A Prospective Phase II Study of Automated Non-Coplanar VMAT for Recurrent Head and Neck Cancer: Initial Report of Feasibility, Safety, and Patient-Reported Outcomes
Abstract
:Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Patient Characteristics
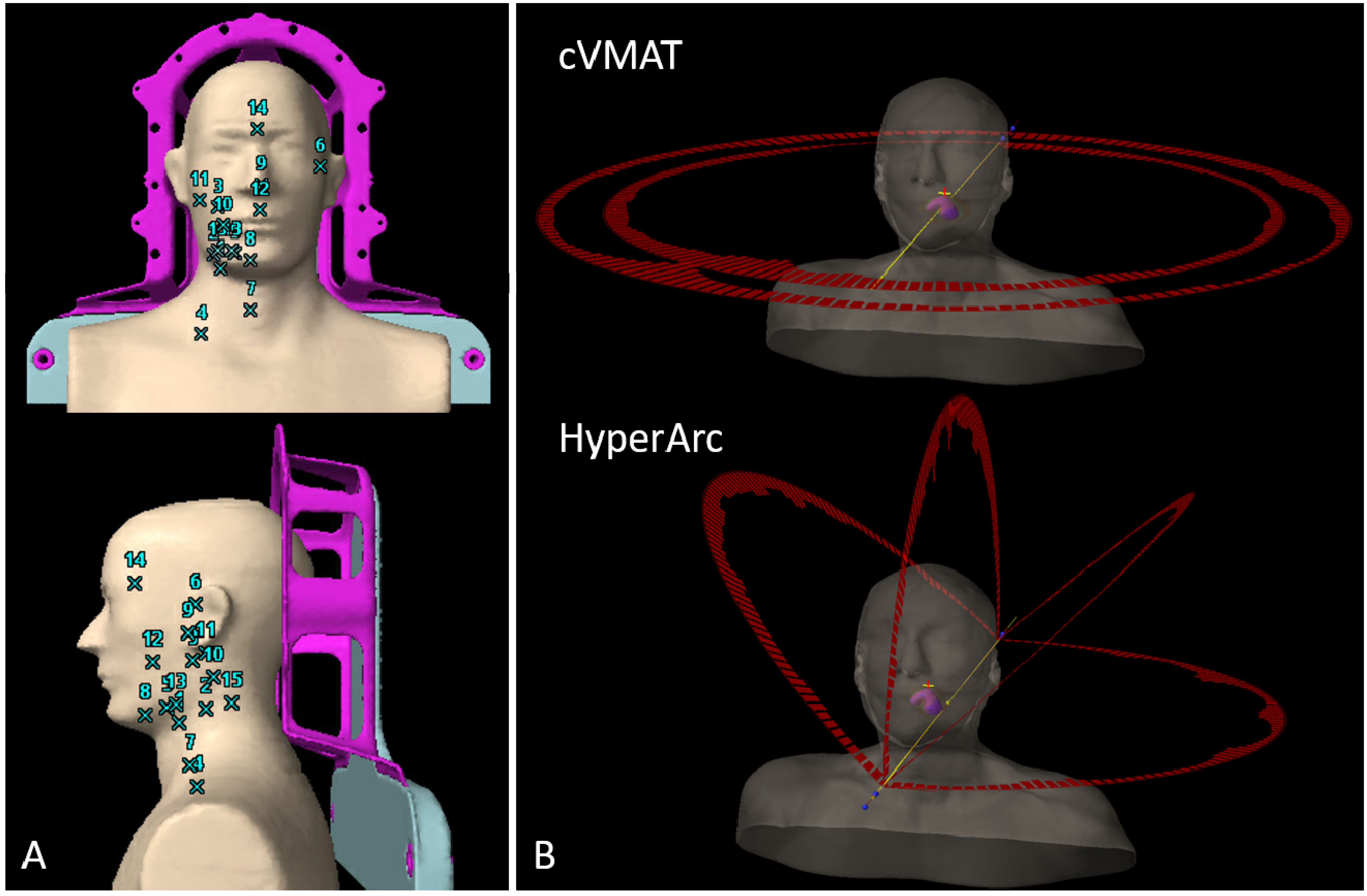
2.2. Simulation and Treatment Planning
2.3. Treatment Delivery
2.4. Clinical Assessment
2.4.1. Toxicity Measurement
2.4.2. Quality of Life Measurement
2.5. Statistical Analysis
3. Results
3.1. Dosimetry
3.2. Treatment Delivery
3.3. Toxicity and Patient-Reported Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Translations (mm) | Rotations (°) | |||||
|---|---|---|---|---|---|---|
| Lateral | Vertical | Longitudinal | Pitch | Yaw | Roll | |
| Shift measurements | 0.5 ± 0.4 | 0.4 ± 0.4 | 0.5 ± 0.3 | 0.2 ± 0.1 | 0.3 ± 0.1 | 0.2 ± 0.1 |
| Systematic error | 0.4 | 0.4 | 0.3 | 0.1 | 0.1 | 0.1 |
| Random error | 0.7 | 0.7 | 0.6 | 0.3 | 0.3 | 0.2 |
| van Herk PTV margin | 1.4 | 1.5 | 1.2 | 0.5 | 0.6 | 0.4 |

References
- Hall, S.F.; Groome, P.A.; Irish, J.; O’Sullivan, B. The natural history of patients with squamous cell carcinoma of the hypopharynx. Laryngoscope 2008, 118, 1362–1371. [Google Scholar] [CrossRef] [PubMed]
- Brockstein, B.; Haraf, D.J.; Rademaker, A.W.; Kies, M.S.; Stenson, K.M.; Rosen, F.; Mittal, B.B.; Pelzer, H.; Fung, B.B.; Witt, M.E.; et al. Patterns of failure, prognostic factors and survival in locoregionally advanced head and neck cancer treated with concomitant chemoradiotherapy: A 9-year, 337-patient, multi-institutional experience. Ann. Oncol. 2004, 15, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. Clinical Practice Guidelines in Oncology: Head and Neck Cancers (Version 3.2021). Available online: https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf (accessed on 11 February 2022).
- Janot, F.; de Raucourt, D.; Benhamou, E.; Ferron, C.; Dolivet, G.; Bensadoun, R.J.; Hamoir, M.; Géry, B.; Julieron, M.; Castaing, M.; et al. Randomized trial of postoperative reirradiation combined with chemotherapy after salvage surgery compared with salvage surgery alone in head and neck carcinoma. J. Clin. Oncol. 2008, 26, 5518–5523. [Google Scholar] [CrossRef] [PubMed]
- Haraf, D.J.; Weichselbaum, R.R.; Vokes, E.E. Re-irradiation with concomitant chemotherapy of unresectable recurrent head and neck cancer: A potentially curable disease. Ann. Oncol. 1996, 7, 913–918. [Google Scholar] [CrossRef]
- De Crevoisier, R.; Bourhis, J.; Domenge, C.; Wibault, P.; Koscielny, S.; Lusinchi, A.; Mamelle, G.; Janot, F.; Julieron, M.; Leridant, A.M.; et al. Full-dose reirradiation for unresectable head and neck carcinoma: Experience at the Gustave-Roussy Institute in a series of 169 patients. J. Clin. Oncol. 1998, 16, 3556–3562. [Google Scholar] [CrossRef]
- Takiar, V.; Garden, A.S.; Ma, D.; Morrison, W.H.; Edson, M.; Zafereo, M.E.; Gunn, G.B.; Fuller, C.D.; Beadle, B.; Frank, S.J.; et al. Reirradiation of Head and Neck Cancers With Intensity Modulated Radiation Therapy: Outcomes and Analyses. Int. J. Radiat. Oncol. Biol. Phys. 2016, 95, 1117–1131. [Google Scholar] [CrossRef]
- Ward, M.C.; Lee, N.Y.; Caudell, J.J.; Zajichek, A.; Awan, M.J.; Koyfman, S.A.; Dunlap, N.E.; Zakem, S.J.; Hassanzadeh, C.; Marcrom, S.; et al. A competing risk nomogram to predict severe late toxicity after modern re-irradiation for squamous carcinoma of the head and neck. Oral. Oncol. 2019, 90, 80–86. [Google Scholar] [CrossRef]
- Salama, J.K.; Vokes, E.E.; Chmura, S.J.; Milano, M.T.; Kao, J.; Stenson, K.M.; Witt, M.E.; Haraf, D.J. Long-term outcome of concurrent chemotherapy and reirradiation for recurrent and second primary head-and-neck squamous cell carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 2006, 64, 382–391. [Google Scholar] [CrossRef]
- Langer, C.J.; Harris, J.; Horwitz, E.M.; Nicolaou, N.; Kies, M.; Curran, W.; Wong, S.; Ang, K. Phase II study of low-dose paclitaxel and cisplatin in combination with split-course concomitant twice-daily reirradiation in recurrent squamous cell carcinoma of the head and neck: Results of Radiation Therapy Oncology Group Protocol 9911. J. Clin. Oncol. 2007, 25, 4800–4805. [Google Scholar] [CrossRef]
- Kress, M.A.; Sen, N.; Unger, K.R.; Lominska, C.E.; Deeken, J.F.; Davidson, B.J.; Newkirk, K.A.; Hwang, J.; Harter, K.W. Safety and efficacy of hypofractionated stereotactic body reirradiation in head and neck cancer: Long-term follow-up of a large series. Head Neck 2015, 37, 1403–1409. [Google Scholar] [CrossRef]
- Heron, D.E.; Ferris, R.L.; Karamouzis, M.; Andrade, R.S.; Deeb, E.L.; Burton, S.; Gooding, W.E.; Branstetter, B.F.; Mountz, J.M.; Johnson, J.T.; et al. Stereotactic body radiotherapy for recurrent squamous cell carcinoma of the head and neck: Results of a phase I dose-escalation trial. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 1493–1500. [Google Scholar] [CrossRef]
- Rwigema, J.C.; Heron, D.E.; Ferris, R.L.; Andrade, R.S.; Gibson, M.K.; Yang, Y.; Ozhasoglu, C.; Argiris, A.E.; Grandis, J.R.; Burton, S.A. The impact of tumor volume and radiotherapy dose on outcome in previously irradiated recurrent squamous cell carcinoma of the head and neck treated with stereotactic body radiation therapy. Am. J. Clin. Oncol. 2011, 34, 372–379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vargo, J.A.; Wegner, R.E.; Heron, D.E.; Ferris, R.L.; Rwigema, J.C.; Quinn, A.; Gigliotti, P.; Ohr, J.; Kubicek, G.J.; Burton, S. Stereotactic body radiation therapy for locally recurrent, previously irradiated nonsquamous cell cancers of the head and neck. Head Neck 2012, 34, 1153–1161. [Google Scholar] [CrossRef]
- Baliga, S.; Kabarriti, R.; Ohri, N.; Haynes-Lewis, H.; Yaparpalvi, R.; Kalnicki, S.; Garg, M.K. Stereotactic body radiotherapy for recurrent head and neck cancer: A critical review. Head Neck 2017, 39, 595–601. [Google Scholar] [CrossRef]
- Ling, D.C.; Vargo, J.A.; Ferris, R.L.; Ohr, J.; Clump, D.A.; Yau, W.W.; Duvvuri, U.; Kim, S.; Johnson, J.T.; Bauman, J.E.; et al. Risk of Severe Toxicity According to Site of Recurrence in Patients Treated With Stereotactic Body Radiation Therapy for Recurrent Head and Neck Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2016, 95, 973–980. [Google Scholar] [CrossRef]
- Rwigema, J.C.; Nguyen, D.; Heron, D.E.; Chen, A.M.; Lee, P.; Wang, P.C.; Vargo, J.A.; Low, D.A.; Huq, M.S.; Tenn, S.; et al. 4pi noncoplanar stereotactic body radiation therapy for head-and-neck cancer: Potential to improve tumor control and late toxicity. Int. J. Radiat. Oncol. Biol. Phys. 2015, 91, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.; Lee, P.; Ruan, D.; Long, T.; Romeijn, E.; Low, D.A.; Kupelian, P.; Abraham, J.; Yang, Y.; Sheng, K. 4π noncoplanar stereotactic body radiation therapy for centrally located or larger lung tumors. Int. J. Radiat. Oncol. Biol. Phys. 2013, 86, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.; Lee, P.; Ruan, D.; Long, T.; Romeijn, E.; Yang, Y.; Low, D.; Kupelian, P.; Sheng, K. 4π non-coplanar liver SBRT: A novel delivery technique. Int. J. Radiat. Oncol. Biol. Phys. 2013, 85, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Woods, K.; Lee, P.; Kaprealian, T.; Yang, I.; Sheng, K. Cochlea-sparing acoustic neuroma treatment with 4pi radiation therapy. Adv. Radiat. Oncol. 2018, 3, 100–107. [Google Scholar] [CrossRef] [Green Version]
- Murzin, V.L.; Woods, K.; Moiseenko, V.; Karunamuni, R.; Tringale, K.R.; Seibert, T.M.; Connor, M.J.; Simpson, D.R.; Sheng, K.; Hattangadi-Gluth, J.A. 4π plan optimization for cortical-sparing brain radiotherapy. Radiother. Oncol. 2018, 127, 128–135. [Google Scholar] [CrossRef]
- Yu, V.Y.; Landers, A.; Woods, K.; Nguyen, D.; Cao, M.; Du, D.; Chin, R.K.; Sheng, K.; Kaprealian, T.B. A Prospective 4π Radiation Therapy Clinical Study in Recurrent High-Grade Glioma Patients. Int. J. Radiat. Oncol. Biol. Phys. 2018, 101, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Wild, E.; Bangert, M.; Nill, S.; Oelfke, U. Noncoplanar VMAT for nasopharyngeal tumors: Plan quality versus treatment time. Med. Phys. 2015, 42, 2157–2168. [Google Scholar] [CrossRef] [PubMed]
- Woods, K.; Chin, R.K.; Cook, K.A.; Sheng, K.; Kishan, A.U.; Hegde, J.V.; Tenn, S.; Steinberg, M.L.; Cao, M. Automated Non-Coplanar VMAT for Dose Escalation in Recurrent Head and Neck Cancer Patients. Cancers 2021, 13, 1910. [Google Scholar] [CrossRef] [PubMed]
- Miften, M.; Olch, A.; Mihailidis, D.; Moran, J.; Pawlicki, T.; Molineu, A.; Li, H.; Wijesooriya, K.; Shi, J.; Xia, P.; et al. Tolerance limits and methodologies for IMRT measurement-based verification QA: Recommendations of AAPM Task Group No. 218. Med. Phys. 2018, 45, e53–e83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Herk, M.; Remeijer, P.; Rasch, C.; Lebesque, J.V. The probability of correct target dosage: Dose-population histograms for deriving treatment margins in radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2000, 47, 1121–1135. [Google Scholar] [CrossRef]
- Ivanova, A.; Qaqish, B.F.; Schell, M.J. Continuous toxicity monitoring in phase II trials in oncology. Biometrics 2005, 61, 540–545. [Google Scholar] [CrossRef]
- Ringash, J.; Bezjak, A. A structured review of quality of life instruments for head and neck cancer patients. Head Neck 2001, 23, 201–213. [Google Scholar] [CrossRef]
- List, M.A.; D’Antonio, L.L.; Cella, D.F.; Siston, A.; Mumby, P.; Haraf, D.; Vokes, E. The Performance Status Scale for Head and Neck Cancer Patients and the Functional Assessment of Cancer Therapy-Head and Neck Scale. A study of utility and validity. Cancer 1996, 77, 2294–2301. [Google Scholar] [CrossRef]
- Cella, D.F.; Tulsky, D.S.; Gray, G.; Sarafian, B.; Linn, E.; Bonomi, A.; Silberman, M.; Yellen, S.B.; Winicour, P.; Brannon, J. The Functional Assessment of Cancer Therapy scale: Development and validation of the general measure. J. Clin. Oncol. 1993, 11, 570–579. [Google Scholar] [CrossRef]
- Ringash, J.; Bezjak, A.; O’Sullivan, B.; Redelmeier, D.A. Interpreting differences in quality of life: The FACT-H&N in laryngeal cancer patients. Qual. Life Res. 2004, 13, 725–733. [Google Scholar] [CrossRef]
- Deleyiannis, F.W.; Weymuller, E.A.; Coltrera, M.D. Quality of life of disease-free survivors of advanced (stage III or IV) oropharyngeal cancer. Head Neck 1997, 19, 466–473. [Google Scholar] [CrossRef]
- Weymuller, E.A.; Yueh, B.; Deleyiannis, F.W.; Kuntz, A.L.; Alsarraf, R.; Coltrera, M.D. Quality of life in patients with head and neck cancer: Lessons learned from 549 prospectively evaluated patients. Arch. Otolaryngol. Head Neck Surg. 2000, 126, 329–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weymuller, E.A.; Alsarraf, R.; Yueh, B.; Deleyiannis, F.W.; Coltrera, M.D. Analysis of the performance characteristics of the University of Washington Quality of Life instrument and its modification (UW-QOL-R). Arch. Otolaryngol. Head Neck Surg. 2001, 127, 489–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hassan, S.J.; Weymuller, E.A. Assessment of quality of life in head and neck cancer patients. Head Neck 1993, 15, 485–496. [Google Scholar] [CrossRef]
- Rogers, S.N.; Gwanne, S.; Lowe, D.; Humphris, G.; Yueh, B.; Weymuller, E.A. The addition of mood and anxiety domains to the University of Washington quality of life scale. Head Neck 2002, 24, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Rogers, S.N.; Lowe, D. Screening for dysfunction to promote multidisciplinary intervention by using the University of Washington Quality of Life Questionnaire. Arch. Otolaryngol. Head Neck Surg. 2009, 135, 369–375. [Google Scholar] [CrossRef] [Green Version]
- Rogers, S.N.; Lowe, D.; Yueh, B.; Weymuller, E.A. The physical function and social-emotional function subscales of the University of Washington Quality of Life Questionnaire. Arch. Otolaryngol. Head Neck Surg. 2010, 136, 352–357. [Google Scholar] [CrossRef] [Green Version]
- Krayenbuehl, J.; Davis, J.B.; Ciernik, I.F. Dynamic intensity-modulated non-coplanar arc radiotherapy (INCA) for head and neck cancer. Radiother. Oncol. 2006, 81, 151–157. [Google Scholar] [CrossRef]
- Gayen, S.; Kombathula, S.H.; Manna, S.; Varshney, S.; Pareek, P. Dosimetric comparison of coplanar and non-coplanar volumetric-modulated arc therapy in head and neck cancer treated with radiotherapy. Radiat. Oncol. J. 2020, 38, 138–147. [Google Scholar] [CrossRef]
- Rwigema, J.-C.M.; Vargo, J.A.; Clump, D.A.; Ferris, R.L.; Ohr, J.; Heron, D.E. Stereotactic Body Radiotherapy in the Management of Head and Neck Malignancies. Curr. Cancer Ther. Rev. 2014, 10, 314–322. [Google Scholar] [CrossRef]
- Vergalasova, I.; Liu, H.; Alonso-Basanta, M.; Dong, L.; Li, J.; Nie, K.; Shi, W.; Teo, B.K.; Yu, Y.; Yue, N.J.; et al. Multi-Institutional Dosimetric Evaluation of Modern Day Stereotactic Radiosurgery (SRS) Treatment Options for Multiple Brain Metastases. Front. Oncol. 2019, 9, 483. [Google Scholar] [CrossRef] [PubMed]
- Lartigau, E.F.; Tresch, E.; Thariat, J.; Graff, P.; Coche-Dequeant, B.; Benezery, K.; Schiappacasse, L.; Degardin, M.; Bondiau, P.Y.; Peiffert, D.; et al. Multi institutional phase II study of concomitant stereotactic reirradiation and cetuximab for recurrent head and neck cancer. Radiother. Oncol. 2013, 109, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Vargo, J.A.; Ferris, R.L.; Ohr, J.; Clump, D.A.; Davis, K.S.; Duvvuri, U.; Kim, S.; Johnson, J.T.; Bauman, J.E.; Gibson, M.K.; et al. A prospective phase 2 trial of reirradiation with stereotactic body radiation therapy plus cetuximab in patients with previously irradiated recurrent squamous cell carcinoma of the head and neck. Int. J. Radiat. Oncol. Biol. Phys. 2015, 91, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Eekers, D.B.P.; Roelofs, E.; Jelen, U.; Kirk, M.; Granzier, M.; Ammazzalorso, F.; Ahn, P.H.; Janssens, G.O.R.J.; Hoebers, F.J.P.; Friedmann, T.; et al. Benefit of particle therapy in re-irradiation of head and neck patients. Results of a multicentric in silico ROCOCO trial. Radiother. Oncol. 2016, 121, 387–394. [Google Scholar] [CrossRef] [Green Version]
- Blanchard, P.; Gunn, G.B.; Lin, A.; Foote, R.L.; Lee, N.Y.; Frank, S.J. Proton Therapy for Head and Neck Cancers. Semin. Radiat. Oncol. 2018, 28, 53–63. [Google Scholar] [CrossRef]
- Kandula, S.; Zhu, X.; Garden, A.S.; Gillin, M.; Rosenthal, D.I.; Ang, K.K.; Mohan, R.; Amin, M.V.; Garcia, J.A.; Wu, R.; et al. Spot-scanning beam proton therapy vs. intensity-modulated radiation therapy for ipsilateral head and neck malignancies: A treatment planning comparison. Med. Dosim. 2013, 38, 390–394. [Google Scholar] [CrossRef]
- Bagley, A.F.; Garden, A.S.; Reddy, J.P.; Moreno, A.C.; Frank, S.J.; Rosenthal, D.I.; Morrison, W.H.; Gunn, G.B.; Fuller, C.D.; Shah, S.J.; et al. Highly conformal reirradiation in patients with prior oropharyngeal radiation: Clinical efficacy and toxicity outcomes. Head Neck 2020, 42, 3326–3335. [Google Scholar] [CrossRef]
- Romesser, P.B.; Cahlon, O.; Scher, E.D.; Hug, E.B.; Sine, K.; DeSelm, C.; Fox, J.L.; Mah, D.; Garg, M.K.; Han-Chih Chang, J.; et al. Proton Beam Reirradiation for Recurrent Head and Neck Cancer: Multi-institutional Report on Feasibility and Early Outcomes. Int. J. Radiat. Oncol. Biol. Phys. 2016, 95, 386–395. [Google Scholar] [CrossRef] [Green Version]
- McDonald, M.W.; Zolali-Meybodi, O.; Lehnert, S.J.; Estabrook, N.C.; Liu, Y.; Cohen-Gadol, A.A.; Moore, M.G. Reirradiation of Recurrent and Second Primary Head and Neck Cancer With Proton Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 808–819. [Google Scholar] [CrossRef]
- Phan, J.; Sio, T.T.; Nguyen, T.P.; Takiar, V.; Gunn, G.B.; Garden, A.S.; Rosenthal, D.I.; Fuller, C.D.; Morrison, W.H.; Beadle, B.; et al. Reirradiation of Head and Neck Cancers With Proton Therapy: Outcomes and Analyses. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 30–41. [Google Scholar] [CrossRef]
- Gordon, K.; Gulidov, I.; Semenov, A.; Golovanova, O.; Koryakin, S.; Makeenkova, T.; Ivanov, S.; Kaprin, A. Proton re-irradiation of unresectable recurrent head and neck cancers. Rep. Pract. Oncol. Radiother. 2021, 26, 203–210. [Google Scholar] [CrossRef] [PubMed]



| Pt | Age (years) | Sex | Initial Primary Tumor/RT Site | Prior Dose (Gy) | Interval between RT (months) | Recurrent Site | Histology | PTV Volume (cm3) | Concurrent Systemic Therapy |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 57 | M | Left floor of mouth | 66 1 | 4 | Right neck | SCC | 77.1 | Pembrolizumab |
| 2 | 59 | M | Right nasopharynx | 70 | 19 | Right neck | Undifferentiated carcinoma | 4.9 * [2.7, 1.2, 1.0] | None |
| 3 | 66 | F | Left floor of mouth | 66 | 3 | Right neck | SCC | 38.8 * [19.1, 19.6] | Pembrolizumab |
| 4 | 70 | F | Right oral tongue | 60 | 5 | Right supraclavicular | SCC | 13.8 | Pembrolizumab |
| 5 | 80 | M | Right base of tongue | 72 | 76 | Right base of tongue | SCC | 9.5 | None |
| 6 | 60 | M | Left parotid | 66 | 15 | Temporalis | Salivary ductal carcinoma | 44.5 | Trastuzumab emtansine |
| 7 | 75 | M | Floor of mouth | 70 | 38 | Inferior hypopharynx | SCC | 2.5 | None |
| 8 | 71 | M | Left oral tongue | 54 | 127 | Tongue | SCC | 9.5 | None |
| 9 | 51 | M | Right oral tongue | 66 | 40 | Palate, sphenoid | SCC | 30.0 * [11.6, 18.4] | None |
| 10 | 87 | M | Right floor of mouth | 66 2 | 33 | Left retropharyngeal LN | SCC | 7.2 | Pembrolizumab |
| 11 | 73 | M | Right base of tongue | 66 | 29 | Right parotid | SCC | 24.6 | None |
| 12 | 77 | M | Right base of tongue | 70 | 179 | Left soft palate | SCC | 15.2 | None |
| 13 | 53 | M | Left lateral tongue | Unk | 168 | Right oral cavity | SCC | 13.5 | Pembrolizumab |
| 14 | 67 | M | Right nasal passage | 60 | 33 | Left ethmoid Sinus | Adenocarcinoma | 43 | None |
| 15 | 85 | F | Hard palate | 66 | 48 | Right mandible | SCC | 9.9 | None |
| Delivery Time, Conformity, and Target Dose | |||||
|---|---|---|---|---|---|
| cVMAT | HyperArc | Absolute difference | p value | ||
| Mean delivery time (min) | 2.5 ± 0.7 | 5.2 ± 2.1 | 2.8 ± 2.2 | <0.001 * | |
| R50% | 4.1 ± 3.0 | 2.9 ± 1.3 | −1.2 ± 2.1 | 0.039 * | |
| Gradient measure | 0.8 ± 0.2 | 0.7 ± 0.1 | −0.1 ± 0.1 | <0.001 * | |
| PTV mean (Gy) | 40.6 ± 5.4 | 56.3 ± 9.1 | 15.7 ± 4.9 | <0.001 * | |
| PTV max (Gy) | 48.0 ± 7.2 | 68.4 ± 8.8 | 20.4 ± 7.0 | <0.001 * | |
| GTV mean (Gy) | 41.1 ± 6.5 | 58.2 ± 11.0 | 17.1 ± 6.0 | <0.001 * | |
| GTV max (Gy) | 47.8 ± 7.4 | 68.4 ± 8.9 | 20.6 ± 7.1 | <0.001 * | |
| Maximum OAR Doses † (Gy) | |||||
| Planning constraint | cVMAT | HyperArc | Absolute difference | p value | |
| Larynx | ≤20 | 3.6 ± 6.6 | 6.0 ± 6.1 | 2.4 ± 1.9 | <0.001 * |
| Cord | ≤8 | 3.7 ± 2.3 | 4.1 ± 1.7 | 0.4 ± 2.1 | 0.506 |
| Mandible | ≤20 | 11.4 ± 7.9 | 13.0 ± 6.3 | 1.6 ± 2.9 | 0.051 |
| Brainstem | ≤8 | 3.4 ± 4.6 | 4.7 ± 4.4 | 1.3 ± 2.4 | 0.047 * |
| Skin | ≤39.5 | 23.0 ± 11.8 | 21.3 ± 12.8 | −1.8 ± 2.2 | 0.009 * |
| Chiasm | ≤8 | 1.6 ± 3.2 | 2.8 ± 3.3 | 1.2 ± 3.1 | 0.167 |
| Right optic nerve | ≤8 | 1.1 ± 2.5 | 3.5 ± 3.0 | 2.4 ± 2.8 | 0.005 * |
| Left optic nerve | ≤8 | 2.9 ± 6.7 | 3.4 ± 4.4 | 0.5 ± 3.4 | 0.567 |
| Right cochlea | ≤25 | 0.8 ± 1.3 | 3.2 ± 2.5 | 2.3 ± 2.5 | 0.003 * |
| Left cochlea | ≤25 | 1.7 ± 3.9 | 3.4 ± 4.5 | 1.7 ± 2.0 | 0.006 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woods, K.E.; Ma, T.M.; Cook, K.A.; Morris, E.D.; Gao, Y.; Sheng, K.; Kishan, A.U.; Hegde, J.V.; Felix, C.; Basehart, V.; et al. A Prospective Phase II Study of Automated Non-Coplanar VMAT for Recurrent Head and Neck Cancer: Initial Report of Feasibility, Safety, and Patient-Reported Outcomes. Cancers 2022, 14, 939. https://doi.org/10.3390/cancers14040939
Woods KE, Ma TM, Cook KA, Morris ED, Gao Y, Sheng K, Kishan AU, Hegde JV, Felix C, Basehart V, et al. A Prospective Phase II Study of Automated Non-Coplanar VMAT for Recurrent Head and Neck Cancer: Initial Report of Feasibility, Safety, and Patient-Reported Outcomes. Cancers. 2022; 14(4):939. https://doi.org/10.3390/cancers14040939
Chicago/Turabian StyleWoods, Kaley E., Ting Martin Ma, Kiri A. Cook, Eric D. Morris, Yu Gao, Ke Sheng, Amar U. Kishan, John V. Hegde, Carol Felix, Vincent Basehart, and et al. 2022. "A Prospective Phase II Study of Automated Non-Coplanar VMAT for Recurrent Head and Neck Cancer: Initial Report of Feasibility, Safety, and Patient-Reported Outcomes" Cancers 14, no. 4: 939. https://doi.org/10.3390/cancers14040939






