A Tool for Rapid Assessment of Functional Outcomes in Patients with Head and Neck Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Identification of HNC-Relevant Functional Domains
2.2. External Criteria for Uniform Scoring and Draft Development
2.3. Draft Revision through Semi-Structured Expert Interviews
2.4. Empirical Validation of the Adapted HNC-FIT Scale
2.4.1. Patients and Controls
2.4.2. Empirical Validity, Reliability, Responsiveness, and Fill-In Time
2.4.3. Sample Size Estimation
2.5. Data Analysis
3. Results
3.1. HNC-Relevant Functional Domains
3.2. External Criteria for Uniform Scoring
3.3. Draft Revision through Semi-Structured Expert Interviews
3.4. Empirical Validation of the Expert Adapted HNC-FIT Scale
3.4.1. Patients and Controls
3.4.2. Empirical Validity, Reliability, Responsiveness, and Fill-In Time
4. Discussion
4.1. Introduction
4.2. Development
4.3. Mode of Administration
4.4. Empirical Validation
4.5. Presentation of Functional Outcomes
4.6. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chow, L.Q.M. Head and neck cancer. N. Engl. J. Med. 2020, 382, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.E.; Burtness, B.; Leemans, C.R.; Lui, V.W.Y.; Bauman, J.E.; Grandis, J.R. Head and neck squamous cell carcinoma. Nat. Rev. Dis. Primers 2020, 6, 92. [Google Scholar] [CrossRef] [PubMed]
- León, X.; Orús, C.; Casasayas, M.; Neumann, E.; Holgado, A.; Quer, M. Trends in disease-specific survival of head and neck squamous cell carcinoma patients treated in a single in-stitution over a 30-year period. Oral Oncol. 2021, 115, 105184. [Google Scholar] [CrossRef]
- Brotherston, D.C.; Poon, I.; Le, T.; Leung, M.; Kiss, A.; Ringash, J.; Balogh, J.; Lee, J.; Wright, J.R. Patient preferences for oropharyngeal cancer treatment de-escalation. Head Neck 2012, 35, 151–159. [Google Scholar] [CrossRef] [PubMed]
- List, M.A.; Rutherford, J.L.; Stracks, J.; Pauloski, B.R.; Logemann, J.A.; Lundy, D.; Sullivan, P.; Goodwin, W.; Kies, M.; Vokes, E.E. Prioritizing treatment outcomes: Head and neck cancer patients versus nonpatients. Head Neck 2004, 26, 163–170. [Google Scholar] [CrossRef] [PubMed]
- List, M.A.; Stracks, J.; Colangelo, L.; Butler, P.; Ganzenko, N.; Lundy, D.; Sullivan, P.; Haraf, D.; Kies, M.; Goodwin, W.; et al. How Do Head and Neck Cancer Patients Prioritize Treatment Outcomes Before Initiating Treatment? J. Clin. Oncol. 2000, 18, 877. [Google Scholar] [CrossRef]
- Ringash, J.; Bernstein, L.; Devins, G.; Dunphy, C.; Giuliani, M.; Martino, R.; McEwen, S. Head and Neck Cancer Survivorship: Learning the Needs, Meeting the Needs. Semin. Radiat. Oncol. 2018, 28, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Osborn, H.A.; Ma, T.A.G.; Varvares, M.A. Assessing functional outcomes in head and neck surgical oncology. Head Neck 2019, 41, 2051–2057. [Google Scholar] [CrossRef]
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). 2017; p. 155. Available online: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm (accessed on 3 November 2021).
- List, M.A.; D’Antonio, L.L.; Cella, D.F.; Siston, A.; Mumby, P.; Haraf, D.; Vokes, E. The performance status scale for head and neck cancer patients and the functional assessment of cancer therapy-head and neck scale: A study of utility and validity. Cancer 1996, 77, 2294–2301. [Google Scholar] [CrossRef]
- Weymuller, E.A., Jr.; Alsarraf, R.; Yueh, B.; Deleyiannis, F.W.B.; Coltrera, M.D. Analysis of the performance characteristics of the University of Washington Quality of Life in-strument and its modification (UW-QOL-R). Arch. Otolaryngol. Head Neck Surg. 2001, 127, 489–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogers, S.N.; Gwanne, S.; Lowe, D.; Humphris, G.; Yueh, B.; Weymuller, E.A., Jr. The addition of mood and anxiety domains to the University of Washington quality of life scale. Head Neck 2002, 24, 521–529. [Google Scholar] [CrossRef]
- Rogers, S.N.; Lowe, D.; Yueh, B.; Weymuller, E.A., Jr. The Physical Function and Social-Emotional Function Subscales of the University of Washington Quality of Life Questionnaire. Arch. Otolaryngol. Head Neck Surg. 2010, 136, 352–357. [Google Scholar] [CrossRef] [Green Version]
- D’Antonio, L.L.; Zimmerman, G.J.; Cella, D.F.; Long, S.A. Quality of Life and Functional Status Measures in Patients with Head and Neck Cancer. Arch. Otolaryngol. Head Neck Surg. 1996, 122, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Bjordal, K.; De Graeff, A.; Fayers, P.M.; Hammerlid, E.; van Pottelsberghe, C.; Curran, D.; Ahlner-Elmqvist, M.; Maher, E.J.; Meyza, J.W.; Bredart, A.; et al. A 12 country field study of the EORTC QLQ-C30 (version 3.0) and the head and neck cancer specific mod-ule (EORTC QLQ-H&N35) in head and neck patients. EORTC Quality of Life Group. Eur. J. Cancer 2000, 36, 1796–1807. [Google Scholar] [PubMed]
- World Health Organization. International Classification of Functioning, Disability and Health: ICF; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Kisser, U.; Adderson-Kisser, C.; Coenen, M.; Stier-Jarmer, M.; Becker, S.; Sabariego, C.; Harréus, U. The development of an ICF-based clinical guideline and screening tool for the standardized assessment and evaluation of functioning after head and neck cancer treatment. Eur. Arch. Oto-Rhino-Laryngol. 2016, 274, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.; Aaronson, N.; Blazeby, J.M.; Bottomley, A.; Fayers, P.; Koller, M.; Kuliś, D.; Ramage, J.; Sprangers, M.; Velikova, G.; et al. Guidelines for Developing Questionnaire Modules: EORTC Quality of Life Group. 2011. Available online: https://www.eortc.org/app/uploads/sites/2/2018/02/guidelines_for_developing_questionnaire-_final.pdf (accessed on 3 November 2021).
- Streiner, D.L.; Norman, G.R.; Cairney, J. Health Measurement Scales: A Practical Guide to Their Development and Use, 5th ed.; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; L. Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical power analyses using G*Power 3.1: Tests for correlation and regression analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef] [Green Version]
- Arifin, W.N. Sample Size Calculator. 2021. Available online: http://wnarifin.github.io (accessed on 29 September 2021).
- Holm, S. A simple sequential rejective method procedure. Scand. J. Stat. 1979, 6, 65–70. [Google Scholar]
- Portney, L.G.; Watkins, M.P. Foundations of Clinical Research: Applications to Practice, 3rd ed.; Prentice Hall: Hoboken, NJ, USA, 2009. [Google Scholar]
- Riedl, D.; Dejaco, D.; Steinbichler, T.; Innerhofer, V.; Gottfried, T.; Bektic-Tadic, L.; Giotakis, A.; Rumpold, G.; Riechelmann, H. Assessment of health-related quality-of-life in patients with chronic Rhinosinusitis—Validation of the German Sino-Nasal Outcome Test-22 (German-SNOT-22). J. Psychosom. Res. 2021, 140, 110316. [Google Scholar] [CrossRef] [PubMed]
- Dejaco, D.; Riedl, D.; Huber, A.; Moschen, R.; Giotakis, A.I.; Bektic-Tadic, L.; Steinbichler, T.; Kahler, P.; Riechelmann, H. The SNOT-22 factorial structure in European patients with chronic rhinosinusitis: New clinical insights. Eur. Arch. Oto-Rhino-Laryngol. 2019, 276, 1355–1365. [Google Scholar] [CrossRef]
- Mendez, A.; Seikaly, H.; Eurich, D.; Dzioba, A.; Aalto, D.; Osswald, M.; Harris, J.R.; O’Connell, D.A.; Lazarus, C.; Urken, M.; et al. Development of a Patient-Centered Functional Outcomes Questionnaire in Head and Neck Cancer. JAMA Otolaryngol. Neck Surg. 2020, 146, 437. [Google Scholar] [CrossRef]
- Bowling, A. Mode of questionnaire administration can have serious effects on data quality. J. Public Health 2005, 27, 281–291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheung, Y.B.; Goh, C.; Thumboo, J.; Khoo, K.-S.; Wee, J. Quality of life scores differed according to mode of administration in a review of three major oncology questionnaires. J. Clin. Epidemiol. 2006, 59, 185–191. [Google Scholar] [CrossRef]
- Kirsh, E.; Naunheim, M.; Ma, C.T.G.; Kammer, R.; Varvares, M.; Goldsmith, T. Patient-reported versus physiologic swallowing outcomes in patients with head and neck cancer after chemoradiation. Laryngoscope 2019, 129, 2059–2064. [Google Scholar] [CrossRef] [PubMed]
- Boyle, G.J. Does item homogeneity indicate internal consistency or item redundancy in psychometric scales? Pers. Individ. Differ. 1991, 12, 291–294. [Google Scholar] [CrossRef]
- Beck, A.C.C.; Kieffer, J.M.; Retèl, V.P.; van Overveld, L.F.; Takes, R.P.; van den Brekel, M.W.; van Harten, W.H.; Stuiver, M.M. Mapping the EORTC QLQ-C30 and QLQ-H&N35 to the EQ-5D for head and neck cancer: Can dis-ease-specific utilities be obtained? PLoS ONE 2019, 14, e0226077. [Google Scholar]
- Baba, K.; John, M.T.; Inukai, M.; Aridome, K.; Igarahsi, Y. Validating an alternate version of the chewing function questionnaire in partially dentate patients. BMC Oral Health 2009, 9, 9. [Google Scholar] [CrossRef] [Green Version]
- Dejaco, D.; Steinbichler, T.; Schartinger, V.H.; Fischer, N.; Anegg, M.; Dudas, J.; Posch, A.; Widmann, G.; Riechelmann, H. Specific growth rates calculated from CTs in patients with head and neck squamous cell carcinoma: A retrospective study performed in Austria. BMJ Open 2019, 9, e025359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Controls (n = 37) | Pretreatment Group (n = 24) | Posttreatment Group (n = 60) | |
|---|---|---|---|
| Sex | |||
| Male | 25 | 21 | 49 |
| Female | 12 | 3 | 11 |
| Age | |||
| ≤50 | 4 | 3 | 3 |
| 51–60 | 13 | 7 | 12 |
| 61–70 | 16 | 7 | 19 |
| 71–80 | 4 | 4 | 19 |
| ≥80 | 0 | 3 | 5 |
| P16 status | |||
| Negative | n.a. 4 | 20 | 49 |
| Positive | n.a. 4 | 4 | 11 |
| Tumor site | |||
| Oral cavity | n.a. 4 | 5 | 12 |
| Oropharynx | n.a. 4 | 6 | 25 |
| Hypopharynx | n.a. 4 | 4 | 8 |
| Larynx | n.a. 4 | 8 | 13 |
| CUP 1 | n.a. 4 | 1 | 2 |
| Clinical UICC stage 2 | |||
| Stage I | n.a. 4 | 10 | 13 |
| Stage II | n.a. 4 | 5 | 14 |
| Stage III | n.a. 4 | 2 | 9 |
| Stage IVa | n.a. 4 | 6 | 21 |
| Stage IVb | n.a. 4 | 1 | 3 |
| Stage IVc | n.a. 4 | 0 | 0 |
| Treatment 3 | |||
| No surgery only | n.a. 4 | 0 | 8 |
| Surgery only | n.a. 4 | 13 | 15 |
| Multimodality | n.a. 4 | 11 | 37 |
| Functional Domain | Controls | Pretreatment Group | Posttreatment Group | p-Value 1 |
|---|---|---|---|---|
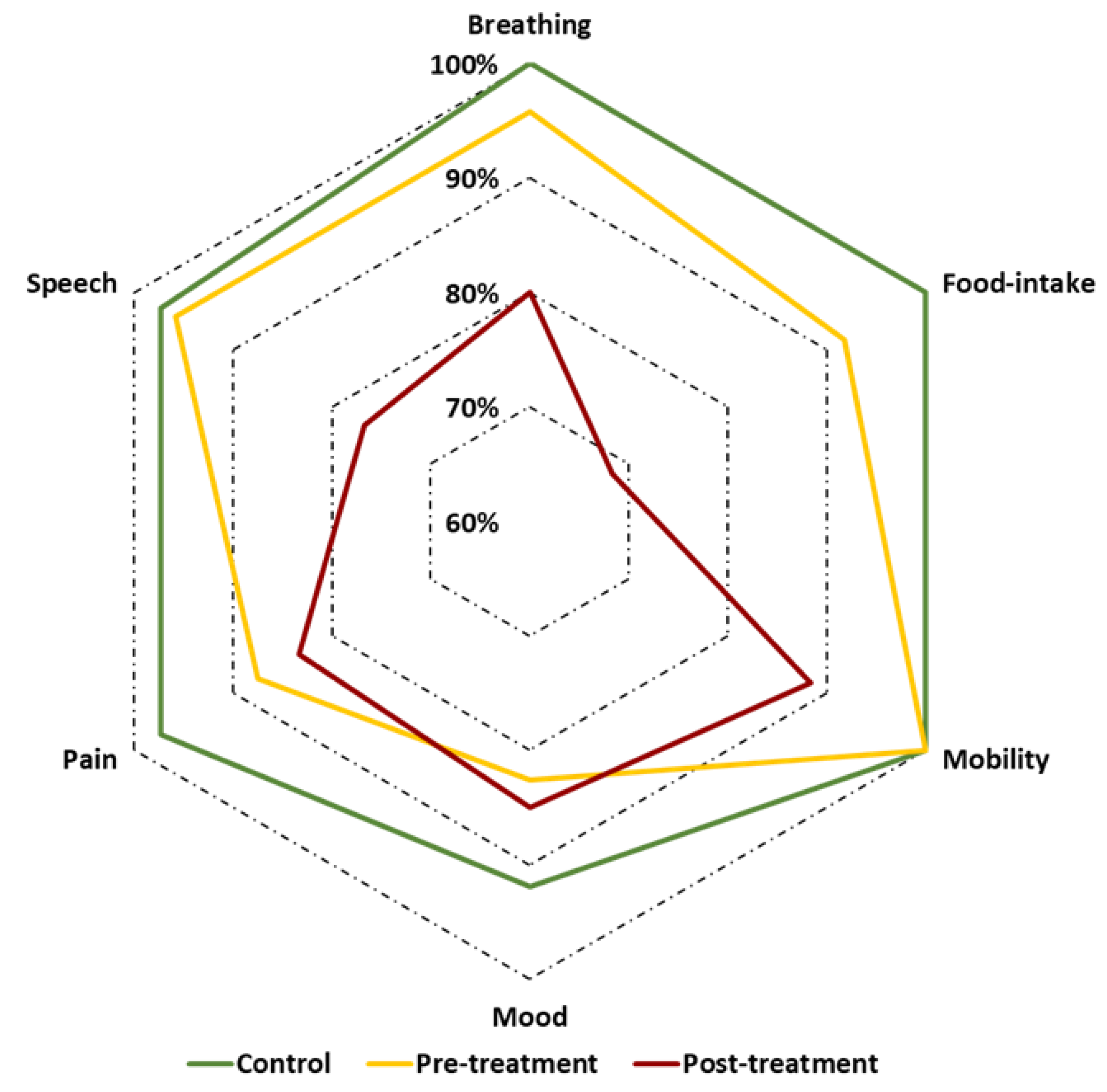
| Food intake | 80.8 | 63.5 | 47.9 | <0.001 |
| Breathing | 71.7 | 67.8 | 51.7 | 0.004 |
| Speech | 74.4 | 65.3 | 50.4 | 0.001 |
| Pain | 68.9 | 56.5 | 57.9 | 0.140 |
| Mood | 69.3 | 56.8 | 56.7 | 0.140 |
| Mobility | 66.8 | 69.8 | 53.9 | 0.027 |
| HNC-FIT Scale | |||||||
|---|---|---|---|---|---|---|---|
| Food Intake | Breathing | Speech | Pain | Mood | Mobility | ||
| EORTC QoL H&N35 | Teeth | −0.16 | −0.01 | −0.17 | −0.13 | −0.32 3 | −0.09 |
| Swallowing | −0.72 1 | −0.20 | −0.31 3 | −0.31 3 | −0.22 | −0.41 2 | |
| Social eating | −0.56 1 | 0.02 | −0.20 | −0.24 | −0.15 | −0.42 2 | |
| Opening mouth | −0.36 2 | −0.05 | −0.21 | −0.24 | −0.26 | −0.40 2 | |
| Dry mouth | −0.29 3 | 0.15 | 0.10 | −0.12 | −0.06 | −0.21 | |
| Sticky saliva | −0.38 2 | −0.13 | −0.12 | −0.11 | −0.26 | −0.32 3 | |
| Nutritional Supplements | −0.35 2 | −0.02 | −0.09 | −0.15 | −0.09 | −0.07 | |
| Feeding tube | −0.73 1 | −0.20 | −0.32 3 | −0.15 | 0.10 | −0.26 | |
| Weight loss | −0.38 2 | −0.15 | −0.21 | −0.26 | −0.01 | −0.02 | |
| Weight gain | 0.04 | −0.26 | −0.26 | 0.05 | −0.16 | −0.23 | |
| Coughed | −0.15 | −0.16 | −0.15 | 0.03 | −0.11 | 0.05 | |
| Speech | −0.34 2 | −0.37 2 | −0.55 1 | −0.35 2 | −0.28 | −0.11 | |
| Pain | −0.36 2 | 0.04 | −0.16 | −0.47 1 | −0.24 | −0.18 | |
| Pain killers | −0.35 2 | −0.05 | −0.16 | −0.61 1 | −0.21 | −0.05 | |
| Social contact | −0.01 | −0.03 | −0.09 | −0.29 3 | −0.25 | −0.08 | |
| Sexuality | −0.27 3 | −0.39 2 | −0.30 3 | −0.05 | −0.25 | −0.22 | |
| Senses | −0.37 2 | −0.19 | −0.25 | −0.19 | −0.19 | −0.26 | |
| Feeling ill | −0.26 3 | −0.14 | −0.09 | −0.11 | −0.30 3 | −0.21 | |
| rICC 1 | 95% CI 2 | p-Value 2 | ||
|---|---|---|---|---|
| Interrater Reliability | Food intake | 0.99 | 0.98–0.99 | 0.006 |
| Breathing | 0.95 | 0.93–0.97 | 0.006 | |
| Speech | 0.96 | 0.94–0.98 | 0.006 | |
| Pain | 0.93 | 0.89–0.95 | 0.006 | |
| Mood | 0.91 | 0.86–0.94 | 0.006 | |
| Shoulder–neck mobility | 0.90 | 0.85–0.93 | 0.006 | |
| Test-Retest-Reliability | Food intake | 0.94 | 0.84–0.98 | 0.006 |
| Breathing | 0.95 | 0.91–0.99 | 0.006 | |
| Speech | 0.97 | 0.94–0.98 | 0.006 | |
| Pain | 0.93 | 0.81–0.98 | 0.006 | |
| Mood | 0.71 | 0.18–0.91 | 0.012 | |
| Dhoulder–neck mobility | 0.98 | 0.93–0.99 | 0.006 |
| Functional Domain | Pretreatment Assessment 1 | Posttreatment Assessment 1 | F-Value | p-Value 2 | η2 |
|---|---|---|---|---|---|
| Food intake | 3.3 ± 1.0 | 2.8 ± 1.1 | 8.64 | 0.040 | 0.313 |
| Breathing | 3.8 ± 0.6 | 3.5 ± 1 | 2.11 | 0.326 | 0.100 |
| Speech | 3.8 ± 0.4 | 3.2 ± 1 | 6.54 | 0.076 | 0.256 |
| Pain | 3.7 ± 0.8 | 2.3 ± 1.1 | 24.18 | 0.006 | 0.560 |
| Mood | 3.4 ± 0.8 | 3.1 ± 0.9 | 3.06 | 0.270 | 0.139 |
| Mobility | 3.8 ± 0.4 | 3.8 ± 0.4 | 0.00 | >0.99 | <0.001 |
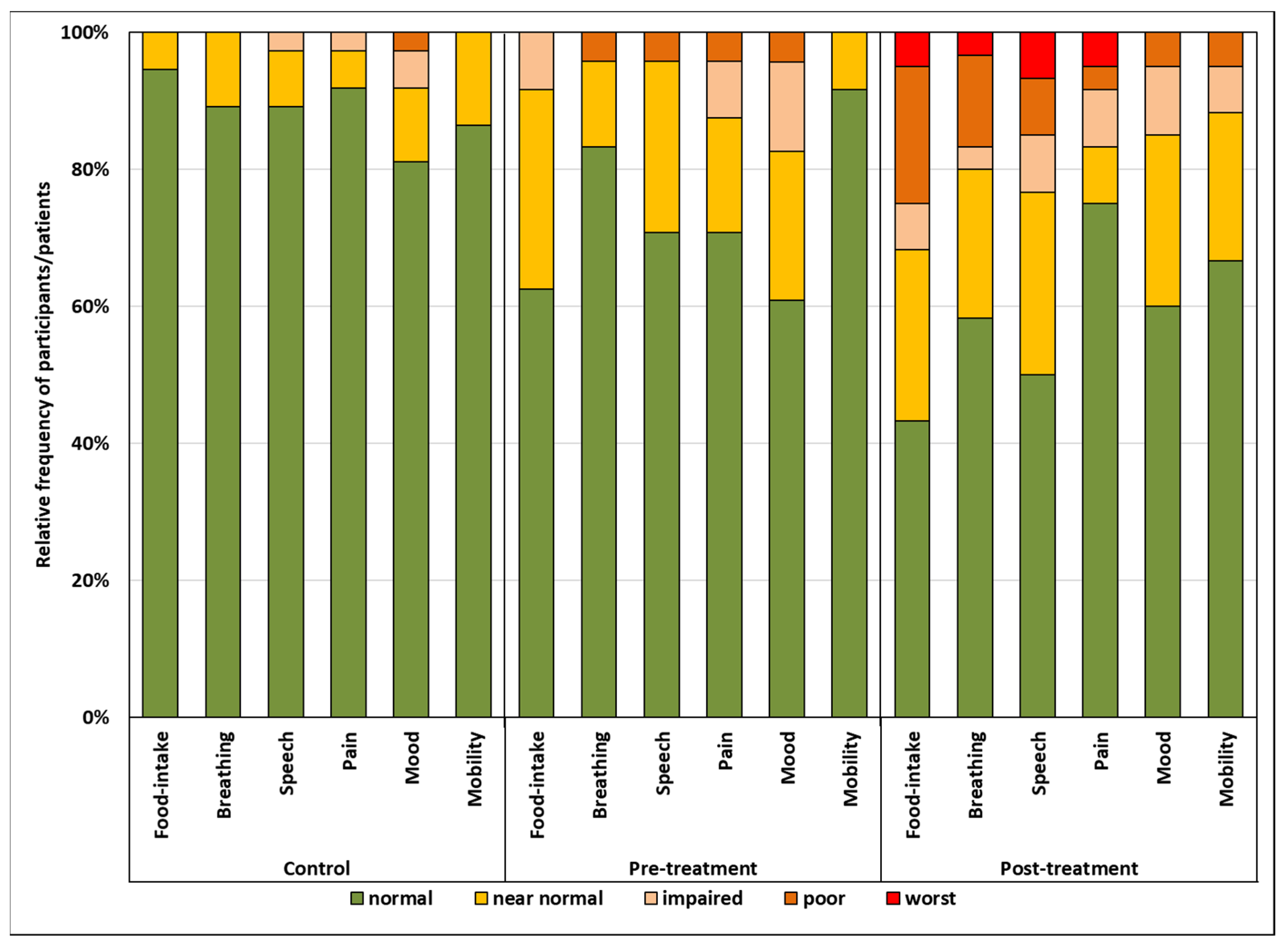
| Score | Verbal Rating | Controls (n = 37) | Pretreatment (n = 24) | Posttreatment (n = 60) | |
|---|---|---|---|---|---|
| Food intake | 0 | No oral feeding; only via gastrostomy tube | 0 | 0 | 3 |
| 1 | Gastrostomy tube needed; some oral feeding possible | 0 | 0 | 12 | |
| 2 | No gastrostomy tube, oral diet, but only liquid/soft food | 0 | 2 | 4 | |
| 3 | No gastrostomy tube, diet/swallowing near-normal | 2 | 7 | 15 | |
| 4 | Normal | 35 | 15 | 26 | |
| Breathing | 0 | Tracheostoma, needs blocked cannula | 0 | 0 | 2 |
| 1 | Tracheostoma, speech cannula no cannula | 0 | 1 | 8 | |
| 2 | No tracheostoma, breathing difficult at rest | 0 | 0 | 2 | |
| 3 | No tracheostoma, breathing difficulties only on exertion | 4 | 3 | 13 | |
| 4 | Normal | 33 | 20 | 35 | |
| Speech | 0 | Not possible/without phonation | 0 | 0 | 4 |
| 1 | Difficult to understand, no phone calls | 0 | 1 | 5 | |
| 2 | Telephoning possible | 1 | 0 | 5 | |
| 3 | Easy to understand, but pronunciation/voice changed | 3 | 6 | 16 | |
| 4 | Normal | 33 | 17 | 30 | |
| Pain | 0 | Pain despite opiate therapy | 0 | 0 | 3 |
| 1 | Controlled with opiates | 0 | 1 | 2 | |
| 2 | Regularly needs non-opioid analgesics | 1 | 2 | 5 | |
| 3 | Needs analgesics from time to time | 2 | 4 | 5 | |
| 4 | Normal | 34 | 17 | 45 | |
| Mood | 0 | Suicidal thoughts | 0 | 0 | 0 |
| 1 | Very depressed despite antidepressants | 1 | 1 | 3 | |
| 2 | With antidepressants overall normal mood, very depressed without antidepressants | 2 | 3 | 6 | |
| 3 | Occasionally depressed, no antidepressants needed | 4 | 5 | 15 | |
| 4 | Normal | 30 | 14 | 36 | |
| Mobility | 0 | Stiff neck and/or shoulder, hardly any movement possible | 0 | 0 | 0 |
| 1 | Can hair hairdly comb, looking backwards in car not possible | 0 | 0 | 3 | |
| 2 | Combing with problems, looking backwards in car difficult | 0 | 0 | 4 | |
| 3 | Combing and looking backwards in car slightly restricted | 5 | 2 | 13 | |
| 4 | Normal | 32 | 22 | 40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dejaco, D.; Riedl, D.; Gasser, S.; Schartinger, V.H.; Innerhofer, V.; Gottfried, T.; Steinbichler, T.B.; Riechelmann, F.; Moschen, R.; Galvan, O.; et al. A Tool for Rapid Assessment of Functional Outcomes in Patients with Head and Neck Cancer. Cancers 2021, 13, 5529. https://doi.org/10.3390/cancers13215529
Dejaco D, Riedl D, Gasser S, Schartinger VH, Innerhofer V, Gottfried T, Steinbichler TB, Riechelmann F, Moschen R, Galvan O, et al. A Tool for Rapid Assessment of Functional Outcomes in Patients with Head and Neck Cancer. Cancers. 2021; 13(21):5529. https://doi.org/10.3390/cancers13215529
Chicago/Turabian StyleDejaco, Daniel, David Riedl, Sebastian Gasser, Volker Hans Schartinger, Veronika Innerhofer, Timo Gottfried, Teresa Bernadette Steinbichler, Felix Riechelmann, Roland Moschen, Oliver Galvan, and et al. 2021. "A Tool for Rapid Assessment of Functional Outcomes in Patients with Head and Neck Cancer" Cancers 13, no. 21: 5529. https://doi.org/10.3390/cancers13215529






