A Treatment Paradigm Shift: Targeted Radionuclide Therapies for Metastatic Castrate Resistant Prostate Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Bone Specific Radiotherapeutics
3. Beta Emitting PSMA Targeted Radiotherapeutics
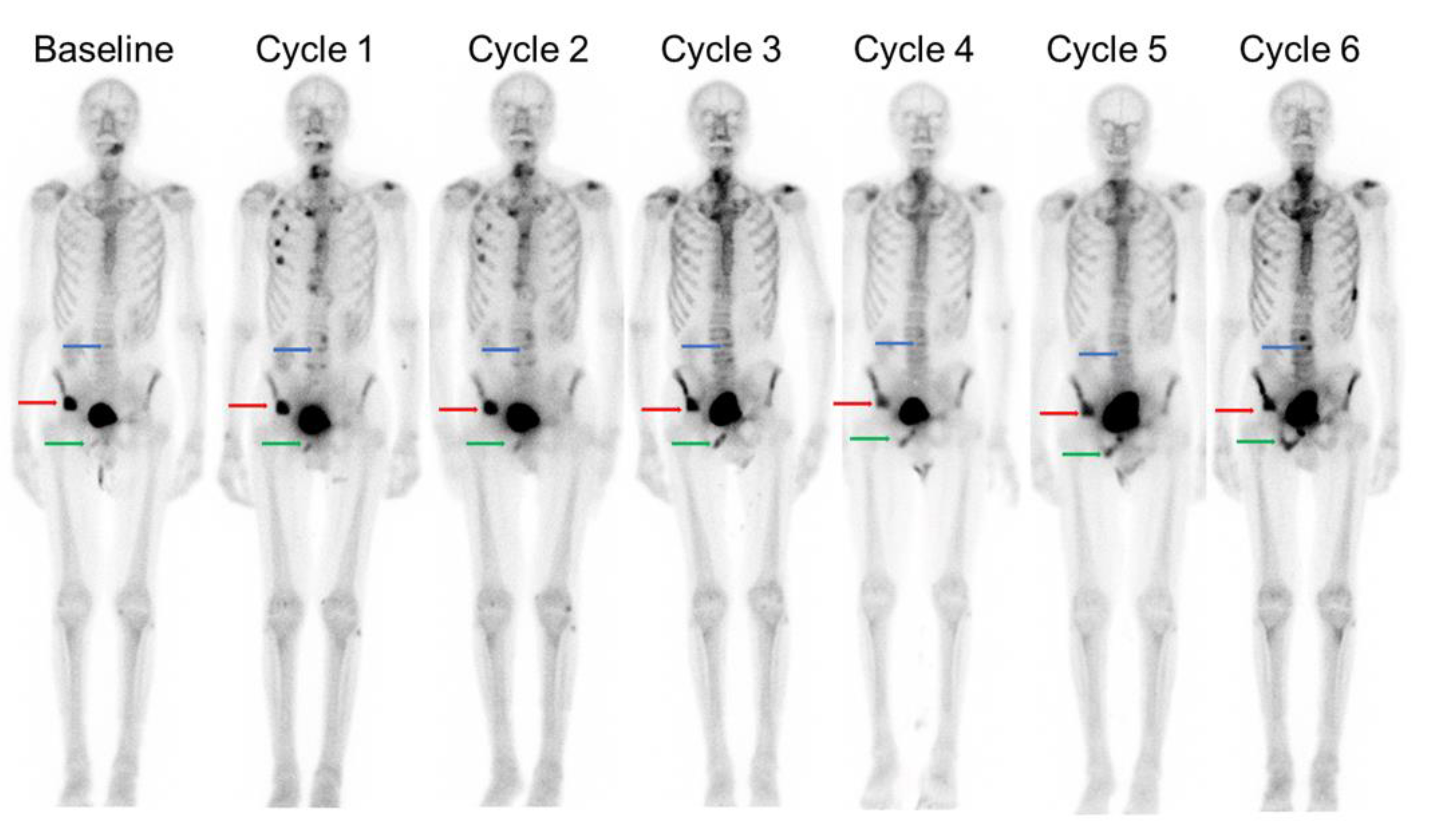
4. Dosimetry and Future Developments of PSMA Targeted Radiotherapeutics
5. Alpha Emitting PSMA Targeted Radiotherapeutics
6. Gastrin-Releasing Peptide Receptor (GRPR) Targeted Radiotherapeutics
7. Somatostatin Targeted Radiotherapeutics
8. Discussion and Clinician’s Perspective
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Mollica, V.; Rizzo, A.; Rosellini, M.; Marchetti, A.; Ricci, A.D.; Cimadamore, A.; Scarpelli, M.; Bonucci, C.; Andrini, E.; Errani, C.; et al. Bone Targeting Agents in Patients with Metastatic Prostate Cancer: State of the Art. Cancers 2021, 13, 546. [Google Scholar] [CrossRef] [PubMed]
- So, A.; Chin, J.; Fleshner, N.; Saad, F. Management of skeletal-related events in patients with advanced prostate cancer and bone metastases: Incorporating new agents into clinical practice. Can. Urol. Assoc. J. 2012, 6, 465–470. [Google Scholar] [CrossRef]
- Rizzo, A.; Mollica, V.; Cimadamore, A.; Santoni, M.; Scarpelli, M.; Giunchi, F.; Cheng, L.; Lopez-Beltran, A.; Fiorentino, M.; Montironi, R.; et al. Is There a Role for Immunotherapy in Prostate Cancer? Cells 2020, 9, 2051. [Google Scholar] [CrossRef]
- Thompson, I.M., Jr.; Goodman, P.J.; Tangen, C.M.; Parnes, H.L.; Minasian, L.M.; Godley, P.A.; Lucia, M.S.; Ford, L.G. Long-term survival of participants in the prostate cancer prevention trial. N. Engl. J. Med. 2013, 369, 603–610. [Google Scholar] [CrossRef]
- Parker, C.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O’Sullivan, J.M.; Fossa, S.D.; Chodacki, A.; Wiechno, P.; Logue, J.; Seke, M.; et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N. Engl. J. Med. 2013, 369, 213–223. [Google Scholar] [CrossRef]
- Smith, M.; Parker, C.; Saad, F.; Miller, K.; Tombal, B.; Ng, Q.S.; Boegemann, M.; Matveev, V.; Piulats, J.M.; Zucca, L.E.; et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. Oncol. 2019, 20, 408–419. [Google Scholar] [CrossRef]
- Agarwal, N.; Nussenzveig, R.; Hahn, A.W.; Hoffman, J.M.; Morton, K.; Gupta, S.; Batten, J.; Thorley, J.; Hawks, J.; Santos, V.S.; et al. Prospective Evaluation of Bone Metabolic Markers as Surrogate Markers of Response to Radium-223 Therapy in Metastatic Castration-resistant Prostate Cancer. Clin. Cancer Res. 2020, 26, 2104–2110. [Google Scholar] [CrossRef]
- Maughan, B.L.; Kessel, A.; McFarland, T.R.; Sayegh, N.; Nussenzveig, R.; Hahn, A.W.; Hoffman, J.M.; Morton, K.; Sirohi, D.; Kohli, M.; et al. Radium-223 plus Enzalutamide Versus Enzalutamide in Metastatic Castration-Refractory Prostate Cancer: Final Safety and Efficacy Results. Oncologist 2021, 26, 1006-e2129. [Google Scholar] [CrossRef]
- Sartor, O.; Vogelzang, N.J.; Sweeney, C.; Fernandez, D.C.; Almeida, F.; Iagaru, A.; Brown, A., Jr.; Smith, M.R.; Agrawal, M.; Dicker, A.P.; et al. Radium-223 Safety, Efficacy, and Concurrent Use with Abiraterone or Enzalutamide: First U.S. Experience from an Expanded Access Program. Oncologist 2017, 23, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Sartor, O.; de Bono, J.; Chi, K.N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S.T.; Nordquist, L.T.; Vaishampayan, N.; El-Haddad, G.; et al. Lutetium-177-PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N. Engl. J. Med. 2021, 385, 1091–1103. [Google Scholar] [CrossRef] [PubMed]
- Hofman, M.S.; Emmett, L.; Sandhu, S.; Iravani, A.; Joshua, A.M.; Goh, J.C.; Pattison, D.A.; Tan, T.H.; Kirkwood, I.D.; Ng, S.; et al. [(177)Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): A randomised, open-label, phase 2 trial. Lancet 2021, 397, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Den, R.B.; George, D.; Pieczonka, C.; McNamara, M. Ra-223 Treatment for Bone Metastases in Castrate-Resistant Prostate Cancer: Practical Management Issues for Patient Selection. Am. J. Clin. Oncol. 2019, 42, 399–406. [Google Scholar] [CrossRef]
- Smith, A.W.; Greenberger, B.A.; Den, R.B.; Stock, R.G. Radiopharmaceuticals for Bone Metastases. Semin. Radiat. Oncol. 2021, 31, 45–59. [Google Scholar] [CrossRef]
- Nilsson, S.; Cislo, P.; Sartor, O.; Vogelzang, N.J.; Coleman, R.E.; O’Sullivan, J.M.; Reuning-Scherer, J.; Shan, M.; Zhan, L.; Parker, C. Patient-reported quality-of-life analysis of radium-223 dichloride from the phase III ALSYMPCA study. Ann. Oncol. 2016, 27, 868–874. [Google Scholar] [CrossRef]
- Parker, C.; Zhan, L.; Cislo, P.; Reuning-Scherer, J.; Vogelzang, N.J.; Nilsson, S.; Sartor, O.; O’Sullivan, J.M.; Coleman, R.E. Effect of radium-223 dichloride (Ra-223) on hospitalisation: An analysis from the phase 3 randomised Alpharadin in Symptomatic Prostate Cancer Patients (ALSYMPCA) trial. Eur. J. Cancer 2017, 71, 1–6. [Google Scholar] [CrossRef]
- Heidenreich, A.; Gillessen, S.; Heinrich, D.; Keizman, D.; O’Sullivan, J.M.; Carles, J.; Wirth, M.; Miller, K.; Reeves, J.; Seger, M.; et al. Radium-223 in asymptomatic patients with castration-resistant prostate cancer and bone metastases treated in an international early access program. BMC Cancer 2019, 19, 12. [Google Scholar] [CrossRef]
- Parker, C.; Heidenreich, A.; Nilsson, S.; Shore, N. Current approaches to incorporation of radium-223 in clinical practice. Prostate Cancer Prostatic Dis. 2018, 21, 37–47. [Google Scholar] [CrossRef]
- Sartor, O.; Coleman, R.E.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O’Sullivan, J.M.; Vogelzang, N.J.; Bruland, O.; Kobina, S.; Wilhelm, S.; et al. An exploratory analysis of alkaline phosphatase, lactate dehydrogenase, and prostate-specific antigen dynamics in the phase 3 ALSYMPCA trial with radium-223. Ann. Oncol. 2017, 28, 1090–1097. [Google Scholar] [CrossRef]
- Sonni, I.; Eiber, M.; Fendler, W.P.; Alano, R.M.; Vangala, S.S.; Kishan, A.U.; Nickols, N.; Rettig, M.B.; Reiter, R.E.; Czernin, J.; et al. Impact of (68)Ga-PSMA-11 PET/CT on Staging and Management of Prostate Cancer Patients in Various Clinical Settings: A Prospective Single-Center Study. J. Nucl. Med. 2020, 61, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Tateishi, U. Prostate-specific membrane antigen (PSMA)-ligand positron emission tomography and radioligand therapy (RLT) of prostate cancer. Jpn. J. Clin. Oncol. 2020, 50, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Weineisen, M.; Schottelius, M.; Simecek, J.; Baum, R.P.; Yildiz, A.; Beykan, S.; Kulkarni, H.R.; Lassmann, M.; Klette, I.; Eiber, M.; et al. 68Ga- and 177Lu-Labeled PSMA I&T: Optimization of a PSMA-Targeted Theranostic Concept and First Proof-of-Concept Human Studies. J. Nucl. Med. 2015, 56, 1169–1176. [Google Scholar] [CrossRef]
- PLUVICTOTM (Lutetium Lu 177 Vipivotide Tetraxetan) Injection, for Intravenous Use [Package Insert]. U.S. Food and Drug Administration. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215833s000lbl.pdf (accessed on 2 August 2022).
- Mokoala, K.; Lawal, I.; Lengana, T.; Kgatle, M.; Giesel, F.L.; Vorster, M.; Sathekge, M. PSMA Theranostics: Science and Practice. Cancers 2021, 13, 3904. [Google Scholar] [CrossRef]
- Kratochwil, C.; Fendler, W.P.; Eiber, M.; Baum, R.; Bozkurt, M.F.; Czernin, J.; Delgado Bolton, R.C.; Ezziddin, S.; Forrer, F.; Hicks, R.J.; et al. EANM procedure guidelines for radionuclide therapy with (177)Lu-labelled PSMA-ligands ((177)Lu-PSMA-RLT). Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2536–2544. [Google Scholar] [CrossRef] [PubMed]
- Ferdinandus, J.; Violet, J.; Sandhu, S.; Hofman, M.S. Prostate-specific membrane antigen theranostics: Therapy with lutetium-177. Curr. Opin. Urol. 2018, 28, 197–204. [Google Scholar] [CrossRef] [PubMed]
- NCCN. NCCN Guidelines Version 4.2022 Prostate Cancer. Available online: https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf (accessed on 30 July 2022).
- Lantheus. Available online: https://investor.lantheus.com/node/13566/pdf (accessed on 30 July 2022).
- Michalski, K.; Ruf, J.; Goetz, C.; Seitz, A.K.; Buck, A.K.; Lapa, C.; Hartrampf, P.E. Prognostic implications of dual tracer PET/CT: PSMA ligand and [(18)F]FDG PET/CT in patients undergoing [(177)Lu]PSMA radioligand therapy. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2024–2030. [Google Scholar] [CrossRef] [PubMed]
- Hotta, M.; Gafita, A.; Czernin, J.; Calais, J. Outcome of patients with PSMA-PET/CT screen failure by VISION criteria and treated with 177Lu-PSMA therapy: A multicenter retrospective analysis. J. Nucl. Med. 2022. [Google Scholar] [CrossRef]
- Chen, R.; Wang, Y.; Zhu, Y.; Shi, Y.; Xu, L.; Huang, G.; Liu, J. The Added Value of (18)F-FDG PET/CT Compared with (68)Ga-PSMA PET/CT in Patients with Castration-Resistant Prostate Cancer. J. Nucl. Med. 2022, 63, 69–75. [Google Scholar] [CrossRef]
- Thang, S.P.; Violet, J.; Sandhu, S.; Iravani, A.; Akhurst, T.; Kong, G.; Ravi Kumar, A.; Murphy, D.G.; Williams, S.G.; Hicks, R.J.; et al. Poor Outcomes for Patients with Metastatic Castration-resistant Prostate Cancer with Low Prostate-specific Membrane Antigen (PSMA) Expression Deemed Ineligible for (177)Lu-labelled PSMA Radioligand Therapy. Eur. Urol. Oncol. 2019, 2, 670–676. [Google Scholar] [CrossRef]
- Hope, T.A.; Abbott, A.; Colucci, K.; Bushnell, D.L.; Gardner, L.; Graham, W.S.; Lindsay, S.; Metz, D.C.; Pryma, D.A.; Stabin, M.G.; et al. NANETS/SNMMI Procedure Standard for Somatostatin Receptor-Based Peptide Receptor Radionuclide Therapy with (177)Lu-DOTATATE. J. Nucl. Med. 2019, 60, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Nautiyal, A.; Jha, A.K.; Mithun, S.; Rangarajan, V. Dosimetry in Lu-177-PSMA-617 prostate-specific membrane antigen targeted radioligand therapy: A systematic review. Nucl. Med. Commun. 2022, 43, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Sadaghiani, M.S.; Sheikhbahaei, S.; Werner, R.A.; Pienta, K.J.; Pomper, M.G.; Solnes, L.B.; Gorin, M.A.; Wang, N.Y.; Rowe, S.P. A Systematic Review and Meta-analysis of the Effectiveness and Toxicities of Lutetium-177-labeled Prostate-specific Membrane Antigen-targeted Radioligand Therapy in Metastatic Castration-Resistant Prostate Cancer. Eur. Urol. 2021, 80, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Rahbar, K.; Ahmadzadehfar, H.; Kratochwil, C.; Haberkorn, U.; Schäfers, M.; Essler, M.; Baum, R.P.; Kulkarni, H.R.; Schmidt, M.; Drzezga, A.; et al. German Multicenter Study Investigating 177Lu-PSMA-617 Radioligand Therapy in Advanced Prostate Cancer Patients. J. Nucl. Med. 2017, 58, 85–90. [Google Scholar] [CrossRef]
- Hofman, M.S.; Violet, J.; Hicks, R.J.; Ferdinandus, J.; Thang, S.P.; Akhurst, T.; Iravani, A.; Kong, G.; Ravi Kumar, A.; Murphy, D.G.; et al. [(177)Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): A single-centre, single-arm, phase 2 study. Lancet Oncol. 2018, 19, 825–833. [Google Scholar] [CrossRef]
- Violet, J.; Sandhu, S.; Iravani, A.; Ferdinandus, J.; Thang, S.P.; Kong, G.; Kumar, A.R.; Akhurst, T.; Pattison, D.A.; Beaulieu, A.; et al. Long-Term Follow-up and Outcomes of Retreatment in an Expanded 50-Patient Single-Center Phase II Prospective Trial of (177)Lu-PSMA-617 Theranostics in Metastatic Castration-Resistant Prostate Cancer. J. Nucl. Med. 2020, 61, 857–865. [Google Scholar] [CrossRef]
- Rathke, H.; Giesel, F.L.; Flechsig, P.; Kopka, K.; Mier, W.; Hohenfellner, M.; Haberkorn, U.; Kratochwil, C. Repeated 177Lu-Labeled PSMA-617 Radioligand Therapy Using Treatment Activities of up to 9.3 GBq. J. Nucl. Med. 2018, 59, 459–465. [Google Scholar] [CrossRef]
- Jackson, P.; Hofman, M.; McIntosh, L.; Buteau, J.P.; Kumar, A.R. Radiation Dosimetry in 177Lu-PSMA-617 Therapy. Semin. Nucl. Med. 2021, 52, 243–254. [Google Scholar] [CrossRef]
- Lawhn-Heath, C.; Hope, T.A.; Martinez, J.; Fung, E.K.; Shin, J.; Seo, Y.; Flavell, R.R. Dosimetry in radionuclide therapy: The clinical role of measuring radiation dose. Lancet Oncol. 2022, 23, e75–e87. [Google Scholar] [CrossRef]
- Violet, J.; Jackson, P.; Ferdinandus, J.; Sandhu, S.; Akhurst, T.; Iravani, A.; Kong, G.; Kumar, A.R.; Thang, S.P.; Eu, P. Dosimetry of 177Lu-PSMA-617 in metastatic castration-resistant prostate cancer: Correlations between pretherapeutic imaging and whole-body tumor dosimetry with treatment outcomes. J. Nucl. Med. 2019, 60, 517–523. [Google Scholar] [CrossRef] [Green Version]
- Gafita, A.; Wang, H.; Robertson, A.; Armstrong, W.R.; Zaum, R.; Weber, M.; Yagubbayli, F.; Kratochwil, C.; Grogan, T.R.; Nguyen, K.; et al. Tumor Sink Effect in (68)Ga-PSMA-11 PET: Myth or Reality? J. Nucl. Med. 2022, 63, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Filss, C.; Heinzel, A.; Miiller, B.; Vogg, A.T.J.; Langen, K.J.; Mottaghy, F.M. Relevant tumor sink effect in prostate cancer patients receiving 177Lu-PSMA-617 radioligand therapy. Nuklearmedizin 2018, 57, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Sathekge, M.; Bruchertseifer, F.; Knoesen, O.; Reyneke, F.; Lawal, I.; Lengana, T.; Davis, C.; Mahapane, J.; Corbett, C.; Vorster, M.; et al. (225)Ac-PSMA-617 in chemotherapy-naive patients with advanced prostate cancer: A pilot study. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Kratochwil, C.; Bruchertseifer, F.; Giesel, F.L.; Weis, M.; Verburg, F.A.; Mottaghy, F.; Kopka, K.; Apostolidis, C.; Haberkorn, U.; Morgenstern, A. 225Ac-PSMA-617 for PSMA-Targeted α-Radiation Therapy of Metastatic Castration-Resistant Prostate Cancer. J. Nucl. Med. 2016, 57, 1941–1944. [Google Scholar] [CrossRef] [PubMed]
- Van der Doelen, M.J.; Mehra, N.; van Oort, I.M.; Looijen-Salamon, M.G.; Janssen, M.J.R.; Custers, J.A.E.; Slootbeek, P.H.J.; Kroeze, L.I.; Bruchertseifer, F.; Morgenstern, A.; et al. Clinical outcomes and molecular profiling of advanced metastatic castration-resistant prostate cancer patients treated with (225)Ac-PSMA-617 targeted alpha-radiation therapy. Urol. Oncol. 2021, 39, e727–e729. [Google Scholar] [CrossRef]
- Satapathy, S.; Sood, A.; Das, C.K.; Mittal, B.R. Evolving role of 225Ac-PSMA radioligand therapy in metastatic castration-resistant prostate cancer—a systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2021, 24, 880–890. [Google Scholar] [CrossRef]
- Kratochwil, C.; Giesel, F.L.; Bruchertseifer, F.; Mier, W.; Apostolidis, C.; Boll, R.; Murphy, K.; Haberkorn, U.; Morgenstern, A. (2)(1)(3)Bi-DOTATOC receptor-targeted alpha-radionuclide therapy induces remission in neuroendocrine tumours refractory to beta radiation: A first-in-human experience. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 2106–2119. [Google Scholar] [CrossRef]
- Kratochwil, C.; Bruchertseifer, F.; Rathke, H.; Bronzel, M.; Apostolidis, C.; Weichert, W.; Haberkorn, U.; Giesel, F.L.; Morgenstern, A. Targeted alpha-Therapy of Metastatic Castration-Resistant Prostate Cancer with (225)Ac-PSMA-617: Dosimetry Estimate and Empiric Dose Finding. J. Nucl. Med. 2017, 58, 1624–1631. [Google Scholar] [CrossRef]
- Feuerecker, B.; Tauber, R.; Knorr, K.; Heck, M.; Beheshti, A.; Seidl, C.; Bruchertseifer, F.; Pickhard, A.; Gafita, A.; Kratochwil, C.; et al. Activity and Adverse Events of Actinium-225-PSMA-617 in Advanced Metastatic Castration-resistant Prostate Cancer After Failure of Lutetium-177-PSMA. Eur. Urol. 2021, 79, 343–350. [Google Scholar] [CrossRef]
- Kratochwil, C.; Bruchertseifer, F.; Rathke, H.; Hohenfellner, M.; Giesel, F.L.; Haberkorn, U.; Morgenstern, A. Targeted alpha-Therapy of Metastatic Castration-Resistant Prostate Cancer with (225)Ac-PSMA-617: Swimmer-Plot Analysis Suggests Efficacy Regarding Duration of Tumor Control. J. Nucl. Med. 2018, 59, 795–802. [Google Scholar] [CrossRef] [Green Version]
- Haberkorn, U.; Giesel, F.; Morgenstern, A.; Kratochwil, C. The Future of Radioligand Therapy: Alpha, beta, or Both? J. Nucl. Med. 2017, 58, 1017–1018. [Google Scholar] [CrossRef] [PubMed]
- Lawal, I.O.; Morgenstern, A.; Vorster, M.; Knoesen, O.; Mahapane, J.; Hlongwa, K.N.; Maserumule, L.C.; Ndlovu, H.; Reed, J.D.; Popoola, G.O.; et al. Hematologic toxicity profile and efficacy of [(225)Ac]Ac-PSMA-617 alpha-radioligand therapy of patients with extensive skeletal metastases of castration-resistant prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 3581–3592. [Google Scholar] [CrossRef] [PubMed]
- Khreish, F.; Ebert, N.; Ries, M.; Maus, S.; Rosar, F.; Bohnenberger, H.; Stemler, T.; Saar, M.; Bartholomä, M.; Ezziddin, S. (225)Ac-PSMA-617/(177)Lu-PSMA-617 tandem therapy of metastatic castration-resistant prostate cancer: Pilot experience. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Pujatti, P.B.; Foster, J.M.; Finucane, C.; Hudson, C.D.; Burnet, J.C.; Pasqualoto, K.F.M.; Mengatti, J.; Mather, S.J.; de Araujo, E.B.; Sosabowski, J.K. Evaluation and comparison of a new DOTA and DTPA-bombesin agonist in vitro and in vivo in low and high GRPR expressing prostate and breast tumor models. Appl. Radiat. Isot. 2015, 96, 91–101. [Google Scholar] [CrossRef]
- Mansi, R.; Fleischmann, A.; Macke, H.R.; Reubi, J.C. Targeting GRPR in urological cancers--from basic research to clinical application. Nat. Rev. Urol. 2013, 10, 235–244. [Google Scholar] [CrossRef]
- Aprikian, A.G.; Tremblay, L.; Han, K.; Chevalier, S. Bombesin stimulates the motility of human prostate-carcinoma cells through tyrosine phosphorylation of focal adhesion kinase and of integrin-associated proteins. Int. J. Cancer 1997, 72, 498–504. [Google Scholar] [CrossRef]
- Dalm, S.U.; Bakker, I.L.; de Blois, E.; Doeswijk, G.N.; Konijnenberg, M.W.; Orlandi, F.; Barbato, D.; Tedesco, M.; Maina, T.; Nock, B.A.; et al. 68Ga/177Lu-NeoBOMB1, a Novel Radiolabeled GRPR Antagonist for Theranostic Use in Oncology. J. Nucl. Med. 2017, 58, 293–299. [Google Scholar] [CrossRef]
- Gourni, E.; Del Pozzo, L.; Kheirallah, E.; Smerling, C.; Waser, B.; Reubi, J.C.; Paterson, B.M.; Donnelly, P.S.; Meyer, P.T.; Maecke, H.R. Copper-64 Labeled Macrobicyclic Sarcophagine Coupled to a GRP Receptor Antagonist Shows Great Promise for PET Imaging of Prostate Cancer. Mol. Pharm. 2015, 12, 2781–2790. [Google Scholar] [CrossRef]
- Liu, F.; Zhu, H.; Yu, J.; Han, X.; Xie, Q.; Liu, T.; Xia, C.; Li, N.; Yang, Z. (68)Ga/(177)Lu-labeled DOTA-TATE shows similar imaging and biodistribution in neuroendocrine tumor model. Tumour Biol. 2017, 39, 1010428317705519. [Google Scholar] [CrossRef]
- Huynh, T.T.; van Dam, E.M.; Sreekumar, S.; Mpoy, C.; Blyth, B.J.; Muntz, F.; Harris, M.J.; Rogers, B.E. Copper-67-Labeled Bombesin Peptide for Targeted Radionuclide Therapy of Prostate Cancer. Pharmaceuticals 2022, 15, 728. [Google Scholar] [CrossRef]
- Bakker, I.L.; Froberg, A.C.; Busstra, M.B.; Verzijlbergen, J.F.; Konijnenberg, M.; van Leenders, G.; Schoots, I.G.; de Blois, E.; van Weerden, W.M.; Dalm, S.U.; et al. GRPr Antagonist (68)Ga-SB3 PET/CT Imaging of Primary Prostate Cancer in Therapy-Naive Patients. J. Nucl. Med. 2021, 62, 1517–1523. [Google Scholar] [CrossRef] [PubMed]
- Maddalena, M.E.; Fox, J.; Chen, J.; Feng, W.; Cagnolini, A.; Linder, K.E.; Tweedle, M.F.; Nunn, A.D.; Lantry, L.E. 177Lu-AMBA biodistribution, radiotherapeutic efficacy, imaging, and autoradiography in prostate cancer models with low GRP-R expression. J. Nucl. Med. 2009, 50, 2017–2024. [Google Scholar] [CrossRef] [PubMed]
- Bodei, L.; Ferrari, M.; Nunn, A.; Llull, J.; Cremonesi, M.; Martano, L.; Laurora, G.; Scardino, E.; Tiberini, S.; Bufi, G.; et al. Lu-177-AMBA Bombesin analogue in hormone refractory prostate cancer patients: A phase I escalation study with single-cycle administrations. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, S221. [Google Scholar]
- Kurth, J.; Krause, B.J.; Schwarzenbock, S.M.; Bergner, C.; Hakenberg, O.W.; Heuschkel, M. First-in-human dosimetry of gastrin-releasing peptide receptor antagonist [(177)Lu]Lu-RM2: A radiopharmaceutical for the treatment of metastatic castration-resistant prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Djaileb, L.; Morgat, C.; van der Veldt, A.; Virgolini, I.; Cortes, F.; Demange, A.; Orlandi, F.; Wegener, A. Preliminary diagnostic performance of [Ga-68]-NeoBOMB1 in patients with gastrin-releasing peptide receptor-positive breast, prostate, colorectal or lung tumors (NeoFIND). J. Nucl. Med. 2020, 61, 346. [Google Scholar]
- Chatalic, K.L.; Kwekkeboom, D.J.; de Jong, M. Radiopeptides for Imaging and Therapy: A Radiant Future. J. Nucl. Med. 2015, 56, 1809–1812. [Google Scholar] [CrossRef]
- Linder, K.E.; Metcalfe, E.; Arunachalam, T.; Chen, J.; Eaton, S.M.; Feng, W.; Fan, H.; Raju, N.; Cagnolini, A.; Lantry, L.E.; et al. In vitro and in vivo metabolism of Lu-AMBA, a GRP-receptor binding compound, and the synthesis and characterization of its metabolites. Bioconjug Chem. 2009, 20, 1171–1178. [Google Scholar] [CrossRef]
- Dumont, R.A.; Tamma, M.; Braun, F.; Borkowski, S.; Reubi, J.C.; Maecke, H.; Weber, W.A.; Mansi, R. Targeted radiotherapy of prostate cancer with a gastrin-releasing peptide receptor antagonist is effective as monotherapy and in combination with rapamycin. J. Nucl. Med. 2013, 54, 762–769. [Google Scholar] [CrossRef]
- Mitran, B.; Rinne, S.S.; Konijnenberg, M.W.; Maina, T.; Nock, B.A.; Altai, M.; Vorobyeva, A.; Larhed, M.; Tolmachev, V.; de Jong, M.; et al. Trastuzumab cotreatment improves survival of mice with PC-3 prostate cancer xenografts treated with the GRPR antagonist (177) Lu-DOTAGA-PEG2 -RM26. Int. J. Cancer 2019, 145, 3347–3358. [Google Scholar] [CrossRef]
- Jimenez, R.E.; Nandy, D.; Qin, R.; Carlson, R.; Tan, W.; Kohli, M. Neuroendocrine differentiation patterns in metastases from advanced prostate cancer. J. Clin. Oncol. 2014, 32, 5085. [Google Scholar] [CrossRef]
- Santoni, M.; Scarpelli, M.; Mazzucchelli, R.; Lopez-Beltran, A.; Cheng, L.; Cascinu, S.; Montironi, R. Targeting prostate-specific membrane antigen for personalized therapies in prostate cancer: Morphologic and molecular backgrounds and future promises. J. Biol. Regul. Homeost Agents 2014, 28, 555–563. [Google Scholar] [PubMed]
- Parimi, V.; Goyal, R.; Poropatich, K.; Yang, X.J. Neuroendocrine differentiation of prostate cancer: A review. Am. J. Clin. Exp. Urol. 2014, 2, 273–285. [Google Scholar] [PubMed]
- Nelson, E.C.; Cambio, A.J.; Yang, J.C.; Ok, J.H.; Lara, P.N., Jr.; Evans, C.P. Clinical implications of neuroendocrine differentiation in prostate cancer. Prostate Cancer Prostatic Dis. 2007, 10, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Borre, M.; Nerstrom, B.; Overgaard, J. Association between immunohistochemical expression of vascular endothelial growth factor (VEGF), VEGF-expressing neuroendocrine-differentiated tumor cells, and outcome in prostate cancer patients subjected to watchful waiting. Clin. Cancer Res. 2000, 6, 1882–1890. [Google Scholar] [PubMed]
- Morichetti, D.; Mazzucchelli, R.; Santinelli, A.; Stramazzotti, D.; Lopez-Beltran, A.; Scarpelli, M.; Bono, A.V.; Cheng, L.; Montironi, R. Immunohistochemical expression and localization of somatostatin receptor subtypes in prostate cancer with neuroendocrine differentiation. Int. J. Immunopathol. Pharm. 2010, 23, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Montironi, R.; Cheng, L.; Mazzucchelli, R.; Morichetti, D.; Stramazzotti, D.; Santinelli, A.; Moroncini, G.; Galosi, A.B.; Muzzonigro, G.; Comeri, G.; et al. Immunohistochemical detection and localization of somatostatin receptor subtypes in prostate tissue from patients with bladder outlet obstruction. Cell Oncol. 2008, 30, 473–482. [Google Scholar] [CrossRef]
- Gabriel, M.; Decristoforo, C.; Kendler, D.; Dobrozemsky, G.; Heute, D.; Uprimny, C.; Kovacs, P.; Von Guggenberg, E.; Bale, R.; Virgolini, I.J. 68Ga-DOTA-Tyr3-octreotide PET in neuroendocrine tumors: Comparison with somatostatin receptor scintigraphy and CT. J. Nucl. Med. 2007, 48, 508–518. [Google Scholar] [CrossRef]
- Alonso, O.; Gambini, J.P.; Lago, G.; Gaudiano, J.; Quagliata, A.; Engler, H. In vivo visualization of somatostatin receptor expression with Ga-68-DOTA-TATE PET/CT in advanced metastatic prostate cancer. Clin. Nucl. Med. 2011, 36, 1063–1064. [Google Scholar] [CrossRef]
- Chen, S.; Cheung, S.K.; Wong, K.N.; Wong, K.K.; Ho, C.L. 68Ga-DOTATOC and 68Ga-PSMA PET/CT Unmasked a Case of Prostate Cancer With Neuroendocrine Differentiation. Clin. Nucl. Med. 2016, 41, 959–960. [Google Scholar] [CrossRef]
- Todorovic-Tirnanic, M.V.; Gajic, M.M.; Obradovic, V.B.; Baum, R.P. Gallium-68 DOTATOC PET/CT in vivo characterization of somatostatin receptor expression in the prostate. Cancer Biother. Radiopharm. 2014, 29, 108–115. [Google Scholar] [CrossRef]
- Gofrit, O.N.; Frank, S.; Meirovitz, A.; Nechushtan, H.; Orevi, M. PET/CT With 68Ga-DOTA-TATE for Diagnosis of Neuroendocrine: Differentiation in Patients With Castrate-Resistant Prostate Cancer. Clin. Nucl. Med. 2017, 42, 1–6. [Google Scholar] [CrossRef]
- Nesari Javan, F.; Aryana, K.; Askari, E. Prostate Cancer With Neuroendocrine Differentiation Recurring After Treatment With 177Lu-PSMA: A Chance for 177Lu-DOTATATE Therapy? Clin. Nucl. Med. 2021, 46, e480–e482. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.; Finkelstein, S.E.; Michalski, J.M.; O’Sullivan, J.M.; Bruland, Ø.; Vogelzang, N.J.; Coleman, R.E.; Nilsson, S.; Sartor, O.; Li, R.; et al. Efficacy and Safety of Radium-223 Dichloride in Symptomatic Castration-resistant Prostate Cancer Patients With or Without Baseline Opioid Use From the Phase 3 ALSYMPCA Trial. Eur. Urol. 2016, 70, 875–883. [Google Scholar] [CrossRef] [PubMed]
- Hoskin, P.; Sartor, O.; O’Sullivan, J.M.; Johannessen, D.C.; Helle, S.I.; Logue, J.; Bottomley, D.; Nilsson, S.; Vogelzang, N.J.; Fang, F.; et al. Efficacy and safety of radium-223 dichloride in patients with castration-resistant prostate cancer and symptomatic bone metastases, with or without previous docetaxel use: A prespecified subgroup analysis from the randomised, double-blind, phase 3 ALSYMPCA trial. Lancet Oncol. 2014, 15, 1397–1406. [Google Scholar] [CrossRef]
- Sartor, A.O.; la Fougère, C.; Essler, M.; Ezziddin, S.; Kramer, G.; Elllinger, J.; Nordquist, L.; Sylvester, J.; Paganelli, G.; Peer, A.; et al. Lutetium-177–prostate-specific membrane antigen ligand following radium-223 treatment in men with bone-metastatic castration-resistant prostate cancer: Real-world clinical experience. J. Nucl. Med. 2021. [Google Scholar] [CrossRef]
- Vaishampayan, N.; Morris, M.J.; Krause, B.J.; Vogelzang, N.J.; Kendi, A.T.; Nordquist, L.T.; Calais, J.; Nagarajah, J.; Beer, T.M.; El-Haddad, G.; et al. [177Lu]Lu-PSMA-617 in PSMA-positive metastatic castration-resistant prostate cancer: Prior and concomitant treatment subgroup analyses of the VISION trial. J. Clin. Oncol. 2022, 40, 5001. [Google Scholar] [CrossRef]
- Hope, T.A.; Truillet, C.; Ehman, E.C.; Afshar-Oromieh, A.; Aggarwal, R.; Ryan, C.J.; Carroll, P.R.; Small, E.J.; Evans, M.J. 68Ga-PSMA-11 PET Imaging of Response to Androgen Receptor Inhibition: First Human Experience. J. Nucl. Med. 2017, 58, 81–84. [Google Scholar] [CrossRef] [PubMed]
- Vaz, S.; Hadaschik, B.; Gabriel, M.; Herrmann, K.; Eiber, M.; Costa, D. Influence of androgen deprivation therapy on PSMA expression and PSMA-ligand PET imaging of prostate cancer patients. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 9–15. [Google Scholar] [CrossRef] [PubMed]
- De Wit, R.; de Bono, J.; Sternberg, C.N.; Fizazi, K.; Tombal, B.; Wülfing, C.; Kramer, G.; Eymard, J.-C.; Bamias, A.; Carles, J.; et al. Cabazitaxel versus Abiraterone or Enzalutamide in Metastatic Prostate Cancer. N. Engl. J. Med. 2019, 381, 2506–2518. [Google Scholar] [CrossRef]
- National Library of Medicine (NLM). Available online: https://www.ClinicalTrials.gov (accessed on 28 August 2022).


| 223RaCl2 | |||
| Alpha Emitter Radium-223 and Survival in Metastatic Prostate Cancer (ALSYMPCA) [7] | 223RaCl2 vs. placebo in mCRPC with bone metastasis | Phase III | 223RaCl2 improved overall survival vs. placebo (median, 14.0 months vs. 11.2 months). |
| Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223) [8] | Abiraterone acetate + prednisone/prednisolone with 223RaCl2 vs. placebo | Phase III | Addition of 223RaCl2 did not improve symptomatic skeletal event-free survival and was associated with increasing frequency of fractures (9% vs. 3%). |
| Prospective Evaluation of Bone Metabolic Markers as Surrogate Markers of Response to Radium-223 Therapy in Metastatic Castration-resistant Prostate Cancer [9,10] | Enzalutamide + 223RaCl2 vs. enzalutamide alone | Phase II | Combination Enzalutamide + 223RaCl2 did not show increase in fractures or other adverse events and showed improved bone metabolic markers. |
| Radium-223 Safety, Efficacy, and Concurrent Use with Abiraterone or Enzalutamide: First U.S. Experience from an Expanded Access Program [11] | 223RaCl2 + concurrent abiraterone acetate or enzalutamide | Phase II | Patients with less advanced disease (<3 prior therapies) were more likely to benefit from 223RaCl2 |
| 177Lu PSMA-617 | |||
| Lutetium-177–PSMA-617 for Metastatic Castration-Resistant Prostate Cancer [12] | 177 Lu PSMA-617 +SOC vs. SOC alone | Phase III | 177Lu PSMA-617 +SOC (compared to SOC alone) improved rPFS (median, 8.7 vs. 3.4 months) and OS (median, 15.3 vs. 11.3 months). |
| [177Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomized, open-label, phase 2 trial [13]. | 177 Lu PSMA-617 vs. cabazitaxel | Phase III | 177Lu PSMA-617 arm had greater PSA response (65%) vs. cabazitaxel (37%) Grade 3–4 adverse events occurred in (33%) of 98 men in the 177Lu PSMA-617 v 45 (53%) of 85 men in the cabazitaxel group. |
| ClinicalTrials.gov Identifier | Name of Study | Study Sponsor | Trials Phase | Location |
|---|---|---|---|---|
| PSMA | ||||
| NCT04443062 | Lutetium-177-PSMA-617 in Oligo-metastatic Hormone Sensitive Prostate Cancer (Bullseye) | Radboud University Medical Center | Phase 2 | The Netherlands |
| NCT05114746 | Study of 177Lu-PSMA-617 In Metastatic Castrate-Resistant Prostate Cancer in Japan | Novartis Pharmaceuticals | Phase 2 | Japan |
| NCT05079698 | A Study of Stereotactic Body Radiotherapy and 177Lu-PSMA-617 for the Treatment of Prostate Cancer | Memorial Sloan Kettering Cancer Center | Phase 1 | New York, USA |
| NCT03454750 | Radiometabolic Therapy (RMT) With 177Lu PSMA 617 in Advanced Castration Resistant Prostate Cancer (CRPC) (LU-PSMA) | Istituto Scientifico Romagnolo per lo Studio e la cura dei Tumori | Phase 2 | Italy |
| NCT05219500 | Targeted Alpha Therapy With 225Actinium-PSMA-I&T of Castration-resISTant Prostate Cancer (TATCIST) | Excel Diagnostics and Nuclear Oncology Center | Phase 2 | Texas, USA |
| NCT04343885 | In Men With Metastatic Prostate Cancer, What is the Safety and Benefit of Lutetium-177PSMA Radionuclide Treatment in Addition to Chemotherapy (UpFrontPSMA) | Peter MacCallum Cancer Centre | Phase 2 | Australia |
| NCT04419402 | Enzalutamide With Lu PSMA-617 Versus Enzalutamide Alone in Men With Metastatic Castration-resistant Prostate Cancer (ENZA-p) | Australian and New Zealand Urogenital and Prostate Cancer Trials Group | Phase 2 | Australia |
| NCT03780075 | 177Lu-EB-PSMA617 Radionuclide Treatment in Patients With Metastatic Castration-resistant Prostate Cancer | Peking Union Medical College Hospital | Phase 1 | China |
| NCT03874884 | 177Lu-PSMA-617 Therapy and Olaparib in Patients With Metastatic Castration Resistant Prostate Cancer (LuPARP) | Peter MacCallum Cancer Centre | Phase 1 | Australia |
| NCT05162573 | EBRT + Lu-PSMA for N1M0 Prostate Cancer (PROQURE-1) | The Netherlands Cancer Institute | Phase 1 | The Netherlands |
| NCT04769817 | ProsTIC Registry of Men Treated With PSMA Theranostics | Peter MacCallum Cancer Centre | Observational | Australia |
| NCT04689828 | 177Lu-PSMA-617 vs. Androgen Receptor-directed Therapy in the Treatment of Progressive Metastatic Castrate Resistant Prostate Cancer (PSMAfore) | Novartis Pharmaceuticals | Phase 3 | Multinational |
| NCT04597411 | Study of 225Ac-PSMA-617 in Men With PSMA-positive Prostate Cancer | Endocyte | Phase 1 | Australia |
| NCT04886986 | 225Ac-J591 Plus 177Lu-PSMA-I&T for mCRPC | Weill Medical College of Cornell University | Phase 1/2 | New York, USA |
| NCT05340374 | Cabazitaxel in Combination With 177Lu-PSMA-617 in Metastatic Castration-resistant Prostate Cancer (LuCAB) | Peter MacCallum Cancer Centre | Phase 1/2 | Australia |
| NCT05204927 | Lu-177-PSMA-I&T for Metastatic Castration-Resistant Prostate Cancer | Curium US LLC | Phase 3 | USA |
| NCT04647526 | Study Evaluating mCRPC Treatment Using PSMA [Lu-177]-PNT2002 Therapy After Second-line Hormonal Treatment (SPLASH) | POINT Biopharma | Phase 3 | Multinational |
| NCT04996602 | Therapeutic Efficiency and Response to 2.0 GBq (55mCi) 177Lu-EB-PSMA in Patients With mCRPC | Peking Union Medical College Hospital | Phase 1 | China |
| NCT04720157 | An International Prospective Open-label, Randomized, Phase III Study Comparing 177Lu-PSMA-617 in Combination With SOC, Versus SOC Alone, in Adult Male Patients With mHSPC (PSMAddition) | Novartis Pharmaceuticals | Phase 3 | Multinational |
| NCT05113537 | Abemaciclib Before 177Lu-PSMA-617 for the Treatment of Metastatic Castrate Resistant Prostate Cancer (UPLIFT) | Vadim S Koshkin | Phase 1 | California, USA |
| NCT04946370 | Maximizing Responses to Anti-PD1 Immunotherapy With PSMA-targeted Alpha Therapy in mCRPC | Weill Medical College of Cornell University | Phase 1/2 | New York, USA |
| NCT04868604 | 64Cu-SAR-bisPSMA and 67Cu-SAR-bisPSMA for Identification and Treatment of PSMA-expressing Metastatic Castrate Resistant Prostate Cancer (SECuRE) | Clarity Pharmaceuticals Ltd. | Phase 1/2 | USA |
| NCT05230251 | Radioligand fOr locAl raDiorecurrent proStaTe cancER (ROADSTER) | Glenn Bauman, Lawson Health Research Institute | Phase 2 | Canada |
| NCT04576871 | Re-treatment 225Ac-J591 for mCRPC | Weill Medical College of Cornell University | Phase 1 | New York, USA |
| NCT04726033 | 64Cu-TLX592 Phase I Safety, PK, Biodistribution and Dosimetry Study (CUPID Study) (CUPID) | Telix International Pty Ltd. | Phase 1 | Australia |
| NCT04506567 | Fractionated and Multiple Dose 225Ac-J591 for Progressive mCRPC | Weill Medical College of Cornell University | Phase 1/2 | New York, USA |
| NCT05150236 | EVOLUTION: 177Lu-PSMA Therapy Versus 177Lu-PSMA in Combination With Ipilimumab and Nivolumab for Men With mCRPC (ANZUP2001) | Australian and New Zealand Urogenital and Prostate Cancer Trials Group | Phase 2 | Australia |
| NCT05413850 | Anti-tumour Activity of (177Lu) rhPSMA-10.1 Injection | Blue Earth Therapeutics Ltd. | Phase 1/2 | Maryland, USA |
| NCT04509557 | [177Lu]Ludotadipep Treatment in Patients With Metastatic Castration-resistant Prostate Cancer. | FutureChem | Phase 1 | Republic of Korea |
| 223RaCl2 | ||||
| NCT04521361 | A Study to Assess How Radium-223 Distributes in the Body of Patients With Prostate Cancer Which Spread to the Bones | Bayer | Phase 1 | Multinational |
| NCT04037358 | RAdium-223 and SABR Versus SABR for Oligometastatic Prostate Cancers (RAVENS) | Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins | Phase 2 | Maryland, USA |
| NCT03574571 | A Study to Test Radium-223 With Docetaxel in Patients With Prostate Cancer | Memorial Sloan Kettering Cancer Center | Phase 3 | Multinational |
| NCT05133440 | A Study of Stereotactic Body Radiation Therapy and Radium (Ra-223) Dichloride in Prostate Cancer That Has Spread to the Bones | Memorial Sloan Kettering Cancer Center | Phase 2 | USA |
| NCT03737370 | Fractionated Docetaxel and Radium 223 in Metastatic Castration-Resistant Prostate Cancer | Tufts Medical Center | Phase 1 | USA |
| NCT04109729 | Study of Nivolumab in Combination w Radium-223 in Men w Metastatic Castration Resistant Prostate Cancer (Rad2Nivo) | University of Utah | Phase 1/2 | Utah, USA |
| NCT04206319 | Radium-223 in Biochemically Recurrent Prostate Cancer | National Cancer Institute (NCI) | Phase 2 | Maryland, USA |
| NCT04597125 | Investigation of Radium-223 Dichloride (Xofigo), a Treatment That Gives Off Radiation That Helps Kill Cancer Cells, Compared to a Treatment That Inactivates Hormones (New Antihormonal Therapy, NAH) in Patients With Prostate Cancer That Has Spread to the Bone Getting Worse on or After Earlier NAH | Bayer | Phase 4 | Multinational |
| NCT03432949 | Radium-223 Combined With Dexamethasone as First-line Therapy in Patients With M+CRPC (TRANCE) | Bayer | Phase 4 | Canada |
| NCT04071236 | Radiation Medication (Radium-223 Dichloride) Versus Radium-223 Dichloride Plus Radiation Enhancing Medication (M3814) Versus Radium-223 Dichloride Plus M3814 Plus Avelumab (a Type of Immunotherapy) for Advanced Prostate Cancer Not Responsive to Hormonal Therapy | National Cancer Institute (NCI) | Phase 1/2 | USA |
| NCT04704505 | Bipolar Androgen Therapy (BAT) and Radium-223 (RAD) in Metastatic Castration-resistant Prostate Cancer (mCRPC) (BAT-RAD) | Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins | Phase 2 | Multinational |
| NCT03361735 | Radium Ra 223 Dichloride, Hormone Therapy and Stereotactic Body Radiation Therapy in Treating Patients With Metastatic Prostate Cancer | City of Hope Medical Center | Phase 2 | California, USA |
| NCT02194842 | Phase III Radium 223 mCRPC-PEACE III (PEACE III) | European Organisation for Research and Treatment of Cancer—EORTC | Phase 3 | Multinational |
| NCT04704505 | Bipolar Androgen Therapy (BAT) and Radium-223 (RAD) in Metastatic Castration-resistant Prostate Cancer (mCRPC) (BAT-RAD) | Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins | Phase 2 | Multinational |
| GRPR | ||||
| NCT05283330 | Safety and Tolerability of ²¹²Pb-DOTAM-GRPR1 ²¹²Pb-DOTAM-GRPR1 in Adult Subjects with Recurrent or Metastatic GRPR- expressing Tumors | Orano Med LLC | Phase 1 | Not yet recruiting |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parent, E.E.; Kase, A.M. A Treatment Paradigm Shift: Targeted Radionuclide Therapies for Metastatic Castrate Resistant Prostate Cancer. Cancers 2022, 14, 4276. https://doi.org/10.3390/cancers14174276
Parent EE, Kase AM. A Treatment Paradigm Shift: Targeted Radionuclide Therapies for Metastatic Castrate Resistant Prostate Cancer. Cancers. 2022; 14(17):4276. https://doi.org/10.3390/cancers14174276
Chicago/Turabian StyleParent, Ephraim E., and Adam M. Kase. 2022. "A Treatment Paradigm Shift: Targeted Radionuclide Therapies for Metastatic Castrate Resistant Prostate Cancer" Cancers 14, no. 17: 4276. https://doi.org/10.3390/cancers14174276








