cancer recurrence in oncologic surgery: Unraveling the puzzle. Cancer Metastasis Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Heaney, A.; Buggy, D.J. Can anaesthetic and analgesic techniques affect cancer recurrence or metastasis? Br. J. Anaesth. 2012, 109 (Suppl. S1), i17–i28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antoni, M.H.; Lutgendorf, S.K.; Cole, S.W.; Dhabhar, F.S.; Sephton, S.E.; McDonald, P.G.; Stefanek, M.; Sood, A.K. The influence of bio-behavioural factors on tumour biology: Pathways and mechanisms. Nat. Rev. Cancer 2006, 6, 240–248. [Google Scholar] [CrossRef]
- Kurosawa, S. Anesthesia in patients with cancer disorders. Curr. Opin. Anesthesiol. 2012, 25, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Sacerdote, P.; Bianchi, M.; Gaspani, L.; Manfredi, B.; Maucione, A.; Terno, G.; Ammatuna, M.; Panerai, A.E. The effects of tramadol and morphine on immune responses and pain after surgery in cancer patients. Anesth. Analg. 2000, 90, 1411–1414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lennard, T.W.J.; Shenton, B.K.; Borzotta, A.; Donnelly, P.K.; White, M.; Gerrie, L.M.; Proud, G.; Taylor, R.M. The influence of surgical operation on components of the human immune system. Br. J. Surg. 1985, 72, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Koenig, A.; Koenig, U.D.; Heicappel, R.; Stoeckel, H. Differences in lymphocyte mitogenic stimulation pattern depending on anesthesia and operative trauma: I. Halothane-nitrouse oxide anesthesia. Eur. J. Anaesthesiol. 1987, 4, 17–24. [Google Scholar] [PubMed]
- Moskowitz, M.; Feuerstein, M.; Todd, B.L. Job stress and physical activity related to elevated symptom clusters in breast cancer survivors at work. J. Occup. Environ. Med. 2013, 55, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Caraceni, A.; Hanks, G.; Kaasa, S.; Bennett, M.I.; Brunelli, C.; Cherny, N.; Dale, O.; De Conno, F.; Fallon, M.; Hanna, M.; et al. European Palliative Care Research Collaborative (EPCRC); European Association for Palliative Care (EAPC). Use of opioid analgesics in the treatment of cancer pain: Evidence-based recommendations from the EAPC. Lancet Oncol. 2012, 13, e58–e68. [Google Scholar] [CrossRef]
- Paice, J.A. Cancer pain management and the opioid crisis in America: How to preserve hard-earned gains in improving the quality of cancer pain management. Cancer 2018, 124, 2491–2497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, J.; Bauer, B.A.; Wahner-Roedler, D.L.; Chon, T.Y.; Xiao, L. The Modified WHO Analgesic Ladder: Is It Appropriate for Chronic Non-Cancer Pain? J. Pain Res. 2020, 13, 411–417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chae, B.K.; Lee, H.W.; Sun, K.; Choi, Y.H.; Kim, H.M. The effect of combined epidural and light general anesthesia on stress hormones in open heart surgery patients. Surg. Today 1998, 28, 727–731. [Google Scholar] [CrossRef] [PubMed]
- González, O.; Cuéllar-Guzmán, L.F.; Soliz, J.; Cata, J.P. Impact of Regional Anesthesia on Recurrence, Metastasis, and Immune Response in Breast Cancer Surgery: A Systematic Review of the Literature. Reg. Anesth. Pain Med. 2017, 42, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The prisma statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef] [Green Version]
- Tai, L.-H.; de Souza, C.T.; Bélanger, S.; Ly, L.; Alkayyal, A.A.; Zhang, J.; Rintoul, J.L.; Ananth, A.A.; Lam, T.; Breitbach, C.J.; et al. Preventing postoperative metastatic disease by inhibiting surgery-induced dysfunction in natural killer cells. Cancer Res. 2013, 73, 97–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mittal, D.; Gubin, M.M.; Schreiber, R.D.; Smyth, M.J. New insights into cancer immunoediting and its three component phases-elimination, equilibrium and escape. Curr. Opin. Immunol. 2014, 27, 16–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shavit, Y.; Ben-Eliyahu, S.; Zeidel, A.; Beilin, B. Effects of fentanyl on natural killer cell activity and on resistance to tumor metastasis in rats. Dose and timing study. Neuroimmunomodulation 2004, 11, 255–260. [Google Scholar] [CrossRef]
- Beilin, B.; Martin, F.C.; Shavit, Y.; Gale, R.P.; Liebeskind, J.C. Suppression of natural killer cell activity by high-dose narcotic anesthesia in rats. Brain Behav. Immun. 1989, 3, 129–137. [Google Scholar] [CrossRef]
- Beilin, B.; Shavit, Y.; Cohn, S.; Kedar, E. Narcotic-induced suppression of natural killer cell activity in ventilated and nonventilated rats. Clin. Immunol. Immunopathol. 1992, 64, 173–176. [Google Scholar] [CrossRef]
- Sultan, S.S. Paravertebral block can attenuate cytokine response when it replaces general anesthesia for cancer breast surgeries. Saudi J. Anaesth. 2013, 7, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Sessler, D.I.; Pei, L.; Huang, Y.; Fleischmann, E.; Marhofer, P.; Kurz, A.; Tey, J.B. Recurrence of breast cancer after regional or general anaesthesia: A randomised controlled trial. Lancet 2019. [Google Scholar] [CrossRef]
- Shavit, Y.; Lewis, J.W.; Terman, G.W.; Gale, R.P.; Liebeskind, J.C. Opioid peptides mediate the suppressive effect of stress on natural killer cell activity. Science 1984, 223, 188–190. [Google Scholar] [CrossRef] [PubMed]
- Snyder, G.L.; Greenberg, S. Effect of anaesthetic technique and other perioperative factors on cancer recurrence. Br. J. Anaesth. 2010, 105, 106–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greenfeld, K.; Avraham, R.; Benish, M.; Goldfarb, Y.; Rosenne, E.; Shapira, Y.; Rudich, T.; Ben-Eliyahu, S. Immune suppression while awaiting surgery and following it: Dissociations between plasma cytokine levels, their induced production, and NK cell cytotoxicity. Brain Behav. Immun. 2007, 21, 503–513. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beilin, B.; Shavit, Y.; Hart, J.; Mordashov, B.; Cohn, S.; Notti, I.; Bessler, H. Effects of Anesthesia Based on Large Versus Small Doses of Fentanyl on Natural Killer Cell Cytotoxicity in the Perioperative Period. Anesth. Analg. 1996, 82, 492–497. [Google Scholar] [CrossRef]
- Ma, J.; Wang, J.; Wan, J.; Charboneau, R.; Chang, Y.; Barke, R.A.; Roy, S. Morphine disrupts interleukin-23 (IL-23)/IL-17-mediated pulmonary mucosal host defense against Streptococcus pneumoniae infection. Infect. Immun. 2010, 78, 830–837. [Google Scholar] [CrossRef] [Green Version]
- Sacerdote, P.; Manfredi, B.; Mantegazza, P.; Panerai, A.E. Antinociceptive and immunosuppressive effects of opiate drugs: A structure-related activity study. Br. J. Pharmacol. 1997, 121, 834–840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pollock, R.E.; Lotzova, E. Surgical-stress-related suppression of natural killer cell activity: A possible role in tumor metastasis. Nat. Immun. Cell Growth Regul. 1987, 6, 269–278. [Google Scholar] [PubMed]
- Desborough, J.P. The stress response to trauma and surgery. Br. J. Anaesth. 2000, 85, 109–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desmond, F.; McCormack, J.; Mulligan, N.; Stokes, M.; Buggy, D.J. Effect of anaesthetic technique on immune cell infiltration in breast cancer: A follow-up pilot analysis of a prospective, randomised, investigator-masked study. Anticancer. Res. 2015, 35, 1311–1319. [Google Scholar]
- Jaura, A.I.; Flood, G.; Gallagher, H.C.; Buggy, D.J. Differential effects of serum from patients administered distinct anaesthetic techniques on apoptosis in breast cancer cells in vitro: A pilot study. Br. J. Anaesth. 2014, 113 (Suppl. S1), i63–i67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Page, G.G.; Blakely, W.P.; Ben-Eliyahu, S. Evidence that postoperative pain is a mediator of the tumor-promoting effects of surgery in rats. Pain 2001, 90, 191–199. [Google Scholar] [CrossRef]
- Page, G.G. Immunologic effects of opioids in the presence or absence of pain. J. Pain Symptom. Manag. 2005, 29, S25–S31. [Google Scholar] [CrossRef] [PubMed]
- Lyden, D.; Hattori, K.; Dias, S.; Costa, C.; Blaikie, P.; Butros, L.; Chadburn, A.; Heissig, B.; Marks, W.; Witte, L.; et al. Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth. Nat. Med. 2001, 7, 1194–1201. [Google Scholar] [CrossRef]
- Gupta, K.; Kshirsagar, S.; Chang, L.; Schwartz, R.; Law, P.Y.; Yee, D.; Hebbel, R.P. Morphine stimulates angiogenesis by activating proangiogenic and survival-promoting signaling and promotes breast tumor growth. Cancer Res. 2002, 62, 4491–4498. [Google Scholar]
- Folkman, J. Angiogenesis in cancer, vascular, rheumatoid, and other diseases. Nat. Med. 1995, 1, 27–31. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef]
- Risau, W.; Flamme, I. Vasculogenesis. Annu. Rev. Cell Dev. Biol. 1995, 11, 73–91. [Google Scholar] [CrossRef] [PubMed]
- Ash, S.A.; Buggy, D.J. Does regional anaesthesia and analgesia or opioid analgesia influence recurrence after primary cancer Surgery? An update of available evidence. Best Pract. Res. Clin. Anaesthesiol. 2013, 27, 441–456. [Google Scholar] [CrossRef] [Green Version]
- Singleton, P.A.; Lingen, M.W.; Fekete, M.J.; Garcia, J.G.; Moss, J. Methylnaltrexone inhibits opiate and VEGF-induced angiogenesis: Role of receptor transactivation. Microvasc. Res. 2006, 72, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Bernabé, D.G.; Tamae, A.C.; Biasoli, É.R.; Oliveira, S.H. Stress hormones increase cell proliferation and regulates interleukin-6 secretion in human oral squamous cell carcinoma cells. Brain Behav. Immun. 2011, 25, 574–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, E.V.; Kim, S.J.; Donovan, E.L.; Chen, M.; Gross, A.C.; Webster Marketon, J.I.; Barsky, S.H.; Glaser, R. Norepinephrine upregulates VEGF, IL-8, and IL-6 expression in human melanoma tumor cell lines: Implications for stress-related enhancement of tumor progression. Brain Behav. Immun. 2009, 23, 267–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marik, P.E.; Flemmer, M. Immunonutrition in the surgical patient. Minerva Anestesiol. 2012, 78, 336–342. [Google Scholar] [CrossRef]
- Sica, A.; Schioppa, T.; Mantovani, A.; Allavena, P. Tumour-associated macrophages are a distinct M2 polarised population promoting tumour progression: Potential targets of anti-cancer therapy. Eur. J. Cancer 2006, 42, 717–727. [Google Scholar] [CrossRef]
- Obermajer, N.; Wong, J.L.; Edwards, R.P.; Odunsi, K.; Moysich, K.; Kalinski, P. PGE2-driven induction and maintenance of cancer-associated myeloid-derived suppressor cells. Immunol. Investig. 2012, 41, 635–657. [Google Scholar] [CrossRef]
- Gach, K.; Szemraj, J.; Wyrebska, A.; Janecka, A. The influence of opioids on matrix metalloproteinase-2 and -9 secretion and mRNA levels in MCF-7 breast cancer cell line. Mol. Biol. Rep. 2011, 38, 1231–1236. [Google Scholar] [CrossRef]
- Al-Hasani, R.; Bruchas, M.R. Molecular mechanisms of opioid receptor-dependent signaling and behavior. Anesthesiology 2011, 115, 1363–1381. [Google Scholar] [CrossRef] [Green Version]
- Bruera, E.; Paice, J.A. Cancer pain management: Safe and effective use of opioids. Am. Soc. Clin. Oncol. Educ. Book 2015, e593–e599. [Google Scholar] [CrossRef] [PubMed]
- Arthur, J.; Bruera, E. Balancing opioid analgesia with the risk of nonmedical opioid use in patients with cancer. Nat. Rev. Clin. Oncol. 2019, 16, 213–226. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Xu, Q.X.; Liao, L.D.; Xu, X.E.; Wu, J.Y.; Wu, Z.Y.; Shen, J.H.; Li, E.M.; Xu, L.Y. Association of mu-opioid receptor expression with lymph node metastasis in esophageal squamous cell carcinoma. Dis. Esophagus 2015, 28, 196–203. [Google Scholar] [CrossRef]
- Weingaertner, I.R.; Koutnik, S.; Ammer, H. Chronic morphine treatment attenuates cell growth of human BT474 breast cancer cells by rearrangement of the ErbB signalling network. PLoS ONE 2013, 8, e53510. [Google Scholar] [CrossRef] [Green Version]
- Lin, X.; Wang, Y.-J.; Li, Q.; Hou, Y.-Y.; Hong, M.-H.; Cao, Y.-L.; Chi, Z.-Q.; Liu, J.-G. Chronic High-Dose Morphine Treatment Promotes SH-SY5Y Cell Apoptosis via c-Jun N-terminal Kinase-Mediated Activation of Mitochondria-Dependent Pathway. FEBS J. 2009, 276, 2022–2036. [Google Scholar] [CrossRef]
- Wang, C.Z.; Li, X.L.; Sun, S.; Xie, J.T.; Aung, H.H.; Tong, R.; McEntee, E.; Yuan, C.S. Methylnaltrexone, a peripherally acting opioid receptor antagonist, enhances tumoricidal effects of 5-Fu on human carcinoma cells. Anticancer. Res. 2009, 29, 2927–2932. [Google Scholar]
- Farooqui, M.; Geng, Z.H.; Stephenson, E.J.; Zaveri, N.; Yee, D.; Gupta, K. Naloxone acts as an antagonist of estrogen receptor activity in MCF-7 cells. Mol. Cancer Ther. 2006, 5, 611–620. [Google Scholar] [CrossRef] [Green Version]
- Maneckjee, R.; Biswas, R.; Vonderhaar, B.K. Binding of opioids to human MCF-7 breast cancer cells and their effects on growth. Cancer Res. 1990, 50, 2234–2238. [Google Scholar]
- McLaughlin, P.J.; Zagon, I.S. Novel treatment for triple-negative breast and ovarian cancer: Endogenous opioid suppression of women’s cancers. Expert Rev. Anticancer. Ther. 2014, 14, 247–250. [Google Scholar] [CrossRef] [Green Version]
- Lysle, D.T.; Coussons, M.E.; Watts, V.J.; Bennett, E.H.; Dykstra, L.A. Morphine-induced Modulation of Immune Status: Evidence for Opioid Receptor Mediation and Compartment Specificity. Adv. Exp. Med. Biol. 1993, 335, 53–59. [Google Scholar] [CrossRef]
- Gach, K.; Wyrebska, A.; Fichna, J.; Janecka, A. The role of morphine in regulation of cancer cell growth. Naunyn Schmiedebergs Arch. Pharmacol. 2011, 384, 221–230. [Google Scholar] [CrossRef] [Green Version]
- Singleton, P.A.; Moss, J. Effect of perioperative opioids on cancer recurrence: A hypothesis. Future Oncol. 2010, 6, 1237–1242. [Google Scholar] [CrossRef]
- Nguyen, J.; Luk, K.; Vang, D.; Soto, W.; Vincent, L.; Robiner, S.; Saavedra, R.; Li, Y.; Gupta, P.; Gupta, K. Morphine stimulates cancer progression and mast cell activation and impairs survival in transgenic mice with breast cancer. Br. J. Anaesth. 2014, 113 (Suppl. S1), i4–i13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cronin-Fenton, D.P.; Heide-Jorgensen, U.; Ahern, T.P.; Lash, T.L.; Christiansen, P.M.; Ejlertsen, B.; Sjøgren, P.; Kehlet, H.; Sørensen, H.T. Opioids and breast cancer recurrence: A Danish population-based cohort study. Cancer 2015, 121, 3507–3514. [Google Scholar] [CrossRef] [Green Version]
- Ekholm, O.; Kurita, G.P.; Hojsted, J.; Juel, K.; Sjogren, P. Chronic pain, opioid prescriptions, and mortality in Denmark: A population-based cohort study. Pain 2014, 155, 2486–2490. [Google Scholar] [CrossRef]
- Blais, L.; Ernst, P.; Suissa, S. Confounding by indication and channeling over time: The risks of beta 2-agonists. Am. J. Epidemiol. 1996, 144, 1161–1169. [Google Scholar] [CrossRef]
- Boudreau, D.M.; Chen, L.; Yu, O.; Bowles, E.J.A.; Chubak, J. Risk of second breast cancer events with chronic opioid use in breast cancer survivors. Pharmacoepidemiol. Drug Saf. 2019, 28, 740–753. [Google Scholar] [CrossRef]
- Exadaktylos, A.K.; Buggy, D.J.; Moriarty, D.C.; Mascha, E.; Sessler, D.I. Can anesthetic technique for primary breast cancer surgery affect recurrence or metastasis? Anesthesiology 2006, 105, 660–664. [Google Scholar] [CrossRef] [Green Version]
- Cata, J.P.; Chavez-MacGregor, M.; Valero, V.; Black, W.; Black, D.M.; Goravanchi, F.; Ifeanyi, I.C.; Hernandez, M.; Rodriguez-Restrepo, A.; Gottumukkala, V. The Impact of Paravertebral Block Analgesia on Breast Cancer Survival After Surgery. Reg. Anesth. Pain Med. 2016, 41, 696–703. [Google Scholar] [CrossRef]
- Forget, P.; Vandenhende, J.; Berliere, M.; Machiels, J.-P.; Nussbaum, B.; Legrand, C.; De Kock, M. Do Intraoperative Analgesics Influence Breast Cancer Recurrence After Mastectomy? A Retrospective Analysis. Anesth. Analg. 2010, 110, 1630–1635. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.H.; Kang, S.H.; Kim, Y.; Kim, H.A.; Kim, B.S. Effects of propofol-based total intravenous anesthesia on recurrence and overall survival in patients after modified radical mastectomy: A retrospective study. Korean J. Anesthesiol. 2016, 69, 126. [Google Scholar] [CrossRef]
- Kim, M.H.; Oh, J.E.; Park, S.; Kim, J.H.; Lee, K.Y.; Bai, S.J.; Song, H.; Hwang, H.J.; Kim, D.W.; Yoo, Y.C. Tramadol use is associated with enhanced postoperative outcomes in breast cancer patients: A retrospective clinical study with in vitro confirmation. Br. J. Anaesth. 2019. [Google Scholar] [CrossRef] [PubMed]
- Montagna, G.; Gupta, H.V.; Hannum, M.; Tan, K.S.; Lee, J.; Scarpa, J.R.; Plitas, G.; Irie, T.; McCormick, P.J.; Fischer, G.W.; et al. Intraoperative opioids are associated with improved recurrence-free survival in triple-negative breast cancer. Br. J. Anaesth. 2021, 126, 367–376. [Google Scholar] [CrossRef] [PubMed]
| Title, Year | Design | Drug | Outcome | Results |
|---|---|---|---|---|
| Shavit et al. [17] 1984 | In vivo study | Morphine | NKCC | High dose of morphine suppresses NKCC |
| Beilin et al. [18] 1989 | In vivo study | Morphine Fentanyl Sufentanil | NKCC | All three drugs suppress NK cytotoxicity, and this effect was blocked by naltrexone |
| Beilin et al. [19] 1992 | In vivo study | Fentanyl | NKCC | Fentanyl significantly suppressed NK cell activity both in ventilated and non-ventilated rats |
| Ma et al. [20] 2010 | In vivo study | Morphine | Neutrophil recruitment | Morphine treatment causes a compromised neutrophil recruitment |
| Sacerdote et al. [21] 1997 | In vivo study | Morphine | Lymphocyte proliferation Cytokines production | Morphine exerts an inhibitory effect on lymphocyte proliferation and IL-2 production |
| Jaura et al. [22] 2014 | In vitro study | Sevoflurane/fentanyl vs. propofol/paravertebral block | Apoptosis in ER-negative breast cancer cells | Serum from patients undergoing PVA produced apoptosis to a greater degree in ER-negative breast cancer cells compared with sevoflurane/opioid anesthesia group |
| Page et al. [23] 2001 | In vivo study | Fentanyl | NKCC | Fentanyl suppressed NK activity at this time point in non-operated rats, but had no effect in operated rats |
| Title, Year | Design | Drug | Outcome | Results |
|---|---|---|---|---|
| Beilin et al. [24] 1996 | Clinical randomized study | Fentanyl | NKCC | LDFA causes prolonged suppression of NK cell function vs. SDFA |
| Sultan et al. [25] 2013 | Clinical randomized study | Sevoflurane/fentanyl vs. propofol/paravertebral block | Cytokines production | Blood samples from patients undergoing PVA (propofol/paravertebral block group) for breast cancer surgery showed lower cytokines levels vs. patients undergoing GA (sevoflurane/fentanyl group) |
| Desmond et al. [26] 2015 | Controlled clinical randomized study | Sevoflurane/fentanyl vs. propofol/paravertebral block | NK and T-helper cells infiltration | Excised breast cancer specimens from the propofol-paravertebral anesthesia group had increased infiltration of NK and T-helper cells compared with sevoflurane/opioid group |
| Title, Year | Design | Drug | Outcome | Results |
|---|---|---|---|---|
| Gupta et al. [34] 2002 | In vitro-in vivo study | Morphine | Angiogenesis and breast tumor growth | Morphine stimulates MAPK/ERK phosphorylation and survival signaling via Act phosphorylation and cyclin D1, promoting in vitro angiogenesis and breast tumor growth in female nude mice |
| Singleton et al. [37] 2006 | In vitro study | MNTX MS | Endothelial cell proliferation and migration | MNTX, a mu opioid antagonist, inhibits morphine-induced endothelial cell proliferation and migration |
| Title, Year | Design | Drug | Outcome | Results |
|---|---|---|---|---|
| Weingaertner et al. [51] 2013 | In vitro study | Morphine | Breast cancer cells growth | Chronic morphine treatment attenuates cell growth of Human BT474 breast cancer cells by rearrangement of the ErbB signaling network |
| Farooqui et al. [53] 2006 | In vitro study | Naloxone | Breast cancer cells growth | Naloxone has inhibitory effects on tumor growth by antagonizing E2- induced MAPK/ERK phosphorylation in breast cancer cell line MCF-7. |
| Maneckjee et al. [54] 1990 | In vitro study | Morphine Naloxone | Breast cancer cells growth | Opioid ligands specific for the different receptor types significantly inhibited the growth of the breast cancer cell line MCF-7 in a dose-dependent manner and this inhibitory effect was reversed by the simultaneous administration of the opioid receptor antagonist, naloxone |
| Nguyen et al. [55] 2014 | In vitro study | Morphine | Cancer progression | Morphine does not affect the onset of tumor development, but it promotes the growth of existing tumors, and reduces overall survival in mice |
| Title, Year | Design | Outcome | Population | Results |
|---|---|---|---|---|
| Sessler et al. [32] 2019 | Prospective cohort study | RFS | Regional anestesia (n = 1043) vs. General anestesia (n = 1065) | Regional anesthesia-analgesia (paravertebral block and propofol) did not reduce breast cancer recurrence after potentially curative surgery compared with volatile anesthesia (sevoflurane) and opioids |
| Cronine-Fenton et al. [61] 2015 | Prospective cohort study | RFS and OS | Opioid Nonusers (n = 18,231) vs. Opioid Users (n = 15,957) | There was no association between ever-use of opioids and breast cancer recurrence. |
| Exadaktylos et al. [65] 2006 | Retrospective study | RFS | GA/PVA (n = 50) vs. GA/opioid anestesia (n = 70) | Reduction in cancer recurrence and metastasis free survival rate during the initial years of follow-up in the group GA/PVA vs. GA/opioid anesthesia |
| Cata et al. [66] 2016 | Retrospective study | RFS and OS | PVB (n = 198) vs. Opioid-based analgesia (n = 594) | The use of PVB is not associated with a significant change in RFS [HR 1.60; 95% CI (0.81–3.16), p = 0.172] or OS [HR 1.28; 95% CI (0.55–3.01) p = 0.567] |
| Forget et al. [67] 2010 | Retrospective study | RFS | Patients n = 327 (Ketorolac n = 175; Sufentanil n = 227; Clonidine n = 184; Ketamine n = 166; Piritramide n = 159; Diclofenac n = 223) | Sufentanil had no deleterious effect on cancer recurrence |
| Lee et al. [68] 2016 | Retrospective study | RFS and OS | Propofol group (n = 173) vs. Sevoflurane group (n = 152) | Propofol-based total intravenous anesthesia for MRM reduced the risk of recurrence of breast cancer during the initial 5 years of follow-up, compared with sevoflurane-based anesthesia |
| Kim et al. [69] 2019 | Retrospective study | RFS and OS | Tramadol use (n = 943) vs. Non-Tramadol (n = 1645) | The use of tramadol as a rescue analgesic after breast cancer surgery was associated with a decreased risk of postoperative disease recurrence rate (7.3% in the tramadol group vs. 9.4% in the control group) and lower mortality (4.7% vs. 8.1%, p = 0.001) |
| Montagna et al. [70] 2021 | Retrospective study | RFS and OS | 1143 TNBC Cases | Higher intraoperative opioid dose was associated with favorable recurrence-free survival, with a hazard ratio of 0.93 (95% confidence interval 0.88–0.99) per 10 oral morphine milligram equivalents increase (p = 0.028), but was not significantly associated with overall survival, with a hazard ratio of 0.96 (95% confidence interval 0.89–1.02) per 10 morphine milligram equivalents increase (p = 0.2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lucia, M.; Luca, T.; Federica, D.P.; Cecilia, G.; Chiara, M.; Laura, D.M.; Carlo, D.R.; Grazia, P.M. Opioids and Breast Cancer Recurrence: A Systematic Review. Cancers 2021, 13, 5499. https://doi.org/10.3390/cancers13215499
Lucia M, Luca T, Federica DP, Cecilia G, Chiara M, Laura DM, Carlo DR, Grazia PM. Opioids and Breast Cancer Recurrence: A Systematic Review. Cancers. 2021; 13(21):5499. https://doi.org/10.3390/cancers13215499
Chicago/Turabian StyleLucia, Merlino, Titi Luca, Del Prete Federica, Galli Cecilia, Mandosi Chiara, De Marchis Laura, Della Rocca Carlo, and Piccioni Maria Grazia. 2021. "Opioids and Breast Cancer Recurrence: A Systematic Review" Cancers 13, no. 21: 5499. https://doi.org/10.3390/cancers13215499






