Biomarker Expression in Multifocal Vulvar High-Grade Squamous Intraepithelial Lesions
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Samples
2.2. Processing of Tissue Blocks
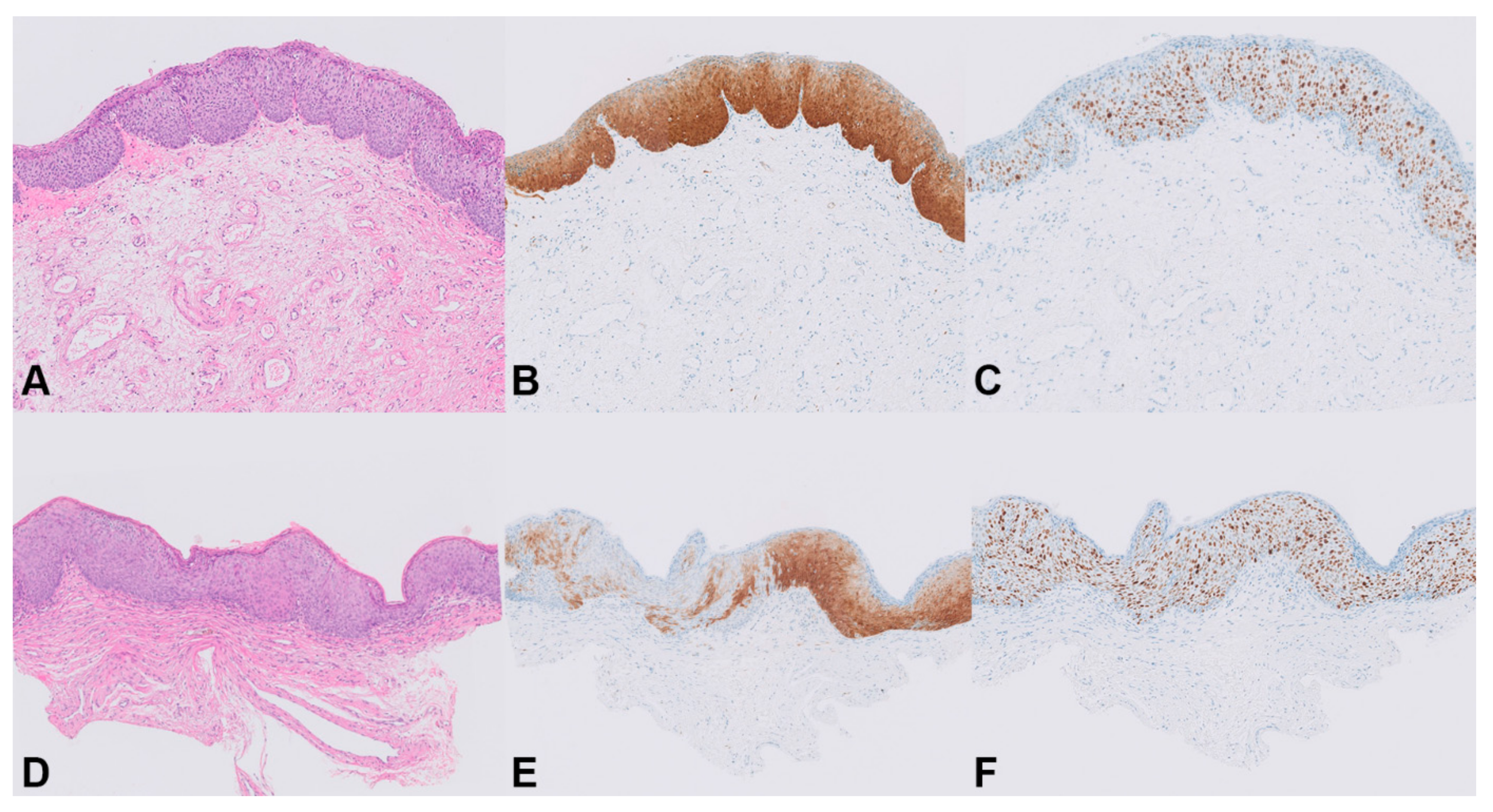
2.3. Histopathology and Immunohistochemistry of p16INK4a and Ki-67
2.4. DNA Isolation
2.5. DNA Methylation Analysis
2.6. Human Papillomavirus (HPV) Testing and Genotyping
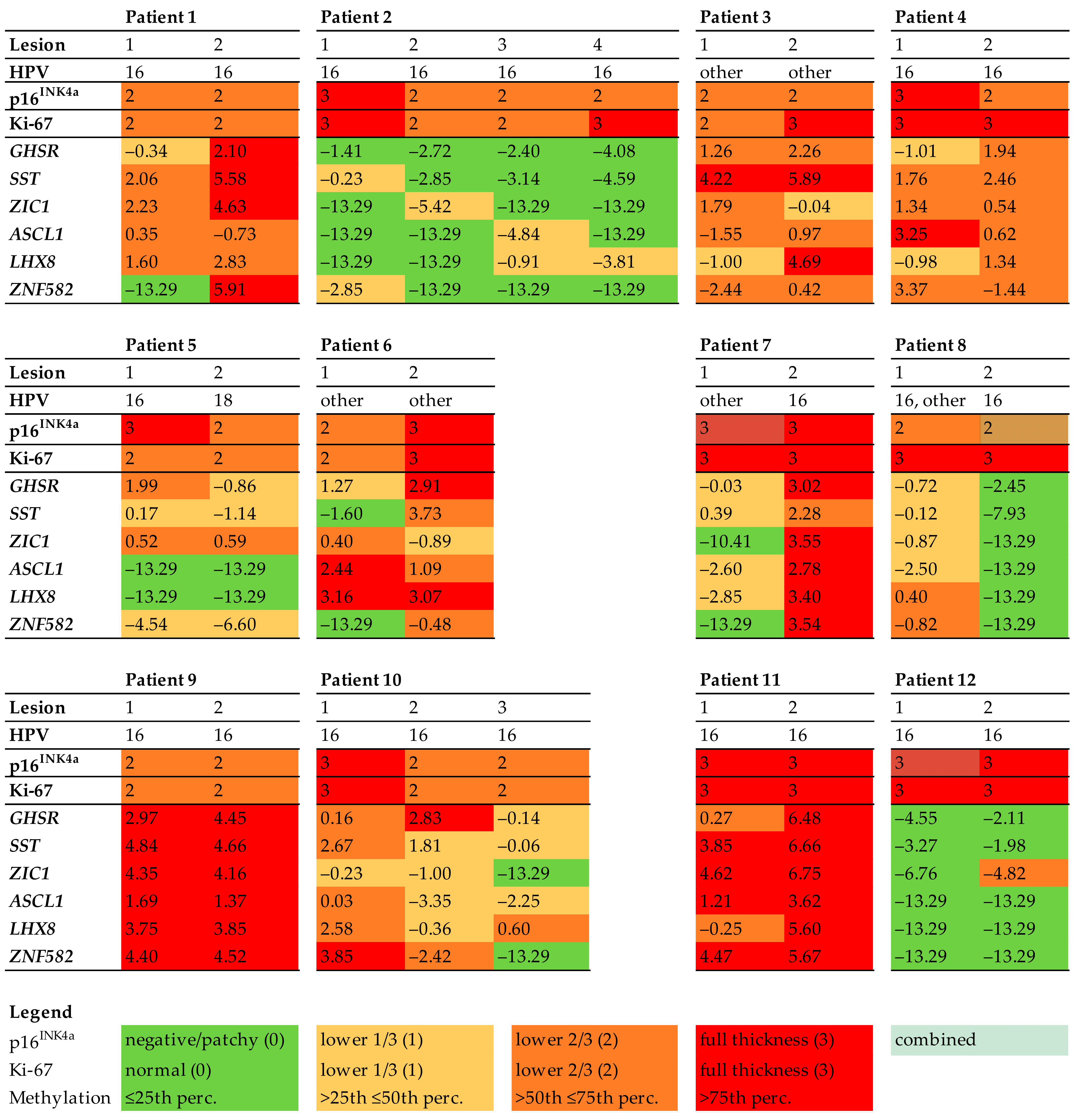
3. Results
3.1. Baseline Characteristics
3.2. Biomarker Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bornstein, J.; Bogliatto, F.; Haefner, H.K.; Stockdale, C.; Preti, M.; Bohl, T.G.; Reutter, J.; Committee, I.T. The 2015 International Society for the Study of Vulvovaginal Disease (ISSVD) Terminology of Vulvar Squamous Intraepithelial Lesions. Obstet. Gynecol. 2016, 127, 264–268. [Google Scholar] [CrossRef]
- De Vuyst, H.; Clifford, G.M.; Nascimento, M.C.; Madeleine, M.M.; Franceschi, S. Prevalence and type distribution of human papillomavirus in carcinoma and intraepithelial neoplasia of the vulva, vagina and anus: A meta-analysis. Int. J. Cancer 2009, 124, 1626–1636. [Google Scholar] [CrossRef]
- Singh, N.; Gilks, C.B. Vulval squamous cell carcinoma and its precursors. Histopathology 2020, 76, 128–138. [Google Scholar] [CrossRef]
- Thuijs, N.B.; Van Beurden, M.; Bruggink, A.H.; Steenbergen, R.D.M.; Berkhof, J.; Bleeker, M.C.G. Vulvar intraepithelial neoplasia: Incidence and long-term risk of vulvar squamous cell carcinoma. Int. J. Cancer 2021, 148, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Al-Ghamdi, A.; Freedman, D.; Miller, D.; Poh, C.; Rosin, M.; Zhang, L.; Gilks, C. Vulvar Squamous Cell Carcinoma in Young Women: A Clinicopathologic Study of 21 Cases. Gynecol. Oncol. 2002, 84, 94–101. [Google Scholar] [CrossRef]
- Preti, M.; Igidbashian, S.; Costa, S.; Cristoforoni, P.; Mariani, L.; Origoni, M.; Sandri, M.T.; Boveri, S.; Spolti, N.; Spinaci, L.; et al. VIN usual type—From the past to the future. Ecancermedicalscience 2015, 9, 531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Seters, M.; van Beurden, M.; de Craen, A.J. Is the assumed natural history of vulvar intraepithelial neoplasia III based on enough evidence? A systematic review of 3322 published patients. Gynecol. Oncol. 2005, 97, 645–651. [Google Scholar] [CrossRef]
- Likes, W.M.; Stegbauer, C.; Tillmanns, T.; Pruett, J. Pilot study of sexual function and quality of life after excision for vulvar intraepithelial neoplasia. J. Reprod. Med. 2007, 52, 23–27. [Google Scholar] [PubMed]
- Hillemanns, P.; Wang, X.; Staehle, S.; Michels, W.; Dannecker, C. Evaluation of different treatment modalities for vulvar intraepithelial neoplasia (VIN): CO2 laser vaporization, photodynamic therapy, excision and vulvectomy. Gynecol. Oncol. 2006, 100, 271–275. [Google Scholar] [CrossRef]
- de Sanjose, S.; Alemany, L.; Ordi, J.; Tous, S.; Alejo, M.; Bigby, S.M.; Joura, E.A.; Maldonado, P.; Laco, J.; Bravo, I.G.; et al. Worldwide human papillomavirus genotype attribution in over 2000 cases of intraepithelial and invasive lesions of the vulva. Eur. J. Cancer 2013, 49, 3450–3461. [Google Scholar] [CrossRef]
- Faber, M.T.; Sand, F.L.; Albieri, V.; Norrild, B.; Kjaer, S.K.; Verdoodt, F. Prevalence and type distribution of human papillomavirus in squamous cell carcinoma and intraepithelial neoplasia of the vulva. Int. J. Cancer 2017, 141, 1161–1169. [Google Scholar] [CrossRef] [Green Version]
- O’Neill, C.J.; McCluggage, W.G. p16 Expression in the Female Genital Tract and Its Value in Diagnosis. Adv. Anat. Pathol. 2006, 13, 8–15. [Google Scholar] [CrossRef]
- Steenbergen, R.; Snijders, P.J.F.; Heideman, D.A.M.; Meijer, C.J.L.M. Clinical implications of (epi)genetic changes in HPV-induced cervical precancerous lesions. Nat. Rev. Cancer 2014, 14, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Swarts, D.R.A.; Voorham, Q.J.M.; Van Splunter, A.P.; Wilting, S.M.; Sie, D.; Pronk, D.; Van Beurden, M.; Heideman, D.A.M.; Snijders, P.J.F.; Meijer, C.J.L.M.; et al. Molecular heterogeneity in human papillomavirus-dependent and -independent vulvar carcinogenesis. Cancer Med. 2018, 7, 4542–4553. [Google Scholar] [CrossRef] [PubMed]
- Thuijs, N.B.; Berkhof, J.; Özer, M.; Duin, S.; van Splunter, A.P.; Snoek, B.C.; Heideman, D.A.M.; van Beurden, M.; Steenbergen, R.D.M.; Bleeker, M.C.G. DNA methylation markers for cancer risk prediction of vulvar intraepithelial neoplasia. Int. J. Cancer 2021, 148, 2481–2488. [Google Scholar] [CrossRef] [PubMed]
- De Strooper, L.M.; Meijer, C.J.; Berkhof, J.; Hesselink, A.T.; Snijders, P.J.; Steenbergen, R.; Heideman, D.A. Methylation Analysis of the FAM19A4 Gene in Cervical Scrapes Is Highly Efficient in Detecting Cervical Carcinomas and Advanced CIN2/3 Lesions. Cancer Prev. Res. 2014, 7, 1251–1257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Zee, R.P.; Richel, O.; van Noesel, C.J.M.; Ciocănea-Teodorescu, I.; van Splunter, A.P.; ter Braak, T.J.; Nathan, M.; Cuming, T.; Sheaff, M.; Kreuter, A.; et al. Cancer Risk Stratification of Anal Intraepithelial Neoplasia in Human Immunodeficiency Virus–Positive Men by Validated Methylation Markers Associated With Progression to Cancer. Clin. Infect. Dis. 2021, 72, 2154–2163. [Google Scholar] [CrossRef] [Green Version]
- Dasgupta, S.; Ewing-Graham, P.; Swagemakers, S.; Bosch, T.V.D.; Atmodimedjo, P.; Verbiest, M.; de Haan, M.; van Doorn, H.; van der Spek, P.; Koljenović, S.; et al. Exploring Differentially Methylated Genes in Vulvar Squamous Cell Carcinoma. Cancers 2021, 13, 3580. [Google Scholar] [CrossRef]
- Bleeker, M.C.; Visser, P.J.; Overbeek, L.I.; Van Beurden, M.; Berkhof, J. Lichen Sclerosus: Incidence and Risk of Vulvar Squamous Cell Carcinoma. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1224–1230. [Google Scholar] [CrossRef] [Green Version]
- Rietbergen, M.M.; Leemans, C.R.; Bloemena, E.; Heideman, D.A.; Braakhuis, B.J.; Hesselink, A.T.; Witte, B.I.; de Jong, R.B.; Meijer, C.J.; Snijders, P.J.; et al. Increasing prevalence rates of HPV attributable oropharyngeal squamous cell carcinomas in the Netherlands as assessed by a validated test algorithm. Int. J. Cancer 2013, 132, 1565–1571. [Google Scholar] [CrossRef] [PubMed]
- Overmeer, R.M.; Henken, E.F.; Bierkens, M.; Wilting, S.; Timmerman, I.; Meijer, C.J.L.M.; Snijders, P.J.F.; Steenbergen, R.D.M. Repression of MAL tumour suppressor activity by promoter methylation during cervical carcinogenesis. J. Pathol. 2009, 219, 327–336. [Google Scholar] [CrossRef] [PubMed]
- van der Zee, R.; Richel, O.; Van Noesel, C.J.M.; Novianti, P.W.; Ciocanea-Teodorescu, I.; Van Splunter, A.P.; Duin, S.; van den Berk, G.E.L.; Meijer, C.J.L.M.; Quint, W.G.V.; et al. Host Cell Deoxyribonucleic Acid Methylation Markers for the Detection of High-grade Anal Intraepithelial Neoplasia and Anal Cancer. Clin. Infect. Dis. 2019, 68, 1110–1117. [Google Scholar] [CrossRef] [PubMed]
- Snellenberg, S.; De Strooper, A.L.M.; Hesselink, A.T.; Meijer, C.J.L.M.; Snijders, P.J.F.; Heideman, D.A.M.; Steenbergen, R.D.M. Development of a multiplex methylation-specific PCR as candidate triage test for women with an HPV-positive cervical scrape. BMC Cancer 2012, 12, 551. [Google Scholar] [CrossRef] [Green Version]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Hesselink, A.T.; Berkhof, J.; Van Der Salm, M.L.; Van Splunter, A.P.; Geelen, T.H.; Van Kemenade, F.J.; Bleeker, M.; Heideman, D.A.M. Clinical Validation of the HPV-Risk Assay, a Novel Real-Time PCR Assay for Detection of High-Risk Human Papillomavirus DNA by Targeting the E7 Region. J. Clin. Microbiol. 2014, 52, 890–896. [Google Scholar] [CrossRef] [Green Version]
- Preti, M.; Van Seters, M.; Sideri, M.; Van Beurden, M. Squamous Vulvar Intraepithelial Neoplasia. Clin. Obstet. Gynecol. 2005, 48, 845–861. [Google Scholar] [CrossRef] [PubMed]
- van der Marel, J.; Berkhof, J.; Ordi, J.; Torné, A.; Del Pino, M.; van Baars, R.; Schiffman, M.; Wentzensen, N.; Jenkins, D.; Quint, W.G. Attributing Oncogenic Human Papillomavirus Genotypes to High-grade Cervical Neoplasia: Which Type Causes the Lesion? Am. J. Surg. Pathol. 2015, 39, 496–504. [Google Scholar] [CrossRef]
- Kleter, B.; van Doorn, L.-J.; Schrauwen, L.; Molijn, A.; Sastrowijoto, S.; ter Schegget, J.; Lindeman, J.; ter Harmsel, B.; Burger, M.; Quint, W. Development and Clinical Evaluation of a Highly Sensitive PCR-Reverse Hybridization Line Probe Assay for Detection and Identification of Anogenital Human Papillomavirus. J. Clin. Microbiol. 1999, 37, 2508–2517. [Google Scholar] [CrossRef] [Green Version]
- Quint, W.G.V.; Scholte, G.; Van Doorn, L.J.; Kleter, B.; Smits, P.H.M.; Lindeman, J. Comparative analysis of human papillomavirus infections in cervical scrapes and biopsy specimens by general SPF10 PCR and HPV genotyping. J. Pathol. 2001, 194, 51–58. [Google Scholar] [CrossRef]
- Spinillo, A.; Dal Bello, B.D.; Gardella, B.; Roccio, M.; Dacco’, M.D.; Silini, E.M. Multiple human papillomavirus infection and high grade cervical intraepithelial neoplasia among women with cytological diagnosis of atypical squamous cells of undetermined significance or low grade squamous intraepithelial lesions. Gynecol. Oncol. 2009, 113, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Demarco, M.; Hyun, N.; Carter-Pokras, O.; Raine-Bennett, T.R.; Cheung, L.; Chen, X.; Hammer, A.; Campos, N.; Kinney, W.; Gage, J.C.; et al. A study of type-specific HPV natural history and implications for contemporary cervical cancer screening programs. EClinicalMedicine 2020, 22, 100293. [Google Scholar] [CrossRef] [PubMed]
- Hoevenaars, B.M.; van der Avoort, I.A.; de Wilde, P.C.; Massuger, L.F.; Melchers, W.J.; de Hullu, J.A.; Bulten, J. A panel of p16INK4A, MIB1 and p53 proteins can distinguish between the 2 pathways leading to vulvar squamous cell carcinoma. Int. J. Cancer 2008, 123, 2767–2773. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, S.; Ewing-Graham, P.C.; Swagemakers, S.M.; van der Spek, P.J.; van Doorn, H.C.; Hegt, N.; Koljenović, S.; van Kemenade, F.J. Precursor lesions of vulvar squamous cell carcinoma—Histology and biomarkers: A systematic review. Crit. Rev. Oncol. Hematol. 2020, 147, 102866. [Google Scholar] [CrossRef]
- Darragh, T.M.; Colgan, T.J.; Cox, J.T.; Heller, D.S.; Henry, M.R.; Luff, R.D.; McCalmont, T.; Nayar, R.; Palefsky, J.M.; Stoler, M.H.; et al. The Lower Anogenital Squamous Terminology Standardization Project for HPV-Associated Lesions: Background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. J. Low. Genit. Tract Dis. 2012, 16, 205–242. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Baseline | Follow-Up | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient | Age (years) | Other Anogenital Conditions | Smoking | Immuno-Compromised | Number | Aspect | Topography | Type of Biopsy | Primary Treatment | Time to Last HSIL Diagnosis (years) | VSCC During Follow-Up | Time to VSCC (years) | Topography VSCC |
| 1 | 42 | AIN3, condylomata acuminata | Unknown | Unknown | 2 | Not specified | Perineum, lab maj R | Diagnostic | Skinning vulvectomy | * | No | NA | NA |
| 2 | 24 | None | Yes | No | 4 | Hyperpigmentation, maculopapulous | Lab maj R and L, lab min R and L | Diagnostic | Skinning vulvectomy | 0.8 | No | NA | NA |
| 3 | 44 | AIN2 | Yes | No | 2 | Leukoplakia | Lab min R and L | Diagnostic | Local excision | 18.5 | No | NA | NA |
| 4 | 37 | CIN3 | Yes | No | 2 | Not specified | 6 and 9 o’clock | Diagnostic | Laser evaporatisation | 16.7 | No | NA | NA |
| 5 | 58 | AIN3, CIN3 | Unknown | No | 2 | Hyperpigmentation, condylomatous brown | Lab min R and L | Diagnostic | None | 0.5 | No | NA | NA |
| 6 | 38 | None | Yes | No | 2 | Not specified | Perineum, lab maj/min R | Therapeutic | Local excision | 20.2 | No | NA | NA |
| 7 | 45 | CIN3, condylomata acuminata | Yes | No | 2 | Hyperpigmentation | Commisura posterior, perianal | Therapeutic | Local excision | 14.8 | Yes | 9.2 | Perianal |
| 8 | 31 | CIN2, VAIN2, condylomata accuminata | Yes | Yes | 2 | Not specified | Lab maj/min L, lab min R | Diagnostic | Local excision | * | No | NA | NA |
| 9 | 44 | None | Unknown | Unknown | 2 | Hypertrophic dystrophic | Lab maj R and L | Diagnostic | Local excision | 3.1 | No | NA | NA |
| 10 | 49 | CIN3 | Yes | No | 3 | Papillomatous, erosive, varyingly pigmented | Perineum, lab maj/min L, lab min R | Diagnostic | Skinning vulvectomy | 19.1 | Yes | 9.3 | Perianal |
| 11 | 38 | Unknown | Unknown | Yes | 2 | Not specified | Lab min R, commissura posterior | Therapeutic | Local excision | 11.4 | Yes | 5.9 | Anterior L |
| 12 | 28 | CIN2, condylomata acuminata | Yes | No | 2 | Hyperpigmentation, condylomatous brown | Lab maj/min L, clitoris | Therapeutic | Skinning vulvectomy | 16.1 | Yes | 11.4 | Posterior L |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thuijs, N.B.; Schonck, W.A.M.; Klaver, L.L.J.; Fons, G.; van Beurden, M.; Steenbergen, R.D.M.; Bleeker, M.C.G. Biomarker Expression in Multifocal Vulvar High-Grade Squamous Intraepithelial Lesions. Cancers 2021, 13, 5646. https://doi.org/10.3390/cancers13225646
Thuijs NB, Schonck WAM, Klaver LLJ, Fons G, van Beurden M, Steenbergen RDM, Bleeker MCG. Biomarker Expression in Multifocal Vulvar High-Grade Squamous Intraepithelial Lesions. Cancers. 2021; 13(22):5646. https://doi.org/10.3390/cancers13225646
Chicago/Turabian StyleThuijs, Nikki B., Willemijn A. M. Schonck, Linde L. J. Klaver, Guus Fons, Marc van Beurden, Renske D. M. Steenbergen, and Maaike C. G. Bleeker. 2021. "Biomarker Expression in Multifocal Vulvar High-Grade Squamous Intraepithelial Lesions" Cancers 13, no. 22: 5646. https://doi.org/10.3390/cancers13225646






