Activation of ABCC Genes by Cisplatin Depends on the CoREST Occurrence at Their Promoters in A549 and MDA-MB-231 Cell Lines
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture and Treatment with Inhibitors
2.3. Cisplatin Resistance Induction
2.4. Resazurin Toxicity Assay
2.5. Quantification of Gene Expression
2.6. Transient Gene Silencing
2.7. Chromatin Immunoprecipitation
2.8. Confocal Microscopy
2.9. Transcription Factor Binding Sites Assessment
2.10. Statistical Analysis
3. Results
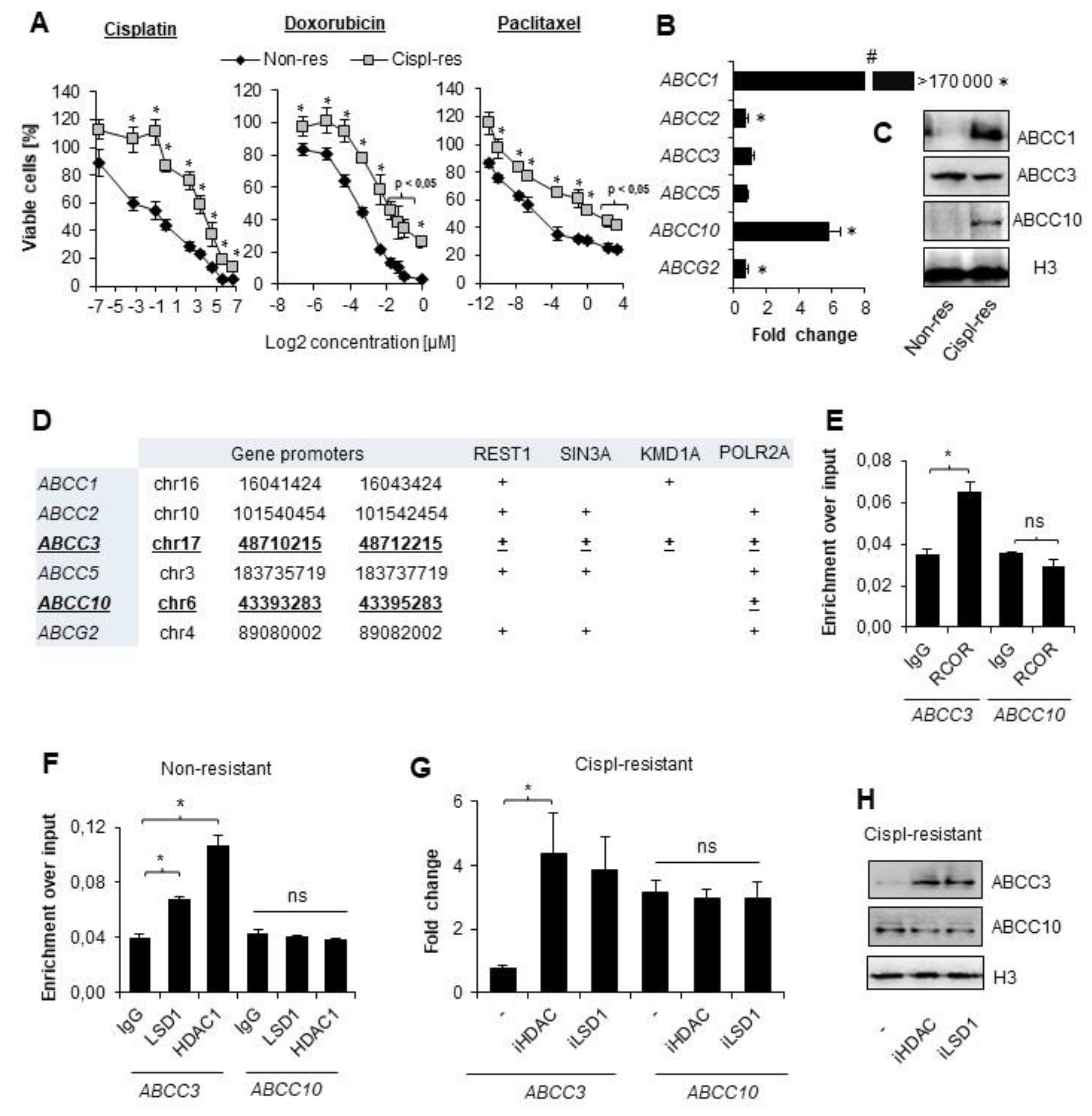
3.1. Non-Small Cell Lung Cancer Cells Exposed to Multiple Doses of Cisplatin Gain Resistance to Drugs of Various Chemical Structures and Activity
3.2. CoREST Complex Subunits Occupy Promoter of ABCC3 That Remains Transcriptionally Irresponsive to Cisplatin in A549
3.3. EP300 Drives ABCC10 Overexpression in Cisplatin-Resistant A549 Cells
3.4. P53 Allows for EP300-Mediated Increase in ABCC10 Transcription after Single Dose of Cisplatin in Non-Resistant A549 Cells
3.5. CoREST-Free Promoter of ABCC10 Responds to Cisplatin in EP300 and p53-Dependent Fashion in TNBC Cell Line—MDA-MB-231
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, Q.; Lu, Q. Bin New Combination Chemotherapy of Cisplatin with an Electron-Donating Compound for Treatment of Multiple Cancers. Sci. Rep. 2021, 11, 788. [Google Scholar] [CrossRef] [PubMed]
- Fennell, D.A.; Summers, Y.; Cadranel, J.; Benepal, T.; Christoph, D.C.; Lal, R.; Das, M.; Maxwell, F.; Visseren-Grul, C.; Ferry, D. Cisplatin in the Modern Era: The Backbone of First-Line Chemotherapy for Non-Small Cell Lung Cancer. Cancer Treat. Rev. 2016, 44, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z.; et al. DrugBank 5.0: A Major Update to the DrugBank Database for 2018. Nucleic Acids Res. 2018, 46, D1074–D1082. [Google Scholar] [CrossRef] [PubMed]
- O’Grady, S.; Finn, S.P.; Cuffe, S.; Richard, D.J.; O’Byrne, K.J.; Barr, M.P. The Role of DNA Repair Pathways in Cisplatin Resistant Lung Cancer. Cancer Treat. Rev. 2014, 40, 1161–1170. [Google Scholar] [CrossRef] [PubMed]
- Dilruba, S.; Kalayda, G.V. Platinum-Based Drugs: Past, Present and Future. Cancer Chemother. Pharmacol. 2016, 77, 1103–1124. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Senovilla, L.; Vitale, I.; Michels, J.; Martins, I.; Kepp, O.; Castedo, M.; Kroemer, G. Molecular Mechanisms of Cisplatin Resistance. Oncogene 2012, 31, 1869–1883. [Google Scholar] [CrossRef] [Green Version]
- Lan, D.; Wang, L.; He, R.; Ma, J.; Bin, Y.; Chi, X.; Chen, G.; Cai, Z. Exogenous Glutathione Contributes to Cisplatin Resistance in Lung Cancer A549 Cells. Am. J. Transl. Res. 2018, 10, 1295–1309. [Google Scholar] [PubMed]
- Rees, D.C.; Johnson, E.; Lewinson, O. ABC Transporters: The Power to Change. Nat. Rev. Mol. Cell Biol. 2009, 10, 218–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hilgeroth, A.; Hemmer, M.; Coburger, C. The Impact of the Induction of Multidrug Resistance Transporters in Therapies by Used Drugs: Recent Studies. Mini Rev. Med. Chem. 2012, 12, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; He, L.R.; Gao, Y.; Zhou, N.N.; Liu, Y.; Zhou, X.K.; Liu, J.F.; Guan, X.Y.; Ma, N.F.; Xie, D. CHD1L Contributes to Cisplatin Resistance by Upregulating the ABCB1–NF-ΚB Axis in Human Non-Small-Cell Lung Cancer. Cell Death Dis. 2019, 10, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Y.; Ling, X.L. Establishment of a Cisplatin-Induced Multidrug Resistance Cell Line SK-Hep1/DDP. Chin. J. Cancer 2010, 29, 167–171. [Google Scholar] [CrossRef] [Green Version]
- Materna, V.; Liedert, B.; Thomale, J.; Lage, H. Protection of Platinum–DNA Adduct Formation and Reversal of Cisplatin Resistance by Anti-MRP2 Hammerhead Ribozymes in Human Cancer Cells. Int. J. Cancer 2005, 115, 393–402. [Google Scholar] [CrossRef]
- Savaraj, N.; Wu, C.; Wangpaichitr, M.; Kuo, M.T.; Lampidis, T.; Robles, C.; Furst, A.J.; Feun, L. Overexpression of Mutated MRP4 in Cisplatin Resistant Small Cell Lung Cancer Cell Line: Collateral Sensitivity to Azidothymidine. Int. J. Oncol. 2003, 23, 173–179. [Google Scholar] [CrossRef]
- Weaver, D.A.; Crawford, E.L.; Warner, K.A.; Elkhairi, F.; Khuder, S.A.; Willey, J.C. ABCC5, ERCC2, XPA and XRCC1 Transcript Abundance Levels Correlate with Cisplatin Chemoresistance in Non-Small Cell Lung Cancer Cell Lines. Mol. Cancer 2005, 4, 18. [Google Scholar] [CrossRef] [Green Version]
- Jaromi, L.; Csongei, V.; Vesel, M.; Abdelwahab, E.M.M.; Soltani, A.; Torok, Z.; Smuk, G.; Sarosi, V.; Pongracz, J.E. Kras and Egfr Mutations Differentially Alter Abc Drug Transporter Expression in Cisplatin-Resistant Non-Small Cell Lung Cancer. Int. J. Mol. Sci. 2021, 22, 5384. [Google Scholar] [CrossRef]
- Fletcher, J.I.; Haber, M.; Henderson, M.J.; Norris, M.D. ABC Transporters in Cancer: More than Just Drug Efflux Pumps. Nat. Rev. Cancer 2010, 10, 147–156. [Google Scholar] [CrossRef]
- Miller, D.S. Regulation of ABC Transporters Blood-Brain Barrier: The Good, the Bad, and the Ugly. Adv. Cancer Res. 2015, 125, 43–70. [Google Scholar] [CrossRef] [Green Version]
- Crawford, R.R.; Potukuchi, P.K.; Schuetz, E.G.; Schuetz, J.D. Beyond Competitive Inhibition: Regulation of ABC Transporters by Kinases and Protein-Protein Interactions as Potential Mechanisms of Drug-Drug Interactions. Drug Metab. Dispos. 2018, 46, 567. [Google Scholar] [CrossRef] [Green Version]
- Bush, J.A.; Li, G. Cancer Chemoresistance: The Relationship between P53 and Multidrug Transporters. Int. J. Cancer 2002, 98, 323–330. [Google Scholar] [CrossRef]
- Scotto, K.W. Transcriptional Regulation of ABC Drug Transporters. Oncogene 2003, 22, 7496–7511. [Google Scholar] [CrossRef] [Green Version]
- Garg, N.; Kumar, P.; Gadhave, K.; Giri, R. The Dark Proteome of Cancer: Intrinsic Disorderedness and Functionality of HIF-1α along with Its Interacting Proteins. Prog. Mol. Biol. Transl. Sci. 2019, 166, 371–403. [Google Scholar] [CrossRef]
- Sobczak, M.; Pietrzak, J.; Płoszaj, T.; Robaszkiewicz, A. BRG1 Activates Proliferation and Transcription of Cell Cycle-Dependent Genes in Breast Cancer Cells. Cancers 2020, 12, 349. [Google Scholar] [CrossRef] [Green Version]
- Ito, A.; Lai, C.H.; Zhao, X.; Saito, S.; Hamilton, M.H.; Appella, E.; Yao, T.P. P300/CBP-Mediated P53 Acetylation Is Commonly Induced by P53-Activating Agents and Inhibited by MDM2. EMBO J. 2001, 20, 1331. [Google Scholar] [CrossRef] [Green Version]
- Chee, J.L.Y.; Saidin, S.; Lane, D.P.; Leong, S.M.; Noll, J.E.; Neilsen, P.M.; Phua, Y.T.; Gabra, H.; Lim, T.M. Wild-Type and Mutant P53 Mediate Cisplatin Resistance through Interaction and Inhibition of Active Caspase-9. Cell Cycle 2013, 12, 278. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, R.; Kaypee, S.; Shasmal, M.; Kundu, T.K.; Roy, S.; Sengupta, J. Tumor Suppressor P53-Mediated Structural Reorganization of the Transcriptional Coactivator P300. Biochemistry 2019, 58, 3434–3443. [Google Scholar] [CrossRef]
- Kaypee, S.; Sahadevan, S.A.; Patil, S.; Ghosh, P.; Roy, N.S.; Roy, S.; Kundu, T.K. Mutant and Wild-Type Tumor Suppressor P53 Induces P300 Autoacetylation. iScience 2018, 4, 260. [Google Scholar] [CrossRef]
- Strachowska, M.; Gronkowska, K.; Michlewska, S.; Robaszkiewicz, A. Cbp/P300 Bromodomain Inhibitor–i–Cbp112 Declines Transcription of the Key Abc Transporters and Sensitizes Cancer Cells to Chemotherapy Drugs. Cancers 2021, 13, 4614. [Google Scholar] [CrossRef]
- Wang, H.; Huang, C.; Zhao, L.; Zhang, H.; Yang, J.M.; Luo, P.; Zhan, B.X.; Pan, Q.; Li, J.; Wang, B.L. Histone Deacetylase Inhibitors Regulate P-Gp Expression in Colorectal Cancer via Transcriptional Activation and MRNA Stabilization. Oncotarget 2016, 7, 49848. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.A.; Zhu, J.; Yennawar, N.; Eek, P.; Tan, S. Crystal Structure of the LSD1/CoREST Histone Demethylase Bound to Its Nucleosome Substrate. Mol. Cell 2020, 78, 903–914.e4. [Google Scholar] [CrossRef]
- Qureshi, I.A.; Gokhan, S.; Mehler, M.F. REST and CoREST Are Transcriptional and Epigenetic Regulators of Seminal Neural Fate Decisions. Cell Cycle 2010, 9, 4477. [Google Scholar] [CrossRef] [Green Version]
- Song, Y.; Dagil, L.; Fairall, L.; Robertson, N.; Wu, M.; Ragan, T.J.; Savva, C.G.; Saleh, A.; Morone, N.; Kunze, M.B.A.; et al. Mechanism of Crosstalk between the LSD1 Demethylase and HDAC1 Deacetylase in the CoREST Complex. Cell Rep. 2020, 30, 2699–2711.e8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Charlier, C.; Kintz, P.; Dubois, N.; Plomteux, G. Fatal Overdosage with Cisplatin. J. Anal. Toxicol. 2004, 28, 138–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pietrzak, J.; Płoszaj, T.; Pułaski, Ł.; Robaszkiewicz, A. EP300-HDAC1-SWI/SNF Functional Unit Defines Transcription of Some DNA Repair Enzymes during Differentiation of Human Macrophages. Biochim. Biophys. Acta-Gene Regul. Mech. 2019, 1862, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.L.; Chen, J.J.; Kumar, P.; Chen, K.; Sodani, K.; Patel, A.; Chen, Y.L.; Chen, S.D.; Jiang, W.Q.; Chen, Z.S. Reversal of MRP7 (ABCC10)-Mediated Multidrug Resistance by Tariquidar. PLoS ONE 2013, 8, 55576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.; Huang, Y.; Shi, J.; Dai, Y.; Wu, L.; Zhou, H. ABCC10 Plays a Significant Role in the Transport of Gefitinib and Contributes to Acquired Resistance to Gefitinib in NSCLC. Front. Pharmacol. 2018, 9, 1312. [Google Scholar] [CrossRef]
- Oguri, T.; Ozasa, H.; Uemura, T.; Bessho, Y.; Miyazaki, M.; Maeno, K.; Maeda, H.; Sato, S.; Ueda, R. MRP7/ABCC10 Expression Is a Predictive Biomarker for the Resistance to Paclitaxel in Non-Small Cell Lung Cancer. Mol. Cancer Ther. 2008, 7, 1150–1155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zappe, K.; Cichna-Markl, M. Aberrant DNA Methylation of ABC Transporters in Cancer. Cells 2020, 9, 2281. [Google Scholar] [CrossRef] [PubMed]
- Toth, M.; Boros, I.M.; Balint, E. Elevated Level of Lysine 9-acetylated Histone H3 at the MDR1 Promoter in Multidrug-resistant Cells. Cancer Sci. 2012, 103, 659. [Google Scholar] [CrossRef]
- Ring, A.; Kaur, P.; Lang, J.E. EP300 Knockdown Reduces Cancer Stem Cell Phenotype, Tumor Growth and Metastasis in Triple Negative Breast Cancer. BMC Cancer 2020, 20, 1–14. [Google Scholar] [CrossRef]
- Li, Q.L.; Wang, D.Y.; Ju, L.G.; Yao, J.; Gao, C.; Lei, P.J.; Li, L.Y.; Zhao, X.L.; Wu, M. The Hyper-Activation of Transcriptional Enhancers in Breast Cancer. Clin. Epigenetics 2019, 11, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Ono, H.; Kato, T.; Murase, Y.; Nakamura, Y.; Ishikawa, Y.; Watanabe, S.; Akahoshi, K.; Ogura, T.; Ogawa, K.; Ban, D.; et al. C646 Inhibits G2/M Cell Cycle-Related Proteins and Potentiates Anti-Tumor Effects in Pancreatic Cancer. Sci. Rep. 2021, 11, 10078. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Hu, Y.; Yang, M.; Jat, P.; Li, K.; Lombardo, Y.; Xiong, D.; Coombes, R.C.; Raguz, S.; Yagüe, E. The MiR-106b∼25 Cluster Promotes Bypass of Doxorubicin-Induced Senescence and Increase in Motility and Invasion by Targeting the E-Cadherin Transcriptional Activator EP300. Cell Death Differ. 2014, 21, 462. [Google Scholar] [CrossRef] [Green Version]
- Ono, H.; Basson, M.D.; Ito, H. P300 Inhibition Enhances Gemcitabine-Induced Apoptosis of Pancreatic Cancer. Oncotarget 2016, 7, 51301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, W.; Zhang, Z.; Kimball, H.; Qu, F.; Berlind, K.; Stopsack, K.H.; Lee, G.S.M.; Choueiri, T.K.; Kantoff, P.W. Abiraterone Acetate Induces CREB1 Phosphorylation and Enhances the Function of the CBP-P300 Complex, Leading to Resistance in Prostate Cancer Cells. Clin. Cancer Res. 2021, 27, 2087. [Google Scholar] [CrossRef] [PubMed]
- Gruber, M.; Ferrone, L.; Puhr, M.; Santer, F.R.; Furlan, T.; Eder, I.E.; Sampson, N.; Schäfer, G.; Handle, F.; Culig, Z. P300 Is Upregulated by Docetaxel and Is a Target in Chemoresistant Prostate Cancer. Endocr. Relat. Cancer 2020, 27, 187. [Google Scholar] [CrossRef] [Green Version]
- Kawamura, T.; Ono, K.; Morimoto, T.; Akao, M.; Iwai-Kanai, E.; Wada, H.; Sowa, N.; Kita, T.; Hasegawa, K. Endothelin-1-Dependent Nuclear Factor of Activated T Lymphocyte Signaling Associates with Transcriptional Coactivator P300 in the Activation of the B Cell Leukemia-2 Promoter in Cardiac Myocytes. Circ. Res. 2004, 94, 1492–1499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Xiao, X.Q.; Qian, Y.H.; Zhou, Z.Y. The CtBP1–P300–FOXO3a Transcriptional Complex Represses the Expression of the Apoptotic Regulators Bax and Bim in Human Osteosarcoma Cells. J. Cell. Physiol. 2019, 234, 22365–22377. [Google Scholar] [CrossRef] [PubMed]
- Tonigold, M.; Rossmann, A.; Meinold, M.; Bette, M.; Märken, M.; Henkenius, K.; Bretz, A.C.; Giel, G.; Cai, C.; Rodepeter, F.R.; et al. A Cisplatin-Resistant Head and Neck Cancer Cell Line with Cytoplasmic P53(Mut) Exhibits ATP-Binding Cassette Transporter Upregulation and High Glutathione Levels. J. Cancer Res. Clin. Oncol. 2014, 140, 1689–1704. [Google Scholar] [CrossRef] [PubMed]
- Phatak, V.; von Grabowiecki, Y.; Janus, J.; Officer, L.; Behan, C.; Aschauer, L.; Pinon, L.; Mackay, H.; Zanivan, S.; Norman, J.C.; et al. Mutant P53 Promotes RCP-Dependent Chemoresistance Coinciding with Increased Delivery of P-Glycoprotein to the Plasma Membrane. Cell Death Dis. 2021, 12, 207. [Google Scholar] [CrossRef]
- Zhang, X.; Qi, Z.; Yin, H.; Yang, G. Interaction between P53 and Ras Signaling Controls Cisplatin Resistance via HDAC4- and HIF-1α-Mediated Regulation of Apoptosis and Autophagy. Theranostics 2019, 9, 1096. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Chang, P.Y.; Chao, C.C.K. CITED2 Silencing Sensitizes Cancer Cells to Cisplatin by Inhibiting P53 Trans-Activation and Chromatin Relaxation on the ERCC1 DNA Repair Gene. Nucleic Acids Res. 2015, 43, 10760. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Negrini, M.; Sabbioni, S.; Haldar, S.; Possati, L.; Castagnoli, A.; Corallini, A.; Barbanti-Brodano, G.; Croce, C.M.; C„, A. Tumor and Growth Suppression of Breast Cancer Cells by Chromosome 17-Associated Functions1. Cancer Res. 1994, 54, 1818–1824. [Google Scholar] [PubMed]
- Walerych, D.; Napoli, M.; Collavin, L.; Del Sal, G. The Rebel Angel: Mutant P53 as the Driving Oncogene in Breast Cancer. Carcinogenesis 2012, 33, 2007–2017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pham, T.H.; Park, H.M.; Kim, J.; Hong, J.T.; Yoon, D.Y. STAT3 and P53: Dual Target for Cancer Therapy. Biomedicines 2020, 8, 637. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.S.; Trovão, F.; Andrade Pinheiro, B.; Freire, F.; Gomes, S.; Oliveira, C.; Domingues, L.; Romão, M.J.; Saraiva, L.; Carvalho, A.L. The Crystal Structure of the R280K Mutant of Human P53 Explains the Loss of DNA Binding. Int. J. Mol. Sci. 2018, 19, 1184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- VCV000376657.7-ClinVar-NCBI. Available online: https://www.ncbi.nlm.nih.gov/clinvar/variation/376657/?new_evidence=false (accessed on 2 February 2022).
- Vrba, L.; Junk, D.J.; Novak, P.; Futscher, B.W. P53 Induces Distinct Epigenetic States at Its Direct Target Promoters. BMC Genomics 2008, 9, 486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Chi, C.H.; Zhang, Y.; Shi, B.; Jia, R.; Wang, B.J. Effects of Histone Deacetylase Inhibitors on ATP-Binding Cassette Transporters in Lung Cancer A549 and Colorectal Cancer HCT116 Cells. Oncol. Lett. 2019, 18, 63–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, B.; Xu, F.F.; Xiang, C.P.; Jia, R.; Yan, C.H.; Ma, S.Q.; Wang, N.; Wang, A.J.; Fan, P. Effect of Sodium Butyrate on ABC Transporters in Lung Cancer A549 and Colorectal Cancer HCT116 Cells. Oncol. Lett. 2020, 20, 148. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sobczak, M.; Strachowska, M.; Gronkowska, K.; Robaszkiewicz, A. Activation of ABCC Genes by Cisplatin Depends on the CoREST Occurrence at Their Promoters in A549 and MDA-MB-231 Cell Lines. Cancers 2022, 14, 894. https://doi.org/10.3390/cancers14040894
Sobczak M, Strachowska M, Gronkowska K, Robaszkiewicz A. Activation of ABCC Genes by Cisplatin Depends on the CoREST Occurrence at Their Promoters in A549 and MDA-MB-231 Cell Lines. Cancers. 2022; 14(4):894. https://doi.org/10.3390/cancers14040894
Chicago/Turabian StyleSobczak, Maciej, Magdalena Strachowska, Karolina Gronkowska, and Agnieszka Robaszkiewicz. 2022. "Activation of ABCC Genes by Cisplatin Depends on the CoREST Occurrence at Their Promoters in A549 and MDA-MB-231 Cell Lines" Cancers 14, no. 4: 894. https://doi.org/10.3390/cancers14040894





