1. Introduction
Cardiac tumors are rare overall. The most common are cardiac myxomas (CM), which are usually localized in the left atrium [
1,
2,
3]. Macroscopically, solid from non-solid or papillary CM are distinguished [
4]. CM do not cause a specific sign of illness. Symptoms such as dyspnea, palpitations, chest pain, and syncope can occur [
5]. When CM are not diagnosed incidentally, e.g., during echocardiography, cerebral emboli are often the first landmark clinical signs [
6].
To prevent embolic complications, patients diagnosed with CM should be transferred to a cardiac surgery center immediately after diagnosis, where cardiac tumor extirpation can be performed promptly. Outcomes after the complete surgical excision of CM appear to be good, and recurrence rates are probably very low [
7]. The bulk of publications on CM are case reports with impressive findings [
3,
8,
9]. Only a few authors have published their general experience in a series of patients with CM in original papers.
It is uncertain to date whether the outcome differs in affected patients in whom CM was discovered incidentally or in whom the diagnosis was established after cerebral tumor embolization. On the basis of this background, the present article describes our experience over 15 years.
2. Materials and Methods
2.1. Patients
All patients who underwent extirpation of a CM between 1 January 2005 and 31 December 2019 at our Department of Cardiovascular Surgery were retrospectively analyzed. The cohort gavewritten informed consent for research with patient data. The study was approved by the local ethics committee (D518/20).
2.2. Diagnostics
All CM were diagnosed by echocardiography and/or computed tomography or magnetic resonance imaging (
Figure 1,
Figure 2 and
Figure 3). After tumor excision, the diagnosis was confirmed by histological examination.
2.3. Surgical Techniques
After a median sternotomy, a heart–lung machine was connected via both the venae cavae and the ascending aorta. In cardioplegic cardiac arrest, the CM was then visualized via the right atrium, transseptally if necessary, and/or via the left atrium, depending on its location and extent (
Figure 4). After the resection of the attachment site and complete tumor removal, bovine pericardium was used to cover the defects of the endocardium or atrial septum, if necessary. This was followed by the closure of the cardiac access pathways and necessary adjunctive cardiac surgery interventions.
In cases with a minimally invasive approach, the connection to the heart–lung machine was via the femoral artery and femoral vein, and, optionally, via the internal jugular vein. The heart was accessed via an anterolateral minithoracotomy in the 5th intercostal space on the right side. The CM was then salvaged in the same manner as conventional surgery, during a period of cardioplegic arrest.
2.4. Follow-Up
The follow-up was conductedby mail. If patients could not be reached, contact was made with the relevant registry offices.
2.5. Statistical Analysis
The statistical analysis was performed using the IBM SPSS Statistics for Windows (Version 28.0., Armonk, NY, USA). Characteristics of the patient groups were presented as mean and standard deviations and compared by unpaired t-tests for approximately normally distributed continuous variables, while not-normally distributed continuous data, as well as ordinal data, were presented as medians with 25th and 75th percentiles and compared by the Mann–Whitney U test. Normal distribution was assessed by the Kolmogorov–Smirnovtest. Categorial data were summarized as absolute (n) and relative (%) frequencies and compared by a Chi2test or Fisher’s exact test, as appropriate. Survival was calculated on right-censored data by Kaplan–Meier analyses and compared for differences between the groups by a log rank test. All tests were 2-sided and a p-value of ≤0.05 was regarded as statistically significant. The primary endpoint was 30-day mortality. Secondary endpoints were intraoperative variables, and postoperative courses (e.g., ventilation time, bleeding, acute renal failure, neurologic complications, and late mortality).
3. Results
A total of 66 consecutive patients underwent cardiac surgery for CM at our hospital between the beginning of 2005 and the end of 2019. Females accounted for 56.1% of patients and the mean age was 61.8 years.
A comparison of patients who had an embolic stroke related to CM before cardiac surgery (SG;
n = 14) with those patients without prior stroke (N-SG;
n = 52) showed no significant differences in preoperative patient characteristics, concomitant cardiac diseases, and diagnostic procedures. The majority of CM were diagnosed by echocardiography (
Table 1,
Table 2 and
Table 3).
Regarding tumor-associated symptoms, neurological dysfunction was prominent in 100% of the SG. In 28.6% of patients, the left middle cerebral artery territory was affected. In the remaining patients (N-SG), dyspnea was the most common symptom in 34.6%, and 42.3% of them had no symptoms before diagnosis (
Table 4).
The time from diagnosis to surgical removal of the CM did not differ significantly between the groups (7 (3–24) days in the SG and 23 (5–55) days in the N-SG;
p = 0.120), and no patient suffered a first-time stroke or a recurrent stroke during the waiting period. Surgical time (215.5 min vs. 192.5 min;
p = 0.046) and intraoperative clamping time (78.5 min vs. 54.5 min;
p = 0.035) were significantly shorter in the SG. The respective proportion of minimally invasive approaches and combined cardiac surgery procedures did not differ significantly between the groups. CM access was significantly more frequent via the right atrium in the N-SG (57.7% vs. 14.3%;
p = 0.007), and exclusively in the N-SG, the resection site on the endocardium was reconstructed using bovine pericardium in 51.9% of cases (
p < 0.001) (
Table 5).
CM sizes of both groups were comparable (3.4 cm vs. 3.8 cm in their maximum lengths) (SG vs. N-SG;
p = 0.538). CM location was not significantly different between both groups (
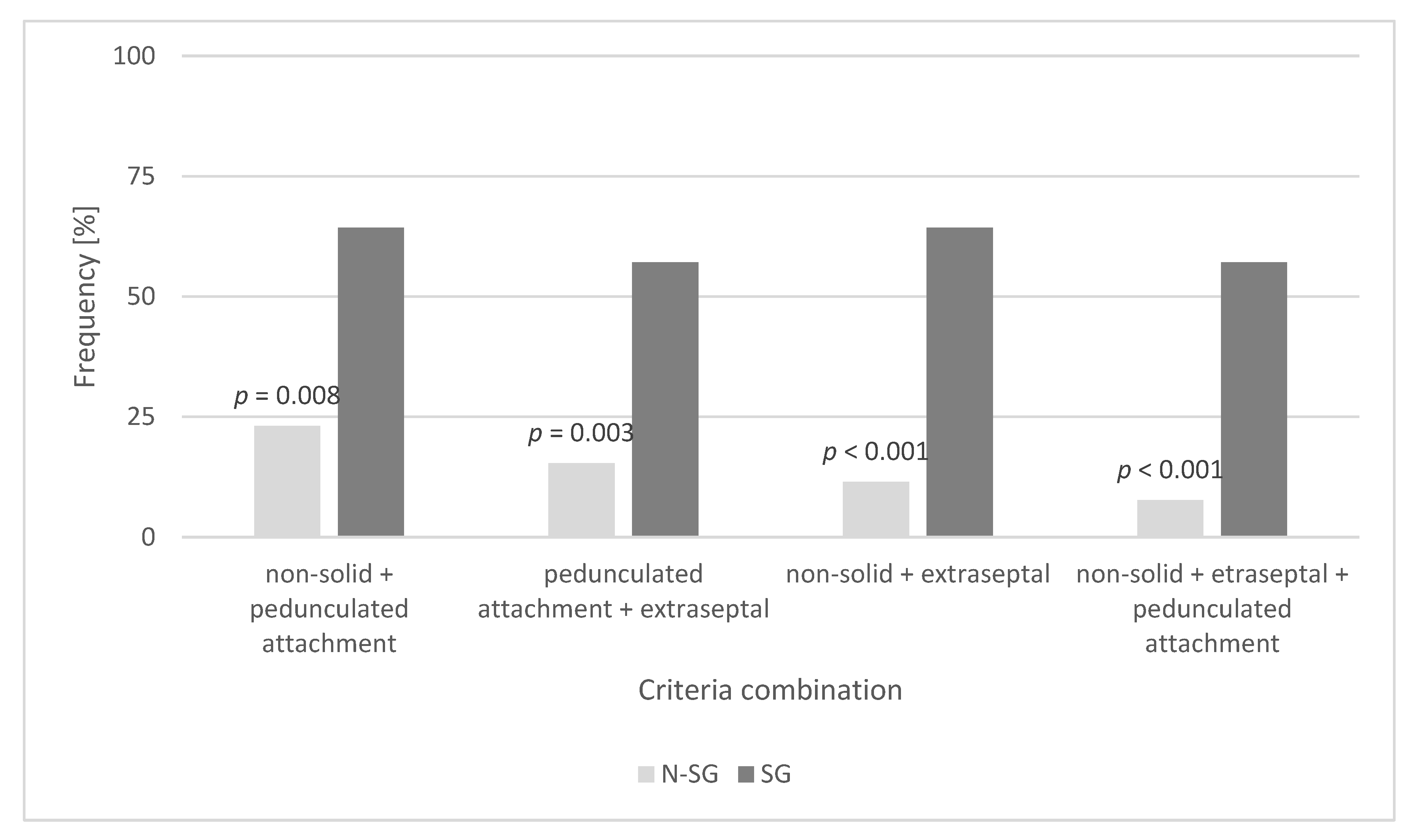
p = 0.436); most CM (92.9% (SG) and 78.8% (N-SG)) were found in the left atrium. In the SG, 57.1% of CM had a non-solid surface, were localized in the left heart, and had a pedunculated attachment away from the fossa ovalis. In the N-SG, 92.3% of CM did not meet all these criteria (
p < 0.001) (
Table 6 and
Figure 5).
Patients in the N-SG required a significantly shorter time in the intensive care unit after surgery (2 vs. 3 days;
p = 0.020). No other significant differences during hospitalization were observed. In the early course after tumor removal, 1.9% of the N-SG suffered their first stroke, and 14.3% of the SG had a cerebral re-infarction (
p = 0.111). The 30-daymortality was 1.9% (N-SG) and 7.1% (SG), with no significant discrepancy (
p = 0.382). One year after operation, the mortality rates were unchanged (
Table 7).
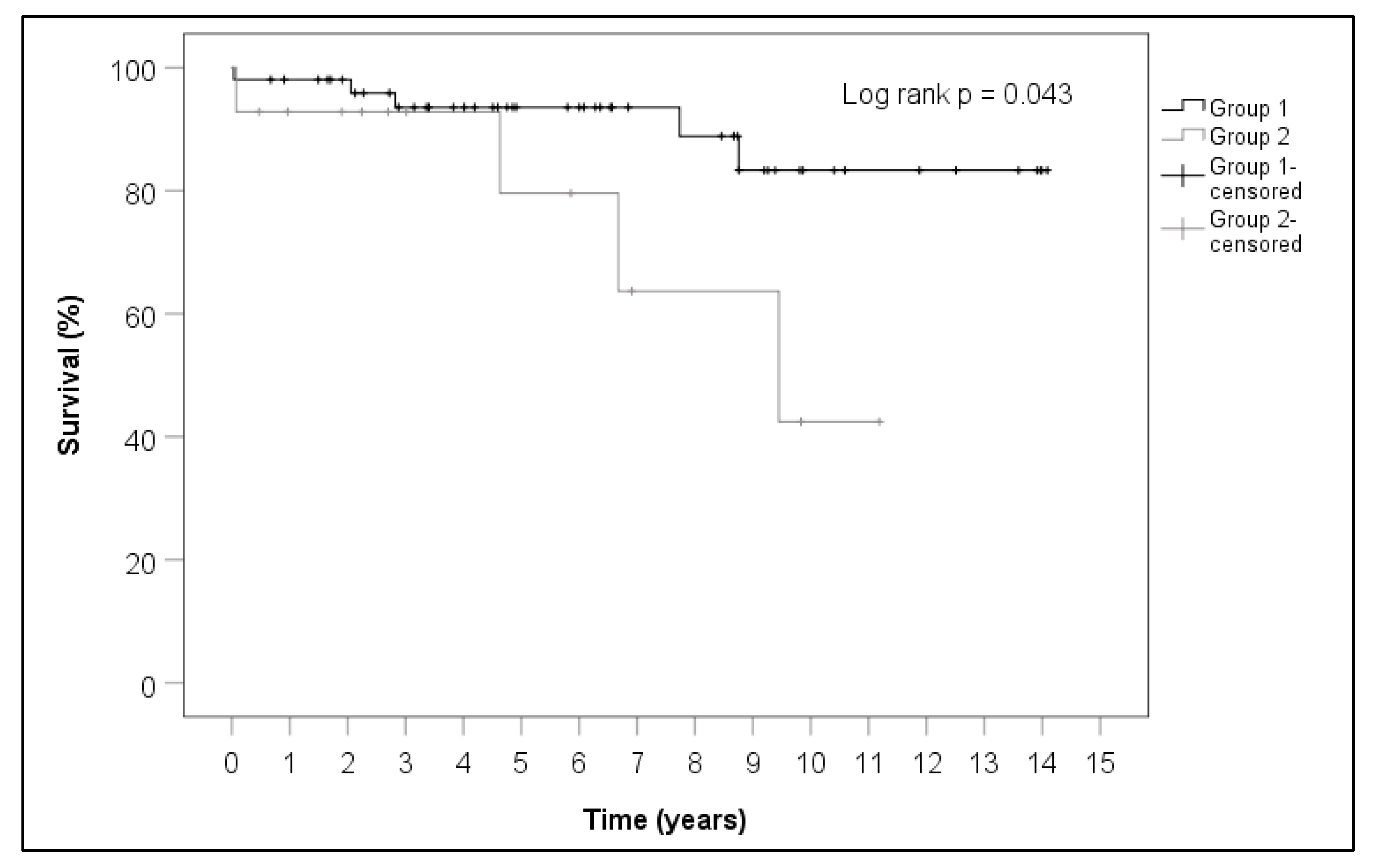
The follow-up showed that one patient in the N-SG underwent a re-operation for a recurrence of CM at our hospital. New strokes in the later course were not documented in eitherstudy group. Mortality was not significantly different at the time of follow-up (28.6% (SG) vs. 9.6% (N-SG);
p = 0.087). The calculations for the Kaplan–Meier curve showed a significant survival advantage for the patients in the N-SG (
p = 0.043) (
Table 8 and
Figure 6). Survival of the N-SG group and the SG group was 98% vs. 92% after one year, 93% vs. 92% after three years and 93% vs. 79% after five years.
4. Discussion
CM were represented in the literature through numerous case reports but in few original papers [
3]. The aim of the present work was to investigate whether the outcomes of patients who underwent removal of CM with or without pre-excision stroke differ. Special attention was paid to CM-related characteristics associated with preoperative stroke.
During the 15-year period considered, a total of 66 consecutive patients, 14 with (SG) and 52 without previous stroke (N-SG), underwent cardiac surgery for CM at our hospital. Considering the low incidence of CM, estimated at 0.5 cases per 1 million individuals per year, the low number of assigned CM patients is not surprising, even in larger cardiac centers [
7]. Females accounted for 56.1% of our studied patients. Other study groups were also more likely to encounter females suffering from CM than males. In the work of Tasoglu et al., as many as 77.6% of CM patients were female [
8]. The ratio of cerebral embolized cardiac myxomas in our collective is similar to the observations of Stefanou et al., who found eight patients with embolic stroke among 52 CM patients in their clinic within 12 years [
10].
CM localized in the left heart can potentially embolize in all cerebral areas. If a stroke was present preoperatively in our group, the left middle cerebral artery territory was most frequently affected (28.6%). Interestingly, Lee et al. also found a particularly frequent embolization of CM into the middle cerebral arteries in their studies [
11]. Our patients in the SG disproportionately (57.1%) had CM that were non-solid and had a pedunculated site of attachment in the left heart away from the fossa ovalis. In N-SG, 92.3% of the diagnosed CM did not meet this combination of the above criteria. Keeling et al. found a significant reason for embolization in terms of tumor consistency in their study [
12]. Swartz and coworkers observed a significant association between an extraseptal attachment site and neurologic events in their collective [
13]. Our results did not provide a single criterion for assessing the risk of embolization. Only the combination of at least two of the criteria we mentioned was significantly associated with cerebral embolization in our collective. With regard to the risk of embolization, the size of the CM per se did not play a role in our evaluations. This was confirmed by Garatti and colleagues [
14]. The significantly longer operative time and clamping time in the N-SG are related to the need for plastic reconstruction of the resection site with bovine pericardium. In our studies, no patients in the SG required such reconstruction, suggesting that there were no particularly deep or wide defects after the complete resection of the CM in this group. In the SG, the CM was explored less frequently via the right atrium, because it is almost localized in all patients in the left heart. Since 17.3% of the patients in N-SG had CM localization in the right atrium, it is plausible that exploration via the right atrium was significantly more frequent in these patients. We do not consider it unusual that patients with a previous stroke require a longer stay in the intensive care unit after cardiac surgery; moreover, the total hospital length of stay was comparable in both of our groups. Basically, cardiac surgery on the heart–lung machine with full heparinization in patients with acute or subacute stroke is associated with the risk of intracerebral hemorrhage during the procedure. Fortunately, however, none of our patients suffered this severe complication.
Most of our patients underwent regular cardiological examinations postoperatively. One patient (1.5%), who belonged to the N-SG, was diagnosed with a recurrence of CM, which required repeat surgical extirpation at our hospital. Other study groups reported similarly low recurrence rates [
11,
14,
15]. Neurologic follow-up was utilized by only one of our patients. In both groups, patients who participated in the follow-up did not experience significantly different rates of new, clinically manifested neurologic events during the course after surgery. Although 1-year mortality was still not significantly different, our patients in SG had a significantly worse long-term prognosis; however, the prognosis was still better than those patients with other malignant cardiac sarcomas, where their prognosis remains very poor and is considered a lethal disease [
16].