Event-Free Survival in Patients with Early HER2-Positive Breast Cancer with a Pathological Complete Response after HER2-Targeted Therapy: A Pooled Analysis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Patient Populations
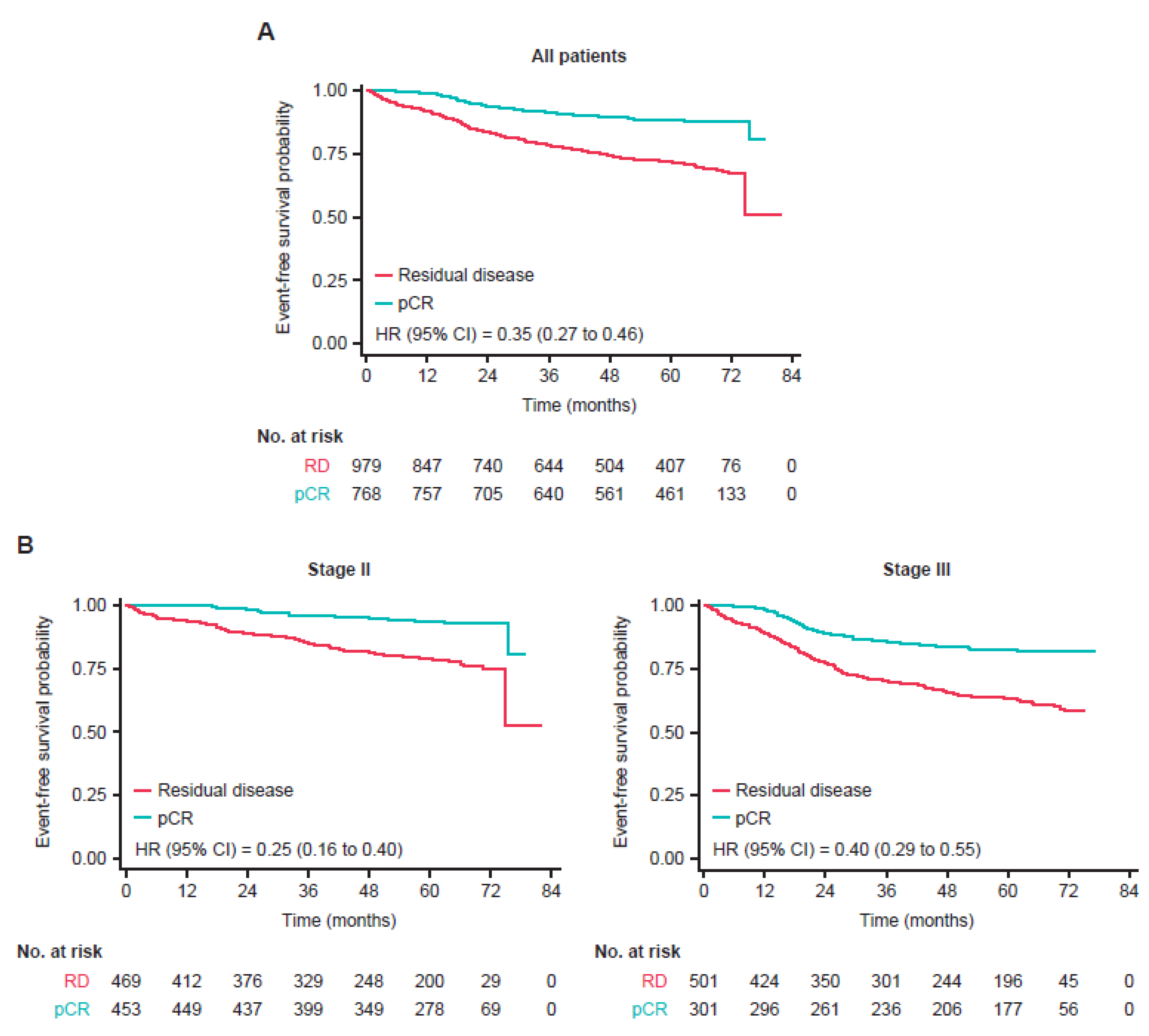
3.2. Event-Free Survival
3.3. Type of Recurrence
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Untch, M.; Fasching, P.A.; Konecny, G.E.; Hasmüller, S.; Lebeau, A.; Kreienberg, R.; Camara, O.; Müller, V.; du Bois, A.; Kühn, T.; et al. Pathologic complete response after neoadjuvant chemotherapy plus trastuzumab predicts favorable survival in human epidermal growth factor receptor 2-overexpressing breast cancer: Results from the TECHNO trial of the AGO and GBG study groups. J. Clin. Oncol. 2011, 29, 3351–3357. [Google Scholar] [CrossRef] [PubMed]
- Cortazar, P.; Zhang, L.; Untch, M.; Mehta, K.; Costantino, J.P.; Wolmark, N.; Bonnefoi, H.; Cameron, D.; Gianni, L.; Valagussa, P.; et al. Pathological complete response and long-term clinical benefit in breast cancer: The CTNeoBC pooled analysis. Lancet 2014, 384, 164–172. [Google Scholar] [CrossRef] [Green Version]
- de Azambuja, E.; Holmes, A.P.; Piccart-Gebhart, M.; Holmes, E.; Di Cosimo, S.; Swaby, R.F.; Untch, M.; Jackisch, C.; Lang, I.; Smith, I.; et al. Lapatinib with trastuzumab for HER2-positive early breast cancer (NeoALTTO): Survival outcomes of a randomised, open-label, multicentre, phase 3 trial and their association with pathological complete response. Lancet Oncol. 2014, 15, 1137–1146. [Google Scholar] [CrossRef] [Green Version]
- Gianni, L.; Eiermann, W.; Semiglazov, V.; Lluch, A.; Tjulandin, S.; Zambetti, M.; Moliterni, A.; Vazquez, F.; Byakhov, M.; Lichinitser, M.; et al. Neoadjuvant and adjuvant trastuzumab in patients with HER2-positive locally advanced breast cancer (NOAH): Follow-up of a randomised controlled superiority trial with a parallel HER2-negative cohort. Lancet Oncol. 2014, 15, 640–647. [Google Scholar] [CrossRef]
- Schneeweiss, A.; Chia, S.; Hickish, T.; Harvey, V.; Eniu, A.; Waldron-Lynch, M.; Eng-Wong, J.; Kirk, S.; Cortés, J. Long-term efficacy analysis of the randomised, phase II TRYPHAENA cardiac safety study: Evaluating pertuzumab and trastuzumab plus standard neoadjuvant anthracycline-containing and anthracycline-free chemotherapy regimens in patients with HER2-positive early breast cancer. Eur. J. Cancer 2018, 89, 27–35. [Google Scholar] [PubMed]
- Loibl, S.; Untch, M.; Buyse, M.; Robidoux, A.; Gianni, L.; Schneeweiss, A.; Conte, P.; Piccart, M.; Bonnefoi, H.; Jackisch, C.; et al. Pathologic complete response (pCR) and prognosis following neoadjuvant chemotherapy plus anti-HER2 therapy of HER2-positive early breast cancer (EBC). Cancer Res. 2020, 80, P5–06. [Google Scholar] [CrossRef]
- Spring, L.M.; Fell, G.; Are, A.; Sharma, C.; Greenup, R.; Reynolds, K.L.; Smith, B.L.; Alexander, B.; Moy, B.; Isakoff, S.J.; et al. Pathologic complete response after neoadjuvant chemotherapy and impact on breast cancer recurrence and survival: A comprehensive meta-analysis. Clin. Cancer Res. 2020, 26, 2838–2848. [Google Scholar] [CrossRef]
- Cardoso, F.; Kyriakides, S.; Ohno, S.; Penault-Llorca, F.; Poortmans, P.; Rubio, I.T.; Zackrisson, S.; Senkus, E.; ESMO Guidelines Committee. Early breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 1674. [Google Scholar] [CrossRef] [Green Version]
- Ditsch, N.; Untch, M.; Thill, M.; Müller, V.; Janni, W.; Albert, U.-S.; Bauerfeind, I.; Blohmer, J.; Budach, W.; Dall, P.; et al. AGO recommendations for he diagnosis and treatment of patients with early breast cancer: Update 2019. Breast Care 2019, 14, 224–245. [Google Scholar] [CrossRef] [Green Version]
- National Comprehensive Cancer Network: NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Breast Cancer.Version 7.2021. 23 August 2021. Available online: https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf (accessed on 25 July 2022).
- Loibl, S.; Poortmans, P.; Morrow, M.; Denkert, C.; Curigliano, G. Breast cancer. Lancet 2021, 397, 1750–1769. [Google Scholar] [CrossRef]
- Thomssen, C.; Balic, M.; Harbeck, N.; Gnant, M.S. Gallen/Vienna 2021: A brief summary of the consensus discussion on customizing therapies for women with early breast cancer. Breast Care 2021, 16, 135–143. [Google Scholar] [CrossRef] [PubMed]
- von Minckwitz, G.; Huang, C.-S.; Mano, M.S.; Loibl, S.; Mamounas, E.P.; Untch, M.; Wolmark, N.; Rastogi, P.; Schneeweiss, A.; Redondo, A.; et al. Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N. Engl. J. Med. 2019, 380, 617–628. [Google Scholar] [CrossRef] [PubMed]
- Gianni, L.; Pienkowski, T.; Im, Y.-H.; Roman, L.; Tseng, L.-M.; Liu, M.-C.; Lluch, A.; Staroslawska, E.; de la Haba-Rodriguez, J.; Im, S.-A.; et al. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): A randomised multicentre, open-label, phase 2 trial. Lancet Oncol. 2012, 13, 25–32. [Google Scholar] [CrossRef]
- Ismael, G.; Hegg, R.; Muehlbauer, S.; Heinzmann, D.; Lum, B.; Kim, S.-B.; Pienkowski, T.; Lichinitser, M.; Semiglazov, V.; Melichar, B.; et al. Subcutaneous versus intravenous administration of (neo)adjuvant trastuzumab in patients with HER2-positive, clinical stage I-III breast cancer (HannaH study): A phase 3, open-label, multicentre, randomised trial. Lancet Oncol. 2012, 13, 869–878. [Google Scholar] [CrossRef]
- Schneeweiss, A.; Chia, S.; Hickish, T.; Harvey, V.; Eniu, A.; Hegg, R.; Tausch, J.; Seo, J.H.; Tsai, Y.-F.; Ratnayake, J.; et al. Pertuzumab plus trastuzumab in combination with standard neoadjuvant anthracycline-containing and anthracycline-free chemotherapy regimens in patients with HER2-positive early breast cancer: A randomized phase II cardiac safety study (TRYPHAENA). Ann. Oncol. 2013, 24, 2278–2284. [Google Scholar] [CrossRef] [PubMed]
- Gianni, L.; Pienkowski, T.; Im, Y.-H.; Tseng, L.-M.; Liu, M.-C.; Lluch, A.; Staroslawska, E.; de la Haba-Rodriguez, J.; Im, S.A.; Pedrini, J.L.; et al. 5-year analysis of neoadjuvant pertuzumab and trastuzumab in patients with locally advanced, inflammatory, or early-stage HER2-positive breast cancer (NeoSphere): A multicentre, open-label, phase 2 randomised trial. Lancet Oncol. 2016, 17, 791–800. [Google Scholar] [CrossRef]
- Hurvitz, S.A.; Martin, M.; Symmans, W.F.; Jung, K.H.; Huang, C.-S.; Thompson, A.M.; Harbeck, N.; Valero, V.; Stroyakovskiy, D.; Wildiers, H.; et al. Neoadjuvant trastuzumab, pertuzumab, and chemotherapy versus trastuzumab emtansine plus pertuzumab in patients with HER2-positive breast cancer (KRISTINE): A randomised, open-label, multicentre, phase 3 trial. Lancet Oncol. 2018, 19, 115–126. [Google Scholar] [CrossRef]
- Swain, S.M.; Ewer, M.S.; Viale, G.; Delaloge, S.; Ferrero, J.-M.; Verrill, M.; Colomer, R.; Vieira, C.; Werner, T.L.; Douthwaite, H.; et al. Pertuzumab, trastuzumab, and standard anthracycline- and taxane-based chemotherapy for the neoadjuvant treatment of patients with HER2-positive localized breast cancer (BERENICE): A phase II, open-label, multicenter, multinational cardiac safety study. Ann. Oncol. 2018, 29, 646–653. [Google Scholar] [CrossRef]
- Hurvitz, S.A.; Martin, M.; Jung, K.H.; Huang, C.-S.; Harbeck, N.; Valero, V.; Stoyakovskiy, D.; Wildiers, H.; Campone, M.; Boileau, J.-F.; et al. Neoadjuvant trastuzumab emtansine and pertuzumab in human epidermal growth factor receptor 2-positive breast cancer: Three-year outcomes from the phase III KRISTINE study. J. Clin. Oncol. 2019, 37, 2206–2216. [Google Scholar] [CrossRef]
- Jackisch, C.; Stroyakovskiy, D.; Pivot, X.; Ahn, J.S.; Melichar, B.; Chen, S.-C.; Meyenberg, C.; Al-Sakaff, N.; Heinzmann, D.; Hegg, R. Subcutaneous vs intravenous trastuzumab for patients with ERBB2-positive early breast cancer: Final analysis of the HannaH phase 3 randomized clinical trial. JAMA. Oncol. 2019, 5, e190339. [Google Scholar] [CrossRef]
- von Minckwitz, G.; Procter, M.; de Azambuja, E.; Zardavas, D.; Benyunes, M.; Viale, G.; Suter, T.; Arahmani, A.; Rouchet, N.; Clark, E.; et al. Adjuvant pertuzumab and trastuzumab in early HER2-positive breast cancer. N. Engl. J. Med. 2017, 377, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Piccart, M.; Procter, M.; Fumagalli, D.; de Azambuja, E.; Clark, E.; Ewer, M.S.; Restuccia, E.; Jerusalem, G.; Dent, S.; Reaby, L.; et al. Adjuvant pertuzumab and trastuzumab in early HER2-positive breast cancer in the APHINITY trial: 6 years’ follow-up. J. Clin. Oncol. 2021, 39, 1448–1457. [Google Scholar] [CrossRef] [PubMed]
- Dang, C.; Ewer, M.S.; Delaloge, S.; Ferrero, J.-M.; Colomer, R.; de la Cruz-Merino, L.; Werner, T.L.; Dadswell, K.; Verrill, M.; Eiger, D.; et al. BERENICE final analysis: Cardiac safety study of neoadjuvant pertuzumab, trastuzumab, and chemotherapy followed by adjuvant pertuzumab and trastuzumab in HER2-positive early breast cancer. Cancers 2022, 14, 2596. [Google Scholar] [CrossRef] [PubMed]
- Hajage, D.; Chauvet, G.; Belin, L.; Lafourcade, A.; Tubach, F.; De Rycke, Y. Closed-form variance estimator for weighted propensity score estimators with survival outcome. Biom. J. 2018, 60, 1151–1163. [Google Scholar] [CrossRef] [PubMed]
- O’Shaughnessy, J.; Robert, N.; Annavarapu, S.; Zhou, J.; Sussell, J.; Cheng, A.; Fung, A. Recurrence rates in patients with HER2+ breast cancer who achieved a pathological complete response after neoadjuvant pertuzumab plus trastuzumab followed by adjuvant trastuzumab: A real-world evidence study. Breast Cancer Res. Treat. 2021, 187, 903–913. [Google Scholar] [CrossRef]
- Bines, J.; Clark, E.; Barton, C.; Restuccia, E.; Procter, M.; Sonnenblick, A.; Fumagalli, D.; Parlier, D.; Arahmani, A.; Baselga, J.; et al. Patient-reported function, health-related quality of life, and symptoms in APHINITY: Pertuzumab plus trastuzumab and chemotherapy in HER2-positive early breast cancer. Br. J. Cancer 2021, 125, 38–47. [Google Scholar] [CrossRef]
- Weiss, A.; Campbell, J.; Ballman, K.V.; Sikov, W.M.; Carey, L.A.; Hwang, E.S.; Poppe, M.M.; Partridge, A.H.; Ollila, D.W.; Golshan, M. Factors associated with nodal pathologic complete response among breast cancer patients treated with neoadjuvant chemotherapy: Results of CALGB 40601 (HER2+) and 40603 (triple-negative) (Alliance). Ann. Surg. Oncol. 2021, 28, 5960–5971. [Google Scholar] [CrossRef]


| Characteristic, n (%) | Overall (n = 1763) | H→H (n = 703) | PH→H (n = 439) | PH→PH (n = 621) |
|---|---|---|---|---|
| Age (years) | ||||
| <40 | 314 (17.8) | 115 (16.4) | 73 (16.6) | 126 (20.3) |
| 40−60 | 1302 (73.9) | 531 (75.5) | 336 (76.5) | 435 (70.0) |
| >60 | 147 (8.3) | 57 (8.1) | 30 (6.8) | 60 (9.7) |
| Clinical stage | ||||
| I | 22 (1.2) | 21 (3.0) | 0 | 1 (0.2) |
| II | 933 (52.9) | 285 (40.5) | 204 (46.5) | 444 (71.5) |
| III | 807 (45.8) | 397 (56.5) | 235 (53.5) | 175 (28.2) |
| Unknown | 1 (0.1) | 0 | 0 | 1 (0.2) |
| Hormone receptor status | ||||
| Negative | 783 (44.4) | 328 (46.7) | 223 (50.8) | 232 (37.4) |
| Positive | 963 (54.6) | 372 (52.9) | 215 (49.0) | 376 (60.5) |
| Unknown | 17 (1.0) | 3 (0.4) | 1 (0.2) | 13 (2.1) |
| Tumor stage at study entry | ||||
| T1 | 72 (4.1) | 43 (6.1) | 0 | 29 (4.7) |
| T2 | 926 (52.5) | 304 (43.2) | 201 (45.8) | 421 (67.8) |
| T3 | 406 (23.0) | 138 (19.6) | 144 (32.8) | 124 (20.0) |
| T4 | 354 (20.1) | 217 (30.9) | 91 (20.7) | 46 (7.4) |
| Unknown | 5 (0.3) | 1 (0.1) | 3 (0.7) | 1 (0.2) |
| Clinical nodal stage at study entry | ||||
| Node-positive | 536 (30.4) | 164 (23.3) | 128 (29.2) | 244 (39.3) |
| Node-negative | 1206 (68.4) | 538 (76.5) | 308 (70.2) | 360 (58.0) |
| Unknown | 21 (1.2) | 1 (0.1) | 3 (0.7) | 17 (2.7) |
| n (%) | pCR | Residual Disease | Total |
|---|---|---|---|
| Tumor stage | |||
| T1 | 36 (50.0) | 36 (50.0) | 72 |
| T2 | 447 (48.3) | 479 (51.7) | 926 |
| T3 | 177 (43.6) | 229 (56.4) | 406 |
| T4 | 111 (31.4) | 243 (68.6) | 354 |
| Unknown | 2 (40.0) | 3 (60.0) | 5 |
| Clinical stage | |||
| 0 | 0 (0) | 1 (100.0) | 1 |
| I | 11 (50.0) | 11 (50.0) | 22 |
| II | 456 (48.9) | 477 (51.1) | 933 |
| III | 306 (37.9) | 501 (62.1) | 807 |
| Hormone receptor status | |||
| Negative | 434 (55.4) | 349 (44.6) | 783 |
| Positive | 331 (34.4) | 632 (65.6) | 963 |
| Unknown | 8 (47.1) | 9 (52.9) | 17 |
| Treatment modality | |||
| H→H | 236 (33.6) | 467 (66.4) | 703 |
| PH→H | 185 (42.1) | 254 (57.9) | 439 |
| PH→PH | 352 (56.7) | 269 (43.3) | 621 |
| All Patients, Regardless of Pathological Complete Response Status | ||||||
| H→H (n = 703) | PH→H (n = 439) | PH→PH (n = 621) | ||||
| Patients with EFS event, % | 217 (30.9) | 79 (18.0) | 53 (8.5) | |||
| Patients without EFS event, % | 486 (69.1) | 360 (82.0) | 568 (91.5) | |||
| Hazard ratio versus H→H (95% CI) | 0.56 (0.43–0.73) | 0.36 (0.26–0.49) | ||||
| Hazard ratio versus PH→H (95% CI) | 0.67 (0.47–0.96) | |||||
| pCR | Residual Disease | |||||
| H→H (n = 236) | PH→H (n = 185) | PH→PH (n = 352) | H→H (n = 467) | PH→H (n = 254) | PH→PH (n = 269) | |
| Patients with EFS event, % | 41 (17.4) | 19 (10.3) | 20 (5.7) | 176 (37.7) | 60 (23.6) | 33 (12.3) |
| Patients without EFS event, % | 195 (82.6) | 166 (89.7) | 332 (94.3) | 291 (62.3) | 194 (76.4) | 236 (87.7) |
| Time to event (months) | ||||||
| Hazard ratio versus H→H (95% CI) | 0.59 (0.34–1.01) | 0.46 (0.26–0.82) | 0.60 (0.45–0.81) | 0.43 (0.30–0.63) | ||
| Hazard ratio versus PH→H (95% CI) | 0.85 (0.44–1.65) | 0.75 (0.49–1.16) | ||||
| 4-Year Event-Free Survival Rate in Patients with pCR | |||
| H→H (n = 236) | PH→H (n = 185) | PH→PH (n = 352) | |
| Patients remaining at risk, n | 179 | 155 | 219 |
| 4-year event-free survival rate, % (95% CI) | 86 (81–89) | 90 (85–94) | 95 (92–97) |
| 4-Year Event-Free Survival Rate in Patients with Residual Disease | |||
| H→H (n = 467) | PH→H (n = 254) | PH→PH (n = 269) | |
| Patients remaining at risk, n | 251 | 176 | 107 |
| 4-year event-free survival rate, % (95% CI) | 64 (59–68) | 80 (75–85) | 87 (82–91) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Swain, S.M.; Macharia, H.; Cortes, J.; Dang, C.; Gianni, L.; Hurvitz, S.A.; Jackisch, C.; Schneeweiss, A.; Slamon, D.; Valagussa, P.; et al. Event-Free Survival in Patients with Early HER2-Positive Breast Cancer with a Pathological Complete Response after HER2-Targeted Therapy: A Pooled Analysis. Cancers 2022, 14, 5051. https://doi.org/10.3390/cancers14205051
Swain SM, Macharia H, Cortes J, Dang C, Gianni L, Hurvitz SA, Jackisch C, Schneeweiss A, Slamon D, Valagussa P, et al. Event-Free Survival in Patients with Early HER2-Positive Breast Cancer with a Pathological Complete Response after HER2-Targeted Therapy: A Pooled Analysis. Cancers. 2022; 14(20):5051. https://doi.org/10.3390/cancers14205051
Chicago/Turabian StyleSwain, Sandra M., Harrison Macharia, Javier Cortes, Chau Dang, Luca Gianni, Sara A. Hurvitz, Christian Jackisch, Andreas Schneeweiss, Dennis Slamon, Pinuccia Valagussa, and et al. 2022. "Event-Free Survival in Patients with Early HER2-Positive Breast Cancer with a Pathological Complete Response after HER2-Targeted Therapy: A Pooled Analysis" Cancers 14, no. 20: 5051. https://doi.org/10.3390/cancers14205051







