Simple Summary
New assays are developed regularly to improve health care for patients. It is important to ensure that assays are performed correctly. Therefore, it is advised to participate in training and proficiency (competence assessment) programs. Tumor infiltrating lymphocytes (TILs) might improve the estimates of response to therapy and prognosis. Herewith, we propose a new training and proficiency program in which each pathologist can train and test themselves regarding TILs (and PDL1) scoring.
Increasing data suggests that an intact immune system is required for improved outcomes in patients with Human Epidermal Growth Factor Receptor 2 (HER2+) and Triple Negative Breast Cancer (TNBC). Tumor Infiltrating Lymphocytes (TILs), an indicator of anti-tumor immune response in the host, are an established prognostic factor [1], and are predictive for response to immunotherapy in TNBC [2]. An accurate and reproducible assessment of TILs is therefore necessary for daily and clinical trial practices.
TILs are currently measured by visual evaluation of hematoxylin-eosin stained (H&E) slides. In addition, there are ongoing efforts for assessment of TILs by machine learning and artificial intelligence-methods [3]. The International Immuno-Oncology Biomarker Working Group (also called the TILs-WG, https://www.tilsinbreastcancer.org/ (accessed on 15 July 2021)) has developed an easy and accessible method to assess TILs using H&E slides [4]. The TILs-WG has also developed a freely available self-training tool, including audio-visual materials (https://www.youtube.com/watch?v=aPa-pXIBBlU (accessed on 7 January 2021)) and has completed several inter-laboratory comparison studies, i.e., ring studies that showed this method to be reproducible [5]. These TIL training materials are freely available on the WG’s website.
The assessment of TILs is increasingly being introduced in daily practices worldwide [4,6]. Hence, high-quality training and continuous competence assessment is crucial for sustained implementation of this biomarker in thousands of pathology laboratories worldwide.
Standardized and reproducible assay results are key to successful biomarker implementation in clinical practice. Inaccurate diagnostic results can have a potential negative impact in patient clinical management. An increasing number of governmental and regulatory bodies in many countries recognize the importance of requiring a minimum level of quality management in clinical laboratories [7]. New diagnostic methods and increasing awareness about assay errors and their consequences emphasize the great importance of quality in health care settings [8]. A laboratory quality management system (QMS), such as compliance with ISO15189 [9] or through the College of American Pathologists (CAP) is a step forward to ensure quality of testing [10].
This ISO-standard, for example, states that “interlaboratory comparison program(s) chosen by the laboratory shall, as far as possible, provide clinically relevant challenges that mimic patient samples […]” [9]. As recognized by the ISO-standard, participation in external quality assessment (EQA) schemes, also called proficiency testing, allows monitoring of laboratory performance, comparisons between laboratories and provides feedback to ensure consistent delivery of services over time. When partaking in EQA schemes organized by, for example, NORDIQC, UK NEQAS, CBQA, etc., participants receive a set of samples they must analyze according to their routine protocols. All participants receive samples from the same source, so results can be compared. After submission of results, participants receive feedback on their performance, as well as information about their performance compared to other participants. This practice arms laboratories/pathologists with knowledge to increase their performance and increases confidence to patients and clinicians in the results provided by the laboratory.
EQA schemes are an essential tool when implementing new types of assessments of biomarkers [11], and participation in EQAs can result in increased performance and better standardization across different laboratories worldwide [12,13]. However, in many parts of the world, certainly in low-to-middle-income resource countries, implementing, for example, an ISO15189-standard in pathology laboratories is not always feasible. Nevertheless, even in these settings, participation in EQAs can be helpful, certainly when the “assay” is the pathologist evaluating a morphological biomarker on an H&E-slide, as is the case with TILs.
A team of experts from the TILs-WG, in collaboration with the Biomedical Quality Assurance Research Unit of the University of Leuven, have set up a worldwide, freely available training- and EQA-scheme using an new developed online platform, to train and investigate the competence of pathologists in evaluating TILs as a new morphologic biomarker in TNBC, and, as PD-L1-assays in breast cancer mostly stain TILs, also PD-L1 (Figure 1; TILs and PD-L1 Training Course: International TILS Working Group (tilsinbreastcancer.org, accessed on 15 July 2021)). The overall purpose of this study is to support pathologists implementing TILs scoring in clinical practice as a prognostic biomarker for TILs, and as a predictive combined TILs/PD-L1-biomarker, as both are associated with improved outcomes in TNBC. For example, in IMpassion130, a phase III clinical trial that assessed nab-paclitaxel with Atezolizumab or placebo for patients with newly diagnosed inoperable and/or metastatic TNBC, the Hazard Ratio (HR) of benefit to immunotherapy is similar for PD-L1 IC+/any TILs (0.65 for PFS and 0.71 for OS), compared to the HR for any PD-L1 IC+ and TILs > 10% (0.64 for PFS and 0.75 for OS), suggesting that both are complementary metrics [14].
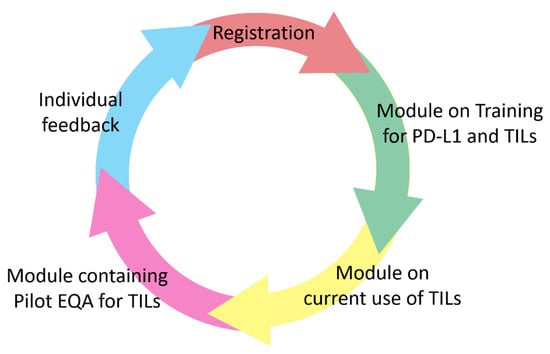
Figure 1.
After registration, through www.tilsinbreastcancer.org (accessed on 15 July 2021), a training module will be available for PD-L1 and TILs. Next, a pilot EQA module will be available to assess the current practices and competences of pathologists to score TILs. Finally, individual feedback will be offered to each participant separately.
TILs are thus increasingly considered to be complementary to PD-L1. Since the immune cell staining is an important component of PD-L1 assays in breast cancer immunotherapy trials, a combined assessment of TILs and PD-L1 might prove to be a successful complementary approach to use in daily practice, the possibility of which will be studied in the EQA scheme mentioned above. In some laboratories, mostly in low-to-middle-income countries, TILs are pre-screened as a cost saving measure, since, if no immune cells are present, any PD-L1-assay assessing the expression of PD-L1 on immune cells will likely be negative [15]. Specifically, for a given TIL percentage, we question how likely is it that the PD-L1 IC score is above the clinically relevant cut-off point.
The EQA scheme introduced here will be organized on an individual level, rather than on a laboratory level, so every pathologist that wants to train themselves can participate, with no previous experience necessary. The scheme will contain multiple modules (Figure 1). After registration on the digital platform (TILS Login Page (agoko.be, accessed on 19 October 2021)), a training module for TILs estimation will be available (e.g., theoretical explanations, comparative exercises, videos, etc.). Another module will probe the pathologists experience and levels of training on TIL estimation as well as the current use of PD-L1 and TILs in clinical practice. A subsequent module will contain an EQA where we ask participants to score the TILs and PD-L1 on digital slides. Digital slides, made in a single center, will be made available. The PD-L1 stained samples were stained with multiple assays (SP142, SP263 and 22C3), all in the same laboratory and according to FDA-approved assay requirements.
After submission of the results, an expert team will analyze the data. If variables are found that influence scoring, there will be a feedback mechanism to the participants. Feedback on the organization and quality of the EQA scheme itself will be collected to learn and improve the platform for future use.
A comparison will be made of PD-L1 scores in different tumor compartments (immune cells and tumor cells, and the combination hereof) to investigate the complementarity of TILs to PD-L1 testing, wherein each variable will be assessed in a continuous manner, including but not limited to the assay-specific scoring-algorithm as developed by the vendor. This will allow us to analyze, in an unbiased manner, the analytical performance of pathologists on TILs and PD-L1, on both tumor cells and immune cells, as well as the variance between pathologists.
Participation in this EQA by pathologists worldwide should be encouraged, as only unbiased EQA-schemes in the scientific community can identify problems and obstacles, and mitigate these issues in further training-modules and subsequent competence assessments. For the sake of our patients, this partnership between academia and industry to perform unbiased EQA and learn from it, is key to the sustained implementation of a biomarker in daily practices worldwide. We need to learn from previous experiences on how biomarkers were introduced and improve the process by which biomarkers become widely adopted in daily practice. This project aims to develop a framework that is in the best interest of our patients [12] and this framework, as proposed by the most important pathology organizations worldwide, should be used for all future biomarker assessments before incorporation in daily practices can be considered.
Author Contributions
Conceptualization, I.N. and R.S.; validation, I.N., R.S., N.L. and E.D., writing—original draft preparation, I.N.; writing—review and editing, N.L., D.P., B.A., C.D., S.M., H.H., K.P.S., S.E., D.Z., R.M., J.B., G.F., J.H., C.H.M.v.D., D.C., E.D. and R.S.; visualization, I.N.; supervision, R.S. and E.D.; funding acquisition, R.S., S.E., D.Z. and R.M. All authors have read and agreed to the published version of the manuscript.
Funding
The training and EQC-platform have been supported by the Breast Cancer Research Foundation and Bristol-Myers Squib (CW3199227).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Denkert, C.; Von Minckwitz, G.; Darb-Esfahani, S.; Lederer, B.; Heppner, B.I.; Weber, K.E.; Budczies, J.; Huober, J.; Klauschen, F.; Furlanetto, J.; et al. Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: A pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol. 2018, 19, 40–50. [Google Scholar] [CrossRef]
- Loi, S.; Adams, S.; Schmid, P.; Cortés, J.; Cescon, D.W.; Winer, E.P.; Toppmeyer, D.L.; Rugo, H.S.; De Laurentiis, M.; Nanda, R.; et al. Relationship between tumor infiltrating lymphocyte (TIL) levels and response to pembrolizumab (pembro) in metastatic triple-negative breast cancer (mTNBC): Results from KEYNOTE-086. Ann. Oncol. 2017, 28, v608. [Google Scholar] [CrossRef]
- Amgad, M.; Stovgaard, E.S.; Balslev, E.; Thagaard, J.; Chen, W.; Dudgeon, S.; Sharma, A.; Kerner, J.K.; Denkert, C.; Yuan, Y.; et al. Report on computational assessment of Tumor Infiltrating Lymphocytes from the International Immuno-Oncology Biomarker Working Group. npj Breast Cancer 2020, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Salgado, R.; Denkert, C.; Demaria, S.; Sirtaine, N.; Klauschen, F.; Pruneri, G.; Wienert, S.; Van den Eynden, G.; Baehner, F.L.; Penault-Llorca, F.; et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: Recommendations by an International TILs Working Group 2014. Ann. Oncol. 2015, 26, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Denkert, C.; Wienert, S.; Poterie, A.; Loibl, S.; Budczies, J.; Badve, S.; Bago-Horvath, Z.; Bane, A.; Bedri, S.; Brock, J.; et al. Standardized evaluation of tumor-infiltrating lymphocytes in breast cancer: Results of the ring studies of the international immuno-oncology biomarker working group. Mod. Pathol. 2016, 29, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.C.; Salgado, R.; Luen, S.J.; Savas, P.; Loi, S. Tumor-Infiltrating Lymphocyctes in Triple-Negative Breast Cancer: Update for 2020. Cancer J. 2021, 27, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Boursier, G.; Vukasovic, I.; Brguljan, P.M.; Lohmander, M.; Ghita, I.; Andreu, F.A.B.; Barrett, E.; Brugnoni, D.; Kroupis, C.; Sprongl, L.; et al. Accreditation process in European countries—An EFLM survey. Clin. Chem. Lab. Med. (CCLM) 2016, 54, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Berner, E.S. Diagnostic Error in Medicine: Implications for Clinical Laboratory Scientists. Ann. Clin. Lab. Sci. 2017, 47, 649–656. [Google Scholar] [PubMed]
- Standardization IOf ISO15189: 2012; Medical Laboratories—Requirements for Quality and Competence. The International Organization for Standardization: Geneva, Switzerland, 2012.
- Tack, V.; Schuuring, E.; Keppens, C.; Hart, N.; Pauwels, P.; Van Krieken, H.; Dequeker, E.M. Accreditation, setting and experience as indicators to assure quality in oncology biomarker testing laboratories. Br. J. Cancer 2018, 119, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Keppens, C.; De Smedt, J.; Mc Dequeker, E. Implementation of Novel Methods, Markers or Sample types in Molecular Pathology–Quality Assurance Aspects. Arch. Clin. Biomed. Res. 2020, 4, 302–324. [Google Scholar] [CrossRef]
- Salgado, R.; Bellizzi, A.M.; Rimm, D.; Bartlett, J.M.S.; Nielsen, T.; Holger, M.; Laenkholm, A.-V.; Quinn, C.; Cserni, G.; Cunha, I.W.; et al. How current assay approval policies are leading to unintended imprecision medicine. Lancet Oncol. 2020, 21, 1399–1401. [Google Scholar] [CrossRef]
- Hoda, R.S.; Brogi, E.; D’Alfonso, T.M.; Grabenstetter, A.; Giri, D.; Hanna, M.G.; Kuba, M.G.; Murray, D.M.P.; Vallejo, C.E.; Zhang, H.; et al. Interobserver Variation of PD-L1 SP142 Immunohistochemistry Interpretation in Breast Carcinoma: A Study of 79 Cases Using Whole Slide Imaging. Arch. Pathol. Lab. Med. 2021, 145, 1132–1137. [Google Scholar] [CrossRef] [PubMed]
- Emens, L.; Molinero, L.; Loi, S.; Rugo, H.S.; Schneeweiss, A.; Diéras, V.; Iwata, H.; Barrios, C.H.; Nechaeva, M.; Duc, A.N.; et al. Atezolizumab and nab-Paclitaxel in Advanced Triple-Negative Breast Cancer: Biomarker Evaluation of the IMpassion130 Study. J. Natl. Cancer Inst. 2021, 113, 1005–1016. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Ericsson, P.I.; Stovgaard, E.S.; Sua, L.F.; Reisenbichler, E.; Kos, Z.; Carter, J.M.; Michiels, S.; Le Quesne, J.; Nielsen, T.O.; Lænkholm, A.V.; et al. The path to a better biomarker: Application of a risk management framework for the implementation of PD-L1 and TILs as immuno-oncology biomarkers in breast cancer clinical trials and daily practice. J. Pathol. 2020, 250, 667–684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).