Second Cancers in Classical Hodgkin Lymphoma and Diffuse Large B-Cell Lymphoma: A Systematic Review by the Fondazione Italiana Linfomi
Abstract
:Simple Summary
Abstract
1. Introduction
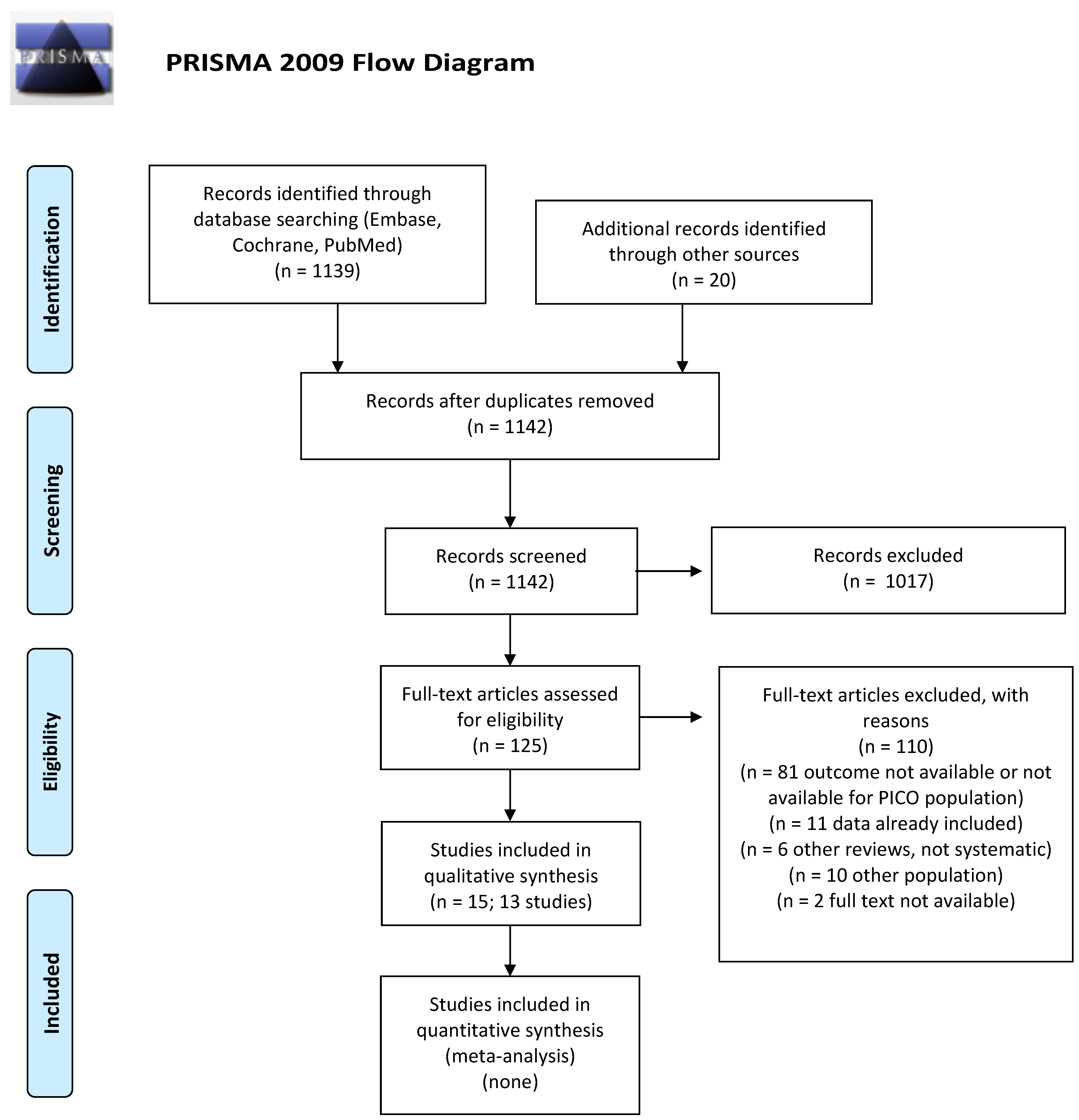
2. Materials and Methods
2.1. Study Identification
2.2. Eligibility Criteria
2.3. Study Selection and Data Extraction
2.4. Risk of Bias and Quality of Evidence Assessment
2.5. Data Synthesis
3. Results
3.1. What Is the Incidence of Second Cancers in cHL or DLBCL Long-Term Survivors after First- or Second-Line Treatments including ASCT?
3.2. Has the Incidence of Second Cancers in cHL or DLBCL Long-Term Survivors Who Underwent First or Second Line CHT and ASCT Changed with the Introduction of Modern RT?
3.3. Are Planned Follow-Up/Screening Schemes Effective in the Management and Early Diagnosis of Second Cancers in cHL or DLBCL Long-Term Survivors Treated, Regardless of the Type of CHT/RT (First and Second Line including ASCT)?
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sehn, L.H.; Salles, G. Diffuse large B-cell lymphoma. N. Engl. J. Med. 2021, 384, 842–858. [Google Scholar] [CrossRef]
- National Cancer Institute: Surveillance, Epidemiology, and End Results Program. Available online: https://seer.cancer.gov/statfacts/html/hodg.html (accessed on 11 November 2021).
- Bonadonna, G.; De Lena, M.; Banfi, A.; Lattuada, A. Secondary neoplasms in malignant lymphomas after intensive therapy. N. Engl. J. Med. 1973, 288, 1242–1243. [Google Scholar] [CrossRef]
- Minoia, C.; Bari, A.; Nassi, L.; Banzi, R.; Gerardi, C.; Lenti, V.; Calabrese, M.; Spina, M.; Guarini, A. Management of lymphoma survivor patients in Italy: An evaluation by Fondazione Italiana Linfomi. Tumori 2021, 107, 91–94. [Google Scholar] [CrossRef] [PubMed]
- Travis, L.B.; Rabkin, C.S.; Brown, L.M.; Allan, J.M.; Alter, B.P.; Ambrosone, C.B.; Begg, C.B.; Caporaso, N.; Chanock, S.; DeMichele, A.; et al. Cancer survivorship–genetic susceptibility and second primary cancers: Research strategies and recommendations. J. Natl. Cancer Inst. 2006, 98, 15–25. [Google Scholar] [CrossRef]
- Bessell, E.M.; Bouliotis, G.; Armstrong, S.; Baddeley, J.; Haynes, A.P.; O’Connor, S.; Nichols-Elliott, H.; Bradley, M. Long-term survival after treatment for Hodgkin’s disease (1973–2002): Improved survival with successive 10-years cohorts. Br. J. Cancer 2012, 107, 531–536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coiffier, B.; Lepage, E.; Brière, J.; Herbrecht, R.; Tilly, H.; Bouabdallah, R.; Morel, P.; Van Den Neste, E.; Salles, G.; Gaulard, P.; et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large B-cell lymphoma. N. Engl. J. Med. 2002, 346, 235–242. [Google Scholar] [CrossRef]
- Fisher, R.I.; Gaynor, E.R.; Dahlberg, S.; Oken, M.M.; Grogan, T.M.; Mize, E.M.; Glick, J.H.; Coltman, C.A.; Miller, T.P. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin’s lymphoma. N. Engl. J. Med. 1993, 328, 1002–1006. [Google Scholar] [CrossRef] [PubMed]
- Duggan, D.B.; Petroni, G.R.; Johnson, J.L.; Glick, J.H.; Fisher, R.I.; Connors, J.M.; Canellos, G.P.; Peterson, B.A. Randomized comparison of ABVD and MOPP/ABV hybrid for the treatment of advanced Hodgkin’s disease: Report of an Intergroup trial. J. Clin. Oncol. 2003, 21, 607–614. [Google Scholar] [CrossRef] [PubMed]
- Specht, L.; Yahalom, J.; Illidge, T.; Berthelsn, A.K.; Constine, L.S.; Eich, H.T.; Girinsky, T.; Hoppe, R.T.; Mauch, P.; Mikhaeel, N.G.; et al. ILROG. Modern radiation therapy for Hodgkin lymphoma: Field and dose guidelines from the International Lymphoma Radiation Oncology Group (ILROG). Int. J. Radiat. Oncol. Biol. Phys. 2014, 89, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Yahalom, J.; Illidge, T.; Specht, L.; Hoppe, R.T.; Li, Y.; Tsang, R.; Wirth, A.; International Lymphoma Radiation Oncology Group. Modern radiation therapy for Hodgkin lymphomas: Field and dose guidelines from the International Lymphoma Radiation Oncology Group. Int. J. Radiat. Oncol. Biol. Phys. 2015, 92, 11–31. [Google Scholar] [CrossRef]
- Chiappella, A.; Martelli, M.; Angelucci, E.; Brusamolino, E.; Evangelista, A.; Carella, A.M.; Stelitano, C.; Rossi, G.; Balzarotti, M.; Merli, F.; et al. Rituximab-dose-dense chemotherapy with or without high-dose chemotherapy plus autologous stem-cell transplantation in high-risk diffuse large B-cell lymhpoma (DLCL04): Final results of a multi-center, open-label, randomized, controlled, phase 3 study. Lancet Oncol. 2017, 18, 1076–1088. [Google Scholar] [CrossRef]
- Van Leeuwen, F.E.; Ng, A.K. Long-term risk of second malignancies and cardiovascular disease after Hodgkin lymphoma treatment. Hematol. Am. Soc. Educ. Program 2016, 2016, 323–330. [Google Scholar] [CrossRef] [Green Version]
- Ciavarella, S.; Minoia, C.; Quinto, A.M.; Oliva, S.; Carbonara, S.; Cormio, C.; Cox, M.C.; Bravo, E.; Santoro, F.; Napolitano, M.; et al. Improving care provision for lymphoma long-term survivors. Clin. Lymphoma Myeloma Leuk. 2017, 17, e1–e9. [Google Scholar] [CrossRef] [PubMed]
- Gerardi, C.; Allocati, E.; Minoia, C.; Guarini, A.; Banzi, R. Long-term follow-up of classical Hodgkin lymphoma and diffuse large B-cell lymphoma survivors: Aims and methodological approach for Fondazione Italiana Linfomi systematic reviews. Cancers 2021, 13, 2976. [Google Scholar] [CrossRef]
- Minoia, C.; Gerardi, C.; Allocati, E.; Daniele, A.; De Sanctis, V.; Bari, A.; Guarini, A. The Impact of Healthy Lifestyles on Late Sequelae in Classical Hodgkin Lymphoma and Diffuse Large B-Cell Lymphoma Survivors. A Systematic Review by the Fondazione Italiana Linfomi. Cancers 2021, 13, 3135. [Google Scholar] [CrossRef]
- Franceschetti, S.; Annunziata, M.A.; Agostinelli, G.; Gerardi, C.; Allocati, E.; Minoia, C.; Guarini, A. Late Neurological and Cognitive Sequelae and Long-Term Monitoring of Classical Hodgkin Lymphoma and Diffuse Large B-Cell Lymphoma Survivors: A Systematic Review by the Fondazione Italiana Linfomi. Cancers 2021, 13, 3401. [Google Scholar] [CrossRef] [PubMed]
- Viviani, S.; Caccavari, V.; Gerardi, C.; Ramadan, S.; Allocati, E.; Minoia, C.; Guarini, A.; Di Russo, A. Male and Female Fertility: Prevention and Monitoring Hodgkin’ Lymphoma and Diffuse Large B-Cell Lymphoma Adult Survivors. A Systematic Review by the Fondazione Italiana Linfomi. Cancers 2021, 13, 2881. [Google Scholar] [CrossRef]
- Minoia, C.; Gerardi, C.; Allocati, E.; De Sanctis, V.; Franceschetti, S.; Viviani, S.; Annunziata, M.A.; Bari, A.; Skrypets, T.; Oliva, S.; et al. Late toxicities and long-term monitoring in classical Hodgkin lymphoma and diffuse large B-cell lymphoma survivors: A series of systematic reviews of the Fondazione Italiana Linfomi. Hematol. Oncol. 2021, 39 (Suppl. S2), 478–479. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA group. Preferred reported items for systematic reviews and meta-analysis. The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [Green Version]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. Br. Med. J. 2017, 358, j4008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Higgins, J.P.; Altman, D.G. The Cochrane Collaboration’s tool for assessing risk of bias. In Cochrane Handbook for Systematic Reviews of Interventions; Higgins, J.P., Green, S., Eds.; John Wiley and Sons: Hoboken, NJ, USA, 2010; pp. 194–202. [Google Scholar]
- Wells Ga, S.B.; O’connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Non-Randomised Studies in Meta-Analyses; Ottawa Health Research Institute: Ottawa, ON, Canada, 1999; Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 11 November 2021).
- Andre, M.P.E.; Carde, P.; Viviani, S.; Bellei, M.; Fortpied, C.; Hutchings, M.; Gianni, A.M.; Brice, P.; Casasnovas, O.; Gobbi, P.G.; et al. Long-term overall survival and toxicities of ABVD vs BEACOPP in advanced Hodgkin lymphoma: A pooled analysis of four randomized trials. Cancer Med. 2020, 9, 6565–6575. [Google Scholar] [CrossRef]
- Torok, J.A.; Wu, Y.; Prosnitz, L.R.; Kim, G.J.; Beaven, A.W.; Diehl, L.F.; Kelsey, C.R. Low-dose consolidation radiation therapy for early stage unfavourable Hodgkin lymphoma. Int. J. Radiat. Oncol. Biol. Phys. 2015, 92, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Engert, A.; Franklin, J.; Eich, H.T.; Brillant, C.; Sehlen, S.; Cartoni, C.; Herrmann, R.; Pfreundschuh, M.; Sieber MTesch, H.; Franke, A.; et al. Two cycles of doxorubicin, bleomycin, vinblastine, and dacarbazine plus extended-field radiotherapy is superior to radiotherapy alone in early favorable Hodgkin’s lymphoma: Final results of the GHSG HD7 trial. J. Clin. Oncol. 2007, 25, 3495–3502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schaapveld, M.; Aleman, B.M.P.; van Eggermond, A.M.; Janus, C.P.M.; Krol, A.D.G.; van del Maazen, R.W.M.; Roesink, J.; Raemaekers, J.M.M.; de Boer, J.P.; Zijlstra, J.M.; et al. Second cancer risk up to 40 years after treatment for Hodgkin’s lymphoma. N. Engl. J. Med. 2015, 373, 2499–2511. [Google Scholar] [CrossRef]
- Frontzek, F.; Ziepert, M.; Nickelsen, M.; Altmann, B.; Glass, B.; Haenel, M.; Truemper, L.; Held, G.; Bentz, M.; Borchmann, P.; et al. Rituximab plus high-dose chemotherapy (MegaCHOEP) or conventional chemotherapy (CHOEP14) in young, high-risk patients with aggressive B-cell lymphoma: 10-year follow-up of a randomized, open-label, phase 3 trial. Lancet Haematol. 2021, 8, e267–e277. [Google Scholar] [CrossRef]
- Coiffier, B.; Thieblemont, C.; Van Den Neste, E.; Lepeu, G.; Plantier, I.; Castaigne, S.; Lefort, S.; Marit, G.; Macro, M.; Sebban, C.; et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: A study by the Groupe d’Etudes des Lymphomes de l’Adulte. Blood 2010, 116, 2040–2045. [Google Scholar] [CrossRef] [PubMed]
- Castellino, A.; Chiappella, A.; LaPlant, B.R.; Pederson, L.D.; Gaidano, G.; Macon, W.R.; Inghirami, G.; Reeder, C.B.; Tucci, A.; King, R.L.; et al. Lenalidomide plus R-CHOP21 in newly diagnosed diffuse large B-cell lymphoma (DLBCL): Long-term follow-up results from a combined analysis from two phase 2 trials. Blood Cancer J. 2018, 8, 108–115. [Google Scholar] [CrossRef]
- Paudel, N.; Schulze, D.; Gentzler, R.D.; Evens, A.M.; Helenowski, I.; Dillehay, G.; Frankfurt, O.; Mehta, J.; Donnelly, E.D.; Gordon, L.I.; et al. Patterns of failure and survival outcomes after total lymphoid irradiation and high-dose chemotherapy with stem cell transplantation for relapsed or refractory classical Hodgkin lymphoma. Int. J. Radiat. Oncol. Biol. Phys. 2019, 104, 436–446. [Google Scholar] [CrossRef] [PubMed]
- Pingali, S.R.; Saliba, R.M.; Anderlini, P.; Hosing, C.; Khouri, I.; Alousi, A.M.; Niego, Y.; Qazilbash, M.H.; Champlin, R.; Popat, U.R. Age over Fifty-Five Years at Diagnosis Increases Risk of Second Malignancies after Autologous Transplantation for Patients with Hodgkin Lymphoma. Biol. Blood Marrow Transplant. 2017, 23, 1059–1063. [Google Scholar] [CrossRef] [Green Version]
- Sibon, D.; Morschhauser, F.; Resche-Rigon, M.; Ghez, D.; Dupuis, J.; Marçais, A.; Deau-Fischer, B.; Boubdallah, R.; Sebban, C.; Salles, G.; et al. Single or tandem autologous stem-cell transplantation for first-relapsed or refractory Hodgkin lymphoma: 10-year follow-up of the prospective H96 trial by the LYSA/SFGM-TC study group. Haematologica 2016, 101, 474–481. [Google Scholar] [CrossRef] [Green Version]
- Minn, A.Y.; Riedel, E.; Halpern, J.; Johnston, L.J.; Horning, S.J.; Hoppe, R.T.; Goodman, K.A. Long-term outcomes after high-dose therapy and autologous haematopoietic cell rescue for refractory/relapsed Hodgkin lymphoma. Br. J. Haematol. 2012, 159, 329–339. [Google Scholar] [CrossRef]
- Tarella, C.; Passera, R.; Magni, M.; Benedetti, F.; Rossi, A.; Gueli, A.; Patti, C.; Parvis, G.; Ciceri, F.; Gallamini, A.; et al. Risk factors for development of secondary malignancy after high-dose chemotherapy and autograft, with or without rituximab: A 20-year retrospective follow-up study in patients with lymphoma. J. Clin. Oncol. 2011, 29, 814–824. [Google Scholar] [CrossRef]
- Franklin, J.; Eichenauer, D.A.; Becker, I.; Monsef, I.; Engert, A. Optimization of chemotherapy and radiotherapy for untreated Hodgkin lymphoma patients with respect to second malignant neoplasms, overall and progression-free survival: Individual participant data-analysis (review). Cochrane Database Syst. Rev. 2017, 9, CD008814. [Google Scholar] [CrossRef]
- Patel, C.G.; Michaelson, E.; Chen, Y.H.; Silver, B.; Marcus, K.J.; Stevenson, M.A.; Mauch, P.; Ng, A.K. Reduced Mortality Risk in the Recent Era in Early-Stage Hodgkin Lymphoma Patients Treated With Radiation Therapy With or Without Chemotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 498–506. [Google Scholar] [CrossRef] [PubMed]
- LeMieux, M.H.; Solanki, A.A.; Mahmood, U.; Chmura, S.J.; Koshy, M. Risk of second malignancies in patients with early-stage classical Hodgkin’s lymphoma treated in a modern era. Cancer Med. 2015, 4, 513–518. [Google Scholar] [CrossRef]
- De Bruin, M.L.; Sparidans, J.; van’t Veer, M.B.; Noordijk, E.M.; Louwman, M.W.J.; Zijlstra, J.M.; van den Berg, H.; Russell, N.S.; Broeks, A.; Baaijens, M.H.A.; et al. Breast Cancer Risk in Female Survivors of Hodgkin’s Lymphoma: Lower Risk After Smaller Radiation Volumes. J. Clin. Oncol. 2009, 27, 4239–4246. [Google Scholar] [CrossRef]
- Conway, J.L.; Connors, J.M.; Tyldesley, S.; Savage, K.J.; Campbell, B.A.; Zheng, Y.Y.; Hamm, J.; Pickles, T. Secondary Breast Cancer Risk by Radiation Volume in Women with Hodgkin Lymphoma. Int. J. Radiat. Oncol. Biol. Phys. 2017, 97, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Franklin, J.; Paus, M.D.; Pluetschow, A.; Specht, L. Chemotherapy, radiotherapy and combined modality for Hodgkin’s disease, with emphasis on second cancer risk (Review). Cochrane Database Syst. Rev. 2005, 4, CD003187. [Google Scholar] [CrossRef]
- Diller, L.; Medeiros Nancarrow, C.; Shaffer, K.; Matulonis, U.; Mauch, P.; Neuberg, D.; Tarbell, N.J.; Litman, H.; Garber, J. Breast cancer screening in women previously treated for Hodgkin’s disease: A prospective cohort study. J. Clin. Oncol. 2002, 20, 2085–2091. [Google Scholar] [CrossRef]
- Kwong, A.; Hancock, S.L.; Bloom, J.R.; Pal, S.; Birdwell, R.L.; Mariscal, C.; Ikeda, D.M. Mammographic screening in women at increased risk of breast cancer after treatment of Hodgkin’s disease. Breast. J. 2008, 14, 39–48. [Google Scholar] [CrossRef]
- Lee, L.; Pintilie, M.; Hodgson, D.C.; Goss, P.E.; Crump, M. Screening mammography for young women treated with supradiaphragmatic radiation for Hodgkin’s lymphoma. Ann. Oncol. 2008, 19, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Howell, S.J.; Searle, C.; Goode, V.; Gardener, T.; Linton, K.; Cowan, R.A.; Harris, M.A.; Hopwood, P.; Swindell, R.; Norman, A.; et al. The UK national breast cancer screening programme for survivors of Hodgkin lymphoma detects breast cancer at an early stage. Br. J. Cancer 2009, 101, 582–588. [Google Scholar] [CrossRef]
- Elkin, E.B.; Klem, M.L.; Gonzales, A.M.; Ishill, N.M.; Hodgson, D.; Ng, A.K.; Marks, L.B.; Weidhaas, J.; Freedman, G.M.; Miller, R.C.; et al. Characteristics and outcomes of breast cancer in women with and without a history of radiation for Hodgkin’s lymphoma: A multi-institutional, matched cohort study. J. Clin. Oncol. 2011, 29, 2466–2473. [Google Scholar] [CrossRef] [PubMed]
- Mariscotti, G.; Durando, M.; Ghione, G.; Luparia, A.; Regini, E.; Alfieri, C.; Campanino, P.P.; Gavarotti, P.; Brignardello, E.; Gandini, G. Breast cancer surveillance in patients treated by radiotherapy for Hodgkin’s lymphoma. Radiol. Med. 2013, 118, 401–414. [Google Scholar] [CrossRef]
- Ng, A.K.; Garber, J.E.; Diller, L.R.; Birdwell, R.L.; Feng, Y.; Neuberg, D.S.; Silver, B.; Fisher, D.C.; Marcus, K.J.; Mauch, P.M. Prospective study of the efficacy of breast magnetic resonance imaging and mammographic screening in survivors of Hodgkin lymphoma. J. Clin. Oncol. 2013, 31, 2282–2288. [Google Scholar] [CrossRef]
- Andre, M.; Fortpied, C.; Viviani, S.; Bellei, M.; Carde, P.; Hutchings, M.; Gianni, A.; Brice, P.; Casasnovas, O.; Gobbi, P.; et al. Overall survival impact of BEACOPP vs ABVD in advanced Hodgkin lymphoma: A pooled analysis of four randomized trials. Haematologica 2016, 101, 19. [Google Scholar]
- Sasse, S.; Bröckelmann, P.J.; Goergen, H.; Plütschow, A.; Müller, H.; Kreissl, S.; Buerkle, C.; Borchmann, S.; Fuchs, M.; Borchmann, P.; et al. Long-term follow-up of contemporary treatment in early-stage Hodgkin lymphoma: Updated analysis of the German Hodgkin Study Group HD7, HD8, HD10, and HD11 trials. J. Clin. Oncol. 2017, 35, 1999–2007. [Google Scholar] [CrossRef] [PubMed]
- Engert, A.; Schiller, P.; Josting, A.; Herrmann, R.; Koch, P.; Sieber, M.; Boissevain, F.; de Wit, M.; Mezger, J.; Dühmke, E.; et al. Involved-field radiotherapy is equally effective and less toxic compared with extended-field radiotherapy after four cycles of chemotherapy in patients with early-stage unfavorable Hodgkin’s lymphoma: Results of the HD8 trial of the German Hodgkin’s Lymphoma Study Group. J. Clin. Oncol. 2003, 21, 3601–3608. [Google Scholar] [CrossRef] [PubMed]
- Engert, A.; Plütschow, A.; Eich, H.T.; Lohri, A.; Dörken, B.; Borchmann, P.; Berger, B.; Greil, R.; Willborn, K.C.; Wilhelm, M.; et al. Reduced treatment intensity in patients with early-stage Hodgkin’s lymphoma. N. Engl. J. Med. 2010, 363, 640–652. [Google Scholar] [CrossRef] [Green Version]
- Eich, H.T.; Diehl, V.; Görgen, H.; Pabst, T.; Markova, J.; Debus, J.; Ho, A.; Dörken, B.; Rank, A.; Grosu, A.L.; et al. Intensified chemotherapy and dose-reduced involved-field radiotherapy in patients with early unfavorable Hodgkin’s lymphoma: Final analysis of the German Hodgkin Study Group HD11 trial. J. Clin. Oncol. 2010, 28, 4199–4206. [Google Scholar] [CrossRef] [PubMed]
- Frontzek, F.; Ziepert, M.; Nickelsen, M.; Altmann, B.; Glass, B.; Haenel, M.; Truemper, L.; Held, G.; Bentz, M.; Borchmann, P.; et al. Conventional chemotherapy (R-CHOEP) vs high-dose immunochemotherapy (R-megaCHOEP) in younger patients with high-risk aggressive B-cell lymphoma: 10-year long-term follow-up of a German Lymphoma Alliance (GLA) Study. Blood 2019, 134, 1589. [Google Scholar] [CrossRef]
- Bloom, J.R.; Stewart, S.L.; Hancock, S.L. Breast Cancer Screening in Women Surviving Hodgkin Disease. Am. J. Clin. Oncol. 2006, 29, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Bonadonna, G.; Zucali, R.; Monfardini, S.; De Lena, M.; Uslenghi, C. Combination chemotherapy of Hodgkin’s disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide versus MOPP. Cancer 1975, 36, 252–259. [Google Scholar] [CrossRef]
- Valagussa, P.; Santoro, A.; Kenda, R.; Fossati Bellani, F.; Franchi, F.; Banfi, A.; Rilke, F.; Bonadonna, G. Second malignancies in Hodgkin’s disease: A complication of certain forms of treatment. Br. Med. J. 1980, 280, 216–219. [Google Scholar] [CrossRef] [Green Version]
- Brusamolino, E.; Anselmo, A.P.; Klersy, C.; Santoro, M.; Orlandi, E.; Pagnucco, G.; Lunghi, F.; Maurizi-Enrici, R.; Baroni, C.D.; Lazzarino, M.; et al. The risk of acute leukemia in patients treated for Hodgkin’s disease is significantly higher after combined modality programs than after chemotherapy alone and is correlated with the extent of radiotherapy and type and duration of chemotherapy: A case-control study. Haematologica 1998, 83, 812–823. [Google Scholar]
- Canellos, G.P.; Anderson, J.R.; Propert, K.J.; Nissen, N.; Cooper, M.R.; Henderson, E.S.; Green, M.R.; Gottlieb, A.; Peterson, B.A. Chemotherapy of advanced Hodgkin’s disease with MOPP, ABVD, or MOPP alternating with ABVD. N. Engl. J. Med. 1992, 327, 1478–1484. [Google Scholar] [CrossRef] [PubMed]
- Casasnovas, R.O.; Bouabdallah, R.; Brice, P.; Lazarovici, J.; Ghesquieres, H.; Stamatoullas, A.; Dupuis, J.; Gac, A.C.; Gastinne, T.; Joly, B.; et al. PET-adapted treatment for newly diagnosed advanced Hodgkin lymphoma (AHL2011): A randomized, multicentre, non-inferiority, phase 3 study. Lancet Haematol. 2019, 20, 202–215. [Google Scholar] [CrossRef]
- Gallamini, A.; Tarella, C.; Viviani, S.; Rossi, A.; Patti, C.; Mulé, A.; Picardi, M.; Romano, A.; Cantonetti, M.; La Nasa, G.; et al. Early chemotherapy intensification with escalated BEACOPP in patients with advanced-stage Hodgkin lymphoma with a positive interim positron emission tomography/computed tomography scan after two ABVD cycles: Long-term results of the GITIL/FIL HD 0607 trial. J. Clin. Oncol. 2018, 36, 454–462. [Google Scholar] [CrossRef]
- Hodgson, D.C.; Koh, E.S.; Tran, T.H.; Heydarian, M.; Tsang, R.; Pintilie, M.; Xu, T.; Huang, L.; Sachs, R.K.; Brenner, D.J. Individualized estimates of second cancer risks after contemporary radiation therapy for Hodgkin lymphoma. Cancer 2007, 110, 2576–2586. [Google Scholar] [CrossRef]
- Koh, E.S.; Tran, T.H.; Heydarian, M.; Sachs, R.K.; Tsang, R.W.; Brenner, D.J.; Pintilie, M.; Xu, T.; Chung, J.; Paul, N.; et al. A comparison of mantle versus involved-field radiotherapy for Hodgkin’s lymphoma: Reduction in normal tissue dose and second cancer risk. Radiat. Oncol. 2007, 2, 13. [Google Scholar] [CrossRef] [Green Version]
- Schneider, U.; Sumila, M.; Robotka, J.; Weber, D.; Gruber, G. Radiation-induced second malignancies after involved-node radiotherapy with deep-inspiration breath-hold technique for early stage Hodgkin Lymphoma: A dosimetric study. Radiat. Oncol. 2014, 9, 58. [Google Scholar] [CrossRef] [Green Version]
- Mazonakis, M.; Lyraraki, E.; Damilakis, J. Second cancer risk assessments after involved-site radiotherapy for mediastinal Hodgkin lymphoma. Med. Phys. 2017, 44, 3866–3874. [Google Scholar] [CrossRef] [PubMed]
- Campbell, B.A.; Hornby, C.; Cunninghame, J.; Burns, M.; MacManus, M.; Ryan, G.; Lau, E.; Seymour, J.F.; Wirth, A. Minimising critical organ irradiation in limited stage Hodgkin lymphoma: A dosimetric study of the benefit of involved node radiotherapy. Ann. Oncol. 2012, 23, 1259–1266. [Google Scholar] [CrossRef]
- Kourinou, K.M.; Mazonakis, M.; Lyraraki, E.; Papadaki, H.A.; Damilakis, J. Probability of carcinogenesis due to involved field and involved site radiation therapy techniques for supra- and infradiaphragmatic Hodgkin’s disease. Phys. Med. 2019, 57, 100–106. [Google Scholar] [CrossRef]
- Aznar, M.C.; Maraldo, M.V.; Schut, D.A.; Lundemann, M.; Brodin, N.P.; Vogelius, I.R.; Berthelsen, A.K.; Specht, L.; Petersen, P.M. Minimizing late effects for patients with mediastinal Hodgkin lymphoma: Deep inspiration breath-hold, IMRT, or both? Int. J. Radiat. Oncol. Biol. Phys. 2015, 92, 169–174. [Google Scholar] [CrossRef]
- Maraldo, M.V.; Specht, L. A decade of comparative dose planning studies for early-stage Hodgkin lymphoma: What can we learn? Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 1126–1135. [Google Scholar] [CrossRef] [PubMed]
- Maraldo, M.V.; Brodin, N.P.; Aznar, M.C.; Vogelius, I.R.; Munck af Rosenschöld, P.; Petersen, P.M.; Specht, L. Estimated risk of cardiovascular disease and secondary cancers with modern highly conformal radiotherapy for early-stage mediastinal Hodgkin lymphoma. Ann. Oncol. 2013, 24, 2113–2118. [Google Scholar] [CrossRef]
- Timlin, C.; Loken, J.; Kruse, J.; Miller, R.; Schneider, U. Comparing second cancer risk for multiple radiotherapy modalities in survivors of hodgkin lymphoma. Br. J. Radiol. 2021, 94, 20200354. [Google Scholar] [CrossRef] [PubMed]
- Filippi, A.R.; Ragona, R.; Piva, C.; Scafa, D.; Fiandra, C.; Fusella, M.; Giglioli, F.R.; Lohr, F.; Ricardi, U. Optimized volumetric modulated arc therapy versus 3D-CRT for early stage mediastinal Hodgkin lymphoma without axillary involvement: A comparison of second cancers and heart disease risk. Int. J. Radiat. Oncol. Biol. Phys. 2015, 92, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.C.; Johanson, S.; Peguret, N.; Cozzi, L.; Olsen, D.R. Predicted risk of radiation-induced cancers after involved field and involved node radiotherapy with or without intensity modulation for early-stage hodgkin lymphoma in female patients. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 490–497. [Google Scholar] [CrossRef]
- Cella, L.; Conson, M.; Pressello, M.C.; Molinelli, S.; Schneider, U.; Donato, V.; Orecchia, R.; Salvatore, M.; Pacelli, R. Hodgkin’s lymphoma emerging radiation treatment techniques: Trade-offs between late radio-induced toxicities and secondary malignant neoplasms. Radiat. Oncol. 2013, 8, 22. [Google Scholar] [CrossRef] [Green Version]
- Filippi, A.R.; Ragona, R.; Fusella, M.; Botticella, A.; Fiandra, C.; Ricardi, U. Changes in breast cancer risk associated with different volumes, doses, and techniques in female Hodgkin lymphoma patients treated with supra-diaphragmatic radiation therapy. Pract. Radiat. Oncol. 2013, 3, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Hill, D.A.; Gilbert, E.; Dores, G.M.; Gospodarowicz, M.; van Leeuwen, F.E.; Holowaty, E.; Glimelius, B.; Andersson, M.; Wiklund, T.; Lynch, C.F.; et al. Breast cancer risk following radiotherapy for Hodgkin lymphoma: Modification by other risk factors. Blood 2005, 106, 3358–3365. [Google Scholar] [CrossRef] [Green Version]
- Filippi, A.R.; Meregalli, S.; DIRusso, A.; Levis, M.; Ciammella, P.; Buglione, M.; Guerini, A.E.; De Marco, G.; De Sanctis, V.; Vagge, S.; et al. Fondazione Italiana Linfomi (FIL) Radiotherapy Committee. Fondazione Italiana Linfomi (FIL) expert consensus on the use of intensity-modulated and image-guided radiotherapy for Hodgkin’s lymphoma involving the mediastinum. Radiat. Oncol. 2020, 15, 62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manem, V.S.K.; Dhawan, A. Modelling recurrence and second cancer risks induced by proton therapy. Math. Med. Biol. 2018, 35, 347–361. [Google Scholar] [CrossRef]
- Horn, S.; Fournier-Bidoz, N.; Pernin, V.; Peurien, D.; Vaillant, M.; Dendale, R.; Fourquet, A.; Kirova, Y.M. Comparison of passive-beam proton therapy, helical tomotherapy and 3D conformal radiation therapy in Hodgkin’s lymphoma female patients receiving involved-field or involved site radiation therapy. Cancer Radiother. 2016, 20, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, D.C.; Gilbert, E.S.; Dores, G.M.; Schonfeld, S.J.; Lynch, C.F.; Storm, H.; Hall, P.; Langmark, F.; Pukkala, E.; Andersson, M.; et al. Long-Term Solid Cancer Risk Among 5-Year Survivors of Hodgkin’s Lymphoma. J. Clin. Oncol. 2007, 25, 1489–1497. [Google Scholar] [CrossRef] [Green Version]
- Dores, G.M.; Metayer, C.; Curtis, R.E.; Lynch, C.F.; Clarke, E.A.; Glimelius, B.; Storm, H.; Pukkala, E.; van Leeuwen, F.E.; Holowaty, E.J.; et al. Second malignant neoplasms among long-term survivors of Hodgkin’s disease: A population-based evaluation over 25 years. J. Clin. Oncol. 2002, 20, 3484–3494. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.; Yasui, Y.; Robison, L.L.; Birch, J.M.; Bogue, M.K.; Diller, L.; DeLaat, C.; Fossati-Bellani, F.; Morgan, E.; Oberlin, O.; et al. Late Effects Study Group. High risk of subsequent neoplasms continues with extended follow-up of childhood Hodgkin’s disease: Report from the Late Effects Study Group. J. Clin. Oncol. 2003, 21, 4386–4394. [Google Scholar] [CrossRef] [PubMed]
- Bröckelmann, P.J.; Eichenauer, D.A.; Jakob, T.; Follmann, M.; Engert, A.; Skoetz, N. Hodgkin Lymphoma in Adults. Dtsch. Arztebl. Int. 2018, 115, 535–540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ha, C.S.; Hodgson, D.C.; Advani, R.; Dabaja, B.S.; Dhakal, S.; Flowers, C.R.; Hoppe, B.S.; Mendenhall, N.P.; Metzger, M.L.; Plastaras, J.P.; et al. ACR appropriateness criteria follow-up of Hodgkin lymphoma. J. Am. Coll. Radiol. 2014, 11, 1026–1033.e3. [Google Scholar] [CrossRef]
- Hodgson, D.C. Hodgkin lymphoma: The follow-up of long-term survivors. Hematol. Oncol. Clin. N. Am. 2008, 22, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, D.C.; Grunfeld, E.; Gunraj, N.; Del Giudice, L. A population-based study of follow-up care for Hodgkin lymphoma survivors: Opportunities to improve surveillance for relapse and late effects. Cancer 2010, 116, 3417–3425. [Google Scholar] [CrossRef] [PubMed]
- Travis, L.B.; Gospodarowicz, M.; Curtis, R.E.; Clarke, E.A.; Andersson, M.; Glimelius, B.; Joensuu, T.; Lynch, C.F.; van Leeuwen, F.E.; Holowaty, E.; et al. Lung cancer following chemotherapy and radiotherapy for Hodgkin’s disease. J. Natl. Cancer Inst. 2002, 94, 182–192. [Google Scholar] [CrossRef]
- Das, P.; Ng, A.K.; Earle, C.C.; Mauch, P.M.; Kuntz, K.M. Computed tomography screening for lung cancer in Hodgkin’s lymphoma survivors: Decision analysis and cost-effectiveness analysis. Ann. Oncol. 2006, 17, 785–793. [Google Scholar] [CrossRef] [PubMed]
- Wattson, D.A.; Hunink, M.G.; DiPiro, P.J.; Das, P.; Hodgson, D.C.; Mauch, P.M.; Ng, A.K. Low-dose chest computed tomography for lung cancer screening among Hodgkin lymphoma survivors: A cost-effectiveness analysis. Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 344–353. [Google Scholar] [CrossRef]
- NCCN Clinical Practice Guidelines in Oncology. Survivorship. Version 3.2021. 23 August 2021. Available online: https://www.nccn.org/professionals/physician_gls/pdf/survivorship.pdf (accessed on 11 November 2021).
- Curry, S.J.; Krist, A.H.; Owens, D.K.; Barry, M.J.; Caughey, A.B.; Davidson, K.W.; Doubeni, C.A.; Epling, J.W., Jr.; Kemper, A.R.; Kubik, M.; et al. US Preventive Services Task Force. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA 2018, 320, 674–686. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Clinical Question | PICOs |
|---|---|
| PICO A: What is the incidence of SM in cHL or DLBCL long-term survivors after first or second line treatments? | P: population of cHL or DLBCL long-term survivors (≥5 years of disease/treatment free) aged ≥ 18 years at diagnosis I: chemotherapy (e.g., ABVD for cHL; R-CHOP for DLBCL), second-line chemotherapy and autologous transplant, radiotherapy C1: none C2: general population equal to age and sex C3: other chemotherapy/radiotherapy treatment regimens O: number of cases of second neoplasms (breast, lung, gastrointestinal, prostate, melanoma, thyroid, etc.) |
| PICO B: Has the incidence of SM in cHL or DLBCL long-term survivors who underwent first or second line chemotherapy and ASCT changed with the introduction of modern radiotherapy? | P: population of cHL or DLBCL long-term survivors (≥5 years of disease/treatment free) aged ≥ 18 years at diagnosis I: new radiotherapy approaches (e.g., 3DCRT, volume reduction, dose reduction, IMRT) C: previous radiotherapy regimens (EFRT, 2DRT, RT dose) O: number of cases of second malignancies (e.g., breast, lung, gastrointestinal, prostate, melanoma, thyroid) |
| PICO C: Are planned follow-up/screening schemes effective for the management and early diagnosis of second cancers in cHL or DLBCL long-term survivors treated, regardless of the type of CHT/RT (first and second line including ASCT)? | P: population of cHL or DLBCL long-term survivors (≥5 years of disease/treatment free) aged ≥18 years at diagnosis I: scheduled follow-up/monitoring schemes for second malignancies, diversified by age, gender/sex, which include laboratory and instrumental tests (e.g., breast mammography/MRI, chest CT, stool occult blood, colonoscopy, dermatoscopy, thyroid US, PSA) C1: no follow up/monitoring planned C2: follow up/monitoring schemes with different intensity (with respect to I, including different times and frequencies) O: early diagnosis of second neoplasms, problems related to overdiagnosis, QoL, mortality |
| Study | Study Design and Sample Size | Intervention and Comparison | Main Outcomes |
|---|---|---|---|
| PICO 1 | |||
| André M, 2020 [24] | Pooled analysis of randomized control trials (1227 patients with advanced stage cHL in first line | BEACOPP vs. ABVD | Incidence of SM per 1000 person-years was 6.3% in ABVD arm vs. 9.6% in BEACOPP arm. Thirteen cases of secondary MDS/AML were reported in the BEACOPP arm, compared to none in the ABVD arm. |
| Torok JA, 2015 [25] | Retrospective cohort study (90 patients with early unfavourable cHL in first line) | CT (ABVD in 88%) plus RT | Seven SM were diagnosed in the cohort. The median time for diagnosis of SM was 10 years (range: 1–14 years), but all hematological SM occurred within 3 years of diagnosis. |
| Engert A, 2007 [26] | Randomized control trial (627 patients with early favourable cHL in first line) | 2 ABVD plus EFRT 30 Gy vs. EFRT 30 Gy plus boost 10 Gy | Total number of SM was 39 (6.2%). Eleven of the solid SM occurred in irradiated areas, three in nonirradiated areas, and for seven cases it was unknown or unclear. There were no significant differences between treatment arms. Between years 2 and 9, the incidence remained constant (0.8% per year); numbers at risk were too small for reliable estimates beyond year 9. |
| Schaapveld M, 2015 [27] | Retrospective cohort study (3905 patients with cHL in first line) | CT and/or RT | A total 1055 SM were observed in 908 patients; a third cancer developed in 130 patients, and a fourth developed in 17. SM risk in cHL treated patients was higher than in the general population (121.8 excess cancers per 10,000 PY). Breast cancer contributed most (24.9 cases per 10,000 PY in the whole cohort, 54.3 cases per 10,000 PY in women. SIR for any SM remained high for at least 35 years after the start of treatment for cHL, whereas the absolute excess risk increased steadily over time. A cumulative procarbazine dose of 4.3 g or more per square meter of body-surface area was associated with a higher risk of gastrointestinal cancer. |
| Frontzek F, 2021 [28] | Randomize control trial (275 patients with high-risk DLBCL in first line) | R-megaCHOEP vs. R-CHOEP14 | Twenty-two SM were reported in the ITT population, 9% in the R-CHOEP14 group and 8% in the R-MegaCHOEP group. The median time to MDS/AML was 44 months; median time to solid SM was 72 months. Age older than 50 years at diagnosis was the only factor associated with significantly increased risk of secondary SM. |
| Coiffier B, 2010 [29] | Randomized control trial (399 patients with DLBCL in first line, 60–80 years old) | R-CHOP21 vs. CHOP21 | Fourty-three patients (10.8%) developed a SM after entering the study, 22 in the CHOP arm and 21 in the R-CHOP arm. Three patients developed a third SM, 1 in the CHOP arm and 2 in the R-CHOP arm. There was no pattern in the type of secondary cancers that occurred. |
| Castellino A, 2018 [30] | Pooled analysis of prospective trial (112 patients with DLBCL in first line) | R-CHOP21 plus lenalidomide | SM were observed in seven patients (6.3%). The median time from the end of treatment to the SM onset was 16.4 months. |
| Paudel N, 2019 [31] | Retrospective cohort study (89 patients with relapsed/refractory cHL) | Total lymphoid irradiation plus carboplatin-cyclophosphamide- etoposide and ASCT | Eight of the 89 patients had developed SM at a median of 5.6 years from the time of transplant. Five patients developed hematologic SM, three were solid SM. Three of the patients with subsequent hematologic SM had received MOPP. |
| Pingali SR, 2017 [32] | Retrospective cohort study (310 patients with relapsed/refractory cHL patients) | ASCT with BEAM conditioning, mobilization with ifosfamide and etoposide | Cumulative incidence of SM was 11% in the entire cohort, with 13 cases of MDS/AML, 5 non-melanoma skin cancers, 1 prostate cancer, 1 MPN, 1 sarcoma. The incidence of SM was significantly higher in pts aged >55 years at diagnosis, even after exclusion of second skin cancers. Time from initial diagnosis to transplantation and exposure to RT were not significant predictors of SM in the study population. MOPP, which was frontline CT in 20 patients, all of whom were <55 years of age, was associated with higher incidence of SM compared with ABVD. |
| Sibon D, 2016 [33] | Retrospective cohort study (245 patients with relapsed/refractory cHL) | Poor risk: tandem ASCT; other patients single ASCT with BEAM conditioning | Sixteen SM occurred. The 10- and 15-year cumulative incidences of SM were 8% and 15%, respectively; the 10-year cumulative incidences of SM were 15% in intermediate-risk patients and 1.5% in poor-risk patients. Considering only patients who did not relapse after completing ASCT, the 10- and 15-year cumulative incidences of SM were 9% and 13%, respectively; the 10- and 15-year cumulative incidences of SM were 16% and 24%, respectively, for the intermediate-risk patients and 2% and 2% for the poor-risk patients. |
| Minn AY, 2012 [34] | Retrospective cohort study (154 patients with relapsed/refractory cHL) | Etoposide + cyclophosmamide with TBI or carmustin or lomustin | There were 20 SM, 18 occurring more than 2 years from ASCT. The 5-, 10-, and 15-year cumulative incidence of SM was 5%, 8%, and 12%, respectively. AER was 160 per 10,000 PY of follow-up. The risk of SM compared with patients with cHL in the SEER registry was not elevated 5–10 years after ASCT, but was higher 2–5 and >10 years after ASCT. The overall risk of SM was 3.0 compared with cHL patients from SEER with an AER of 123 per 10,000 PY of follow-up. Overall risk of SM among patients receiving ASCT was not elevated compared with the non-ASCT population at Stanford. |
| Tarella C, 2011 [35] | Retrospective cohort study (234 patients with relapsed/refractory cHL; 569 patients with high-risk DLBCL in first line or relapsed/refractory DLBCL) | High-dose sequential chemotherapy | The cumulative incidence of MDS/AML in cHL at 5-, 10, and 15-years was 4.1%, 5%, and 15%, respectively, while the cumulative incidence of solid SM was 1.8, 5.5, and 6.9%, respectively. Risk analyses for MDS/AML and solid SM were performed for the whole populations and not for cHL patients only. The overall SIR for MDS/AML was 2.6, with a significantly higher risk for patients aged younger than 45 years (SIR 7.2) and 45 to 65 years of age (SIR 2.1) compared to the age-matched Italian population. Overall, the SIR for solid SM was 3.2; again, increased risk compared to the Italian population was documented for patients aged younger than 45 years (SIR 7.6) and 45 to 65 years of age (SIR 2.7). |
| Franklin J, 2017 [36] | Systematic review (9498 patients with cHL in first line) | Different therapies including CT and RT | In patients treated with CT the omission of additional RT probably reduces SM incidence, corresponding to an estimated reduction of 8-year SM risk from 8% to 4%. The authors observed that the decrease would be particularly true for AML. The authors investigated the role of the number of CT courses, which probably has little or no effect on SM risk, and the role of intensified CT in patients with advanced stage disease, with insufficient evidence to determine the effect on SM. In patients who received intensified CT, the rate of secondary AML (and for younger patients, all SM) was probably higher than in patients treated with standard-dose ABVD-like protocols. |
| PICO 2 | |||
| Patel CG, 2018 [37] | Retrospective cohort study (1541 patients with early stage cHL in first line) | IFRT or EFRT | A trend of lower risk of death from SM in more recently-treated patients (smaller fields) was observed, though this did not reach statistical significance, likely due to the small number of events within each treatment era. |
| LeMieux MH, 2015 [38] | Retrospective cohort study (8807 patients with early stage cHL in first line) | IFRT or EFRT | The authors suggests that patients treated with RT prior to 2000 (larger fields) had a slightly higher risk of SM compared to treatment in 2000 and later. |
| Schaapveld M, 2015 [27] | Retrospective cohort study (3905 patients with cHL in first line) | CT and/or RT | Patients who received supradiaphragmatic RT not including the axilla had a lower risk of a solid SM than those who received complete mantle-field RT, largely due to a lower risk of breast cancer. |
| De Bruin ML, 2009 [39] | Retrospective cohort study (1022 female patients with cHL in first line) | Mantle field RT or smaller field RT | Mantle field RT (involving the axillary, mediastinal, and neck nodes) was associated with a 2.7-fold increased risk compared with similarly dosed (36 to 44 Gy) mediastinal RT alone. |
| Conway JL, 2017 [40] | Retrospective cohort study (734 patients with cHL in first line) | Mantle field RT or smaller field RT | The 20-year cumulative incidences for secondary breast cancer, after accounting for death and loss to follow-up as competing risks, were 7.5% in mantle field and 3.1% in smaller field RT, compared to 2% in CT only. |
| Franklin J, 2017 [36] | Systematic review (9498 patients with cHL in first line) | Different therapies including CT and RT | There is insufficient evidence to determine whether smaller radiation field reduces SM risk and the impact on OS and PFS, with the bias of a too short follow-up of the included studies to record the occurrence of solid tumors. |
| Franklin J, 2005 [41] | Systematic review (9312 patients with cHL in first line) | Different therapies including CT and RT | No difference in second tumor incidence between EFRT and IFRT. |
| PICO 3 | |||
| Diller L, 2002 [42] | Prospective cohort study (90 partecipating out of 167 patients with cHL treated with RT) | Mx | Ten out of 90 women had a breast cancer, 8 unilateral and 2 bilateral cancer. All cancers were detectable with Mx. Most of the tumors were small and without evidence of nodal involvement. Nine non-breast cancers have developed in members of this cohort during the follow-up period. Screening by Mx can detect small, node-negative breast cancers in these patients. Multivariate analysis revealed that older patients who understood that they were at high risk and received risk information from an oncologist were seven times more likely than patients without this profile to have had a mammogram in the previous 2 years |
| Kwong A, 2008 [43] | Randomized trial (167 responding out of 297 patients with cHL treated with RT) | Early vs. late telephone counseling | Completion of Mx during the period of the study was reported by 115 of the 167 subjects: 99 Mx were reviewed, 17 recalled; 7 of the 17 women with abnormal Mx were recommended for biopsy. Four out of 115 (3.5%) women who reported completion of Mx (3.5%) were diagnosed with DCIS and two of these had, at least, microscopic evidence for invasive cancer. Three of these patients were diagnosed solely because of calcifications observed on mammography. In the fourth, mammography confirmed suspicious findings noted on self-examination and professional clinical examination. |
| Lee L, 2008 [44] | Prospective cohort study (100 responding out of 360 patients with cHL treated with RT) | Clinical exam and Mx | Twelve of the 100 participating women (12%) were diagnosed with breast cancer after a median of 5 years of surveillance. Seven cancers presented as palpable masses (six invasive, one DCIS), five were detected by Mx (one invasive, four DCIS). Screening Mx may be effective at detecting DCIS, but may be inadequate for early invasive BC detection in this high-risk population. Evaluation of more intensive screening and the contribution of MRI for earlier detection is warranted. |
| Howell SJ, 2009 [45] | Report of screening (243 screening reports in patients with cHL treated with RT) | No imaging or breast MRI/US or Mx/US or Mx | Of 417 women, 23 (5.5%) have been diagnosed with breast cancer. Five of them were diagnosed within the screening program, none of them involved axillary lymph nodes compared with 7 of 13 (54%) diagnosed outside the program. |
| Elkin EB, 2011 [46] | Observational case-control study (253 patients with cHL treated with RT matched 741 breast cancer patients with no history of cHL) | Various surveillance methods | cHL survivors were more likely to have breast cancer detected by screening Mx (40% vs. 33%), were more likely to be diagnosed at an earlier stage (61% vs. 42%), were less likely to have axillary lymphnode involvement (25% vs. 39%), and were more likely to present with bilateral disease (6% vs. 2%). |
| Mariscotti G, 2013 [47] | Retrospective cohort trial (54 patients with cHL treated with RT) | Clinical exam/US or clinical exam/US/Mx | The authors concluded that patients treated with RT have a higher risk of developing breast cancer and that they need to undergo adequate breast surveillance protocols. They suggested to use of MRI in patients ≤30 years of age and as a complementary method to US/Mx in patients >30, as suggested for women with BRCA mutations. |
| Ng AK, 2013 [48] | Prospective cohort study (148 patients with cHL treated with RT) | MRI and Mx surveillance | The overall sensitivity of Mx for breast cancer detection was 68%, as compared with 67% for breast MRI. The use of both screening modalities increased the sensitivity to 94% |
| Monitoring strategies should be individualized, depending on RT dose, type of CT regimen, age at therapy, and predisposing factors (family history, sex, behavioral risk factors). |
| No evidence of screening programs for DLBCL survivors, |
| Breast cancer: for patients treated with >10 Gy RT on the chest: start at age 40 or 8 years after RT, whichever comes first, by annual Mx, add annual breast MRI for women who received chest RT between ages 10–30 years. |
| Lung cancer: annual chest LDCT scan for smokers treated with alkylators/RT. |
| Skin cancer: annual skin evaluation of the irradiated skin areas. |
| Thyroid cancer: neck ultra-sound for pts treated with neck RT. |
| Colorectal cancer: annual FOB and colonoscopy every 10 years (based on findings) for pts treated with abdominal RT (≥20 Gy), starting from the age of 30 years or 5 years after RT. |
| MDS/AML: annual blood cell count evaluation. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nassi, L.; De Sanctis, V.; Loseto, G.; Gerardi, C.; Allocati, E.; Ciavarella, S.; Minoia, C.; Guarini, A.; Bari, A. Second Cancers in Classical Hodgkin Lymphoma and Diffuse Large B-Cell Lymphoma: A Systematic Review by the Fondazione Italiana Linfomi. Cancers 2022, 14, 519. https://doi.org/10.3390/cancers14030519
Nassi L, De Sanctis V, Loseto G, Gerardi C, Allocati E, Ciavarella S, Minoia C, Guarini A, Bari A. Second Cancers in Classical Hodgkin Lymphoma and Diffuse Large B-Cell Lymphoma: A Systematic Review by the Fondazione Italiana Linfomi. Cancers. 2022; 14(3):519. https://doi.org/10.3390/cancers14030519
Chicago/Turabian StyleNassi, Luca, Vitaliana De Sanctis, Giacomo Loseto, Chiara Gerardi, Eleonora Allocati, Sabino Ciavarella, Carla Minoia, Attilio Guarini, and Alessia Bari. 2022. "Second Cancers in Classical Hodgkin Lymphoma and Diffuse Large B-Cell Lymphoma: A Systematic Review by the Fondazione Italiana Linfomi" Cancers 14, no. 3: 519. https://doi.org/10.3390/cancers14030519







