Landscape of Epidermal Growth Factor Receptor Heterodimers in Brain Metastases
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
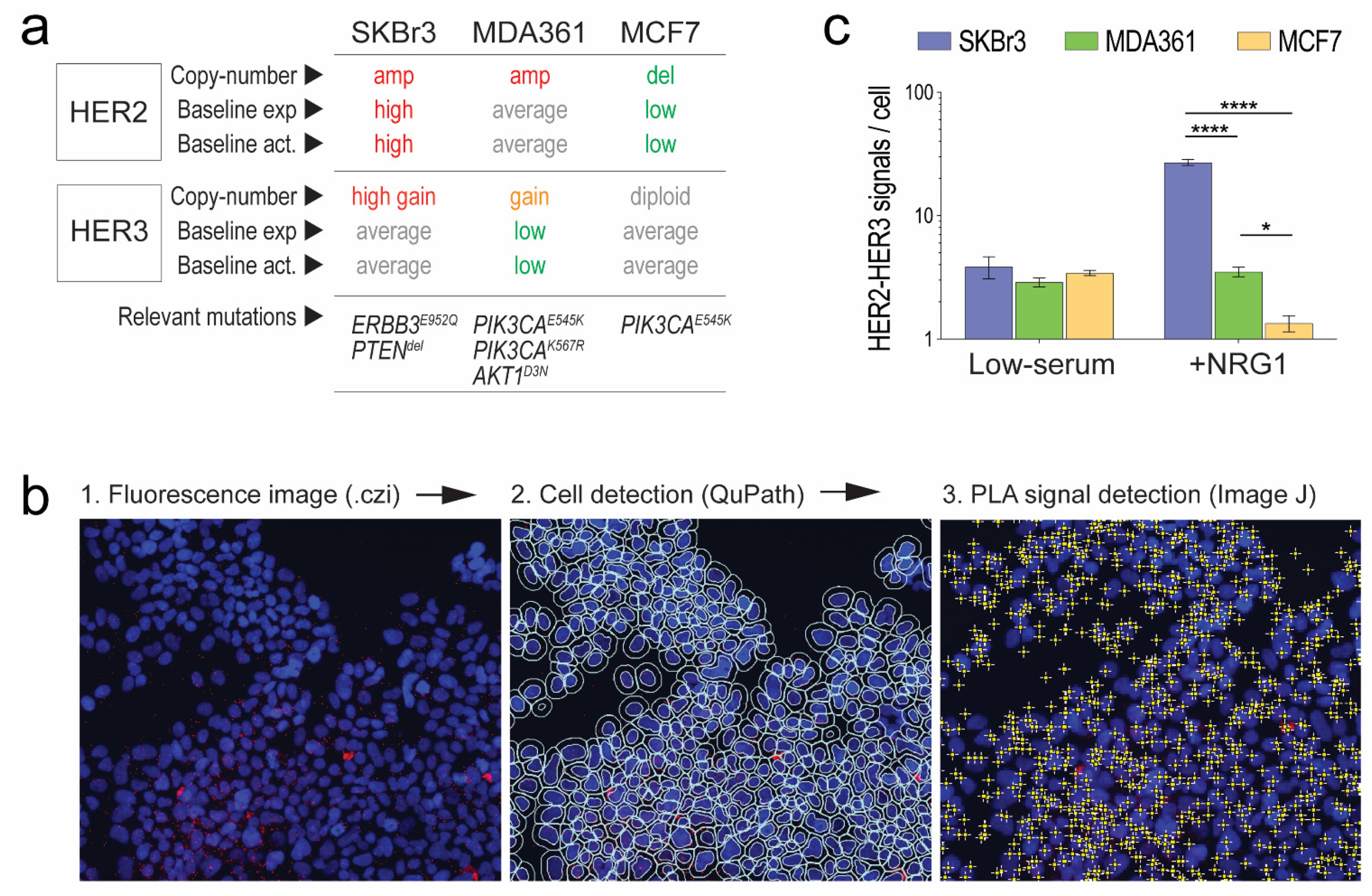
2.1. Cell Line Selection and Cell Culture
2.2. Clinical Samples and Human Research Ethics Committee Approval
- Mixed BM cohort: various histological origin: 48 from lung cancer, 33 breast, 30 melanoma, 19 renal cell, 17 colorectal, 5 prostate, 4 ovarian, 1 adenoid cystic, 1 neuroendocrine and 1 thyroid cancer case. HER2 status for these tumours was determined previously using immunohistochemistry (IHC) and diagnostic scoring criteria [30].
- Breast-BM cohort: 41 brain-metastatic breast cancer cases (34 primary breast tumours and 44 BM, of which 28 are ‘matched pairs’) [31]. HER2 status was extracted from primary breast cancer pathology reports, determined by in situ hybridization (ISH).
2.3. Immunofluorescence
2.4. Fluorescent In Situ Proximity Ligation Assay (PLA)
2.5. Digital Image Analysis
3. Results
3.1. Validation of Fluorescent In Situ PLA and Development of a Data Extraction Pipeline
- SKBr3, a model of genomically driven HER2-HER3-PI3K pathway activation with ERBB2 and ERBB3 copy-number gains, high expression, and strong activation under baseline culture conditions, a HER3 tyrosine kinase domain mutation of unknown significance, and homozygous deletion of PTEN. PTEN exerts negative feedback on PI3K and is frequently lost in breast cancer BM [30,52,53].
- MDA(-MB-)361, a breast cancer BM-derived line with ERBB2 and ERBB3 gene copy-number gains but average to low baseline expression and activation. This line has dual PIK3CA mutations (E545K, K567R): helical domain alterations that decouple PI3K’s catalytic and regulatory domains, disrupting negative feedback [54,55,56]. Its growth in the brain is more dependent on PI3K and HER3 than HER2 [27].
- MCF7, a model of luminal-like breast cancer with below average baseline expression and activation of HER2 and HER3, ERBB2 copy number loss and PIK3CAE545K.
3.2. HER2-HER3 Complexes Are Prevalent in Brain Metastases with Different Histological Origins, but Do Not Correlate Reliably with Protein Level or Phosphorylation
3.3. HER2-HER3 Dimers Are Induced in BM Compared to Matched Primary Breast Tumours
3.4. Effects of HER2-Targeted mAbs on the HER2/3 Heterodimer Landscape
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- AIWH. Cancer in Australia: An Overview 2014. In Cancer Series No 90; Cat. no. CAN 88; AIHW: Canberra, Australia, 2014. [Google Scholar]
- Pestalozzi, B.C.; Zahrieh, D.; Price, K.N.; Holmberg, S.B.; Lindtner, J.; Collins, J.; Crivellari, D.; Fey, M.F.; Murray, E.; Pagani, O.; et al. Identifying breast cancer patients at risk for Central Nervous System (CNS) metastases in trials of the International Breast Cancer Study Group (IBCSG). Ann. Oncol. 2006, 17, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, E.M.; Shim, B.; Goodman, S.; Amonkar, M.M. Epidemiology and economic burden of brain metastases among patients with primary breast cancer: Results from a US claims data analysis. Breast Cancer Res. Treat. 2007, 108, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhu, Y.; Liu, X.; Liao, X.; He, J.; Niu, L. The Clinicopathological features and survival outcomes of patients with different metastatic sites in stage IV breast cancer. BMC Cancer 2019, 19, 1091. [Google Scholar] [CrossRef] [Green Version]
- Patel, A.; Unni, N.; Peng, Y. The Changing Paradigm for the Treatment of HER2-Positive Breast Cancer. Cancers 2020, 12, 2081. [Google Scholar] [CrossRef]
- Palmieri, D.; Bronder, J.L.; Herring, J.M.; Yoneda, T.; Weil, R.J.; Stark, A.M.; Kurek, R.; Vega-Valle, E.; Feigenbaum, L.; Halverson, D.; et al. Her-2 Overexpression Increases the Metastatic Outgrowth of Breast Cancer Cells in the Brain. Cancer Res. 2007, 67, 4190–4198. [Google Scholar] [CrossRef] [Green Version]
- Martin, A.M.; Cagney, D.N.; Catalano, P.J.; Warren, L.E.; Bellon, J.R.; Punglia, R.S.; Claus, E.B.; Lee, E.Q.; Wen, P.Y.; Haas-Kogan, D.A.; et al. Brain Metastases in Newly Diagnosed Breast Cancer: A Population-Based Study. JAMA Oncol. 2017, 3, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Berghoff, A.S.; Schur, S.; Füreder, L.M.; Gatterbauer, B.; Dieckmann, K.; Widhalm, G.; Hainfellner, J.; Zielinski, C.C.; Birner, P.; Bartsch, R.; et al. Descriptive statistical analysis of a real life cohort of 2419 patients with brain metastases of solid cancers. ESMO Open 2016, 1, e000024. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsieh, A.C.; Moasser, M.M. Targeting HER proteins in cancer therapy and the role of the non-target HER3. Br. J. Cancer 2007, 97, 453–457. [Google Scholar] [CrossRef] [Green Version]
- Hohensee, I.; Lamszus, K.; Riethdorf, S.; Meyer-Staeckling, S.; Glatzel, M.; Matschke, J.; Witzel, I.; Westphal, M.; Brandt, B.; Müller, V.; et al. Frequent Genetic Alterations in EGFR- and HER2-Driven Pathways in Breast Cancer Brain Metastases. Am. J. Pathol. 2013, 183, 83–95. [Google Scholar] [CrossRef]
- Saunus, J.M.; Reed, A.E.M.; Lim, M.; Lakhani, S. Breast Cancer Brain Metastases: Clonal Evolution in Clinical Context. Int. J. Mol. Sci. 2017, 18, 152. [Google Scholar] [CrossRef]
- Park, Y.H.; Park, M.J.; Ji, S.H.; Yi, S.Y.; Lim, D.H.; Nam, D.H.; Lee, J.-I.; Park, W.; Choi, D.H.; Huh, S.J.; et al. Trastuzumab treatment improves brain metastasis outcomes through control and durable prolongation of systemic extracranial disease in HER2-overexpressing breast cancer patients. Br. J. Cancer 2009, 100, 894–900. [Google Scholar] [CrossRef] [PubMed]
- Von Minckwitz, G.; Procter, M.; de Azambuja, E.; Zardavas, D.; Benyunes, M.; Viale, G.; Suter, T.; Arahmani, A.; Rouchet, N.; Clark, E.; et al. Adjuvant Pertuzumab and Trastuzumab in Early HER2-Positive Breast Cancer. N. Engl. J. Med. 2017, 377, 122–131. [Google Scholar] [CrossRef]
- Olson, E.M.; Abdel-Rasoul, M.; Maly, J.; Wu, C.S.; Lin, N.U.; Shapiro, C.L. Incidence and risk of central nervous system metastases as site of first recurrence in patients with HER2-positive breast cancer treated with adjuvant trastuzumab. Ann. Oncol. 2013, 24, 1526–1533. [Google Scholar] [CrossRef]
- Brufsky, A.M.; Mayer, M.; Rugo, H.S.; Kaufman, P.A.; Tan-Chiu, E.; Tripathy, D.; Tudor, I.C.; Wang, L.I.; Brammer, M.G.; Shing, M.; et al. Central Nervous System Metastases in Patients with HER2-Positive Metastatic Breast Cancer: Incidence, Treatment, and Survival in Patients from registHER. Clin. Cancer Res. 2011, 17, 4834–4843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Von Minckwitz, G.; Huang, C.S.; Mano, M.S.; Loibl, S.; Mamounas, E.P.; Untch, M.; Wolmark, N.; Rastogi, P.; Schneeweiss, A.; Redondo, A.; et al. Trastuzumab Emtansine for Residual Invasive HER2-Positive Breast Cancer. N. Engl. J. Med. 2019, 380, 617–628. [Google Scholar] [CrossRef] [PubMed]
- Swain, S.M.; Baselga, J.; Miles, D.; Im, Y.-H.; Quah, C.; Lee, L.F.; Cortes, J. Incidence of central nervous system metastases in patients with HER2-positive metastatic breast cancer treated with pertuzumab, trastuzumab, and docetaxel: Results from the randomized phase III study CLEOPATRA. Ann. Oncol. 2014, 25, 1116–1121. [Google Scholar] [CrossRef]
- Murthy, R.K.; Loi, S.; Okines, A.; Paplomata, E.; Hamilton, E.; Hurvitz, S.A.; Lin, N.U.; Borges, V.; Abramson, V.; Anders, C.; et al. Tucatinib, Trastuzumab, and Capecitabine for HER2-Positive Metastatic Breast Cancer. N. Engl. J. Med. 2020, 382, 597–609. [Google Scholar] [CrossRef]
- Stemmler, H.J.; Kahlert, S.; Siekiera, W.; Untch, M.; Heinrich, B.; Heinemann, V. Characteristics of patients with brain metastases receiving trastuzumab for HER2 overexpressing meta-static breast cancer. Breast 2006, 15, 219–225. [Google Scholar] [CrossRef]
- Stavrou, E.; Winer, E.; Lin, N. How we treat HER2-positive brain metastases. ESMO Open 2021, 6, 100256. [Google Scholar] [CrossRef]
- Taskar, K.S.; Rudraraju, V.; Mittapalli, R.K.; Samala, R.; Thorsheim, H.R.; Lockman, J.; Gril, B.; Hua, E.; Palmieri, D.; Polli, J.; et al. Lapatinib Distribution in HER2 Overexpressing Experimental Brain Metastases of Breast Cancer. Pharm. Res. 2011, 29, 770–781. [Google Scholar] [CrossRef] [Green Version]
- Lockman, P.R.; Mittapalli, R.K.; Taskar, K.S.; Rudraraju, V.; Gril, B.; Bohn, K.A.; Adkins, C.E.; Roberts, A.; Thorsheim, H.R.; Gaasch, J.A.; et al. Heterogeneous blood-tumor barrier permeability determines drug efficacy in experimental brain metas-tases of breast cancer. Clin. Cancer Res. 2010, 16, 5664–5678. [Google Scholar] [CrossRef] [Green Version]
- Mortimer, J.E.; Bading, J.R.; Colcher, D.M.; Conti, P.S.; Frankel, P.H.; Carroll, M.I.; Tong, S.; Poku, E.; Miles, J.K.; Shively, J.E.; et al. Functional imaging of HER2-positive metastatic breast cancer using (64)Cu-DOTA-trastuzumab PET. J. Nucl. Med. 2014, 55, 23–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dijkers, E.C.; Oude Munnink, T.H.; Kosterink, J.G.; Brouwers, A.H.; Jager, P.L.; De Jong, J.R.; Van Dongen, G.A.; Schroder, C.P.; Lub-de Hooge, M.N.; de Vries, E.G. Biodistribution of 89Zr-trastuzumab and PET Imaging of HER2-Positive Lesions in Patients With Metastatic Breast Cancer. Clin. Pharmacol. Ther. 2010, 87, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Kurihara, H.; Yonemori, K.; Tsuda, H.; Suzuki, J.; Kono, Y.; Honda, N.; Kodaira, M.; Yamamoto, H.; Yunokawa, M.; et al. 64Cu-DOTA-Trastuzumab PET Imaging in Patients with HER2-Positive Breast Cancer. J. Nucl. Med. 2013, 54, 1869–1875. [Google Scholar] [CrossRef] [Green Version]
- Kodack, D.P.; Askoxylakis, V.; Ferraro, G.B.; Fukumura, D.; Jain, R.K. Emerging Strategies for Treating Brain Metastases from Breast Cancer. Cancer Cell 2015, 27, 163–175. [Google Scholar] [CrossRef] [Green Version]
- Kodack, D.P.; Askoxylakis, V.; Ferraro, G.B.; Sheng, Q.; Badeaux, M.; Goel, S.; Qi, X.; Shankaraiah, R.; Cao, Z.A.; Ramjiawan, R.R.; et al. The brain microenvironment mediates resistance in luminal breast cancer to PI3K inhibition through HER3 activation. Sci. Transl. Med. 2017, 9, eaal4682. [Google Scholar] [CrossRef] [Green Version]
- Lampson, L.A. Monoclonal antibodies in neuro-oncology: Getting past the blood-brain barrier. MAbs 2011, 3, 153–160. [Google Scholar] [CrossRef] [Green Version]
- Sun, M.; Behrens, C.; Feng, L.; Ozburn, N.; Tang, X.; Yin, G.; Komaki, R.; Varella-Garcia, M.; Hong, W.K.; Aldape, K.D.; et al. HER Family Receptor Abnormalities in Lung Cancer Brain Metastases and Corresponding Primary Tumors. Clin. Cancer Res. 2009, 15, 4829–4837. [Google Scholar] [CrossRef] [Green Version]
- Saunus, J.M.; Quinn, M.C.; Patch, A.-M.; Pearson, J.V.; Bailey, P.J.; Nones, K.; Reed, A.E.M.; Miller, D.; Wilson, P.J.; Al-Ejeh, F.; et al. Integrated genomic and transcriptomic analysis of human brain metastases identifies alterations of potential clinical significance. J. Pathol. 2015, 237, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Kalita-de Croft, P.; Lim, M.; Chittoory, H.; De Luca, X.M.; Kutasovic, J.R.; Day, B.W.; Al-Ejeh, F.; Simpson, P.T.; Reed, A.M.; Lakhani, S.R.; et al. Clinicopathologic significance of nuclear HER4 and phospho-YAP(S127) in human breast cancers and matching brain metastases. Ther. Adv. Med. Oncol. 2020, 12, 1758835920946259. [Google Scholar] [CrossRef]
- Da Silva, L.; Simpson, P.T.; Smart, C.E.; Cocciardi, S.; Waddell, N.; Lane, A.; Morrison, B.J.; Vargas, A.C.; Healey, S.; Beesley, J.; et al. HER3 and downstream pathways are involved in colonization of brain metastases from breast cancer. Breast Cancer Res. 2010, 12, R46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sliwkowski, M.X.; Schaefer, G.; Akita, R.W.; Lofgren, J.A.; Fitzpatrick, V.D.; Nuijens, A.; Fendly, B.M.; Cerione, R.A.; Vandlen, R.L.; Carraway, K.L. Coexpression of erbB2 and erbB3 proteins reconstitutes a high affinity receptor for heregulin. J. Biol. Chem. 1994, 269, 14661–14665. [Google Scholar] [CrossRef]
- Soltoff, S.P.; Carraway, K.L., 3rd; Prigent, S.A.; Gullick, W.G.; Cantley, L.C. ErbB3 is involved in activation of phosphatidylinositol 3-kinase by epidermal growth factor. Mol. Cell. Biol. 1994, 14, 3550–3558. [Google Scholar] [PubMed] [Green Version]
- Baselga, J.; Swain, S.M. Novel anticancer targets: Revisiting ERBB2 and discovering ERBB3. Nat. Cancer 2009, 9, 463–475. [Google Scholar] [CrossRef]
- Clark, A.S.; West, K.; Streicher, S.; A Dennis, P. Constitutive and inducible Akt activity promotes resistance to chemotherapy, trastuzumab, or tamoxifen in breast cancer cells. Mol. Cancer Ther. 2002, 1, 707–717. [Google Scholar]
- Xia, W.; Petricoin, E.F.; Zhao, S.; Liu, L.; Osada, T.; Cheng, Q.; Wulfkuhle, J.D.; Gwin, W.R.; Yang, X.; Gallagher, R.I.; et al. An heregulin-EGFR-HER3 autocrine signaling axis can mediate acquired lapatinib resistance in HER2+ breast cancer models. Breast Cancer Res. 2013, 15, R85. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.D.; Ma, L.; Wong, M.P.; Lee, V.H.F.; Yan, H. Contribution of EGFR and ErbB-3 Heterodimerization to the EGFR Mutation-Induced Gefitinib- and Erlo-tinib-Resistance in Non-Small-Cell Lung Carcinoma Treatments. PLoS ONE 2015, 10, e0128360. [Google Scholar]
- Ma, J.; Lyu, H.; Huang, J.; Liu, B. Targeting of erbB3 receptor to overcome resistance in cancer treatment. Mol. Cancer 2014, 13, 105. [Google Scholar] [CrossRef] [Green Version]
- Yao, E.; Zhou, W.; Lee-Hoeflich, S.T.; Truong, T.; Haverty, P.M.; Eastham-Anderson, J.; Lewin-Koh, N.; Gunter, B.; Belvin, M.; Murray, L.J.; et al. Suppression of HER2/HER3-mediated growth of breast cancer cells with combinations of GDC-0941 PI3K in-hibitor, trastuzumab, and pertuzumab. Clin. Cancer Res. 2009, 15, 4147–4156. [Google Scholar] [CrossRef] [Green Version]
- Koutras, A.K.; Fountzilas, G.; Kalogeras, K.T.; Starakis, I.; Iconomou, G.; Kalofonos, H.P. The upgraded role of HER3 and HER4 receptors in breast cancer. Crit. Rev. Oncol. 2010, 74, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Sergina, N.V.; Rausch, M.; Wang, D.; Blair, J.; Hann, B.; Shokat, K.M.; Moasser, M.M. Escape from HER-family tyrosine kinase inhibitor therapy by the kinase-inactive HER3. Nature 2007, 445, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Narayan, M.; Wilken, J.A.; Harris, L.N.; Baron, A.; Kimbler, K.D.; Maihle, N.J. Trastuzumab-Induced HER Reprogramming in “Resistant” Breast Carcinoma Cells. Cancer Res. 2009, 69, 2191–2194. [Google Scholar] [CrossRef] [Green Version]
- Liu, B.; Ordonez-Ercan, D.; Fan, Z.; Edgerton, S.M.; Yang, X.; Thor, A.D. Downregulation of erbB3 abrogates erbB2-mediated tamoxifen resistance in breast cancer cells. Int. J. Cancer 2007, 120, 1874–1882. [Google Scholar] [CrossRef] [PubMed]
- Haikala, H.M.; Jänne, P.A. Thirty Years of HER3: From Basic Biology to Therapeutic Interventions. Clin. Cancer Res. 2021, 27, 3528–3539. [Google Scholar] [CrossRef] [PubMed]
- Barretina, J.; Caponigro, G.; Stransky, N.; Venkatesan, K.; Margolin, A.A.; Kim, S.; Wilson, C.J.; Lehár, J.; Kryukov, G.V.; Sonkin, D.; et al. Addendum: The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature 2018, 565, E5–E6. [Google Scholar] [CrossRef]
- The Cancer Cell Line Encyclopedia Consortium; The Genomics of Drug Sensitivity in Cancer Consortium. Pharmacogenomic agreement between two cancer cell line data sets. Nature 2015, 528, 84–87. [Google Scholar] [CrossRef]
- Rondón-Lagos, M.; Di Cantogno, L.V.; Rangel, N.; Mele, T.; Ramírez-Clavijo, S.R.; Scagliotti, G.; Marchio, C.; Sapino, A. Unraveling the chromosome 17 patterns of FISH in interphase nuclei: An in-depth analysis of the HER2 amplicon and chromosome 17 centromere by karyotyping, FISH and M-FISH in breast cancer cells. BMC Cancer 2014, 14, 922. [Google Scholar] [CrossRef] [Green Version]
- Saunus, J.M.; Smart, C.E.; Kutasovic, J.R.; Johnston, R.; Croft, P.K.-D.; Miranda, M.; Rozali, E.N.; Vargas, A.C.; Reid, L.E.; Lorsy, E.; et al. Multidimensional phenotyping of breast cancer cell lines to guide preclinical research. Breast Cancer Res. Treat. 2017, 167, 289–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Söderberg, O.; Gullberg, M.; Jarvius, M.; Ridderstråle, K.; Leuchowius, K.-J.; Jarvius, J.; Wester, K.; Hydbring, P.; Bahram, F.; Larsson, L.-G.; et al. Direct observation of individual endogenous protein complexes in situ by proximity ligation. Nat. Methods 2006, 3, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Bankhead, P.; Loughrey, M.B.; Fernández, J.A.; Dombrowski, Y.; McArt, D.G.; Dunne, P.D.; McQuaid, S.; Gray, R.T.; Murray, L.J.; Coleman, H.G.; et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 2017, 7, 16878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wikman, H.; Lamszus, K.; Detels, N.; Uslar, L.; Wrage, M.; Benner, C.; Hohensee, I.; Ylstra, B.; Eylmann, K.; Zapatka, M.; et al. Relevance of PTEN loss in brain metastasis formation in breast cancer patients. Breast Cancer Res. 2012, 14, R49. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Zhang, S.; Yao, J.; Lowery, F.J.; Zhang, Q.; Huang, W.-C.; Li, P.; Li, M.; Wang, X.; Zhang, C.; et al. Microenvironment-induced PTEN loss by exosomal microRNA primes brain metastasis outgrowth. Nature 2015, 527, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, A.; Rexer, B.N.; Wang, S.E.; Cook, R.S.; Engelman, J.A.; Arteaga, C.L.; Carlos, L.A. H1047R phosphatidylinositol 3-kinase mutant enhances HER2-mediated transformation by heregulin production and activation of HER3. Oncogene 2010, 29, 5193–5203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leontiadou, H.; Galdadas, I.; Athanasiou, C.; Cournia, Z. Insights into the mechanism of the PIK3CA E545K activating mutation using MD simulations. Sci. Rep. 2018, 8, 15544. [Google Scholar] [CrossRef] [PubMed]
- Blair, B.G.; Wu, X.; Zahari, M.S.; Mohseni, M.; Cidado, J.; Wong, H.Y.; Beaver, J.A.; Cochran, R.L.; Zabransky, D.J.; Croessmann, S.; et al. A phosphoproteomic screen demonstrates differential dependence on HER3 for MAP kinase pathway acti-vation by distinct PIK3CA mutations. Proteomics 2015, 15, 318–326. [Google Scholar] [CrossRef] [Green Version]
- Uhlén, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Tissue-Based Map of the Human Proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Savinainen, K.J.; Saramäki, O.R.; Linja, M.J.; Bratt, O.; Tammela, T.L.; Isola, J.J.; Visakorpi, T. Expression and Gene Copy Number Analysis of ERBB2 Oncogene in Prostate Cancer. Am. J. Pathol. 2002, 160, 339–345. [Google Scholar] [CrossRef] [Green Version]
- Calvo, B.F.; Levine, A.M.; Marcos, M.; Collins, Q.F.; Iacocca, M.V.; Caskey, L.S.; Gregory, C.W.; Lin, Y.; Whang, Y.E.; Earp, H.S.; et al. Human epidermal receptor-2 expression in prostate cancer. Clin. Cancer Res. 2003, 9, 1087. [Google Scholar]
- Guven, D.C.; Kaya, M.B.; Fedai, B.; Ozden, M.; Yildirim, H.C.; Kosemehmetoglu, K.; Kertmen, N.; Dizdar, O.; Uner, A.; Aksoy, S. HER2-low breast cancer could be associated with an increased risk of brain metastasis. Int. J. Clin. Oncol. 2021, 1–8. [Google Scholar] [CrossRef]
- Schwarz, L.; E Hutchinson, K.E.; Rexer, B.N.; Estrada, M.V.; Ericsson, P.G.; E Sanders, M.E.; Dugger, T.C.; Formisano, L.; Guerrero-Zotano, A.; Red-Brewer, M.; et al. An ERBB1-3 Neutralizing Antibody Mixture with High Activity against Drug-Resistant HER2+ Breast Cancers with ERBB Ligand Overexpression. J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef] [Green Version]
- Jhaveri, K.; Drago, J.Z.; Shah, P.D.; Wang, R.; Pareja, F.; Ratzon, F.; Iasonos, A.; Patil, S.; Rosen, N.; Fornier, M.N.; et al. A Phase I Study of Alpelisib in Combination with Trastuzumab and LJM716 in Patients with PIK3CA-Mutated HER2-Positive Metastatic Breast Cancer. Clin. Cancer Res. 2021, 27, 3867–3875. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, K.L.; Bedard, P.L.; Lee, S.-H.; Lin, C.-C.; Tabernero, J.; Alsina, M.; Cohen, E.; Baselga, J.; Blumenschein, G., Jr.; Graham, D.M.; et al. A phase I open-label dose-escalation study of the anti-HER3 monoclonal antibody LJM716 in patients with advanced squamous cell carcinoma of the esophagus or head and neck and HER2-overexpressing breast or gastric cancer. BMC Cancer 2017, 17, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Lua, W.H.; Ling, W.L.; Yeo, J.Y.; Poh, J.J.; Lane, D.P.; Gan, S.K. The effects of Antibody Engineering CH and CL in Trastuzumab and Pertuzumab recombinant models: Impact on antibody production and antigen-binding. Sci. Rep. 2018, 8, 718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brastianos, P.K.; Carter, S.L.; Santagata, S.; Cahill, D.; Taylor-Weiner, A.; Jones, R.T.; Van Allen, E.M.; Lawrence, M.S.; Horowitz, P.; Cibulskis, K.; et al. Genomic Characterization of Brain Metastases Reveals Branched Evolution and Potential Therapeutic Targets. Cancer Discov. 2015, 5, 1164–1177. [Google Scholar] [CrossRef] [Green Version]
- Steinkamp, M.P.; Low-Nam, S.T.; Yang, S.; Lidke, K.A.; Lidke, D.S.; Wilson, B.S. erbB3 is an Active Tyrosine Kinase Capable of Homo- and Heterointeractions. Mol. Cell. Biol. 2014, 34, 965–977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, J.; Kim, W.; Kim, L.K.; VanHouten, J.; Wysolmerski, J.J. HER2 signaling regulates HER2 localization and membrane retention. PLoS ONE 2017, 12, e0174849. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Dimer Complex | Antibody 1 (Host: Rabbit) | Antibody 2 (Host: Mouse) | Conditions * | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Supplier (Cat No.) | Clone | Host | Dilution | Supplier (Cat No.) | Clone | Host | Dilution | ||
| In vitroExperiments (Fixed Cells on Coverslips) | |||||||||
| (1)HER3-(2)EGFR | CST (12708) | D22C5 | Rabbit | 1/100 | SC (sc73511) | R2 | Mouse | 1/500 | O/N, 4 °C |
| (1)HER3-(2)HER2 | CST (12708) | D22C5 | Rabbit | 1/100 | Abcam (ab16901) | 3B5 | Mouse | 1/2000 | O/N, 4 °C |
| (1)HER3-(2)HER4 | CST (12708) | D22C5 | Rabbit | 1/100 | SC (sc-71070) | 3F168 | Mouse | 1/100 | O/N, 4 °C |
| (1)HER2-(2)EGFR | DAKO (A0485) | Poly | Rabbit | 1/2500 | SC (sc73511) | R2 | Mouse | 1/500 | O/N, 4 °C |
| (1)HER2-(2)HER4 | DAKO (A0485) | Poly | Rabbit | 1/2500 | SC (sc-71070) | 3F168 | Mouse | 1/100 | O/N, 4 °C |
| In situ Analysis (FFPE Tissue Sections) | |||||||||
| (1)HER2-(2)HER3 | DAKO (A0485) | Poly | Rabbit | 1/2500 | MP (05-390) | 2F12 | Mouse | 1/200 | 10 mM Tris-EDTA (pH 8.8) 95 °C; 30′ O/N 4 °C |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, M.; Nguyen, T.H.; Niland, C.; Reid, L.E.; Jat, P.S.; Saunus, J.M.; Lakhani, S.R. Landscape of Epidermal Growth Factor Receptor Heterodimers in Brain Metastases. Cancers 2022, 14, 533. https://doi.org/10.3390/cancers14030533
Lim M, Nguyen TH, Niland C, Reid LE, Jat PS, Saunus JM, Lakhani SR. Landscape of Epidermal Growth Factor Receptor Heterodimers in Brain Metastases. Cancers. 2022; 14(3):533. https://doi.org/10.3390/cancers14030533
Chicago/Turabian StyleLim, Malcolm, Tam H. Nguyen, Colleen Niland, Lynne E. Reid, Parmjit S. Jat, Jodi M. Saunus, and Sunil R. Lakhani. 2022. "Landscape of Epidermal Growth Factor Receptor Heterodimers in Brain Metastases" Cancers 14, no. 3: 533. https://doi.org/10.3390/cancers14030533







