Colorectal Cancer Progression Is Potently Reduced by a Glucose-Free, High-Protein Diet: Comparison to Anti-EGFR Therapy
Abstract
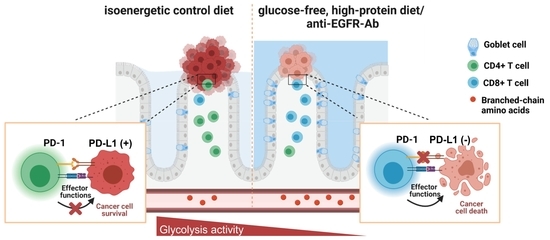
:Simple Summary
Abstract
1. Introduction
2. Results
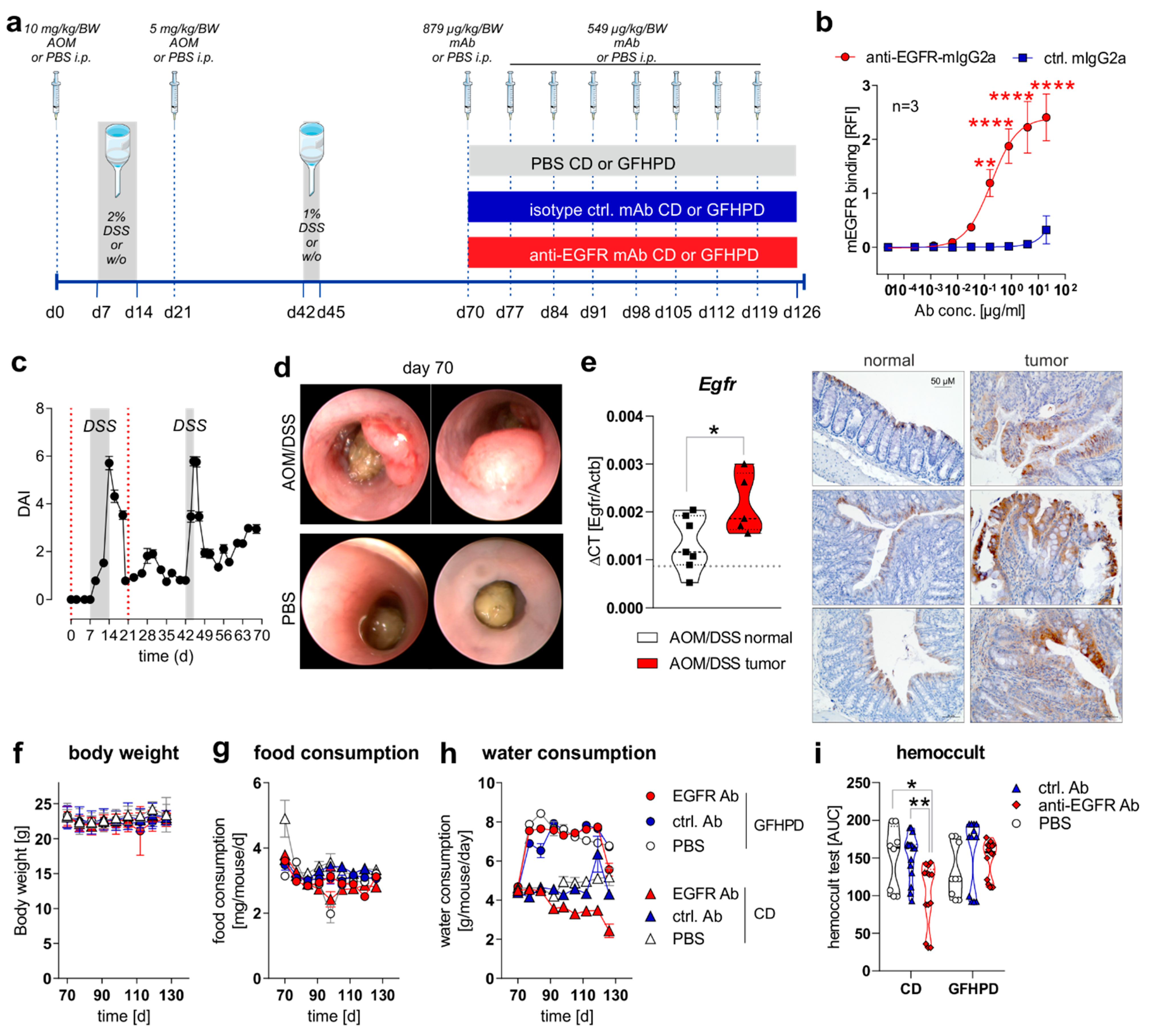
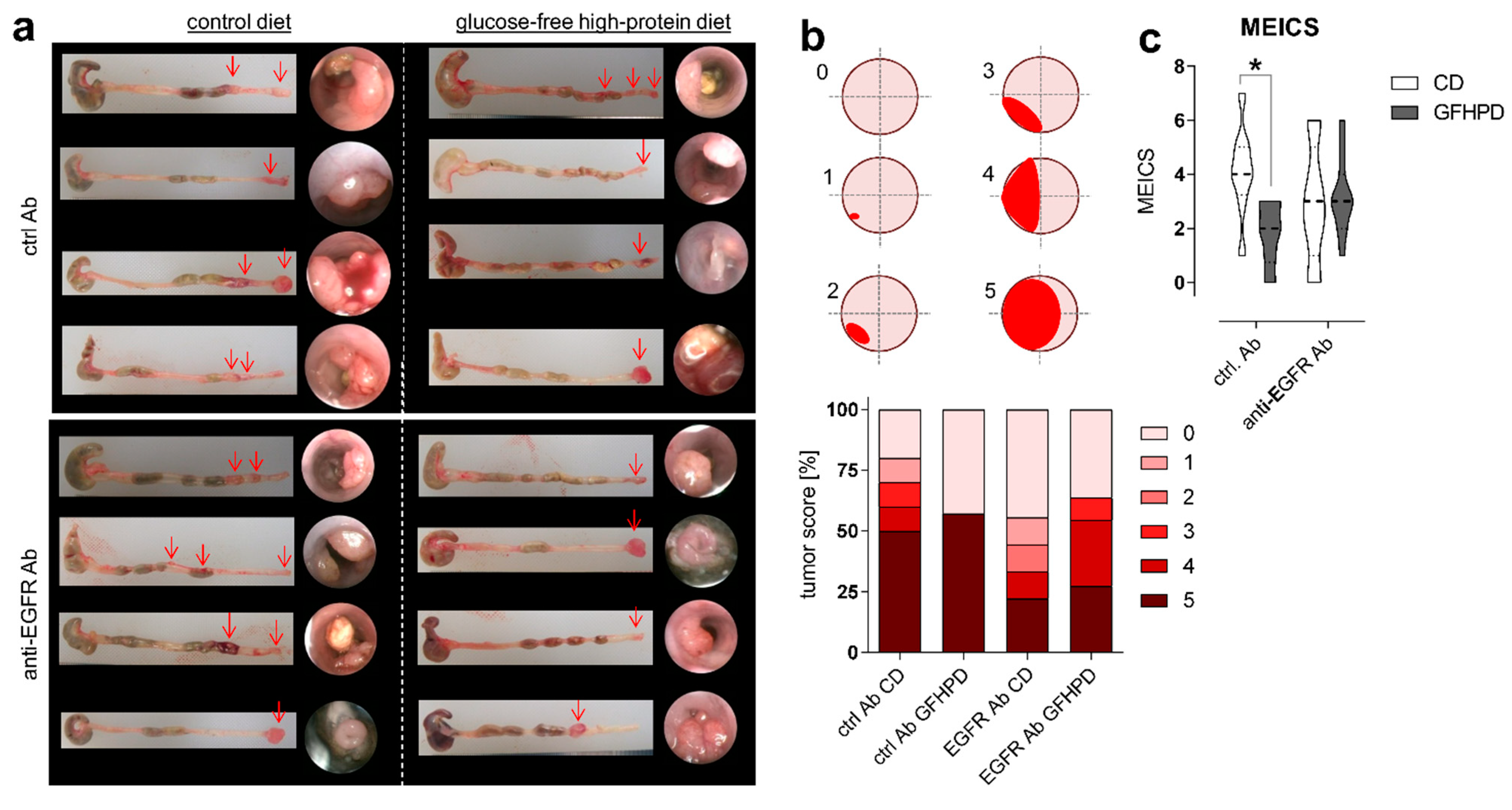
2.1. Anti-EGFR Ab Therapy or Glucose-Free, High-Protein Nutritional Intervention Efficaciously Prevents CRC Progression
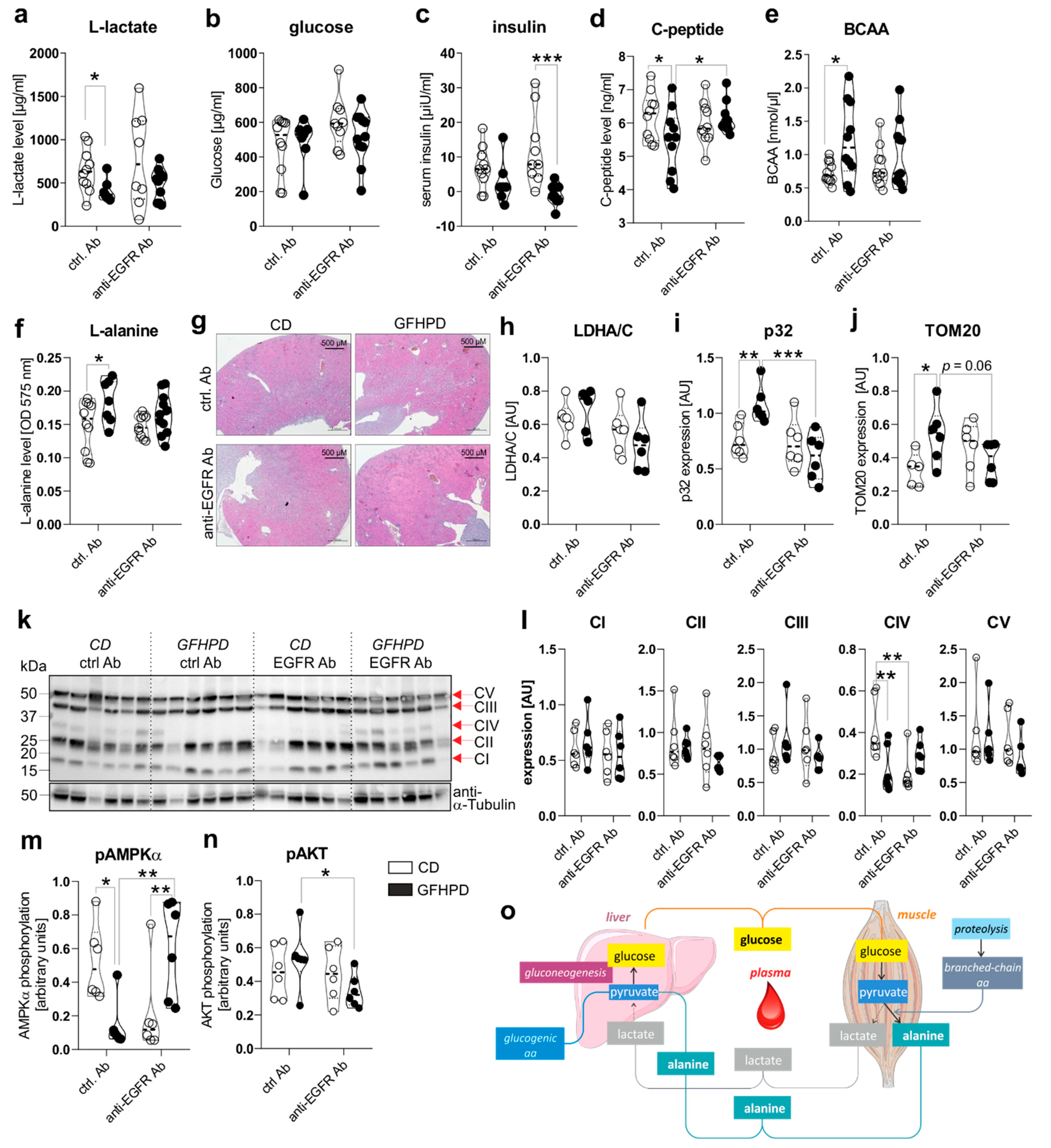
2.2. A Glucose-Free, High-Protein Diet Increases Amino Acid Metabolism and Decreases Lactate and Insulin Production
2.3. Goblet Cell Differentiation Is Enhanced by GFHP Diet or anti-EGFR Ab Therapy in AOM/DSS-Treated Mice
2.4. Increased Amino Acid Metabolism Enhances Goblet Cell Differentiation in Colorectal Tumor Cells In Vitro
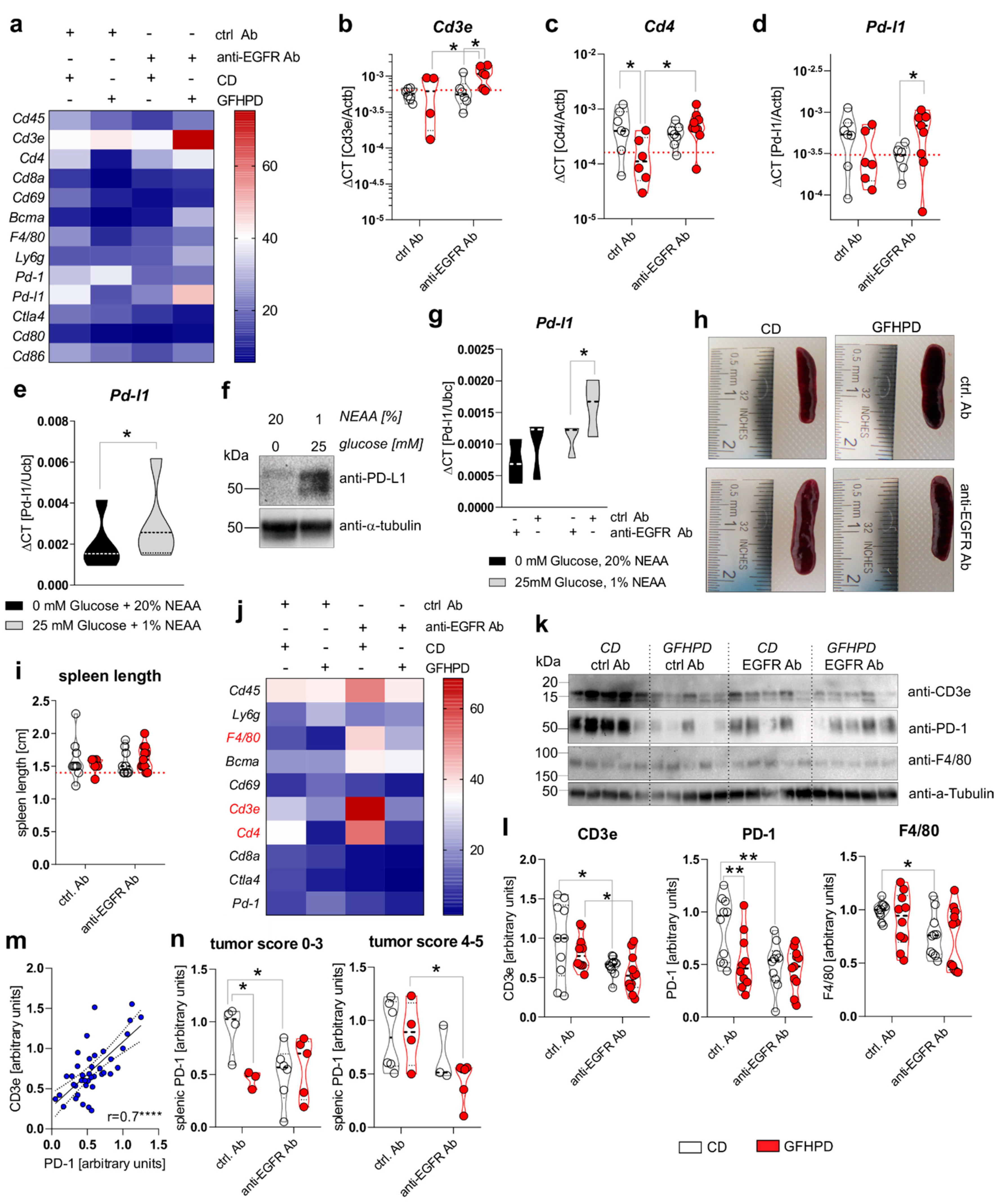
2.5. A Metabolic Switch from Glucose to Amino Acid Metabolism as Well as Anti-EGFR Ab Treatment Decreases PD-L1 Expression in CRC Cells and Colonic CD4+ T-Cell Count
2.6. PD-1 Expression on Splenic T Cells Is Reduced under GFHP Diet or Anti-EGFR Ab Treatment in AOM/DSS-Treated Mice
3. Discussion
4. Materials and Methods
4.1. Animal Experiments
4.2. Histology and Microscopy Analyses
4.3. Metabolic Analyses in Serum Samples
4.4. Cell Culture
4.5. RNA Extraction, cDNA Synthesis and Quantitative PCR
4.6. Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) and Immunoblotting
4.7. Neutral Red Cytotoxicity Assay
4.8. Muc2 and Muc5AC ELISA
4.9. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vuik, F.E.; Nieuwenburg, S.A.; Bardou, M.; Lansdorp-Vogelaar, I.; Dinis-Ribeiro, M.; Bento, M.J.; Zadnik, V.; Pellise, M.; Esteban, L.; Kaminski, M.F.; et al. Increasing incidence of colorectal cancer in young adults in Europe over the last 25 years. Gut 2019, 68, 1820–1826. [Google Scholar] [CrossRef] [PubMed]
- Fatemi, S.R.; Pourhoseingholi, M.A.; Asadi, F.; Vahedi, M.; Pasha, S.; Alizadeh, L.; Zali, M.R. Recurrence and Five-Year Survival in Colorectal Cancer Patients After Surgery. Iran. J. Cancer Prev. 2015, 8, e3439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Francescangeli, F.; De Angelis, M.L.; Zeuner, A. Dietary Factors in the Control of Gut Homeostasis, Intestinal Stem Cells, and Colorectal Cancer. Nutrients 2019, 11, 2936. [Google Scholar] [CrossRef] [Green Version]
- GBD 2017 Colorectal Cancer Collaborators. The global, regional, and national burden of colorectal cancer and its attributable risk factors in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2019, 4, 913–933. [Google Scholar] [CrossRef] [Green Version]
- Terzic, J.; Grivennikov, S.; Karin, E.; Karin, M. Inflammation and colon cancer. Gastroenterology 2010, 138, 2101–2114.e5. [Google Scholar] [CrossRef]
- Cooks, T.; Pateras, I.S.; Tarcic, O.; Solomon, H.; Schetter, A.J.; Wilder, S.; Lozano, G.; Pikarsky, E.; Forshew, T.; Rosenfeld, N.; et al. Mutant p53 prolongs NF-kappaB activation and promotes chronic inflammation and inflammation-associated colorectal cancer. Cancer Cell 2013, 23, 634–646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, J.; Nagahashi, M.; Kim, E.Y.; Harikumar, K.B.; Yamada, A.; Huang, W.C.; Hait, N.C.; Allegood, J.C.; Price, M.M.; Avni, D.; et al. Sphingosine-1-phosphate links persistent STAT3 activation, chronic intestinal inflammation, and development of colitis-associated cancer. Cancer Cell 2013, 23, 107–120. [Google Scholar] [CrossRef] [Green Version]
- Mendelsohn, J. Targeting the epidermal growth factor receptor for cancer therapy. J. Clin. Oncol. 2002, 20, 1S–13S. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [Green Version]
- Ciardiello, F.; Tortora, G. EGFR antagonists in cancer treatment. N. Engl. J. Med. 2008, 358, 1160–1174. [Google Scholar] [CrossRef] [Green Version]
- Reichert, J.M.; Dhimolea, E. The future of antibodies as cancer drugs. Drug Discov. Today 2012, 17, 954–963. [Google Scholar] [CrossRef]
- Baselga, J.; Mendelsohn, J. Epidermal growth factor receptor targeting in cancer. Sem. Oncol. 2006, 33, 369–385. [Google Scholar] [CrossRef]
- Sinha, A.; Nightingale, J.; West, K.P.; Berlanga-Acosta, J.; Playford, R.J. Epidermal growth factor enemas with oral mesalamine for mild-to-moderate left-sided ulcerative colitis or proctitis. N. Engl. J. Med. 2003, 349, 350–357. [Google Scholar] [CrossRef] [Green Version]
- Dubé, P.; Polk, D. P-186 YI Opposing Roles for EGFR in Colitis-Associated Cancer: The Presence of Active Inflammation Determines a Protective Versus Tumorigenic Role for EGFR. Inflamm. Bowel Dis. 2012, 18, S89–S90. [Google Scholar] [CrossRef]
- Dube, P.E.; Yan, F.; Punit, S.; Girish, N.; McElroy, S.J.; Washington, M.K.; Polk, D.B. Epidermal growth factor receptor inhibits colitis-associated cancer in mice. J. Clin. Investig. 2012, 122, 2780–2792. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Vander Heiden, M.G.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reitzer, L.J.; Wice, B.M.; Kennell, D. Evidence that glutamine, not sugar, is the major energy source for cultured HeLa cells. J. Biol. Chem. 1979, 254, 2669–2676. [Google Scholar] [CrossRef]
- Rossignol, R.; Gilkerson, R.; Aggeler, R.; Yamagata, K.; Remington, S.J.; Capaldi, R.A. Energy substrate modulates mitochondrial structure and oxidative capacity in cancer cells. Cancer Res. 2004, 64, 985–993. [Google Scholar] [CrossRef] [Green Version]
- Lunt, S.Y.; Vander Heiden, M.G. Aerobic glycolysis: Meeting the metabolic requirements of cell proliferation. Annu. Rev. Cell Dev. Biol. 2011, 27, 441–464. [Google Scholar] [CrossRef] [Green Version]
- Dang, C.V. Links between metabolism and cancer. Genes Dev. 2012, 26, 877–890. [Google Scholar] [CrossRef] [Green Version]
- Yang, P.; Li, Z.; Zhang, L.; Li, H.; Li, Z. Analysis of metabonomic profiling alterations in a mouse model of colitis-associated cancer and 2-deoxy-d-glucose treatment. RSC Adv. 2016, 6, 58862–58870. [Google Scholar] [CrossRef]
- Lim, S.O.; Li, C.W.; Xia, W.; Lee, H.H.; Chang, S.S.; Shen, J.; Hsu, J.L.; Raftery, D.; Djukovic, D.; Gu, H.; et al. EGFR Signaling Enhances Aerobic Glycolysis in Triple-Negative Breast Cancer Cells to Promote Tumor Growth and Immune Escape. Cancer Res. 2016, 76, 1284–1296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garrido, G.; Sanchez, B.; Rodriguez, H.M.; Lorenzano, P.; Alonso, D.; Fernandez, L.E. 7A7 MAb: A new tool for the pre-clinical evaluation of EGFR-based therapies. Hybrid. Hybridomics 2004, 23, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Van Buren, G., 2nd; Yang, A.D.; Dallas, N.A.; Gray, M.J.; Lim, S.J.; Xia, L.; Fan, F.; Somcio, R.; Wu, Y.; Hicklin, D.J.; et al. Effect of molecular therapeutics on liver regeneration in a murine model. J. Clin. Oncol. 2008, 26, 1836–1842. [Google Scholar] [CrossRef]
- Garrido, G.; Lorenzano, P.; Sanchez, B.; Beausoleil, I.; Alonso, D.F.; Perez, R.; Fernandez, L.E. T cells are crucial for the anti-metastatic effect of anti-epidermal growth factor receptor antibodies. Cancer Immunol. Immunother. 2007, 56, 1701–1710. [Google Scholar] [CrossRef]
- Garrido, G.; Rabasa, A.; Sanchez, B.; Lopez, M.V.; Blanco, R.; Lopez, A.; Hernandez, D.R.; Perez, R.; Fernandez, L.E. Induction of immunogenic apoptosis by blockade of epidermal growth factor receptor activation with a specific antibody. J. Immunol. 2011, 187, 4954–4966. [Google Scholar] [CrossRef]
- De la Cruz-Lopez, K.G.; Castro-Munoz, L.J.; Reyes-Hernandez, D.O.; Garcia-Carranca, A.; Manzo-Merino, J. Lactate in the Regulation of Tumor Microenvironment and Therapeutic Approaches. Front. Oncol. 2019, 9, 1143. [Google Scholar] [CrossRef] [Green Version]
- Neufert, C.; Becker, C.; Neurath, M.F. An inducible mouse model of colon carcinogenesis for the analysis of sporadic and inflammation-driven tumor progression. Nat. Protoc. 2007, 2, 1998–2004. [Google Scholar] [CrossRef]
- Neufert, C.; Heichler, C.; Brabletz, T.; Scheibe, K.; Boonsanay, V.; Greten, F.R.; Neurath, M.F. Inducible mouse models of colon cancer for the analysis of sporadic and inflammation-driven tumor progression and lymph node metastasis. Nat. Protoc. 2021, 16, 61–85. [Google Scholar] [CrossRef]
- Tanaka, T.; Kohno, H.; Suzuki, R.; Yamada, Y.; Sugie, S.; Mori, H. A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci. 2003, 94, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Kohno, H.; Sugie, S.; Nakagama, H.; Tanaka, T. Strain differences in the susceptibility to azoxymethane and dextran sodium sulfate-induced colon carcinogenesis in mice. Carcinogenesis 2006, 27, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Becker, C.; Fantini, M.C.; Wirtz, S.; Nikolaev, A.; Kiesslich, R.; Lehr, H.A.; Galle, P.R.; Neurath, M.F. In vivo imaging of colitis and colon cancer development in mice using high resolution chromoendoscopy. Gut 2005, 54, 950–954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Park, J.S.; Deng, J.H.; Bai, Y. Cytochrome c oxidase subunit IV is essential for assembly and respiratory function of the enzyme complex. J. Bioenerg. Biomembr. 2006, 38, 283–291. [Google Scholar] [CrossRef] [Green Version]
- Kadenbach, B.; Huttemann, M.; Arnold, S.; Lee, I.; Bender, E. Mitochondrial energy metabolism is regulated via nuclear-coded subunits of cytochrome c oxidase. Free Radic. Biol. Med. 2000, 29, 211–221. [Google Scholar] [CrossRef]
- Zhao, Y.; Hu, X.; Liu, Y.; Dong, S.; Wen, Z.; He, W.; Zhang, S.; Huang, Q.; Shi, M. ROS signaling under metabolic stress: Cross-talk between AMPK and AKT pathway. Mol. Cancer 2017, 16, 79. [Google Scholar] [CrossRef] [Green Version]
- Sünderhauf, A.; Hicken, M.; Schlichting, H.; Skibbe, K.; Ragab, M.; Raschdorf, A.; Hirose, M.; Schaffler, H.; Bokemeyer, A.; Bettenworth, D.; et al. Loss of Mucosal p32/gC1qR/HABP1 Triggers Energy Deficiency and Impairs Goblet Cell Differentiation in Ulcerative Colitis. Cell. Mol. Gastroenterol. Hepatol. 2021, 12, 229–250. [Google Scholar] [CrossRef]
- Sünderhauf, A.; Raschdorf, A.; Hicken, M.; Schlichting, H.; Fetzer, F.; Brethack, A.K.; Perner, S.; Kemper, C.; Ghebrehiwet, B.; Sina, C.; et al. GC1qR Cleavage by Caspase-1 Drives Aerobic Glycolysis in Tumor Cells. Front. Oncol. 2020, 10, 575854. [Google Scholar] [CrossRef]
- Sünderhauf, A.; Skibbe, K.; Preisker, S.; Ebbert, K.; Verschoor, A.; Karsten, C.M.; Kemper, C.; Huber-Lang, M.; Basic, M.; Bleich, A.; et al. Regulation of epithelial cell expressed C3 in the intestine—Relevance for the pathophysiology of inflammatory bowel disease? Mol. Immunol. 2017, 90, 227–238. [Google Scholar] [CrossRef]
- Bronte, V.; Pittet, M.J. The spleen in local and systemic regulation of immunity. Immunity 2013, 39, 806–818. [Google Scholar] [CrossRef] [Green Version]
- Sugihara, K.; Jass, J.R. Colorectal goblet cell sialomucin heterogeneity: Its relation to malignant disease. J. Clin. Pathol. 1986, 39, 1088–1095. [Google Scholar] [CrossRef] [Green Version]
- Milosevic, V.; Vukmirovic, F.; Zindovic, M.; Krstic, M.; Milenkovic, S.; Jancic, S. Interplay between expression of leptin receptors and mucin histochemical aberrations in colorectal adenocarcinoma. Rom. J. Morphol. Embryol. 2015, 56, 709–716. [Google Scholar] [PubMed]
- Kasprzak, A.; Seraszek-Jaros, A.; Jagielska, J.; Helak-Lapaj, C.; Siodla, E.; Szmeja, J.; Kaczmarek, E. The Histochemical Alterations of Mucin in Colorectal Carcinoma Quantified by Two Efficient Algorithms of Digital Image Analysis. Int. J. Mol. Sci. 2019, 20, 4580. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raschdorf, A.; Sunderhauf, A.; Skibbe, K.; Ghebrehiwet, B.; Peerschke, E.I.; Sina, C.; Derer, S. Heterozygous P32/C1QBP/HABP1 Polymorphism rs56014026 Reduces Mitochondrial Oxidative Phosphorylation and Is Expressed in Low-grade Colorectal Carcinomas. Front. Oncol. 2020, 10, 631592. [Google Scholar] [CrossRef] [PubMed]
- Tajan, M.; Vousden, K.H. Dietary Approaches to Cancer Therapy. Cancer Cell 2020, 37, 767–785. [Google Scholar] [CrossRef] [PubMed]
- Tannenbaum, A. The dependence of tumor formation on the composition of the calorie-restricted diet as well as on the degree of restriction. Cancer Res. 1945, 5, 616–625. [Google Scholar]
- Ho, V.W.; Leung, K.; Hsu, A.; Luk, B.; Lai, J.; Shen, S.Y.; Minchinton, A.I.; Waterhouse, D.; Bally, M.B.; Lin, W.; et al. A low carbohydrate, high protein diet slows tumor growth and prevents cancer initiation. Cancer Res. 2011, 71, 4484–4493. [Google Scholar] [CrossRef] [Green Version]
- Glaire, M.A.; Domingo, E.; Sveen, A.; Bruun, J.; Nesbakken, A.; Nicholson, G.; Novelli, M.; Lawson, K.; Oukrif, D.; Kildal, W.; et al. Tumour-infiltrating CD8(+) lymphocytes and colorectal cancer recurrence by tumour and nodal stage. Br. J. Cancer 2019, 121, 474–482. [Google Scholar] [CrossRef]
- Galon, J.; Costes, A.; Sanchez-Cabo, F.; Kirilovsky, A.; Mlecnik, B.; Lagorce-Pages, C.; Tosolini, M.; Camus, M.; Berger, A.; Wind, P.; et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 2006, 313, 1960–1964. [Google Scholar] [CrossRef] [Green Version]
- Konjar, S.; Veldhoen, M. Dynamic Metabolic State of Tissue Resident CD8 T Cells. Front. Immunol. 2019, 10, 1683. [Google Scholar] [CrossRef] [Green Version]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef] [Green Version]
- Topalian, S.L.; Taube, J.M.; Anders, R.A.; Pardoll, D.M. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat. Rev. Cancer 2016, 16, 275–287. [Google Scholar] [CrossRef]
- Zeboudj, L.; Maitre, M.; Guyonnet, L.; Laurans, L.; Joffre, J.; Lemarie, J.; Bourcier, S.; Nour-Eldine, W.; Guerin, C.; Friard, J.; et al. Selective EGF-Receptor Inhibition in CD4(+) T Cells Induces Anergy and Limits Atherosclerosis. J. Am. Coll. Cardiol. 2018, 71, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Derer, S.; Bauer, P.; Lohse, S.; Scheel, A.H.; Berger, S.; Kellner, C.; Peipp, M.; Valerius, T. Impact of epidermal growth factor receptor (EGFR) cell surface expression levels on effector mechanisms of EGFR antibodies. J. Immunol. 2012, 189, 5230–5239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pirker, R.; Pereira, J.R.; von Pawel, J.; Krzakowski, M.; Ramlau, R.; Park, K.; de Marinis, F.; Eberhardt, W.E.; Paz-Ares, L.; Storkel, S.; et al. EGFR expression as a predictor of survival for first-line chemotherapy plus cetuximab in patients with advanced non-small-cell lung cancer: Analysis of data from the phase 3 FLEX study. Lancet Oncol. 2012, 13, 33–42. [Google Scholar] [CrossRef]
- Lievre, A.; Bachet, J.B.; Boige, V.; Cayre, A.; Le Corre, D.; Buc, E.; Ychou, M.; Bouche, O.; Landi, B.; Louvet, C.; et al. KRAS mutations as an independent prognostic factor in patients with advanced colorectal cancer treated with cetuximab. J. Clin. Oncol. 2008, 26, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Allegra, C.J.; Jessup, J.M.; Somerfield, M.R.; Hamilton, S.R.; Hammond, E.H.; Hayes, D.F.; McAllister, P.K.; Morton, R.F.; Schilsky, R.L. American Society of Clinical Oncology provisional clinical opinion: Testing for KRAS gene mutations in patients with metastatic colorectal carcinoma to predict response to anti-epidermal growth factor receptor monoclonal antibody therapy. J. Clin. Oncol. 2009, 27, 2091–2096. [Google Scholar] [CrossRef] [Green Version]
- Derer, S.; Berger, S.; Schlaeth, M.; Schneider-Merck, T.; Klausz, K.; Lohse, S.; Overdijk, M.B.; Dechant, M.; Kellner, C.; Nagelmeier, I.; et al. Oncogenic KRAS impairs EGFR antibodies’ efficiency by C/EBPbeta-dependent suppression of EGFR expression. Neoplasia 2012, 14, 190–205. [Google Scholar] [CrossRef] [Green Version]
- Kalantar-Zadeh, K.; Moore, L.W.; Tortorici, A.R.; Chou, J.A.; St-Jules, D.E.; Aoun, A.; Rojas-Bautista, V.; Tschida, A.K.; Rhee, C.M.; Shah, A.A.; et al. North American experience with Low protein diet for Non-dialysis-dependent chronic kidney disease. BMC Nephrol. 2016, 17, 90. [Google Scholar] [CrossRef] [Green Version]
- Fouque, D.; Aparicio, M. Eleven reasons to control the protein intake of patients with chronic kidney disease. Nat. Clin. Pract. Nephrol. 2007, 3, 383–392. [Google Scholar] [CrossRef]
- Ko, G.J.; Obi, Y.; Tortorici, A.R.; Kalantar-Zadeh, K. Dietary protein intake and chronic kidney disease. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Moskal, A.; Freisling, H.; Byrnes, G.; Assi, N.; Fahey, M.T.; Jenab, M.; Ferrari, P.; Tjonneland, A.; Petersen, K.E.; Dahm, C.C.; et al. Main nutrient patterns and colorectal cancer risk in the European Prospective Investigation into Cancer and Nutrition study. Br. J. Cancer 2016, 115, 1430–1440. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Diet Composition | Isoenergetic Control Diet | Glucose Free High Protein Diet |
|---|---|---|
| Casein | 20% | 60% |
| Brewer’s yeast | - | 2% |
| Corn starch | 28% | - |
| Maltodextrin | 14.5% | - |
| Sucrose | 10% | - |
| Cellulose powder | 15% | 19.5% |
| L-Cystine | 0.3% | 0.3% |
| Vitamin premixture | 1% | 1% |
| Minerals and trace elements | 6% | 6% |
| Choline chloride | 0.2% | 0.2% |
| Soybean oil | 5% | 11% |
| Crude protein | 17.7% | 53.4% |
| Crude fat | 5.1% | 11.4% |
| Crude fiber | 15.8% | 20.3% |
| Crude ash | 5.4% | 5.9% |
| Starch | 26.9% | 0.1% |
| Sugar | 9.9% | - |
| NfE | 51.7% | 1.9% |
| Physiological fuel value | 13.6 MJ/kg | 13.6 MJ/kg |
| Protein | 22 kcal% | 66 kcal% |
| Fat | 14 kcal% | 32 kcal% |
| Carbohydrates | 64 kcal% | 2 kcal% |
| Target Transcripts | Forward Primer (5′–3′) | Reverse Primer (5′–3′) |
|---|---|---|
| Atoh1 | GTGGGGTTGTAGTGGACGAG | GTTGCTCTCCGACATTGGG |
| Actb | GATGCTCCCCGGGCTGTATT | GGGGTACTTCAGGGTCAGGA |
| Klf4 | GATTAAGCAAGAGGCGGT | GGTAAGGTTTCTCGCCTGTG |
| Lgr5 | CGGCAACAGTGTGGACGACCT | GCGAGCACTGCACCGAGTGA |
| Muc2 | GCTGACGAGTGGTTGGTGAATG | GATGAGGTGGCAGACAGGAGAC |
| Spdef1 | GGAGAAGGCAGCATCAGGA | CCAGGGTCTGCTGTGATGT |
| Hes1 | CAAACCAAAGACGGCCTCT | GTCACCTCGTTCATGCACTC |
| Egfr | ATGTGATCCAAGCTGTCCCA | CTGATAGGTGGTGGGGTTGT |
| Caspase-1 | CTTCAATCAGCTCCATCAGC | CACGGCATGCCTGAATAATG |
| C3 | GACGCCACTATGTCCATC | GAAGGTCAGGCAGTCTTC |
| PD-L1 | GCTACGGGCGTTTACTATCAC | CTGAATCACTTGCTCATCTTCC |
| Cd80 | CAAGTGTCTTCAGATGTTGATGAAC | GAACGACACAGCTGTATGTGC |
| Cd86 | CTCAGATGCTGTTTCCGTGG | GTTCCTGTCAAAGCTCGTGC |
| Cd3e | AGTTGACGTGCCCTCTAGAC | ATCAGCAAGCCCAGAGTGAT |
| Cd4 | TTTGCAGAGGAAAACGGGTG | CTGCTTCAGGGTCAGTCTCA |
| Cd45 | TCATGGTCACACGATGTGAAGA | AGCCCGAGTGCCTTCCT |
| Cd8a | GAAGTGTTGGGGTCCGTTTC | TCACCGAGTTGCTGATGACT |
| Cd69 | CAGAAGGACCATGGCACCA | CGTCAAGTTGAACCAGCTG |
| Bcma | GGTGCTCTGGATCTTCTTGG | CCGTAGTCACCCGTTTTTGT |
| F4/80 | CTGTAACCGGATGGCAAACT | CATCACTGCCTCCACTAGCA |
| Ly6g | GATGGATTTTGCGTTGCTCTG | GGACTGAAACCAGGCTGAAC |
| Pd-1 | GTGGTAACAGAGAGAATCCTGGA | ACTAGGGACAGGTGCTGCTG |
| Ctla-4 | GAAGCCATACAGGTGACCCA | TGAATGTCGTGGCACAGACC |
| Primary Antibodies | Species | Company | Working Concentration |
| Anti-AMPKα (#2532) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Anti-β-Actin (#4967) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Anti-α-TUBULIN (#2125) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Anti-Caspase-1 (ab179515) | rabbit | Abcam, Cambridge, UK | 1 µg/mL |
| Anti-MUC2 (C3) (GTX100664) | rabbit | Genetex, Alton Pkwy Irvine, CA, USA | 1 µg/mL |
| Anti-Muc5AC (clone 45M1) (GTX11335) | mouse | Bioss, Woburn, MA, USA | 1 µg/mL |
| Anti-pAMPKα (Thr172, #2535) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Anti-pAkt (Ser473) (D9E) (#4060) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Anti-Akt (#9272) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Anti-EGFR (EP38Y) (ab52894) | rabbit | Abcam, Cambridge, UK | 1:1000 a |
| Anti-PD-L1 (B7-H1) (MAB90781) | rabbit | R&D Systems, Minneapolis, MN, USA | 1 µg/mL |
| Anti-p32 (clone EPR8871) (ab131284) | rabbit | Abcam, Cambridge, UK | 0.4 µg/mL |
| Anti-Tom20 (D8T4N) (#42406) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Anti-LDHA/C (C28H7) (#3558) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Anti-CD3ε (ab5690) | rabbit | Abcam, Cambridge, UK | 1 µg/mL |
| Anti-F4/80 (MA5-16363) | rabbit | Thermo Fisher Scientific Inc., Waltham, MA, USA | 0.2 µg/mL |
| Anti-mEGFR (7A7-mIgG2a) (Ab00134-23.0) | mouse | Absolute Antibody NA, Boston, MA, USA | |
| CD19-mIgG2a (FMC63) (Ab00613-2.0) | mouse | Absolute Antibody NA, Boston, MA, USA | |
| Anti-OXPHOS complexes I-V | mouse | Thermo Fisher Scientific Inc., Waltham, MA, USA | 6 µ/mL |
| Anti-LC3B (#2775) | rabbit | Cell Signaling, Danvers, MA, USA | 1:1000 a |
| Secondary Antibodies | Company | Working Concentration | |
| Anti-mouse IgG HRP | Cell Signaling, Danvers, MA, USA | 1:1000 (ELISA) a | |
| Anti-rabbit IgG HRP | Cell Signaling, Danvers, MA, USA | 1:2000 (ELISA) a; 1:4000 (WB) a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skibbe, K.; Brethack, A.-K.; Sünderhauf, A.; Ragab, M.; Raschdorf, A.; Hicken, M.; Schlichting, H.; Preira, J.; Brandt, J.; Castven, D.; et al. Colorectal Cancer Progression Is Potently Reduced by a Glucose-Free, High-Protein Diet: Comparison to Anti-EGFR Therapy. Cancers 2021, 13, 5817. https://doi.org/10.3390/cancers13225817
Skibbe K, Brethack A-K, Sünderhauf A, Ragab M, Raschdorf A, Hicken M, Schlichting H, Preira J, Brandt J, Castven D, et al. Colorectal Cancer Progression Is Potently Reduced by a Glucose-Free, High-Protein Diet: Comparison to Anti-EGFR Therapy. Cancers. 2021; 13(22):5817. https://doi.org/10.3390/cancers13225817
Chicago/Turabian StyleSkibbe, Kerstin, Ann-Kathrin Brethack, Annika Sünderhauf, Mohab Ragab, Annika Raschdorf, Maren Hicken, Heidi Schlichting, Joyce Preira, Jennifer Brandt, Darko Castven, and et al. 2021. "Colorectal Cancer Progression Is Potently Reduced by a Glucose-Free, High-Protein Diet: Comparison to Anti-EGFR Therapy" Cancers 13, no. 22: 5817. https://doi.org/10.3390/cancers13225817