Non-Melanoma Skin Cancer: A Genetic Update and Future Perspectives
Abstract
Simple Summary
Abstract
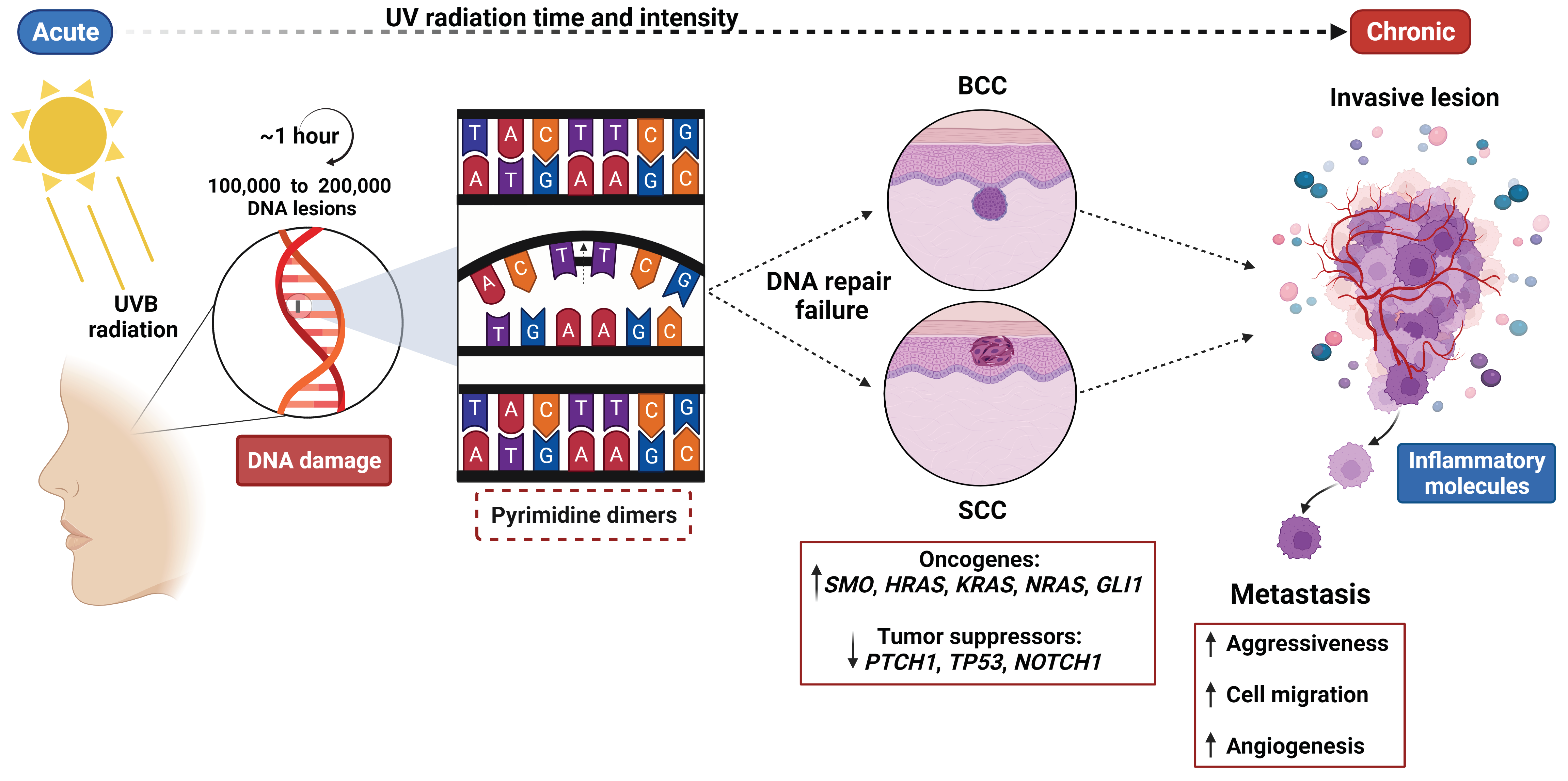
1. Introduction
2. Genetics of Non-Melanoma Skin Cancer and New Candidate Genes
3. Tumor Microenvironment, Cellular Components and Genetic Implications
4. Epigenetic Regulation in Non-Melanoma Skin Cancer
5. Perspectives: About Known Mechanism and New Alternatives
6. Skin Cancer and the COVID-19 Pandemic
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jones, O.T.; Ranmuthu, C.K.I.; Hall, P.N.; Funston, G.; Walter, F.M. Recognising Skin Cancer in Primary Care. Adv. Ther. 2020, 37, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Gruber, P.; Zito, P.M. Skin Cancer. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Das, K.; Cockerell, C.J.; Patil, A.; Pietkiewicz, P.; Giulini, M.; Grabbe, S.; Goldust, M. Machine Learning and Its Application in Skin Cancer. Int. J. Env. Res. Public Health 2021, 18, 13409. [Google Scholar] [CrossRef] [PubMed]
- Apalla, Z.; Nashan, D.; Weller, R.B.; Castellsagué, X. Skin Cancer: Epidemiology, Disease Burden, Pathophysiology, Diagnosis, and Therapeutic Approaches. Dermatol. Ther. 2017, 7, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Campagna, R.; Salvolini, E.; Pompei, V.; Pozzi, V.; Salvucci, A.; Molinelli, E.; Brisigotti, V.; Sartini, D.; Campanati, A.; Offidani, A.; et al. Nicotinamide N-Methyltransferase Gene Silencing Enhances Chemosensitivity of Melanoma Cell Lines. Pigment Cell Melanoma Res. 2021, 34, 1039–1048. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Cives, M.; Mannavola, F.; Lospalluti, L.; Sergi, M.C.; Cazzato, G.; Filoni, E.; Cavallo, F.; Giudice, G.; Stucci, L.S.; Porta, C.; et al. Non-Melanoma Skin Cancers: Biological and Clinical Features. Int. J. Mol. Sci. 2020, 21, 5394. [Google Scholar] [CrossRef]
- Pawlowska, E.; Wysokinski, D.; Blasiak, J. Nucleotide Excision Repair and Vitamin D--Relevance for Skin Cancer Therapy. Int. J. Mol. Sci. 2016, 17, 372. [Google Scholar] [CrossRef]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef]
- Diepgen, T.L.; Fartasch, M.; Drexler, H.; Schmitt, J. Occupational Skin Cancer Induced by Ultraviolet Radiation and Its Prevention. Br. J. Derm. 2012, 167, 76–84. [Google Scholar] [CrossRef]
- Mohania, D.; Chandel, S.; Kumar, P.; Verma, V.; Digvijay, K.; Tripathi, D.; Choudhury, K.; Mitten, S.K.; Shah, D. Ultraviolet Radiations: Skin Defense-Damage Mechanism. Adv. Exp. Med. Biol. 2017, 996, 71–87. [Google Scholar] [CrossRef]
- Suozzi, K.; Turban, J.; Girardi, M. Cutaneous Photoprotection: A Review of the Current Status and Evolving Strategies. Yale J. Biol. Med. 2020, 93, 55–67. [Google Scholar] [PubMed]
- Pellegrini, C.; Maturo, M.G.; Di Nardo, L.; Ciciarelli, V.; Gutiérrez García-Rodrigo, C.; Fargnoli, M.C. Understanding the Molecular Genetics of Basal Cell Carcinoma. Int. J. Mol. Sci. 2017, 18, 2485. [Google Scholar] [CrossRef] [PubMed]
- Bacchetti, T.; Salvolini, E.; Pompei, V.; Campagna, R.; Molinelli, E.; Brisigotti, V.; Togni, L.; Lucarini, G.; Sartini, D.; Campanati, A.; et al. Paraoxonase-2: A Potential Biomarker for Skin Cancer Aggressiveness. Eur. J. Clin. Investig. 2021, 51, e13452. [Google Scholar] [CrossRef] [PubMed]
- Teng, Y.; Yu, Y.; Li, S.; Huang, Y.; Xu, D.; Tao, X.; Fan, Y. Ultraviolet Radiation and Basal Cell Carcinoma: An Environmental Perspective. Front. Public Health 2021, 9, 666528. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Ratnakumar, K.; Hung, K.-F.; Rokunohe, D.; Kawasumi, M. Deciphering UV-Induced DNA Damage Responses to Prevent and Treat Skin Cancer. Photochem. Photobiol. 2020, 96, 478–499. [Google Scholar] [CrossRef] [PubMed]
- Vechtomova, Y.L.; Telegina, T.A.; Buglak, A.A.; Kritsky, M.S. UV Radiation in DNA Damage and Repair Involving DNA-Photolyases and Cryptochromes. Biomedicines 2021, 9, 1564. [Google Scholar] [CrossRef]
- Chiang, A.; Tan, C.Z.; Kuonen, F.; Hodgkinson, L.M.; Chiang, F.; Cho, R.J.; South, A.P.; Tang, J.Y.; Chang, A.L.S.; Rieger, K.E.; et al. Genetic Mutations Underlying Phenotypic Plasticity in Basosquamous Carcinoma. J. Investig. Derm. 2019, 139, 2263–2271.e5. [Google Scholar] [CrossRef]
- Mhamdi-Ghodbani, M.; Starzonek, C.; Degenhardt, S.; Bender, M.; Said, M.; Greinert, R.; Volkmer, B. UVB Damage Response of Dermal Stem Cells as Melanocyte Precursors Compared to Keratinocytes, Melanocytes, and Fibroblasts from Human Foreskin. J. Photochem. Photobiol. B 2021, 220, 112216. [Google Scholar] [CrossRef]
- Knatko, E.V.; Praslicka, B.; Higgins, M.; Evans, A.; Purdie, K.J.; Harwood, C.A.; Proby, C.M.; Ooi, A.; Dinkova-Kostova, A.T. Whole-Exome Sequencing Validates a Preclinical Mouse Model for the Prevention and Treatment of Cutaneous Squamous Cell Carcinoma. Cancer Prev. Res. 2017, 10, 67–75. [Google Scholar] [CrossRef]
- Majoros, H.; Ujfaludi, Z.; Borsos, B.N.; Hudacsek, V.V.; Nagy, Z.; Coin, F.; Buzas, K.; Kovács, I.; Bíró, T.; Boros, I.M.; et al. SerpinB2 Is Involved in Cellular Response upon UV Irradiation. Sci. Rep. 2019, 9, 2753. [Google Scholar] [CrossRef]
- Whitley, M.J.; Suwanpradid, J.; Lai, C.; Jiang, S.W.; Cook, J.L.; Zelac, D.E.; Rudolph, R.; Corcoran, D.L.; Degan, S.; Spasojevic, I.; et al. ENTPD1 (CD39) Expression Inhibits UVR-Induced DNA Damage Repair through Purinergic Signaling and Is Associated with Metastasis in Human Cutaneous Squamous Cell Carcinoma. J. Investig. Derm. 2021, 141, 2509–2520. [Google Scholar] [CrossRef] [PubMed]
- Campagna, R.; Pozzi, V.; Sartini, D.; Salvolini, E.; Brisigotti, V.; Molinelli, E.; Campanati, A.; Offidani, A.; Emanuelli, M. Beyond Nicotinamide Metabolism: Potential Role of Nicotinamide N-Methyltransferase as a Biomarker in Skin Cancers. Cancers 2021, 13, 4943. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.G.; Kuo, K.Y.; Li, S.; Bailey, I.; Aasi, S.; Chang, A.L.S.; Oro, A.E.; Tang, J.Y.; Sarin, K.Y. Frequent Basal Cell Cancer Development Is a Clinical Marker for Inherited Cancer Susceptibility. JCI Insight 2018, 3, 122744. [Google Scholar] [CrossRef] [PubMed]
- Van Haren, R.; Feldman, D.; Sinha, A.A. Systematic Comparison of Nonmelanoma Skin Cancer Microarray Datasets Reveals Lack of Consensus Genes. Br. J. Derm. 2009, 161, 1278–1287. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, H.; Bian, Q. Identification of Potential Biomarkers Associated with Basal Cell Carcinoma. Biomed. Res. Int. 2020, 2020, 2073690. [Google Scholar] [CrossRef]
- Oldak, M.; Grzela, T.; Lazarczyk, M.; Malejczyk, J.; Skopinski, P. Clinical Aspects of Disrupted Hedgehog Signaling (Review). Int. J. Mol. Med. 2001, 8, 445–452. [Google Scholar]
- Lacour, J.P. Carcinogenesis of Basal Cell Carcinomas: Genetics and Molecular Mechanisms. Br. J. Derm. 2002, 146, 17–19. [Google Scholar] [CrossRef]
- Reifenberger, J.; Wolter, M.; Knobbe, C.B.; Köhler, B.; Schönicke, A.; Scharwächter, C.; Kumar, K.; Blaschke, B.; Ruzicka, T.; Reifenberger, G. Somatic Mutations in the PTCH, SMOH, SUFUH and TP53 Genes in Sporadic Basal Cell Carcinomas. Br. J. Derm. 2005, 152, 43–51. [Google Scholar] [CrossRef]
- Lindström, E.; Shimokawa, T.; Toftgård, R.; Zaphiropoulos, P.G. PTCH Mutations: Distribution and Analyses. Hum. Mutat. 2006, 27, 215–219. [Google Scholar] [CrossRef]
- Verkouteren, B.J.A.; Wakkee, M.; van Geel, M.; van Doorn, R.; Winnepenninckx, V.J.; Korpershoek, E.; Mooyaart, A.L.; Reyners, A.K.L.; Terra, J.B.; Aarts, M.J.B.; et al. Molecular Testing in Metastatic Basal Cell Carcinoma. J. Am. Acad. Derm. 2021, 85, 1135–1142. [Google Scholar] [CrossRef]
- Maturo, M.G.; Rachakonda, S.; Heidenreich, B.; Pellegrini, C.; Srinivas, N.; Requena, C.; Serra-Guillen, C.; Llombart, B.; Sanmartin, O.; Guillen, C.; et al. Coding and Noncoding Somatic Mutations in Candidate Genes in Basal Cell Carcinoma. Sci. Rep. 2020, 10, 8005. [Google Scholar] [CrossRef] [PubMed]
- Di Nardo, L.; Pellegrini, C.; Di Stefani, A.; Ricci, F.; Fossati, B.; Del Regno, L.; Carbone, C.; Piro, G.; Corbo, V.; Delfino, P.; et al. Molecular Alterations in Basal Cell Carcinoma Subtypes. Sci. Rep. 2021, 11, 13206. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Ananthaswamy, H.N.; Muller, H.K.; Kripke, M.L. P53 Protects against Skin Cancer Induction by UV-B Radiation. Oncogene 1999, 18, 4247–4253. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Capell, B.C.; Parekh, V.; O’Day, C.; Atillasoy, C.; Bashir, H.M.; Yeh, C.; Shim, E.-H.; Prouty, S.M.; Dentchev, T.; et al. Whole-Exome and Transcriptome Analysis of UV-Exposed Epidermis and Carcinoma In Situ Reveals Early Drivers of Carcinogenesis. J. Investig. Derm. 2021, 141, 295–307.e13. [Google Scholar] [CrossRef] [PubMed]
- Reichrath, J.; Reichrath, S. The Impact of Notch Signaling for Carcinogenesis and Progression of Nonmelanoma Skin Cancer: Lessons Learned from Cancer Stem Cells, Tumor Angiogenesis, and Beyond. Adv. Exp. Med. Biol. 2021, 1287, 123–154. [Google Scholar] [CrossRef]
- Katarkar, A.; Bottoni, G.; Clocchiatti, A.; Goruppi, S.; Bordignon, P.; Lazzaroni, F.; Gregnanin, I.; Ostano, P.; Neel, V.; Dotto, G.P. NOTCH1 Gene Amplification Promotes Expansion of Cancer Associated Fibroblast Populations in Human Skin. Nat. Commun. 2020, 11, 5126. [Google Scholar] [CrossRef]
- Prior, I.A.; Hood, F.E.; Hartley, J.L. The Frequency of Ras Mutations in Cancer. Cancer Res. 2020, 80, 2969–2974. [Google Scholar] [CrossRef]
- Fadul, J.; Zulueta-Coarasa, T.; Slattum, G.M.; Redd, N.M.; Jin, M.F.; Redd, M.J.; Daetwyler, S.; Hedeen, D.; Huisken, J.; Rosenblatt, J. KRas-Transformed Epithelia Cells Invade and Partially Dedifferentiate by Basal Cell Extrusion. Nat. Commun. 2021, 12, 7180. [Google Scholar] [CrossRef]
- Bottomley, M.J.; Thomson, J.; Harwood, C.; Leigh, I. The Role of the Immune System in Cutaneous Squamous Cell Carcinoma. Int. J. Mol. Sci. 2019, 20, 2009. [Google Scholar] [CrossRef]
- Gerlini, G.; Romagnoli, P.; Pimpinelli, N. Skin Cancer and Immunosuppression. Crit. Rev. Oncol. Hematol. 2005, 56, 127–136. [Google Scholar] [CrossRef]
- Fujimura, T.; Aiba, S. Significance of Immunosuppressive Cells as a Target for Immunotherapies in Melanoma and Non-Melanoma Skin Cancers. Biomolecules 2020, 10, 1087. [Google Scholar] [CrossRef] [PubMed]
- Rangwala, S.; Tsai, K.Y. Roles of the Immune System in Skin Cancer. Br. J. Derm. 2011, 165, 953–965. [Google Scholar] [CrossRef] [PubMed]
- Alamartine, E.; Berthoux, P.; Mariat, C.; Cambazard, F.; Berthoux, F. Interleukin-10 Promoter Polymorphisms and Susceptibility to Skin Squamous Cell Carcinoma after Renal Transplantation. J. Investig. Derm. 2003, 120, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Sobjanek, M.; Zabłotna, M.; Bień, E.; Gleń, J.; Sokołowska-Wojdyło, M.; Ruckemann-Dziurdzińska, K.; Nowicki, R. Clinical Significance of IL-2 and IL-10 Gene Polymorphisms and Serum Levels in Patients with Basal-Cell Carcinoma. Biomark. Med. 2016, 10, 185–195. [Google Scholar] [CrossRef]
- Koronellos, N.; Yapijakis, C.; Katoulis, A.; Avgoustidis, D.; Vylliotis, A.; Papakosta, V.; Diamantopoulou, S.; Zografos, O.; Vairaktari, G.; Vairaktaris, E.; et al. Association Study Indicates Combined Effect of Interleukin-10 and Angiotensin-Converting Enzyme in Basal Cell Carcinoma Development. Arch. Derm. Res. 2021, 313, 373–380. [Google Scholar] [CrossRef]
- Basile, J.; Thiers, B.; Maize, J.; Lathers, D.M.R. Chemokine Receptor Expression in Non-Melanoma Skin Cancer. J. Cutan. Pathol. 2008, 35, 623–629. [Google Scholar] [CrossRef]
- Sartini, D.; Campagna, R.; Lucarini, G.; Pompei, V.; Salvolini, E.; Mattioli-Belmonte, M.; Molinelli, E.; Brisigotti, V.; Campanati, A.; Bacchetti, T.; et al. Differential Immunohistochemical Expression of Paraoxonase-2 in Actinic Keratosis and Squamous Cell Carcinoma. Hum. Cell 2021, 34, 1929–1931. [Google Scholar] [CrossRef]
- Fathi, F.; Ebrahimi, M.; Eslami, A.; Hafezi, H.; Eskandari, N.; Motedayyen, H. Association of Programmed Death-1 Gene Polymorphisms with the Risk of Basal Cell Carcinoma. Int. J. Immunogenet. 2019, 46, 444–450. [Google Scholar] [CrossRef]
- Goltz, D.; Gevensleben, H.; Vogt, T.J.; Dietrich, J.; Golletz, C.; Bootz, F.; Kristiansen, G.; Landsberg, J.; Dietrich, D. CTLA4 Methylation Predicts Response to Anti-PD-1 and Anti-CTLA-4 Immunotherapy in Melanoma Patients. JCI Insight 2018, 3, 96793. [Google Scholar] [CrossRef]
- Lajoie, J.E.; Lau, K.W.; Adam, A.P.; Wladis, E.J. Cytotoxic T-Lymphocyte-Associated Protein-4 and Lymphocyte Activation Gene-3 Expression in Orbitally-Invasive Versus Nodular Basal Cell Carcinoma. Ophthalmic Plast. Reconstr. Surg. 2021, 37, S109–S111. [Google Scholar] [CrossRef]
- Sławińska, M.; Zabłotna, M.; Nowicki, R.J.; Sobjanek, M. FOXP3 and CTLA-4 Genetic Variants’ Influence on the Susceptibility and Clinical Course of Basal Cell Carcinoma. Postepy Derm. Alergol. 2021, 38, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yang, J.; Wu, W. Seven Key Hub Genes Identified by Gene Co-Expression Network in Cutaneous Squamous Cell Carcinoma. BMC Cancer 2021, 21, 852. [Google Scholar] [CrossRef] [PubMed]
- Zou, D.-D.; Xu, D.; Deng, Y.-Y.; Wu, W.-J.; Zhang, J.; Huang, L.; He, L. Identification of Key Genes in Cutaneous Squamous Cell Carcinoma: A Transcriptome Sequencing and Bioinformatics Profiling Study. Ann. Transl. Med. 2021, 9, 1497. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lee, M.; Lee, Y.B.; Yu, D.S. Investigation of Genetic Mutations in High-Risk and Low-Risk Basal Cell Carcinoma in a Non-Caucasian Population by Whole Exome Sequencing. Acta Derm. Venereol. 2021, 101, adv00458. [Google Scholar] [CrossRef] [PubMed]
- Sławińska, M.; Zabłotna, M.; Gleń, J.; Lakomy, J.; Nowicki, R.J.; Sobjanek, M. STAT3 Polymorphisms and IL-6 Polymorphism Are Associated with the Risk of Basal Cell Carcinoma in Patients from Northern Poland. Arch. Derm. Res. 2019, 311, 697–704. [Google Scholar] [CrossRef]
- Madonna, S.; Scarponi, C.; Morelli, M.; Sestito, R.; Scognamiglio, P.L.; Marasco, D.; Albanesi, C. SOCS3 Inhibits the Pathological Effects of IL-22 in Non-Melanoma Skin Tumor-Derived Keratinocytes. Oncotarget 2017, 8, 24652–24667. [Google Scholar] [CrossRef]
- Mercuţ, I.M.; Simionescu, C.E.; Stepan, A.E.; Andreiana, B.C.; Ciurea, A.M.; Mercuţ, R.; Ciurea, M.E. The Immunoexpression of MMP-1 and MMP-13 in Eyelid Basal Cell Carcinoma. Rom. J. Morphol. Embryol. 2020, 61, 1221–1226. [Google Scholar] [CrossRef]
- Quan, Z.; Zhang, B.-B.; Yin, F.; Du, J.; Zhi, Y.-T.; Xu, J.; Song, N. DDX5 Silencing Suppresses the Migration of Basal Cell Carcinoma Cells by Downregulating JAK2/STAT3 Pathway. Technol. Cancer Res. Treat. 2019, 18, 1533033819892258. [Google Scholar] [CrossRef]
- Kim, H.S.; Shin, M.S.; Cheon, M.S.; Kim, J.W.; Lee, C.; Kim, W.H.; Kim, Y.S.; Jang, B.G. GREM1 Is Expressed in the Cancer-Associated Myofibroblasts of Basal Cell Carcinomas. PLoS ONE 2017, 12, e0174565. [Google Scholar] [CrossRef]
- Olafsdottir, T.; Stacey, S.N.; Sveinbjornsson, G.; Thorleifsson, G.; Norland, K.; Sigurgeirsson, B.; Thorisdottir, K.; Kristjansson, A.K.; Tryggvadottir, L.; Sarin, K.Y.; et al. Loss-of-Function Variants in the Tumor-Suppressor Gene PTPN14 Confer Increased Cancer Risk. Cancer Res. 2021, 81, 1954–1964. [Google Scholar] [CrossRef]
- Smirnov, A.; Anemona, L.; Montanaro, M.; Mauriello, A.; Annicchiarico-Petruzzelli, M.; Campione, E.; Melino, G.; Candi, E. Transglutaminase 3 Is Expressed in Basal Cell Carcinoma of the Skin. Eur. J. Derm. 2019, 29, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Gualandi, M.; Iorio, M.; Engeler, O.; Serra-Roma, A.; Gasparre, G.; Schulte, J.H.; Hohl, D.; Shakhova, O. Oncogenic ALK F1174L Drives Tumorigenesis in Cutaneous Squamous Cell Carcinoma. Life Sci. Alliance 2020, 3, e201900601. [Google Scholar] [CrossRef] [PubMed]
- Chu, Z.; Zhang, X.; Li, Q.; Hu, G.; Lian, C.G.; Geng, S. CDC20 Contributes to the Development of Human Cutaneous Squamous Cell Carcinoma through the Wnt/Β-catenin Signaling Pathway. Int. J. Oncol. 2019, 54, 1534–1544. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Yang, Y.; Zhang, H.-B.; Zheng, X.-H.; Li, H.-R.; Wen, J. Identification of CDK1 as a Candidate Marker in Cutaneous Squamous Cell Carcinoma by Integrated Bioinformatics Analysis. Transl. Cancer Res. 2021, 10, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Viiklepp, K.; Nissinen, L.; Ojalill, M.; Riihilä, P.; Kallajoki, M.; Meri, S.; Heino, J.; Kähäri, V.-M. C1r Upregulates Production of Matrix Metalloproteinase-13 and Promotes Invasion of Cutaneous Squamous Cell Carcinoma. J. Investig. Derm. 2021, 142, 1478–1488. [Google Scholar] [CrossRef]
- Jackson, W.D.; Gulino, A.; Fossati-Jimack, L.; Castro Seoane, R.; Tian, K.; Best, K.; Köhl, J.; Belmonte, B.; Strid, J.; Botto, M. C3 Drives Inflammatory Skin Carcinogenesis Independently of C5. J. Investig. Derm. 2021, 141, 404–414.e6. [Google Scholar] [CrossRef]
- Gao, D.; Chen, H.-Q. Specific Knockdown of HOXB7 Inhibits Cutaneous Squamous Cell Carcinoma Cell Migration and Invasion While Inducing Apoptosis via the Wnt/β-Catenin Signaling Pathway. Am. J. Physiol. Cell Physiol. 2018, 315, C675–C686. [Google Scholar] [CrossRef]
- Lobl, M.; Georgesen, C.; Black, J.D.; Lum, M.A.; Lauer, S.; Wysong, A. LRP1B Expression Is Correlated with Age and Perineural Invasion in Metastatic Cutaneous Squamous Cell Carcinoma: A Pilot Study. Am. J. Derm. 2022, 44, 49–53. [Google Scholar] [CrossRef]
- Li, Y.; Huang, C.; Yang, X. Characterization of TCF4-Mediated Oncogenic Role in Cutaneous Squamous Cell Carcinoma. Int. J. Clin. Exp. Pathol. 2019, 12, 3583–3594. [Google Scholar]
- Procopio, M.-G.; Laszlo, C.; Dotto, G.P. CSL-P53: From Senescence to CAF Activation. Cell Cycle 2016, 15, 485–486. [Google Scholar] [CrossRef][Green Version]
- Lipson, E.J.; Lilo, M.T.; Ogurtsova, A.; Esandrio, J.; Xu, H.; Brothers, P.; Schollenberger, M.; Sharfman, W.H.; Taube, J.M. Basal Cell Carcinoma: PD-L1/PD-1 Checkpoint Expression and Tumor Regression after PD-1 Blockade. J. Immunother. Cancer 2017, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Beksaç, B.; İlter, N.; Erdem, Ö.; Çakmak, P.; Çenetoğlu, S.; Yapar, D. Sparsity of Dendritic Cells and Cytotoxic T Cells in Tumor Microenvironment May Lead to Recurrence in Basal Cell Carcinoma. Int. J. Derm. 2020, 59, 1258–1263. [Google Scholar] [CrossRef] [PubMed]
- Bellei, B.; Caputo, S.; Carbone, A.; Silipo, V.; Papaccio, F.; Picardo, M.; Eibenschutz, L. The Role of Dermal Fibroblasts in Nevoid Basal Cell Carcinoma Syndrome Patients: An Overview. Int. J. Mol. Sci. 2020, 21, 720. [Google Scholar] [CrossRef] [PubMed]
- Toutfaire, M.; Dumortier, E.; Fattaccioli, A.; Van Steenbrugge, M.; Proby, C.M.; Debacq-Chainiaux, F. Unraveling the Interplay between Senescent Dermal Fibroblasts and Cutaneous Squamous Cell Carcinoma Cell Lines at Different Stages of Tumorigenesis. Int. J. Biochem. Cell Biol. 2018, 98, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R. The Biology and Function of Fibroblasts in Cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef]
- Liu, F.; Qi, L.; Liu, B.; Liu, J.; Zhang, H.; Che, D.; Cao, J.; Shen, J.; Geng, J.; Bi, Y.; et al. Fibroblast Activation Protein Overexpression and Clinical Implications in Solid Tumors: A Meta-Analysis. PLoS ONE 2015, 10, e0116683. [Google Scholar] [CrossRef]
- El Khoury, J.; Kurban, M.; Kibbi, A.-G.; Abbas, O. Fibroblast-Activation Protein: Valuable Marker of Cutaneous Epithelial Malignancy. Arch. Derm. Res. 2014, 306, 359–365. [Google Scholar] [CrossRef]
- Abbas, O.; Richards, J.E.; Mahalingam, M. Fibroblast-Activation Protein: A Single Marker That Confidently Differentiates Morpheaform/Infiltrative Basal Cell Carcinoma from Desmoplastic Trichoepithelioma. Mod. Pathol 2010, 23, 1535–1543. [Google Scholar] [CrossRef]
- Chan, J.S.K.; Tan, M.J.; Sng, M.K.; Teo, Z.; Phua, T.; Choo, C.C.; Li, L.; Zhu, P.; Tan, N.S. Cancer-Associated Fibroblasts Enact Field Cancerization by Promoting Extratumoral Oxidative Stress. Cell Death Dis. 2017, 8, e2562. [Google Scholar] [CrossRef]
- Omland, S.H.; Wettergren, E.E.; Mollerup, S.; Asplund, M.; Mourier, T.; Hansen, A.J.; Gniadecki, R. Cancer Associated Fibroblasts (CAFs) Are Activated in Cutaneous Basal Cell Carcinoma and in the Peritumoural Skin. BMC Cancer 2017, 17, 675. [Google Scholar] [CrossRef]
- Zhang, G.; He, Y.; Liu, Y.; Du, Y.; Yang, C.; Gao, F. Reduced Hyaluronan Cross-Linking Induces Breast Cancer Malignancy in a CAF-Dependent Manner. Cell Death Dis. 2021, 12, 586. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Sugai, T.; Ishida, K.; Osakabe, M.; Amano, H.; Kimura, H.; Sakuraba, M.; Kashiwa, K.; Kobayashi, S. Analysis of Cancer-Associated Fibroblasts and the Epithelial-Mesenchymal Transition in Cutaneous Basal Cell Carcinoma, Squamous Cell Carcinoma, and Malignant Melanoma. Hum. Pathol. 2018, 79, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jena, B.C.; Rout, L.; Dey, A.; Mandal, M. Active Autophagy in Cancer-Associated Fibroblasts: Recent Advances in Understanding the Novel Mechanism of Tumor Progression and Therapeutic Response. J. Cell Physiol. 2021, 236, 7887–7902. [Google Scholar] [CrossRef] [PubMed]
- Chisari, A.; Golán, I.; Campisano, S.; Gélabert, C.; Moustakas, A.; Sancho, P.; Caja, L. Glucose and Amino Acid Metabolic Dependencies Linked to Stemness and Metastasis in Different Aggressive Cancer Types. Front. Pharm. 2021, 12, 723798. [Google Scholar] [CrossRef]
- Yan, Y.; Chen, X.; Wang, X.; Zhao, Z.; Hu, W.; Zeng, S.; Wei, J.; Yang, X.; Qian, L.; Zhou, S.; et al. The Effects and the Mechanisms of Autophagy on the Cancer-Associated Fibroblasts in Cancer. J. Exp. Clin. Cancer Res. 2019, 38, 171. [Google Scholar] [CrossRef]
- Micke, P.; Kappert, K.; Ohshima, M.; Sundquist, C.; Scheidl, S.; Lindahl, P.; Heldin, C.-H.; Botling, J.; Ponten, F.; Ostman, A. In Situ Identification of Genes Regulated Specifically in Fibroblasts of Human Basal Cell Carcinoma. J. Investig. Derm. 2007, 127, 1516–1523. [Google Scholar] [CrossRef]
- Siljamäki, E.; Rappu, P.; Riihilä, P.; Nissinen, L.; Kähäri, V.-M.; Heino, J. H-Ras Activation and Fibroblast-Induced TGF-β Signaling Promote Laminin-332 Accumulation and Invasion in Cutaneous Squamous Cell Carcinoma. Matrix Biol. 2020, 87, 26–47. [Google Scholar] [CrossRef]
- Lacina, L.; Plzak, J.; Kodet, O.; Szabo, P.; Chovanec, M.; Dvorankova, B.; Smetana, K. Cancer Microenvironment: What Can We Learn from the Stem Cell Niche. Int. J. Mol. Sci. 2015, 16, 24094–24110. [Google Scholar] [CrossRef]
- Brandi, J.; Manfredi, M.; Speziali, G.; Gosetti, F.; Marengo, E.; Cecconi, D. Proteomic Approaches to Decipher Cancer Cell Secretome. Semin. Cell Dev. Biol. 2018, 78, 93–101. [Google Scholar] [CrossRef]
- Hoesl, C.; Zanuttigh, E.; Fröhlich, T.; Philippou-Massier, J.; Krebs, S.; Blum, H.; Dahlhoff, M. The Secretome of Skin Cancer Cells Activates the MTOR/MYC Pathway in Healthy Keratinocytes and Induces Tumorigenic Properties. Biochim. Biophys. Acta Mol. Cell Res. 2020, 1867, 118717. [Google Scholar] [CrossRef]
- Azimi, A.; Kaufman, K.L.; Ali, M.; Arthur, J.; Kossard, S.; Fernandez-Penas, P. Differential Proteomic Analysis of Actinic Keratosis, Bowen’s Disease and Cutaneous Squamous Cell Carcinoma by Label-Free LC-MS/MS. J. Derm. Sci. 2018, 91, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Botchkarev, V.A.; Gdula, M.R.; Mardaryev, A.N.; Sharov, A.A.; Fessing, M.Y. Epigenetic Regulation of Gene Expression in Keratinocytes. J. Investig. Derm. 2012, 132, 2505–2521. [Google Scholar] [CrossRef] [PubMed]
- García-Nieto, P.E.; Schwartz, E.K.; King, D.A.; Paulsen, J.; Collas, P.; Herrera, R.E.; Morrison, A.J. Carcinogen Susceptibility Is Regulated by Genome Architecture and Predicts Cancer Mutagenesis. EMBO J. 2017, 36, 2829–2843. [Google Scholar] [CrossRef]
- Penta, D.; Somashekar, B.S.; Meeran, S.M. Epigenetics of Skin Cancer: Interventions by Selected Bioactive Phytochemicals. Photodermatol. Photoimmunol. Photomed. 2018, 34, 42–49. [Google Scholar] [CrossRef]
- Kashyap, M.P.; Sinha, R.; Mukhtar, M.S.; Athar, M. Epigenetic Regulation in the Pathogenesis of Non-Melanoma Skin Cancer. Semin. Cancer Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Gerloff, D.; Sunderkötter, C.; Wohlrab, J. Importance of MicroRNAs in Skin Oncogenesis and Their Suitability as Agents and Targets for Topical Therapy. Ski. Pharm. Physiol. 2020, 33, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Mi, X.; Lai, K.; Yan, L.; Xie, S.; Qiu, X.; Xiao, S.; Wei, S. MiR-18a Expression in Basal Cell Carcinoma and Regulatory Mechanism on Autophagy through MTOR Pathway. Clin. Exp. Derm. 2020, 45, 1027–1034. [Google Scholar] [CrossRef]
- Fernández-Figueras, M.T.; Carrato, C.; Saenz-Sardà, X.; Musulén, E.; Fuente, M.J.; Puig, L. MicroRNA31 and MMP-1 Contribute to the Differentiated Pathway of Invasion -with Enhanced Epithelial-to-Mesenchymal Transition- in Squamous Cell Carcinoma of the Skin. Arch. Derm. Res. 2021. [Google Scholar] [CrossRef]
- Lohcharoenkal, W.; Li, C.; Das Mahapatra, K.; Lapins, J.; Homey, B.; Sonkoly, E.; Pivarcsi, A. MiR-130a Acts as a Tumor Suppressor MicroRNA in Cutaneous Squamous Cell Carcinoma and Regulates the Activity of the BMP/SMAD Pathway by Suppressing ACVR1. J. Investig. Derm. 2021, 141, 1922–1931. [Google Scholar] [CrossRef]
- Chen, I.-P.; Bender, M.; Spassova, I.; Henning, S.; Kubat, L.; Fan, K.; Degenhardt, S.; Mhamdi-Ghodbani, M.; Sriram, A.; Volkmer, B.; et al. UV-Type Specific Alteration of MiRNA Expression and Its Association with Tumor Progression and Metastasis in SCC Cell Lines. J. Cancer Res. Clin. Oncol. 2020, 146, 3215–3231. [Google Scholar] [CrossRef]
- Matos, L.L.; Menderico Junior, G.M.; Theodoro, T.R.; Pasini, F.S.; de Menezes Ishikawa, M.; Ribeiro, A.A.B.; de Mello, E.S.; da Silva Pinhal, M.A.; Moyses, R.A.; Kulcsar, M.A.V.; et al. Cancer-Associated Fibroblast Regulation by MicroRNAs Promotes Invasion of Oral Squamous Cell Carcinoma. Oral Oncol. 2020, 110, 104909. [Google Scholar] [CrossRef] [PubMed]
- Fan, K.; Spassova, I.; Gravemeyer, J.; Ritter, C.; Horny, K.; Lange, A.; Gambichler, T.; Ødum, N.; Schrama, D.; Schadendorf, D.; et al. Merkel Cell Carcinoma-Derived Exosome-Shuttle MiR-375 Induces Fibroblast Polarization by Inhibition of RBPJ and P53. Oncogene 2021, 40, 980–996. [Google Scholar] [CrossRef] [PubMed]
- Verduci, L.; Tarcitano, E.; Strano, S.; Yarden, Y.; Blandino, G. CircRNAs: Role in Human Diseases and Potential Use as Biomarkers. Cell Death Dis. 2021, 12, 468. [Google Scholar] [CrossRef] [PubMed]
- Hatibaruah, A.; Rahman, M.; Agarwala, S.; Singh, S.A.; Shi, J.; Gupta, S.; Paul, P. Circular RNAs in Cancer and Diabetes. J. Genet. 2021, 100, 21. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Li, L. Circular RNA Hsa_Circ_0005795 Mediates Cell Proliferation of Cutaneous Basal Cell Carcinoma via Sponging MiR-1231. Arch. Derm. Res. 2021, 313, 773–782. [Google Scholar] [CrossRef]
- Li, X.; Kong, Y.; Li, H.; Xu, M.; Jiang, M.; Sun, W.; Xu, S. CircRNA Circ_0067772 Aggravates the Malignant Progression of Cutaneous Squamous Cell Carcinoma by Regulating MiR-1238-3p/FOXG1 Axis. Genes Genom. 2021, 43, 491–501. [Google Scholar] [CrossRef]
- Hu, P.; Ma, L.; Wu, Z.; Zheng, G.; Li, J. Expression of MiR-34a in Basal Cell Carcinoma Patients and Its Relationship with Prognosis. J. BUON 2019, 24, 1283–1288. [Google Scholar]
- Sun, H.; Jiang, P. MicroRNA-451a Acts as Tumor Suppressor in Cutaneous Basal Cell Carcinoma. Mol. Genet. Genom. Med. 2018, 6, 1001–1009. [Google Scholar] [CrossRef]
- Fan, Z.-X.; Xi, W.; Miao, X.-Y.; Li, L.-Y.; Miao, G.-Y. Circ_NCKAP1 Promotes Skin Basal Cell Carcinoma Progression by Sponging the MiR-148b-5p/HSP90 Axis. Eur. Rev. Med. Pharm. Sci. 2021, 25, 5355–5364. [Google Scholar] [CrossRef]
- Wimmer, M.; Zauner, R.; Ablinger, M.; Piñón-Hofbauer, J.; Guttmann-Gruber, C.; Reisenberger, M.; Lettner, T.; Niklas, N.; Proell, J.; Sajinovic, M.; et al. A Cancer Stem Cell-like Phenotype Is Associated with MiR-10b Expression in Aggressive Squamous Cell Carcinomas. Cell Commun. Signal. 2020, 18, 61. [Google Scholar] [CrossRef]
- Das Mahapatra, K.; Pasquali, L.; Søndergaard, J.N.; Lapins, J.; Nemeth, I.B.; Baltás, E.; Kemény, L.; Homey, B.; Moldovan, L.-I.; Kjems, J.; et al. A Comprehensive Analysis of Coding and Non-Coding Transcriptomic Changes in Cutaneous Squamous Cell Carcinoma. Sci. Rep. 2020, 10, 3637. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Liu, X.; Ma, G.; Li, C. Upregulated Circular RNA Circ_0070934 Facilitates Cutaneous Squamous Cell Carcinoma Cell Growth and Invasion by Sponging MiR-1238 and MiR-1247-5p. Biochem. Biophys. Res. Commun. 2019, 513, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.-W.; Wu, H.-Y.; Zhu, C.-R.; Wu, D.-D. CircRNA Hsa_circ_0070934 Functions as a Competitive Endogenous RNA to Regulate HOXB7 Expression by Sponging MiR-1236-3p in Cutaneous Squamous Cell Carcinoma. Int. J. Oncol. 2020, 57, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Li, C.; Huang, H.; Liang, L.; Zhang, J.; Li, Q.; Wang, Q.; Zhang, S.; Zeng, K.; Zhang, X.; et al. Circular RNA Profiles and the Potential Involvement of Down-Expression of Hsa_circ_0001360 in Cutaneous Squamous Cell Carcinogenesis. FEBS Open Bio 2021, 11, 1209–1222. [Google Scholar] [CrossRef]
- Wilken, R.; Criscito, M.; Pavlick, A.C.; Stevenson, M.L.; Carucci, J.A. Current Research in Melanoma and Aggressive Nonmelanoma Skin Cancer. Facial Plast. Surg. 2020, 36, 200–210. [Google Scholar] [CrossRef]
- Patrinely, J.R.; Dewan, A.K.; Johnson, D.B. The Role of Anti-PD-1/PD-L1 in the Treatment of Skin Cancer. BioDrugs 2020, 34, 495–503. [Google Scholar] [CrossRef]
- Stratigos, A.J.; Sekulic, A.; Peris, K.; Bechter, O.; Prey, S.; Kaatz, M.; Lewis, K.D.; Basset-Seguin, N.; Chang, A.L.S.; Dalle, S.; et al. Cemiplimab in Locally Advanced Basal Cell Carcinoma after Hedgehog Inhibitor Therapy: An Open-Label, Multi-Centre, Single-Arm, Phase 2 Trial. Lancet Oncol. 2021, 22, 848–857. [Google Scholar] [CrossRef]
- Schöffski, P.; Tan, D.S.W.; Martín, M.; Ochoa-de-Olza, M.; Sarantopoulos, J.; Carvajal, R.D.; Kyi, C.; Esaki, T.; Prawira, A.; Akerley, W.; et al. Phase I/II Study of the LAG-3 Inhibitor Ieramilimab (LAG525) ± Anti-PD-1 Spartalizumab (PDR001) in Patients with Advanced Malignancies. J. Immunother. Cancer 2022, 10, e003776. [Google Scholar] [CrossRef]
- Zhang, L.; Ji, Z.; Zhang, J.; Yang, S. Photodynamic Therapy Enhances Skin Cancer Chemotherapy Effects through Autophagy Regulation. Photodiagnosis Photodyn. 2019, 28, 159–165. [Google Scholar] [CrossRef]
- Li, S.; Wang, P.; Zhang, G.; Ji, J.; Lv, T.; Wang, X.; Wang, H. The Effect of ALA-PDT on Reversing the Activation of Cancer-Associated Fibroblasts in Cutaneous Squamous Cell Carcinoma. Photodiagnosis Photodyn. 2019, 27, 234–240. [Google Scholar] [CrossRef]
- Zamarrón, A.; García, M.; Río, M.D.; Larcher, F.; Juarranz, Á. Effects of Photodynamic Therapy on Dermal Fibroblasts from Xeroderma Pigmentosum and Gorlin-Goltz Syndrome Patients. Oncotarget 2017, 8, 77385–77399. [Google Scholar] [CrossRef] [PubMed]
- Domogauer, J.D.; de Toledo, S.M.; Howell, R.W.; Azzam, E.I. Acquired Radioresistance in Cancer Associated Fibroblasts Is Concomitant with Enhanced Antioxidant Potential and DNA Repair Capacity. Cell Commun. Signal. 2021, 19, 30. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.E.; Herzog, C.; Jones, A.; Leavy, O.C.; Evans, I.; Knapp, S.; Reisel, D.; Nazarenko, T.; Kim, Y.-N.; Franchi, D.; et al. The WID-BC-Index Identifies Women with Primary Poor Prognostic Breast Cancer Based on DNA Methylation in Cervical Samples. Nat. Commun. 2022, 13, 449. [Google Scholar] [CrossRef] [PubMed]
- Du-Harpur, X.; Watt, F.M.; Luscombe, N.M.; Lynch, M.D. What Is AI? Applications of Artificial Intelligence to Dermatology. Br. J. Derm. 2020, 183, 423–430. [Google Scholar] [CrossRef]
- Wells, A.; Patel, S.; Lee, J.B.; Motaparthi, K. Artificial Intelligence in Dermatopathology: Diagnosis, Education, and Research. J. Cutan. Pathol. 2021, 48, 1061–1068. [Google Scholar] [CrossRef]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Cancer Statistics for the Year 2020: An Overview. Int. J. Cancer 2021, 149, 778–789. [Google Scholar] [CrossRef]
- Andrew, T.W.; Alrawi, M.; Lovat, P. Reduction in Skin Cancer Diagnoses in the UK during the COVID-19 Pandemic. Clin. Exp. Derm. 2021, 46, 145–146. [Google Scholar] [CrossRef]
- Baumann, B.C.; MacArthur, K.M.; Brewer, J.D.; Mendenhall, W.M.; Barker, C.A.; Etzkorn, J.R.; Jellinek, N.J.; Scott, J.F.; Gay, H.A.; Baumann, J.C.; et al. Management of Primary Skin Cancer during a Pandemic: Multidisciplinary Recommendations. Cancer 2020, 126, 3900–3906. [Google Scholar] [CrossRef]
- American Cancer Society. Basal & Squamous Cell Skin Cancer Statistics. Available online: https://www.cancer.org/cancer/basal-and-squamous-cell-skin-cancer/about/key-statistics.html (accessed on 3 May 2022).
- Eigentler, T.K.; Leiter, U.; Häfner, H.-M.; Garbe, C.; Röcken, M.; Breuninger, H. Survival of Patients with Cutaneous Squamous Cell Carcinoma: Results of a Prospective Cohort Study. J. Investig. Derm. 2017, 137, 2309–2315. [Google Scholar] [CrossRef]
- Tagliaferri, L.; Di Stefani, A.; Schinzari, G.; Fionda, B.; Rossi, E.; Del Regno, L.; Gentileschi, S.; Federico, F.; Valentini, V.; Tortora, G.; et al. Skin Cancer Triage and Management during COVID-19 Pandemic. J. Eur. Acad. Derm. Venereol. 2020, 34, 1136–1139. [Google Scholar] [CrossRef]
- Earnshaw, C.H.; Hunter, H.J.A.; McMullen, E.; Griffiths, C.E.M.; Warren, R.B. Reduction in Skin Cancer Diagnosis, and Overall Cancer Referrals, during the COVID-19 Pandemic. Br. J. Derm. 2020, 183, 792–794. [Google Scholar] [CrossRef] [PubMed]
- Roseleur, J.; Gonzalez-Chica, D.A.; Emery, J.; Stocks, N.P. Skin Checks and Skin Cancer Diagnosis in Australian General Practice before and during the COVID-19 Pandemic, 2011–2020. Br. J. Derm. 2021, 185, 853–855. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, A.; Agnihotri, S.; Mehta, A.; Rawal, S. COVID-19 and Cancer: Sailing through the Tides. Pathol. Res. Pract. 2021, 221, 153417. [Google Scholar] [CrossRef] [PubMed]
- Goldust, M.; Agarwal, K.; Podder, I.; Navarini, A.A. Skin Cancer and COVID-19. Derm. Ther. 2020, 33, e14050. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Jin, R.; Zhao, J.; Li, W.; Shen, H. Risk of COVID-19 for Patients with Cancer. Lancet Oncol. 2020, 21, e180. [Google Scholar] [CrossRef]
- Griffith, C.F. Skin Cancer in Immunosuppressed Patients. JAAPA 2022, 35, 19–27. [Google Scholar] [CrossRef]
- Liang, W.; Guan, W.; Chen, R.; Wang, W.; Li, J.; Xu, K.; Li, C.; Ai, Q.; Lu, W.; Liang, H.; et al. Cancer Patients in SARS-CoV-2 Infection: A Nationwide Analysis in China. Lancet Oncol. 2020, 21, 335–337. [Google Scholar] [CrossRef]



| Genes | Methods | Findings | Reference |
|---|---|---|---|
| Tumor suppressor | |||
| LRP1B | Tissue | Most frequently mutated gene in 50% of analyzed samples Candidate BCC driver gene in Korean patients | [55] |
| Immune system | |||
| IL-6 | Peripheral blood | Variants in IL-6 are associated with higher risk of BCC | [56] |
| SOCS3 | Cell line Murine model | Use of peptides mimicking the action of SOCS3 may inhibit the proliferative effect of IL-22 upon transformed keratinocytes | [57] |
| Extracellular matrix degradation | |||
| MMP1 MMP3 | Tissue | Matrix metallopeptidases are involved in tumor progression through extracellular matrix degradation | [58] |
| Cell processes | |||
| DDX5 | Tissue Cell line | Gene knockdown increased apoptosis and suppressed migration and invasion of BCC cells | [59] |
| GREM1 | Tissue | Invasive cancer cells may induce GREM1 expression in fibroblasts and BCC subtypes can be determinants for protein expression levels | [60] |
| PTPN14 | Large-scale next generation sequence data | BCC predisposition gene through germline loss of function variants | [61] |
| Protein crosslinking | |||
| TGM3 | Tissue | Overexpression of TGM3 in BCC tissue New potential specific marker for BCC | [62] |
| Genes | Methods | Findings | Reference |
|---|---|---|---|
| Cellular communication | |||
| ALK | Murine model | Variants driving constitutively active function trigger oncogenic signaling for SCC | [63] |
| Cell cycle | |||
| CDC20 | Cell line | Inhibition of SCC proliferation through gene silencing. Novel target for diagnosis and treatment | [64] |
| CDK1 | Bioinformatics analysis research | Higher expression in SCC Gene expression analysis provides a predictive tool in tumor development and progression understanding | [65] |
| Immune system | |||
| C1R | Murine model Cell line | Role of C1r in SCC tumor growth and invasion by increasing matrix metallopeptidases production | [66] |
| C3 | Cell line | Tumorgenic effect of C3 in SCC during chronic inflammation in the skin | [67] |
| SOCS3 | Cell line Murine model | Use of peptides mimicking the action of SOCS3 may inhibit the proliferative effect of IL-22 upon transformed keratinocytes | [57] |
| Cell proliferation and differentiation | |||
| HOXB7 | Cell line | Gene knockdown accelerates apoptosis, suppresses cell migration and tumor progression | [68] |
| Tumor suppressor | |||
| LRP1B | Tissue | Increased expression in metastatic SCC Possible predictive value for immunotherapy response | [69] |
| Transcription factor | |||
| TCF4 | Cell line | Interference of TCF4 expression played an important tumor-repressive role in SCC by the inhibition of signaling pathways activation | [70] |
| Non-Coding RNA | Samples and Methods | Status | Proposed Role | Limitations | Reference |
|---|---|---|---|---|---|
| miRNAs | |||||
| miR-34a | 86 Serum | Downregulation | Tumor suppressor | Small sample size | [108] |
| miRNA-451a | 22 Tissue Murine model | Downregulation | Tumor suppressor | Small sample size | [109] |
| circRNAs | |||||
| Circ_NCKAP1 | 3 Tissue Cell line | Upregulated | Tumor promoter | Limited sample size Lack of in vivo model | [110] |
| Non-Coding RNA | Samples and Methods | Status | Proposed Role | Limitations | Reference |
|---|---|---|---|---|---|
| miRNAs | |||||
| miR-10b | - Cell line | Upregulated | Tumor promoter | Lack of evidence on animal models | [111] |
| lncRNAs | |||||
| RP11-493L12.5 | 28 Tissue | Upregulated | Unknown | Small sample size | [112] |
| KB-1410C5.3/lnc-GRHL2 | Downregulated | Unknown | |||
| circRNAs | |||||
| circ_0070934 | 38 Tissue Cell line | Upregulated | Tumor promoter | Downstream targets of miRNAs sponged by this circRNA are still unknown in SCC | [113] |
| - Cell line | Upregulated | Tumor promoter | Tissue from patients is needed to asses clinical value | [114] | |
| circ_EPSTI | 28 Tissue | Upregulated | Unknown | Small sample size | [112] |
| circ_IFFO2 | Downregulated | Tumor promoter | |||
| circ_0001360 | 3 Tissue | Downregulated | Tumor suppressor | Small sample size | [115] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zambrano-Román, M.; Padilla-Gutiérrez, J.R.; Valle, Y.; Muñoz-Valle, J.F.; Valdés-Alvarado, E. Non-Melanoma Skin Cancer: A Genetic Update and Future Perspectives. Cancers 2022, 14, 2371. https://doi.org/10.3390/cancers14102371
Zambrano-Román M, Padilla-Gutiérrez JR, Valle Y, Muñoz-Valle JF, Valdés-Alvarado E. Non-Melanoma Skin Cancer: A Genetic Update and Future Perspectives. Cancers. 2022; 14(10):2371. https://doi.org/10.3390/cancers14102371
Chicago/Turabian StyleZambrano-Román, Marianela, Jorge R. Padilla-Gutiérrez, Yeminia Valle, José F. Muñoz-Valle, and Emmanuel Valdés-Alvarado. 2022. "Non-Melanoma Skin Cancer: A Genetic Update and Future Perspectives" Cancers 14, no. 10: 2371. https://doi.org/10.3390/cancers14102371
APA StyleZambrano-Román, M., Padilla-Gutiérrez, J. R., Valle, Y., Muñoz-Valle, J. F., & Valdés-Alvarado, E. (2022). Non-Melanoma Skin Cancer: A Genetic Update and Future Perspectives. Cancers, 14(10), 2371. https://doi.org/10.3390/cancers14102371









