Toxicity and Clinical Results after Proton Therapy for Pediatric Medulloblastoma: A Multi-Centric Retrospective Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
3.1. Population
3.2. Toxicity Analysis
3.2.1. Acute and Subacute (<6 Months) Toxicities
3.2.2. Hematological Toxicity
3.2.3. Preliminary Data on Neuroendocrine Toxicities
3.2.4. Preliminary Data on Ototoxicity
3.2.5. Preliminary Data on Late Toxicities
3.3. Preliminary Data on Disease Control and Survival
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Northcott, P.A.; Robinson, G.W.; Kratz, C.P.; Mabbott, D.J.; Pomeroy, S.L.; Clifford, S.C.; Rutkowski, S.; Ellison, D.W.; Malkin, D.; Taylor, M.D.; et al. Medulloblastoma. Nat. Rev. Dis. Prim. 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.J.; Kim, H.; Park, H.J.; Kim, D.S.; Ra, Y.S.; Shin, H.Y. Neurocognitive Function and Health-Related Quality of Life in Pediatric Korean Survivors of Medulloblastoma. J. Korean Med. Sci. 2016, 31, 1726–1734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Veneroni, L.; Boschetti, L.; Barretta, F.; Clerici, C.A.; Simonetti, F.; Schiavello, E.; Biassoni, V.; Spreafico, F.; Gandola, L.; Pecori, E.; et al. Quality of life in long-term survivors treated for metastatic medulloblastoma with a hyperfractionated accelerated radiotherapy (HART) strategy. Childs. Nerv. Syst. 2017, 33, 1969–1976. [Google Scholar] [CrossRef] [PubMed]
- King, A.A.; Seidel, K.; Di, C.; Leisenring, W.M.; Perkins, S.M.; Krull, K.R.; Sklar, C.A.; Green, D.M.; Armstrong, G.T.; Zeltzer, L.K.; et al. Long-term neurologic health and psychosocial function of adult survivors of childhood medulloblastoma/PNET: A report from the Childhood Cancer Survivor Study. Neuro-Oncology 2017, 19, 689–698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moxon-Emre, I.; Bouffet, E.; Taylor, M.D.; Laperriere, N.; Scantlebury, N.; Law, N.; Spiegler, B.J.; Malkin, D.; Janzen, L.; Mabbott, D. Impact of craniospinal dose, boost volume, and neurologic complications on intellectual outcome in patients with medulloblastoma. J. Clin. Oncol. 2014, 32, 1760–1768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spiegler, B.J.; Bouffet, E.; Greenberg, M.L.; Rutka, J.T.; Mabbott, D.J. Change in neurocognitive functioning after treatment with cranial radiation in childhood. J. Clin. Oncol. 2004, 22, 706–713. [Google Scholar] [CrossRef]
- Frange, P.; Alapetite, C.; Gaboriaud, G.; Bours, D.; Zucker, J.M.; Zerah, M.; Brisse, H.; Chevignard, M.; Mosseri, V.; Bouffet, E.; et al. From childhood to adulthood: Long-term outcome of medulloblastoma patients. The Institut Curie experience (1980–2000). J. Neurooncol. 2009, 95, 271–279. [Google Scholar] [CrossRef]
- Ris, M.D.; Packer, R.; Goldwein, J.; Jones-Wallace, D.; Boyett, J.M. Intellectual outcome after reduced-dose radiation therapy plus adjuvant chemotherapy for medulloblastoma: A Children’s Cancer Group study. J. Clin. Oncol. 2001, 19, 3470–3476. [Google Scholar] [CrossRef]
- Ramaswamy, V.; Taylor, M.D. Medulloblastoma: From myth to molecular. J. Clin. Oncol. 2017, 35, 2355–2363. [Google Scholar] [CrossRef]
- Packer, R.J.; Zhou, T.; Holmes, E.; Vezina, G.; Gajjar, A. Survival and secondary tumors in children with medulloblastoma receiving radiotherapy and adjuvant chemotherapy: Results of Children’s Oncology Group trial A9961. Neuro-Oncology 2013, 15, 97–103. [Google Scholar] [CrossRef] [Green Version]
- Packer, R.J.; Gajjar, A.; Vezina, G.; Rorke-Adams, L.; Burger, P.C.; Robertson, P.L.; Bayer, L.; LaFond, D.; Donahue, B.R.; Marymont, M.A.H.; et al. Phase III study of craniospinal radiation therapy followed by adjuvant chemotherapy for newly diagnosed average-risk medulloblastoma. J. Clin. Oncol. 2006, 24, 4202–4208. [Google Scholar] [CrossRef] [PubMed]
- Padovani, L.; Horan, G.; Ajithkumar, T. Radiotherapy Advances in Paediatric Medulloblastoma Treatment. Clin. Oncol. 2019, 31, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Decker, A.L.; Szulc, K.U.; Bouffet, E.; Laughlin, S.; Chakravarty, M.M.; Skocic, J.; de Medeiros, C.B.; Mabbott, D.J. Smaller hippocampal subfield volumes predict verbal associative memory in pediatric brain tumor survivors. Hippocampus 2017, 27, 1140–1154. [Google Scholar] [CrossRef]
- Glass, J.O.; Ogg, R.J.; Hyun, J.W.; Harreld, J.H.; Schreiber, J.E.; Palmer, S.L.; Li, Y.; Gajjar, A.J.; Reddick, W.E. Disrupted development and integrity of frontal white matter in patients treated for pediatric medulloblastoma. Neuro-Oncology 2017, 19, 1408–1418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moxon-Emre, I.; Bouffet, E.; Taylor, M.D.; Laperriere, N.; Sharpe, M.B.; Laughlin, S.; Bartels, U.; Scantlebury, N.; Law, N.; Malkin, D.; et al. Vulnerability of white matter to insult during childhood: Evidence from patients treated for medulloblastoma. J. Neurosurg. Pediatr. 2016, 18, 29–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ajithkumar, T.; Price, S.; Horan, G.; Burke, A.; Jefferies, S. Prevention of radiotherapy-induced neurocognitive dysfunction in survivors of paediatric brain tumours: The potential role of modern imaging and radiotherapy techniques. Lancet Oncol. 2017, 18, e91–e100. [Google Scholar] [CrossRef] [Green Version]
- Rombi, B.; Vennarini, S.; Vinante, L.; Ravanelli, D.; Amichetti, M. Proton radiotherapy for pediatric tumors: Review of first clinical results. Ital. J. Pediatr. 2014, 40. [Google Scholar] [CrossRef]
- Johnstone, P.A.S.; McMullen, K.P.; Buchsbaum, J.C.; Douglas, J.G.; Helft, P. Pediatric CSI: Are protons the only ethical approach? Int. J. Radiat. Oncol. Biol. Phys. 2013, 87, 228–230. [Google Scholar] [CrossRef]
- Lundkvist, J.; Ekman, M.; Ericsson, S.R.; Jönsson, B.; Glimelius, B. Cost-effectiveness of proton radiation in the treatment of childhood medulloblastoma. Cancer 2005, 103, 793–801. [Google Scholar] [CrossRef]
- Journy, N.; Indelicato, D.J.; Withrow, D.R.; Akimoto, T.; Alapetite, C.; Araya, M.; Chang, A.; Chang, J.H.C.; Chon, B.; Confer, M.E.; et al. Patterns of proton therapy use in pediatric cancer management in 2016: An international survey. Radiother. Oncol. 2019, 132, 155–161. [Google Scholar] [CrossRef]
- Kahalley, L.S.; Peterson, R.; Douglas Ris, M.; Janzen, L.; Fatih Okcu, M.; Grosshans, D.R.; Ramaswamy, V.; Paulino, A.C.; Hodgson, D.; Mahajan, A.; et al. Superior intellectual outcomes after proton radiotherapy compared with photon radiotherapy for pediatric medulloblastoma. J. Clin. Oncol. 2020, 38, 454–461. [Google Scholar] [CrossRef]
- Kahalley, L.S.; Ris, M.D.; Grosshans, D.R.; Okcu, M.F.; Paulino, A.C.; Chintagumpala, M.; Moore, B.D.; Guffey, D.; Minard, C.G.; Stancel, H.H.; et al. Comparing intelligence quotient change after treatment with proton versus photon radiation therapy for pediatric brain tumors. J. Clin. Oncol. 2016, 34, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.J.; Zhai, H.; Both, S.; Tochner, Z.; Lustig, R.; Hill-Kayser, C. Breast Cancer Screening for Childhood Cancer Survivors after Craniospinal Irradiation with Protons Versus X-rays. J. Pediatr. Hematol. Oncol. 2013, 35. [Google Scholar] [CrossRef] [PubMed]
- Eaton, B.R.; Esiashvili, N.; Kim, S.; Patterson, B.; Weyman, E.A.; Thornton, L.T.; Mazewski, C.; MacDonald, T.J.; Ebb, D.; MacDonald, S.M.; et al. Endocrine outcomes with proton and photon radiotherapy for standard risk medulloblastoma. Neuro-Oncology 2016, 18, 881–887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paulino, A.C.; Mahajan, A.; Ye, R.; Grosshans, D.R.; Fatih Okcu, M.; Su, J.; McAleer, M.F.; McGovern, S.; Mangona, V.A.; Chintagumpala, M. Ototoxicity and cochlear sparing in children with medulloblastoma: Proton vs. photon radiotherapy. Radiother. Oncol. 2018, 128, 128–132. [Google Scholar] [CrossRef]
- Yock, T.I.; Yeap, B.Y.; Ebb, D.H.; Weyman, E.; Eaton, B.R.; Sherry, N.A.; Jones, R.M.; MacDonald, S.M.; Pulsifer, M.B.; Lavally, B.; et al. Long-term toxic effects of proton radiotherapy for paediatric medulloblastoma: A phase 2 single-arm study. Lancet Oncol. 2016, 17, 287–298. [Google Scholar] [CrossRef]
- Zhang, R.; Howell, R.M.; Homann, K.; Giebeler, A.; Taddei, P.J.; Mahajan, A.; Newhauser, W.D. Predicted risks of radiogenic cardiac toxicity in two pediatric patients undergoing photon or proton radiotherapy. Radiat. Oncol. 2013, 8, 184. [Google Scholar] [CrossRef] [Green Version]
- Pérez-Andújar, A.; Newhauser, W.D.; Taddei, P.J.; Mahajan, A.; Howell, R.M. The predicted relative risk of premature ovarian failure for three radiotherapy modalities in a girl receiving craniospinal irradiation. Phys. Med. Biol. 2013, 58, 3107–3123. [Google Scholar] [CrossRef] [Green Version]
- Cochran, D.M.; Yock, T.I.; Adams, J.A.; Tarbell, N.J. Radiation Dose to the Lens During Craniospinal Irradiation—An Improvement in Proton Radiotherapy Technique. Int. J. Radiat. Oncol. 2008, 70, 1336–1342. [Google Scholar] [CrossRef]
- Song, S.; Park, H.J.; Yoon, J.H.; Kim, D.W.; Park, J.; Shin, D.; Shin, S.H.; Kang, H.J.; Kim, S.-K.; Phi, J.H.; et al. Proton beam therapy reduces the incidence of acute haematological and gastrointestinal toxicities associated with craniospinal irradiation in pediatric brain tumors. Acta Oncol. (Madr). 2014, 53, 1158–1164. [Google Scholar] [CrossRef]
- Suneja, G.; Poorvu, P.D.; Hill-Kayser, C.; Lustig, R.A. Acute toxicity of proton beam radiation for pediatric central nervous system malignancies. Pediatr. Blood Cancer 2013, 60, 1431–1436. [Google Scholar] [CrossRef] [PubMed]
- Gnekow, A.K. Recommendations of the brain tumor subcommittee for the reporting of trials. Med. Pediatr. Oncol. 1995, 24, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Farace, P.; Bizzocchi, N.; Righetto, R.; Fellin, F.; Fracchiolla, F.; Lorentini, S.; Widesott, L.; Algranati, C.; Rombi, B.; Vennarini, S.; et al. Supine craniospinal irradiation in pediatric patients by proton pencil beam scanning. Radiother. Oncol. 2017, 123, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Fellin, F.; Fracchiolla, F.; Rombi, B.; Lipparini, M.; Vennarini, S.; Farace, P. An advanced junction concept in pediatric craniospinal irradiation by proton pencil beam scanning. Phys. Med. 2019, 60, 168–173. [Google Scholar] [CrossRef]
- Ajithkumar, T.; Horan, G.; Padovani, L.; Thorp, N.; Timmermann, B.; Alapetite, C.; Gandola, L.; Ramos, M.; Van Beek, K.; Christiaens, M.; et al. SIOPE–Brain tumor group consensus guideline on craniospinal target volume delineation for high-precision radiotherapy. Radiother. Oncol. 2018, 128, 192–197. [Google Scholar] [CrossRef]
- How, J.; Blattner, M.; Fowler, S.; Wang-Gillam, A.; Schindler, S.E. Chemotherapy-associated Posterior Reversible Encephalopathy Syndrome: A Case Report and Review of the Literature. Neurologist 2016, 21, 112–117. [Google Scholar] [CrossRef]
- Ramirez-Fort, M.K.; Zeng, J.; Feily, A.; Ramirez-Pacheco, L.A.; Jenrette, J.M.; Mayhew, D.L.; Syed, T.; Cooper, S.L.; Linden, C.; Graybill, W.S.; et al. Radiotherapy-induced reactivation of neurotrophic human herpes viruses: Overview and management. J. Clin. Virol. 2018, 98, 18–27. [Google Scholar] [CrossRef]
- Liu, K.X.; Ioakeim-Ioannidou, M.; Susko, M.S.; Rao, A.D.; Yeap, B.Y.; Snijders, A.M.; Ladra, M.M.; Vogel, J.; Zaslowe-Dude, C.; Marcus, K.J.; et al. A Multi-institutional Comparative Analysis of Proton and Photon Therapy-Induced Hematologic Toxicity in Patients With Medulloblastoma. Int. J. Radiat. Oncol. Biol. Phys. 2021, 109, 726–735. [Google Scholar] [CrossRef]
- Jefferies, S.; Rajah, B.; Ashley, S.; Traish, D.; Brada, M. Haematological toxicity of cranio-spinal irradiation. Radiother. Oncol. 1998, 48, 23–27. [Google Scholar] [CrossRef]
- Chang, E.L.; Allen, P.; Wu, C.; Ater, J.; Kuttesch, J.; Maor, M.H. Acute toxicity and treatment interruption related to electron and photon craniospinal irradiation in pediatric patients treated at the University of Texas M. D. Anderson Cancer Center. Int. J. Radiat. Oncol. Biol. Phys. 2002, 52, 1008–1016. [Google Scholar] [CrossRef]
- Wong, K.K.; Ragab, O.; Tran, H.N.; Pham, A.; All, S.; Waxer, J.; Olch, A.J. Acute toxicity of craniospinal irradiation with volumetric-modulated arc therapy in children with solid tumors. Pediatr. Blood Cancer 2018, 65, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.P.; Barney, C.L.; Grosshans, D.R.; McAleer, M.F.; De Groot, J.F.; Puduvalli, V.K.; Tucker, S.L.; Crawford, C.N.; Khan, M.; Khatua, S.; et al. Proton beam craniospinal irradiation reduces acute toxicity for adults with medulloblastoma. Int. J. Radiat. Oncol. Biol. Phys. 2013, 86, 277–284. [Google Scholar] [CrossRef] [Green Version]
- Bernier, V.; Klein, O. Late effects of craniospinal irradiation for medulloblastomas in paediatric patients. Neurochirurgie 2021, 67, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Chemaitilly, W.; Sklar, C.A. Childhood Cancer Treatments and Associated Endocrine Late Effects: A Concise Guide for the Pediatric Endocrinologist. Horm. Res. Paediatr. 2019, 91, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Vatner, R.E.; Niemierko, A.; Misra, M.; Weyman, E.A.; Goebel, C.P.; Ebb, D.H.; Jones, R.M.; Huang, M.S.; Mahajan, A.; Grosshans, D.R.; et al. Endocrine Deficiency As a Function of Radiation Dose to the Hypothalamus and Pituitary in Pediatric and Young Adult Patients With Brain Tumors. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2018, 36, 2854–2862. [Google Scholar] [CrossRef] [PubMed]
- Paulino, A.C.; Lobo, M.; Teh, B.S.; Okcu, M.F.; South, M.; Butler, E.B.; Su, J.; Chintagumpala, M. Ototoxicity after intensity-modulated radiation therapy and cisplatin-based chemotherapy in children with medulloblastoma. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 1445–1450. [Google Scholar] [CrossRef] [PubMed]
- Hua, C.; Bass, J.K.; Khan, R.; Kun, L.E.; Merchant, T.E. Hearing loss after radiotherapy for pediatric brain tumors: Effect of cochlear dose. Int. J. Radiat. Oncol. Biol. Phys. 2008, 72, 892–899. [Google Scholar] [CrossRef]
- Di Giannatale, A.; Morana, G.; Rossi, A.; Cama, A.; Bertoluzzo, L.; Barra, S.; Nozza, P.; Milanaccio, C.; Consales, A.; Garrè, M.L.; et al. Natural history of cavernous malformations in children with brain tumors treated with radiotherapy and chemotherapy. J. Neurooncol. 2014, 117, 311–320. [Google Scholar] [CrossRef]
- Passos, J.; Nzwalo, H.; Valente, M.; Marques, J.; Azevedo, A.; Netto, E.; Mota, A.; Borges, A.; Nunes, S.; Salgado, D. Microbleeds and cavernomas after radiotherapy for paediatric primary brain tumours. J. Neurol. Sci. 2017, 372, 413–416. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Preston, R.; Thomas, S.M.; Brackenridge, A.; Carroll, P.V. Cerebral cavernoma: An emerging long-term consequence of external beam radiation in childhood. Clin. Endocrinol. (Oxf.) 2010, 73, 555–560. [Google Scholar] [CrossRef]
- Patet, G.; Bartoli, A.; Meling, T.R. Natural history and treatment options of radiation-induced brain cavernomas: A systematic review. Neurosurg. Rev. 2021. [Google Scholar] [CrossRef]
- Neu, M.A.; Tanyildizi, Y.; Wingerter, A.; Henninger, N.; El Malki, K.; Alt, F.; Doerr, B.; Schmidberger, H.; Stockinger, M.; Wagner, W.; et al. Susceptibility-weighted magnetic resonance imaging of cerebrovascular sequelae after radiotherapy for pediatric brain tumors. Radiother. Oncol. 2018, 127, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Roongpiboonsopit, D.; Kuijf, H.J.; Charidimou, A.; Xiong, L.; Vashkevich, A.; Martinez-Ramirez, S.; Shih, H.A.; Gill, C.M.; Viswanathan, A.; Dietrich, J. Evolution of cerebral microbleeds after cranial irradiation in medulloblastoma patients. Neurology 2017, 88, 789–796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanyildizi, Y.; Keweloh, S.; Neu, M.A.; Russo, A.; Wingerter, A.; Weyer-Elberich, V.; Stockinger, M.; Schmidberger, H.; Brockmann, M.A.; Faber, J. Radiation-induced vascular changes in the intracranial irradiation field in medulloblastoma survivors: An MRI study. Radiother. Oncol. 2019, 136, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Kralik, S.F.; Mereniuk, T.R.; Grignon, L.; Shih, C.S.; Ho, C.Y.; Finke, W.; Coleman, P.W.; Watson, G.A.; Buchsbaum, J.C. Radiation-Induced Cerebral Microbleeds in Pediatric Patients With Brain Tumors Treated With Proton Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 1465–1471. [Google Scholar] [CrossRef] [Green Version]
- Passos, J.; Nzwalo, H.; Marques, J.; Azevedo, A.; Netto, E.; Nunes, S.; Salgado, D. Late cerebrovascular complications after radiotherapy for childhood primary central nervous system tumors. Pediatr. Neurol. 2015, 53, 211–215. [Google Scholar] [CrossRef]
- Roddy, E.; Sear, K.; Felton, E.; Tamrazi, B.; Gauvain, K.; Torkildson, J.; Del Buono, B.; Samuel, D.; Haas-Kogan, D.A.; Chen, J.; et al. Presence of cerebral microbleeds is associated with worse executive function in pediatric brain tumor survivors. Neuro-Oncology 2016, 18, 1548–1558. [Google Scholar] [CrossRef]
- Jakacki, R.I.; Goldwein, J.W.; Larsen, R.L.; Barber, G.; Silber, J.H. Cardiac dysfunction following spinal irradiation during childhood. J. Clin. Oncol. 1993, 11, 1033–1038. [Google Scholar] [CrossRef]
- Huang, T.T.; Chen, Y.; Dietz, A.C.; Yasui, Y.; Donaldson, S.S.; Stokes, D.C.; Stovall, M.; Leisenring, W.M.; Sklar, C.A.; Diller, L.R.; et al. Pulmonary outcomes in survivors of childhood central nervous system malignancies: A report from the childhood cancer survivor study. Pediatr. Blood Cancer 2014, 61, 319–325. [Google Scholar] [CrossRef] [Green Version]
- Endicott, T.J.; Fisher, B.J.; Wong, E.; Paterson, N.A.M.; Gaspar, L.E.; Bauman, M. Pulmonary sequelae after electron spinal irradiation. In Proceedings of the Radiotherapy and Oncology. Radiother. Oncol. 2001, 60, 267–272. [Google Scholar] [CrossRef]
- Jakacki, R.I.; Schramm, C.M.; Donahue, B.R.; Haas, F.; Allen, J.C. Restrictive lung disease following treatment for malignant brain tumors: A potential late effect of craniospinal irradiation. J. Clin. Oncol. 1995, 13, 1478–1485. [Google Scholar] [CrossRef] [PubMed]
- King, M.T.; Modlin, L.; Million, L.; Donaldson, S.S.; Gibbs, I.C.; Choi, C.Y.H.; Soltys, S.G. The Parotid Gland is an Underrecognized Organ at Risk for Craniospinal Irradiation. Technol. Cancer Res. Treat. 2016, 15, 472–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christopherson, K.M.; Rotondo, R.L.; Bradley, J.A.; Pincus, D.W.; Wynn, T.T.; Fort, J.A.; Morris, C.G.; Mendenhall, N.P.; Marcus, R.B.; Indelicato, D.J. Late toxicity following craniospinal radiation for early-stage medulloblastoma. Acta Oncol. (Madr). 2014, 53, 471–480. [Google Scholar] [CrossRef] [Green Version]
- Murphy, E.S.; Merchant, T.E.; Wu, S.; Xiong, X.; Lukose, R.; Wright, K.D.; Qaddoumi, I.; Armstrong, G.T.; Broniscer, A.; Gajjar, A. Necrosis after craniospinal irradiation: Results from a prospective series of children with central nervous system embryonal tumors. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plimpton, S.R.; Stence, N.; Hemenway, M.; Hankinson, T.C.; Foreman, N.; Liu, A.K. Cerebral radiation necrosis in pediatric patients. Pediatr. Hematol. Oncol. 2015, 32, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Indelicato, D.J.; Rotondo, R.L.; Uezono, H.; Sandler, E.S.; Aldana, P.R.; Ranalli, N.J.; Beier, A.D.; Morris, C.G.; Bradley, J.A. Outcomes Following Proton Therapy for Pediatric Low-Grade Glioma. Int. J. Radiat. Oncol. Biol. Phys. 2019, 104, 149–156. [Google Scholar] [CrossRef] [Green Version]
- Indelicato, D.J.; Bradley, J.A.; Sandler, E.S.; Aldana, P.R.; Sapp, A.; Gains, J.E.; Crellin, A.; Rotondo, R.L. Clinical outcomes following proton therapy for children with central nervous system tumors referred overseas. Pediatr. Blood Cancer 2017, 64, e26654. [Google Scholar] [CrossRef]
- Goitein, M. Trials and tribulations in charged particle radiotherapy. Radiother. Oncol. 2010, 95, 23–31. [Google Scholar] [CrossRef]
- Sheehan, M.; Timlin, C.; Peach, K.; Binik, A.; Puthenparampil, W.; Lodge, M.; Kehoe, S.; Brada, M.; Burnet, N.; Clarke, S.; et al. Position statement on ethics, equipoise and research on charged particle radiation therapy. J. Med. Ethics 2014, 40, 572–575. [Google Scholar] [CrossRef]
- Gornall, J. Proton beam therapy: More than a leap of faith? BMJ 2012, 345, e4917. [Google Scholar] [CrossRef]
- Gajjar, A.; Robinson, G.W.; Smith, K.S.; Lin, T.; Merchant, T.E.; Chintagumpala, M.; Mahajan, A.; Su, J.; Bouffet, E.; Bartels, U.; et al. Outcomes by Clinical and Molecular Features in Children With Medulloblastoma Treated With Risk-Adapted Therapy: Results of an International Phase III Trial (SJMB03). J. Clin. Oncol. 2021, 39, 822–835. [Google Scholar] [CrossRef] [PubMed]

| Patient Characteristics and Radiation | Total Cohort (n = 43) | HR Group (n = 26) | SR Group (n = 14) | Ex-Infant (n = 3) | ||||
|---|---|---|---|---|---|---|---|---|
| Sex | No. | % | No. | % | No. | % | No. | % |
| Male | 23 | 53.5% | 18 | 69.2% | 4 | 26.7% | 1 | 33.3% |
| Female | 20 | 46.5% | 8 | 30.8% | 10 | 73.3% | 2 | 66.7% |
| Histological subtype | ||||||||
| Classic | 31 | 72.1% | 18 | 69.2% | 12 | 85.7% | 1 | 33.3% |
| Desmoplastic | 3 | 7.0% | 1 | 3.8% | 1 | 7.1% | 1 | 33.3% |
| Large cell/Anaplastic | 7 | 16.3% | 7 | 27.0% | - | - | - | - |
| Extensive nodularity | 1 | 2.3% | - | - | - | - | 1 | 33.3% |
| NOS | 1 | 2.3% | - | - | 1 | 7.1% | - | - |
| Molecular subgroup | ||||||||
| Non-WNT/Non-SHH | 29 | 67.4% | 21 | 80.8% | 7 | 50.0% | 1 | 33.3% |
| SHH | 8 | 18.6% | 4 | 15.4% | 2 | 14.3% | 2 | 66.7% |
| WNT | 3 | 7.0% | - | - | 3 | 21.4% | - | - |
| Unavailable | 3 | 7.0% | 1 | 3.8% | 2 | 14.3% | - | - |
| MYC amplification | 2 | 4.7% | 2 | 7.7% | - | - | - | - |
| Chang Stage at PT | ||||||||
| M0 | 28 | 65.1% | 12 | 46.2% | 14 | 100.0% | 2 | 66.7% |
| M1 | 2 | 4.7% | 2 | 7.7% | - | - | - | - |
| M2 | 2 | 4.7% | 2 | 7.7% | - | - | - | - |
| M3 | 11 | 25.6% | 10 | 38.4% | - | - | 1 | 33.3% |
| M4 | - | - | - | - | - | - | - | - |
| Median (IQR) | Min–Max | Median (IQR) | Min–Max | Median (IQR) | Min–Max | Median (IQR) | Min–Max | |
| CSI dose, GyRBE | 36 (12.6) | 23.4–39.6 | 36.0 (0.0) | 24.0 *–39.6 | 23.4 (0.0) | 23.4–25.2 | 36 (6.3) | 23.4–36.0 |
| Total TB dose, GyRBE | 54.0 (0.0) | 54.0–55.8 | 54.0 (0.0) | 54.0–55.8 | 54.0 (0.0) | 54.0–55.8 | 54.0 (0.0) | 54.0–54.0 |
| Boost on mets, GyRBE | 12.6 (9.0) | 9.0–18.0 | 14.4 (9.0) | 9.0–18.0 | - | - | 9.0 (0.0) | 9.0–9.0 |
| Age at diagnosis, years | 8.7 (6.6) | 1.8–18.6 | 10.1 (7.2) | 2.5–18.6 | 6.6 (4.0) | 4.0–17.8 | 1.9 (0.1) | 1.8–2.1 |
| Age at PT start, years | 8.9 (6.8) | 3.0–19.0 | 10.3 (7.3) | 3.0–19.0 | 6.7 (3.9) | 4.1–18.0 | 3.8 (0.1) | 3.6–3.8 |
| Treatment Characteristics, Response and Follow-Up | Total Sample (n = 43) | HR Group (n = 26) | SR Group (n = 14) | Ex-Infant (n = 3) | ||||
|---|---|---|---|---|---|---|---|---|
| Extent of resection | ||||||||
| Partial | 20 (46.5%) | 15 (57.7%) | 2 (14.3%) | 3 (100.0%) | ||||
| Near-total | 2 (4.7%) | - | 2 (14.3%) | - | ||||
| Gross-total | 21 (48.8%) | 11 (42.3%) | 10 (71.4%) | - | ||||
| HD-CHT with aHSCT prior to PT | 14 (32.6%) | 11 (42.3%) | - | 3 (100.0%) | ||||
| Concomitant CHT to PT | 18 (41.9%) | 13 (50.0%) | 5 (35.7%) | - | ||||
| Treatment response | ||||||||
| CR | 35 (81.4%) | 20 (76.9%) | 12 (85.7%) | 3 | 100.0% | |||
| SD | 3 (7.0%) | 3 (11.5%) | - | - | - | |||
| PD | 5 (11.6%) | 3 (11.5%) | 2 (14.2%) | - | - | |||
| Survival at latest FU | ||||||||
| Alive | 41 (95.3%) | 24 (92.3%) | 14 (100.0%) | 3 | 100.0% | |||
| Dead | 2 (4.7%) | 2 (7.7%) | - | - | - | |||
| Median (IQR) | Min–Max | Median (IQR) | Min–Max | Median (IQR) | Min–Max | Median (IQR) | Min–Max | |
| FU duration, months | 26.0 (30.5) | 2.0–67.0 | 20.0 (31.3) | 5.0–67.0 | 28.5 (22.8) | 2.0–66.0 | 24.0 (21.5) | 3.0–46.0 |
| Type of Toxicity | Total Sample (n = 43) | High-Dose CSI (n = 27) | Standard-Dose CSI (n = 16) | |||
|---|---|---|---|---|---|---|
| G1–G2 | G3 | G1–G2 | G3 | G1–G2 | G3 | |
| Radiation dermatitis | 28 (65.1%) | 1 (2.3%) | 17 (63.0%) | 1 (3.7%) | 11 (68.8%) | - |
| Pharyngeal mucositis | 22 (51.2%) | 2 (4.7%) | 15 (55.6%) | 1 (3.7%) | 7 (43.8%) | 1 (6.3%) |
| Nausea/Vomiting | 18 (41.9%) | 1 (2.3%) | 10 (37.0%) | - | 8 (50.0%) | 1 (6.3%) |
| Alopecia | 17 * (94.4%) | - | 3 (100%) | - | 14 (93.3%) | - |
| Anorexia | 16 (37.2%) | 1 (2.3%) | 8 (29.6%) | - | 8 (50.0%) | 1 (6.3%) |
| Fatigue | 15 (34.9%) | - | 8 (29.6%) | - | 7 (43.8%) | - |
| Herpes Zoster | 7 (16.3%) | - | 4 (14.8%) | 3 (18.8%) | ||
| Headache | 5 (11.6%) | 1 (2.3%) | 2 (7.4%) | 1 (3.7%) | 3 (18.8%) | - |
| Upper airway infection | 3 (7.0%) | - | 2 (7.4%) | - | 1 (6.3%) | - |
| Insomnia | 2 (4.7%) | - | 1 (3.7%) | - | 1 (6.3%) | - |
| Fever | 3 (7.0%) | - | 1 (3.7%) | - | 2 (12.5%) | - |
| Cough | 1 (2.3%) | - | - | - | 1 (6.3%) | - |
| Diarrhea | 1 (2.3%) | - | - | - | 1 (6.3%) | - |
| Myalgia | 1 (2.3%) | - | - | - | 1 (6.3%) | - |
| PRES | - | 1 (2.3%) | - | 1 (3.7%) | - | - |
| Cavernoma | 1 (2.3%) | - | 1 (3.7%) | - | - | - |
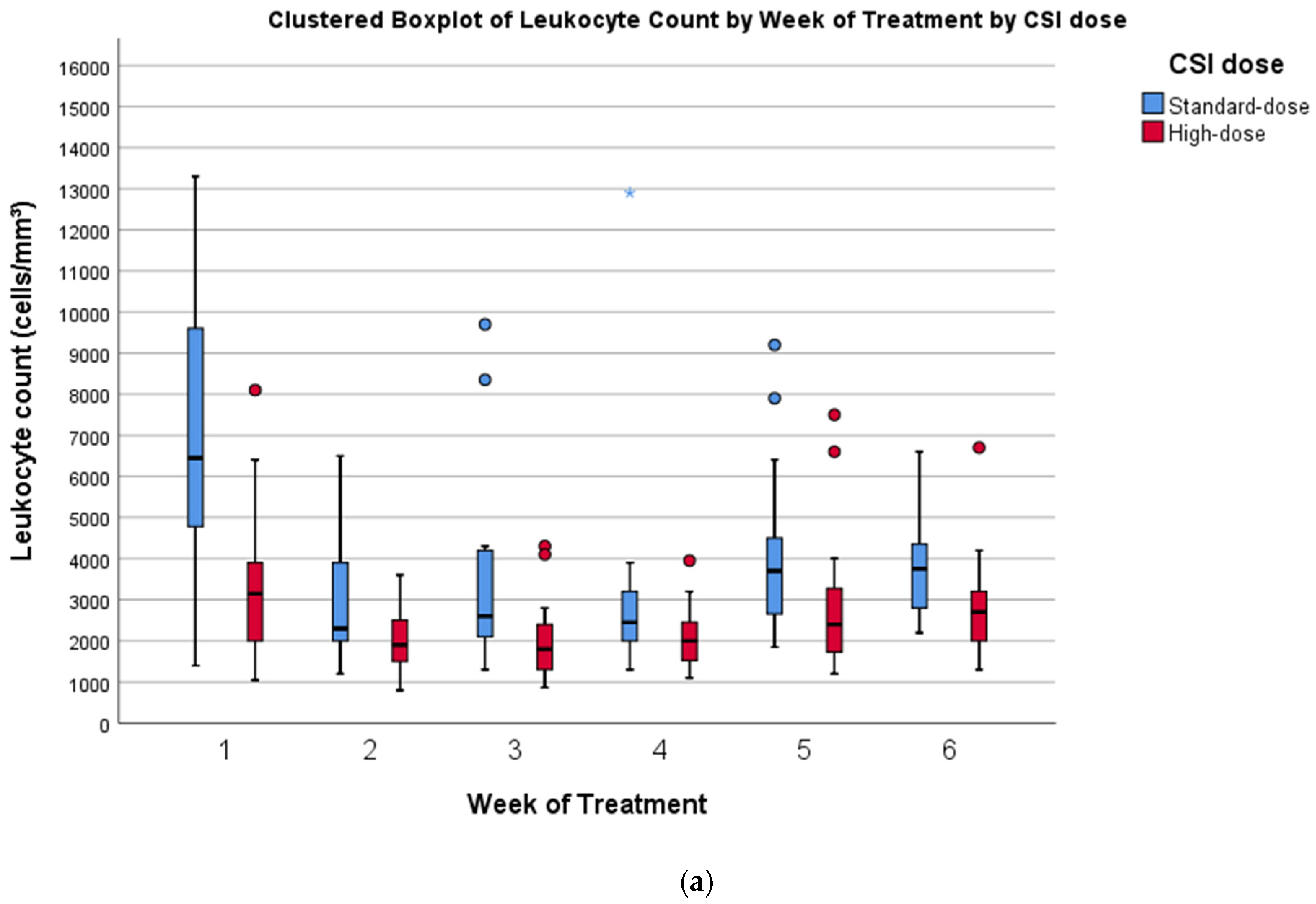
| Comparison of High-Dose vs. Standard-Dose CSI | High-Dose CSI (n = 27) | Standard-Dose CSI (n = 16) | Difference (SE) | 95% CI | p |
|---|---|---|---|---|---|
| Leukocyte Count (Cells/mm³) | Mean (SD) | Mean (SD) | |||
| Week 1 | 3339 (1587) | 6679 (3393) | 3340 (932) | 1382–5298 | <0.00 |
| Week 2 | 1997 (698) | 3118 (1655) | 1121 (463) | 140–2103 | 0.03 |
| Week 3 | 1972 (879) | 3463 (2411) | 1492 (648) | 119–2865 | 0.04 |
| Week 4 | 2117 (738) | 3107 (2804) | 990 (738) | −584–2564 | 0.20 |
| Week 5 | 2814 (1484) | 4022 (2157) | 1208 (561) | 74–2342 | 0.04 |
| Week 6 | 3468 (3641) | 3856 (1548) | 388 (831) | −1299–2074 | 0.64 |
| Hemoglobin value (g/dL) | |||||
| Week 1 | 11.0 (1.0) | 11.6 (1.6) | 0.5 (0.5) | −0.4–1.5 | 0.25 |
| Week 2 | 11.1 (1.2) | 11.3 (1.6) | 0.2 (0.5) | −0.7–1.1 | 0.63 |
| Week 3 | 11.0 (1.3) | 11.2 (1.6) | 0.2 (0.5) | −0.7–1.2 | 0.65 |
| Week 4 | 11.1 (1.0) | 11.2 (1.6) | 0.1 (0.5) | −0.9–1.1 | 0.87 |
| Week 5 | 11.2 (1.0) | 11.2 (1.7) | 0.1 (0.5) | −0.9–1.0 | 0.92 |
| Week 6 | 11.2 (0.7) | 11.2 (1.5) | -0.1 (0.4) | −0.9–0.8 | 0.92 |
| Platelet count (cells/mm³) | |||||
| Week 1 | 21,5240 (69,388) | 311,300 (103,713) | 96,060 (27,333) | 40,728–151,392 | <0.00 |
| Week 2 | 135,590 (41,774) | 203,750 (55,424) | 68,160 (15,545) | 36,691–99,629 | <0.00 |
| Week 3 | 137,353 (50,476) | 192,800 (92,686) | 55,447 (25,974) | 1083–109,810 | 0.04 |
| Week 4 | 141,436 (54,175) | 218,267 (96,355) | 76,831 (27,053) | 20,209–133,452 | 0.01 |
| Week 5 | 156,385 (54,281) | 278,750 (103,357) | 122,365 (27,946) | 64,070–180,660 | <0.00 |
| Week 6 | 161,936 (56,438) | 276,767 (71,235) | 114,831 (20,153) | 74,067–155,595 | <0.00 |
| Therapy | Cases | % |
|---|---|---|
| Any non-preexisting HRT | 6 | 14.0 |
| Monotherapy | 4 | 9.3 |
| Multi-drug HRT | 2 | 4.7 |
| Specific hormones: | ||
| Thyroxine | 5 | 11.6 |
| Hydrocortisone | 3 | 7.0 |
| Desmopressin | 1 | 2.3 |
| Testosterone | 1 | 2.3 |
| Median | Min–Max | |
| Hypothalamic/Pituitary dose, GyRBE | 41.24 | 36.0–55.0 |
| Latency to HRT | 9.5 months | 3.0–25.0 months |
| Patient | CSI/TB dose (GyRBE) | Grade | Laterality | Cochlear Dmean (GyRBE) | Previous Chemotherapy | |
|---|---|---|---|---|---|---|
| Right | Left | |||||
| 1 | 23.4/30.6 | G1 | Bilateral | 35 | 28 | No |
| 2 | 36.0/18.0 | G2 | Bilateral | 37 | 37 | Yes |
| 3 | 36.0/18.0 | G2 | Bilateral | 37 | 42 | Yes |
| 4 | 23.4/30.6 | G2 | Bilateral | 23 | 26 | No |
| 5 | 36.0/18.0 | G3 | Right | 53 | 36 | Yes |
| 6 | 23.4/30.6 | G3 | Bilateral | 32 | 31 | No |
| 7 | 23.4/30.6 | G3 | Right | 35 | 42 | Yes |
| Toxicity | High-Dose CSI (n = 27) | Standard-Dose CSI (n = 16) | ||
|---|---|---|---|---|
| No. of Cases (%) | Grade | No. of Cases (%) | Grade | |
| Cavernoma | 8 (29.6%) | G1 | 1 (6.3%) | G1 |
| 1 (3.7%) | G2 | - | - | |
| Intracranial bleeding | 1 (3.7%) | G2 | - | - |
| Loss of visual acuity | - | - | 2 (12.5%) | G1 |
| Osteoporosis | 1 (3.7%) | G2 | ||
| CMV Encephalitis | 1 (3.7%) | G4 | - | - |
| Stroke | 1 (3.7%) | G1 | - | - |
| RBC Transfusion | 1 (3.7%) | G3 | - | - |
| VII CN Paralysis | - | - | 1 (6.3%) | G1 |
| Chronic headache | - | - | 1 (6.3%) | G1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruggi, A.; Melchionda, F.; Sardi, I.; Pavone, R.; Meneghello, L.; Kitanovski, L.; Zaletel, L.Z.; Farace, P.; Zucchelli, M.; Scagnet, M.; et al. Toxicity and Clinical Results after Proton Therapy for Pediatric Medulloblastoma: A Multi-Centric Retrospective Study. Cancers 2022, 14, 2747. https://doi.org/10.3390/cancers14112747
Ruggi A, Melchionda F, Sardi I, Pavone R, Meneghello L, Kitanovski L, Zaletel LZ, Farace P, Zucchelli M, Scagnet M, et al. Toxicity and Clinical Results after Proton Therapy for Pediatric Medulloblastoma: A Multi-Centric Retrospective Study. Cancers. 2022; 14(11):2747. https://doi.org/10.3390/cancers14112747
Chicago/Turabian StyleRuggi, Alessandro, Fraia Melchionda, Iacopo Sardi, Rossana Pavone, Linda Meneghello, Lidija Kitanovski, Lorna Zadravec Zaletel, Paolo Farace, Mino Zucchelli, Mirko Scagnet, and et al. 2022. "Toxicity and Clinical Results after Proton Therapy for Pediatric Medulloblastoma: A Multi-Centric Retrospective Study" Cancers 14, no. 11: 2747. https://doi.org/10.3390/cancers14112747






