1. Introduction
Biomarkers that help to monitor the efficacy of treatment may be extremely useful for predicting prognosis, guiding the therapy and assessing the early response to it [
1]. There has been growing interest in the extent to which tumor markers may be of value for predicting the prognosis and treatment response in individual patients and for monitoring therapy.
One of these biomarkers is MUC1, which is overexpressed in many malignancies, particularly in breast cancer (BC) [
2,
3]. It comprises two subunits, a transmembrane (MUC1-C) and an extracellular subunit (MUC1-N). MUC1-C has been described as having oncogenic functions, interacting with receptor tyrosine kinases and activating several pathways including the PI3K-, MAPK-, NFκB-, and Wnt/β catenin pathways [
3].
MUC1-N can be shed, is soluble in serum, and can be measured with antibodies directed against epitopes of this subunit. The shed and soluble subunit is often referred to using the name of the antigen (CA27.29 or CA15-3)—i.e., the target of the analytic assay for the measurement of MUC1 levels [
2].
MUC1 is a prognostic marker in both early [
4,
5,
6,
7,
8,
9,
10,
11,
12,
13,
14,
15,
16,
17,
18] and advanced BC [
19,
20]. In most studies in patients with non-metastatic BC, blood for MUC1 assessment has been obtained preoperatively. Clear associations were found between tumor size and the soluble MUC1-level. While large studies have shown that soluble MUC1 is a predictor of prognosis for both early and advanced tumor stages independently of the tumor stage [
7,
9,
15], it may be hypothesized that the close association between tumor stage and MUC1 is due to the fact that MUC1 levels mirror the tumor burden in BC patients. However, testing of postoperative levels and additional assessment after chemotherapy might be more informative for analyzing the usefulness of MUC1 in relation to the efficacy of chemotherapy and other biological effects, such as its role in early systemic tumor spread.
The aim of this study was therefore to assess the association with disease-free survival and MUC1 as measured using a CA27.29 assay before and after adjuvant chemotherapy, taking into account established prognostic factors.
2. Materials and Methods
2.1. Study Design
In the SUCCESS-A open-label phase 3 trial, patients were randomly assigned at a ratio of 1:1 to either an anthracycline/taxane-based chemotherapy or to this chemotherapy regimen plus gemcitabine [
21]. Further treatment specifications are provided in the
Supplementary methods. The SUCCESS-A study was conducted as an investigator-initiated and led trial in Germany approved by all the ethics committees responsible and was conducted in accordance with the Declaration of Helsinki. All of the patients provided written informed consent before entering the study.
2.2. Participants
Patients were eligible if they were aged 18 or older and had a diagnosis of early, non-metastasized, high-risk invasive BC, defined by tumor stage, tumor grade, hormone receptor status or age. The full inclusion/exclusion criteria are provided in Additional
Table 1.
2.3. End Points, Follow-Up, and Data Capture
Disease-free survival was defined as the time from the last chemotherapy administration to the earliest date of disease progression (distant metastasis, local recurrence, death from any cause) or the date of censoring. Patients who were lost to follow-up before the maximum observation time of 5.5 years or were disease-free after the maximum observation time were censored at the last date on which they were known to be disease-free or at the maximum observation time. The maximum observation period of 5.5 years consists of six months of chemotherapy and (up to) five years of follow-up thereafter.
For survival and recurrence assessment, the patients were followed at the study sites at 3-month intervals for the first three years and every six months thereafter. The follow-up included clinical examinations (at each visit), mammography (every six months), and symptom-driven examinations if necessary. High quality of the data was ensured by electronic data management, including automated plausibility checks and regular monitoring visits to the study site by an independent clinical research organization (Alcedis GmbH, Giessen, Germany) and a data monitoring committee (DMC).
2.4. Assessment of Soluble MUC1 with the CA27.29 Assay
Laboratory analysis was performed centrally in the Department of Gynecology and Obstetrics at Munich University Hospital [
22]. Approximately 10 mL of peripheral blood was drawn by peripheral vein puncture in standard serum tubes and centrifuged (10 min, 2000×
g, room temperature) within 24–72 h following the collection time to remove clots. The serum was immediately transferred to an immunoreaction cup from the ST AIA-Pack 27.29 series (Tosoh Bioscience, Tessenderlo, Belgium) for further analysis. The CA27.29 serum concentration was measured using the AIA-600 II automated enzyme immunoassay system (Tosoh Bioscience, Tessenderlo, Belgium) in accordance with the manufacturer’s instructions. In brief, serum samples were combined with a diluent (1:20) and were transferred to an immunoreaction cup from the ST-AIA-Pack 27.29 series. CA27.29 was immobilized using magnetic beads conjugated to antibodies. Enzyme-labeled antibodies attached to a different epitope were then bound to the CA27.29 antigen in a sandwich manner. The samples were then incubated at 37 °C, followed by a washing step to remove any unbound antibody. The fluorogenic substrate 4-methylumbelliferyl phosphate was added to the test cup, and enzyme activity was measured on the basis of the amount of fluorescence.
2.5. Statistical Methods
The primary objective was to study whether information about CA27.29 before and after chemotherapy improves the ability to predict disease-free survival for each patient, in addition to other well-known predictors. For this purpose, Cox regression analyses were performed as described in the
Supplementary methods [
23,
24,
25,
26,
27,
28]. In brief, a Cox regression model for commonly known predictors was compared with a further model containing those variables and additionally CA27.29 before and after chemotherapy and all relevant interaction terms for those variables (“full model”) using the likelihood ratio test (LRT). In case of significance, the full model was tested for the relevance of the included interaction terms by comparing the full model with a model without interaction terms using a second LRT. In the case of significance, a variable selection procedure was carried out to identify relevant interaction terms (“final model”). Hazard ratios (HRs) and survival rates were estimated using the final model. Furthermore, the predictive ability of CA27.29 before and after chemotherapy was compared.
All of the tests were two-sided, and a
p value of <0.05 was regarded as statistically significant. Calculations were carried out using R statistical software (V3.0.1, 2013, The R Foundation for Statistical Computing, Vienna, Austria). Remark criteria were used to report tumor marker data [
29].
3. Results
3.1. Patient Characteristics
In total, 3754 patients were randomly assigned in the SUCCESS-A study. For inclusion in the present analysis, patients were required to have serum samples for CA27.29 analysis at the time of study inclusion and after chemotherapy (
n = 2687). Complete information on all variables as listed in
Table 1 was available for 98.2% of these patients. The percentage of values missing for each variable was less than 0.5%, with the exception of HER2 (1.5%). The characteristics of the patients are shown in
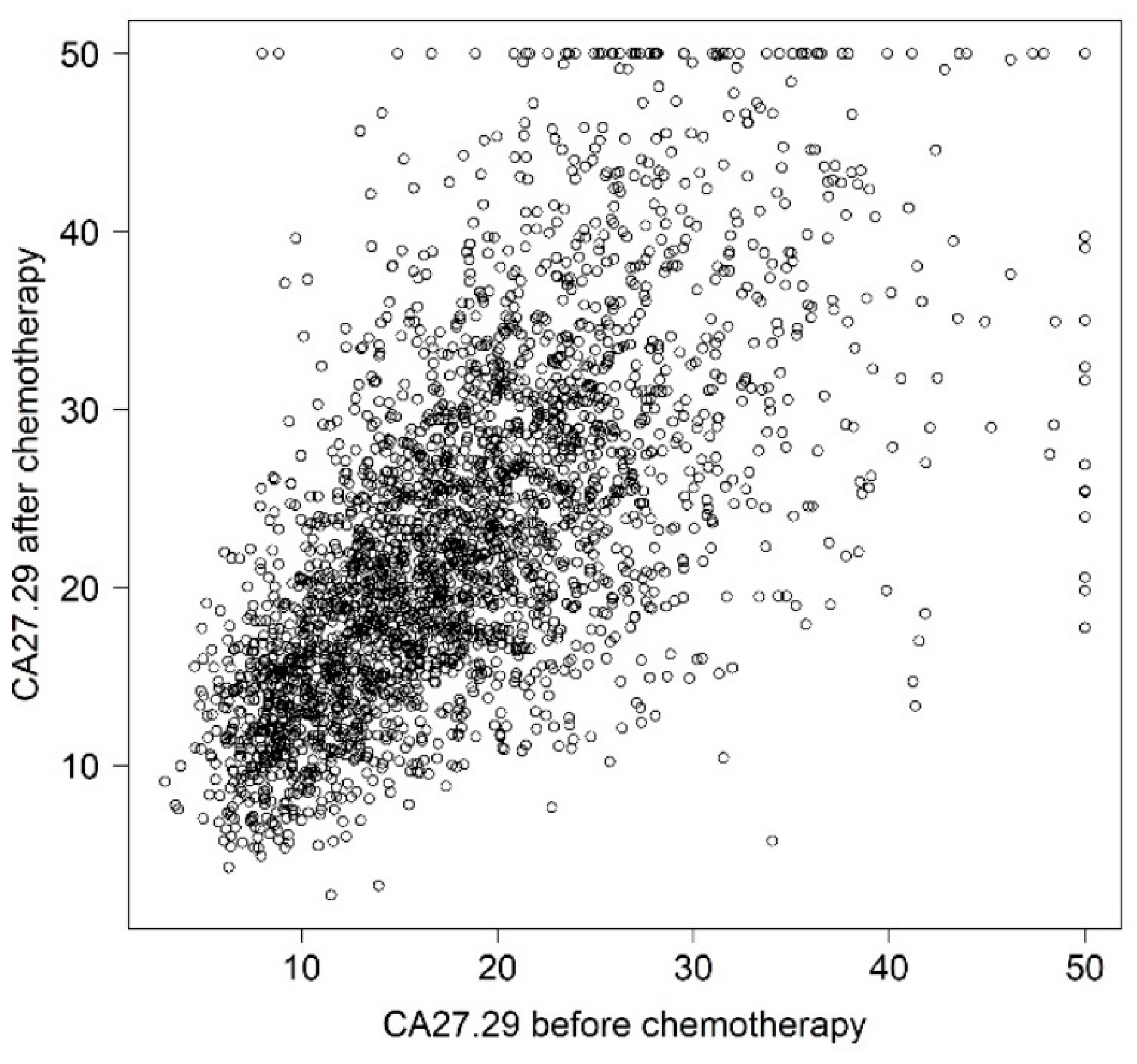
Table 1. CA27.29 values before and after chemotherapy were strongly correlated (
Figure 1).
3.2. Prediction of Disease-Free Survival
CA27.29 was significantly associated with disease-free survival additionally to other predictors (p < 0.000001, first LRT). The effect of CA27.29 on survival differed between patient subgroups (p < 0.001, second LRT). The variable selection process resulted in a final Cox regression model that included, besides the predictors of the reduced model, the interactions of CA27.29 before chemotherapy relative to lymph node status (pN) and tumor size (pT). CA27.29 before chemotherapy was nonlinearly associated with survival, with two degrees of freedom, whereas CA27.29 after chemotherapy was best described as a linear predictor.
The expected probability of 5-year survival was calculated for each patient using the final model. Many patients were found to have a high probability of living free of disease for at least five years, whereas only a few patients had a low probability (
Figure S1). The mean and median probabilities were 85.9% and 88.6%, respectively. One quarter of all patients had a likelihood less than 82.6%, and one quarter had a likelihood greater than 92.5%.
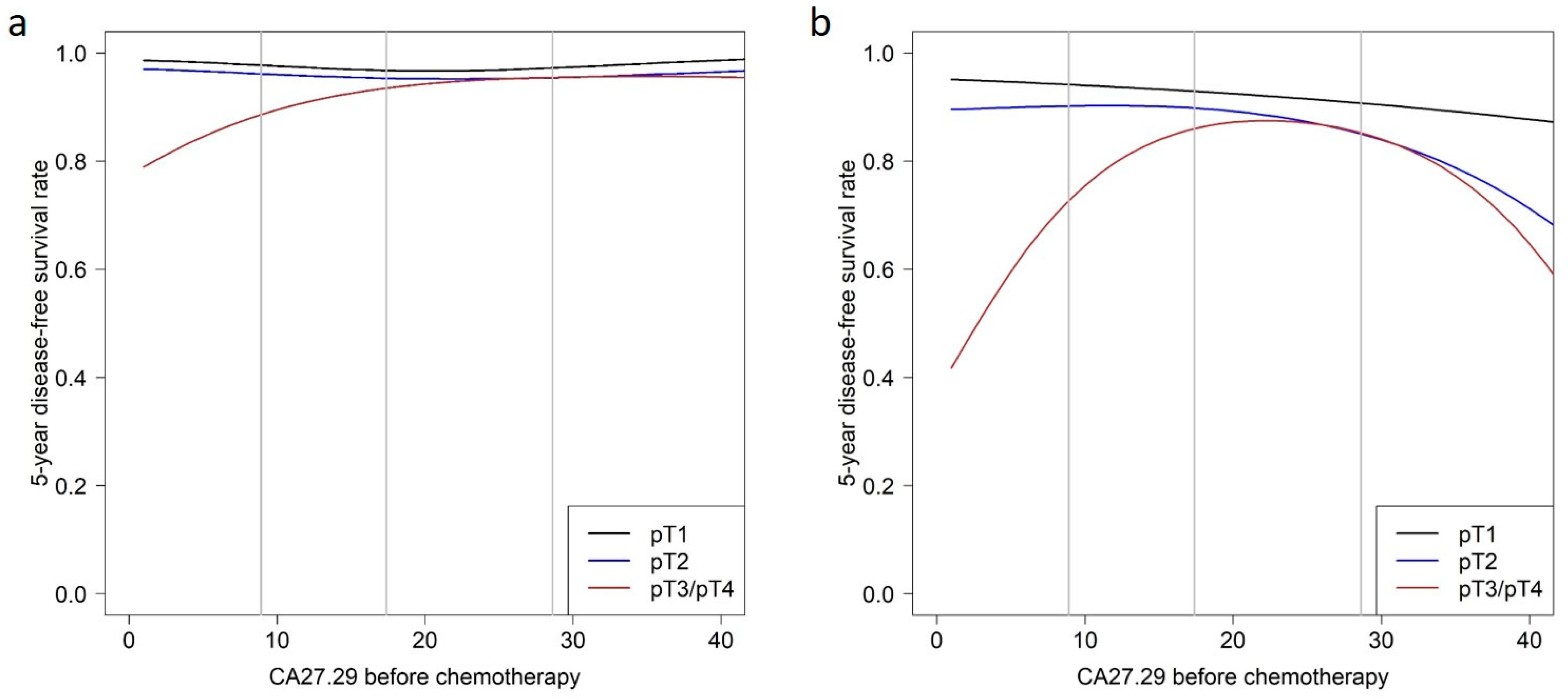
Patient subgroups were defined based on the two predictors pN and pT. pN0 is defined as no cancer cells in any nearby nodes and pN+ as the presence of cancer cells in lymph nodes. pT1 means that the tumor is 2 cm across or less, pT2 means that the tumor size is more than 2 cm but no more than 5 cm, and pT3/4 means that the tumor is bigger than 5 cm or of any size growing into the chest wall or skin. No association of CA27.29 before chemotherapy with survival was observed in patients with lymph node-negative tumors (
Table 2 and
Figure 2a).
In patients with lymph node-positive tumors, CA27.29 only showed a minor association with survival in patients with pT1 tumors. For each CA27.29 value before chemotherapy, the survival rate for patients with pT2 tumors was lower than that of patients with pT1 tumors with the same CA27.29 value. In patients with pT2 tumors, survival rates were constantly high when CA27.29 was below the median, but the survival rates decreased with increasing CA27.29 values. The survival prognosis in patients with large tumors (pT3 or pT4) improved with increasing CA27.29 values, but only up to values of 25 U/mL. Patients with CA27.29 values higher than 25 U/mL had a prognosis similar to that in patients with pT2 tumors—the higher the CA27.29 value, the poorer the prognosis. Moreover, the survival rates of patients with pT2 tumors were similar to those of patients with pT3/4 tumors in this range of above-average CA27.29 (
Table 2 and
Figure 2b and
Figure 3).
No association between CA27.29 after chemotherapy and the disease-free survival was found. The adjusted HR per 10 U/mL increase of CA27.29 was 0.95 (95% CI, 0.83–1.10).
In summary, a “high-risk patient” is a patient with a lymph-node positive tumor with at least pT2 and high CA27.29 values before chemotherapy or pT3/4 and low or high but not intermediate CA27.29 values before chemotherapy. The survival prognosis is further increased or decreased, respectively, by histology, grading, ER, PR, and HER status. For instance, a patient with a tumor classified as T2, G3, ER+, PR+, and HER− is predicted to have a 5-year disease-free survival with a likelihood of 85% if the preoperative CA27.29 level was low and a likelihood of 75% if the preoperative CA27.29 was high.
Table 3 shows the predicted 5-year survival rates obtained from the final model.
Both the full and final models performed better than the basic model without CA27.29 predictors with regard to distinguishing between patients with and without events up to two and five years of follow-up, respectively (
Supplementary Table S2, cross-validated AUC). The full model, however, performed better than the final model; the difference in the cross-validated AUC was 0.041 at two years and 0.06 at five years, implying a certain joint influence of predictors that are poorly predictive on their own. A comparison between the apparent and cross-validated AUC shows that the final model was less overfitted than the full model. The addition of CA27.29 before chemotherapy to the basic model improved prediction, whereas the addition of CA27.29 after chemotherapy did not, confirming the results of the main analysis.
4. Discussion
This study showed that CA27.29 after surgery but before chemotherapy provides prognostic information additional to that available with commonly known prognostic factors. CA27.29 after chemotherapy did not add any prognostic information in addition to commonly known prognostic factors and CA27.29 before chemotherapy. The impact of CA27.29 differed relative to tumor size and nodal status—mainly confining the association of CA27.29 and prognosis to node-positive patients.
In this setting, serial use of CA27.29 did not increase its usefulness as a biomarker. However, there have been several reports in which it was not a single, static soluble MUC1 value that was used for prognostic or predictive purposes, but rather serial measurements with adjustment of later values by the earlier ones [
19,
30,
31,
32,
33,
34,
35,
36,
37,
38,
39,
40]. There are several potential reasons why the two values in the present study were not superior to the one before chemotherapy alone. Firstly, the two assessments were highly correlated (
Figure 1). Secondly, the values after chemotherapy may reflect a baseline value for MUC1 rather than an activity marker for the disease [
30], assuming that at the second time point, the time interval is long enough to exclude any influence of the primary disease on the MUC1 value, and the likelihood of recurrence is rather low. The prognostic information provided by CA27.29 after chemotherapy may therefore be rather low.
Before chemotherapy, two interactions were observed: one between tumor size and CA27.29, and one between nodal status and CA27.29. Patients with a negative nodal status had an excellent prognosis. However, no association between CA27.29 and the prognosis was observed. This might be due to general better prognosis of node-negative BC patients resulting in less impact of CA27.29 levels [
41,
42].
An effect of CA27.29 on the prognosis was seen in node-positive patients, with the exception of patients with advanced tumor stages (pT3/4). In the latter, low CA27.29 values were associated with an unfavorable prognosis. Interactions between molecular subtypes were not analyzed since the assumptions for the proportional hazard function were not met and there was a strong correlation between ER and PR. Several larger studies have reported associations between tumor size, nodal status, and MUC1 [
7,
9,
12,
17]. Patients with larger tumors and positive lymph-node status had higher preoperative MUC1 values. Postoperative values were assessed in only one of these studies [
15], but no association between tumor size, nodal status and CA15-3 was reported. In the present study, there was an association between tumor size and postoperative/before chemotherapy CA27.29 values, but not with nodal status, a finding that was also reported by Rack et al. [
43].
Only one study has investigated the association of postoperative MUC1 assessments in relation to other prognostic factors (tumor size, lymph-node status, histological grading, and hormone receptor status) [
15]. Carcinoembryonic antigen as well as CA15-3 was included but did not have a prognostic effect in the multivariable model. The study also found that postoperative values differ from preoperative ones [
15]. Approximately 18% of all patients had a decrease in CA15-3 values of more than 33%. The decrease was even more prominent in node-positive patients [
15]. This might be an indication that MUC1 plays a different role in node-positive patients than in node-negative ones, but it might also be due to the fact that node-negative patients do not have elevated MUC1 levels initially and can therefore not achieve a decrease after surgery that easily.
MUC1 has been widely discussed as a prognostic biomarker and potential target for anticancer treatment [
44,
45]. In addition to this, however, MUC1 has been linked to immune regulatory mechanisms such as protection from infections, regulation of inflammatory response and, in particular, its ability to function as a T cell regulator and checkpoint molecule [
46,
47,
48,
49]. With respect to the latter, co-stimulatory and co-inhibitory abilities are discussed, which might influence the activity of regulatory T cells, but also CD4+ or CD8+ cells in general [
47,
48]. Thus, the association of low CA27.29 values and poor prognosis of node-positive patients with advanced tumor stage (pT3/4) might be due to the loss of the MUC1-regulated immune response. A lack of co-stimulatory and co-inhibitory MUC1 activities might particularly impact patients with advanced tumor stage and cancer-affected lymph nodes. Thus, for future studies, it could be of interest to evaluate the proliferation, differentiation, and activation of circulating immune cells in addition to CA27.29 values [
49].
MUC1 has been prioritized as a promising target for anticancer therapies [
44,
45]. Vaccination strategies appear to be of particular interest, as MUC1 is overexpressed in BC and other cancer histologies and has been reported to have a high level of immunogenicity [
45,
50]. The present study shows that node-negative patients may not be ideal for an anti-MUC1 therapeutic study. Patients should potentially be node-positive with high MUC1 serum levels to derive benefit.
This study has several strengths and weaknesses. It is the first large-scale study in which samples have been analyzed in a multicenter prospective phase III chemotherapy investigation. CA27.29 was assessed centrally, ensuring high-quality data and standardized analytic methods. It therefore appears to be justifiable to generalize the results, although the study was restricted to a high-risk group undergoing chemotherapy. While the overall sample size was reasonably high with more than 2600 patients, the unfavorable prognostic effect in patients with pT3/4 and low CA27.29 may be imprecise due the small sample size for that specific patient group.
5. Conclusions
In conclusion, this study did not show additional prognostic value for serial CA27.29 assessment before and after chemotherapy alongside commonly used prognostic factors and postoperative CA27.29 measurements. In combination with tumor size and nodal status, as well as other prognostic factors, however, serial CA27.29 measurements can identify patients with an unfavorable prognosis. This patient population may be suitable for further anti-MUC1 treatment after standard therapies.
Supplementary Materials
The following supporting information can be downloaded at:
https://www.mdpi.com/article/10.3390/cancers14071721/s1, Figure S1: Distribution of the predicted 5-year disease-free survival probability (0–100%) in the study population (
n = 2687); Table S1: Full Inclusion and Exclusion Criteria; Table S2: Performance of Cox Regression Models [
23,
24,
25,
26,
27,
28].
Author Contributions
Conceptualization, V.M., A.S., H.T., T.N.F., M.W.B., P.A.F., W.J. and B.R.; data curation, L.H.; formal analysis, H.H., L.H., V.M., I.S., H.F., V.F., I.B., S.M., J.J., N.N., A.S., H.T., S.Y.B., J.-U.B., T.N.F., G.H. and M.R.; investigation, R.L., H.F., V.F., F.S., I.B., S.M., J.J., N.N., S.Y.B., J.-U.B., G.H. and M.R.; methodology, P.A.F.; project administration, F.S.; resources, I.S., R.L., I.B., A.S., T.N.F., M.W.B. and B.R.; supervision, P.A.F., W.J. and B.R.; visualization, H.H. and L.H.; writing—original draft, H.H., L.H. and P.A.F.; writing—review & editing, V.M., I.S., R.L., H.F., V.F., F.S., I.B., S.M., J.J., N.N., A.S., H.T., S.Y.B., J.-U.B., T.N.F., G.H., M.R., M.W.B., W.J. and B.R. All authors revised the manuscript critically for important intellectual content. All authors have approved the submitted version and have agreed both to be personally accountable for the author’s own contributions and to ensure that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and the resolution documented in the literature. All authors have read and agreed to the published version of the manuscript.
Funding
SUCCESS-A was in part supported by grants from Novartis, AstraZeneca, Chugai, SanofiAventis, and Lilly. The study was investigator-initiated and investigator-led, and the bodies providing financial support played no part in the study design, data collection, data analysis, data interpretation, or writing of the report. The authors had full access to all of the data and had final responsibility for the decision to submit for publication. We acknowledge financial support by Deutsche Forschungsgemeinschaft and Friedrich-Alexander-Universität Erlangen-Nürnberg within the funding programme “Open Access Publication Funding”.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional the ethical board of the Ludwig-Maximilian-University of Munich (Project Nr. 076-05).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request. The data are not publicly available due to ethical restrictions.
Acknowledgments
We would like to thank all patients and the staff involved in the study at the participating study sites.
Conflicts of Interest
P.A.F.: honoraria from Novartis, Amgen, Pfizer, Celgene, Roche, Puma. V.M.: received speaker honoraria from Amgen, Astra Zeneca, Celgene, Daiichi-Sankyo, Eisai, Pfizer, MSD, Novartis, Roche, Teva, and consultancy honoraria from Genomic Health, Hexal, Roche, Pierre Fabre, Amgen, ClinSol, Novartis, MSD, Daiichi-Sankyo, Eisai, Lilly, Tesaro and Nektar. Institutional research support from Novartis, Roche, Seattle Genetics, Genentech. A.S.: honoraria from Roche, Celgene. H.T.: honoraria from Novartis, Roche, AMGEN, Pfizer, Astra Zeneca, Eisai, MSD, Lilly. S.M. has received research support, advisory board, honoraria and travel expenses from AbbVie, AstraZeneca, Clovis, Eisai, GlaxoSmithKline, Medac, MSD, Novartis, Olympus, PharmaMar, Roche, Sensor Kinesis, Teva, Tesaro. S.Y.B.’s honoraria from Novartis, Pfizer, Roche, MSD, Teva. J.-U.B.: honoraria from Roche, Novartis, Pfizer, AstraZeneca and MSD. M.W.B.’s institution conducts research with funding from Novartis, Pfizer. W.J.: Sanofi-Aventis, Novartis, Pfizer. B.R.: honoraria from AstraZeneca, Chugai, Lilly, Novartis, Sanofi-Aventis, Janssen-Cilag. N.N.: is currently an employee of Novartis and has received travel support from Novartis and TEVA in the past. F.S.: Speaker honoraria from Roche, Novartis, Pfizer, GSK, AstraZenica und Clovis. Travel expenses: Lilly, Pfizer, Roche, Novartis. All other authors have no conflict of interest.
Abbreviations
| AE | Adverse event |
| ANC | Absolute neutrophil count |
| BC | Breast cancer |
| BMI | Body mass index |
| CI | Confidence interval |
| CMF | Cyclophosphamide, methotrexate and fluorouracil |
| CTC | Circulating tumor cells |
| DFS | Disease-free survival |
| Doc | Docetaxel |
| ER | Estrogen receptor |
| FEC | 5-fluoroucacil, epirubicin and cyclophosphamide |
| 5-FU | 5-fluorouracil |
| G-CSF | Granulocyte-colony stimulating factor |
| Gem | Gemcitabine |
| HER2 | Human epidermal growth factor receptor 2 |
| HR | Hazard ratio |
| HRS | Hormone receptor status |
| MAPK | Mitogen-activated protein kinase |
| MUC1-C | Carboxy-terminal transmembrane subunit |
| MUC1-N | Amino-terminal extracellular subunit |
| NFκB | Nuclear factor kappa B |
| OS | Overall survival |
| PI3K | Phosphatidylinositol-3′-kinase |
| PR | Progesterone receptor |
| SGPT | Serum glutamate pyruvate transaminase |
References
- Hartkopf, A.D.; Muller, V.; Wockel, A.; Lux, M.P.; Janni, W.; Ettl, J.; Belleville, E.; Schutz, F.; Fasching, P.A.; Kolberg, H.C.; et al. Translational Highlights in Breast and Ovarian Cancer 2019—Immunotherapy, DNA Repair, PI3K Inhibition and CDK4/6 Therapy. Geburtshilfe Frauenheilkd 2019, 79, 1309–1319. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.J.; Evoy, D.; McDermott, E.W. CA 15-3: Uses and limitation as a biomarker for breast cancer. Clin. Chim. Acta 2010, 411, 1869–1874. [Google Scholar] [CrossRef] [PubMed]
- Kufe, D.W. MUC1-C oncoprotein as a target in breast cancer: Activation of signaling pathways and therapeutic approaches. Oncogene 2013, 32, 1073–1081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, Y.; Sun, X.; He, Y.; Liu, C.; Liu, H. Elevated Levels of Serum Tumor Markers CEA and CA15-3 Are Prognostic Parameters for Different Molecular Subtypes of Breast Cancer. PLoS ONE 2015, 10, e0133830. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.G.; He, Z.Y.; Zhou, J.; Sun, J.Y.; Li, F.Y.; Lin, Q.; Guo, L.; Lin, H.X. Serum levels of CEA and CA15-3 in different molecular subtypes and prognostic value in Chinese breast cancer. Breast 2014, 23, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chen, K.; Su, F.; Song, E.; Gong, C. Preoperative CA 15-3 levels predict the prognosis of nonmetastatic luminal A breast cancer. J. Surg. Res. 2014, 189, 48–56. [Google Scholar] [CrossRef]
- Lee, J.S.; Park, S.; Park, J.M.; Cho, J.H.; Kim, S.I.; Park, B.W. Elevated levels of preoperative CA 15-3 and CEA serum levels have independently poor prognostic significance in breast cancer. Ann. Oncol. 2013, 24, 1225–1231. [Google Scholar] [CrossRef]
- Ali, H.Q.; Mahdi, N.K.; Al-Jowher, M.H. The value of CA15-3 in diagnosis, prognosis and treatment response in women with breast cancer. JPMA J. Pak. Med. Assoc. 2013, 63, 1138–1141. [Google Scholar]
- Sandri, M.T.; Salvatici, M.; Botteri, E.; Passerini, R.; Zorzino, L.; Rotmensz, N.; Luini, A.; Mauro, C.; Bagnardi, V.; Cassatella, M.C.; et al. Prognostic role of CA15.3 in 7942 patients with operable breast cancer. Breast Cancer Res. Treat. 2012, 132, 317–326. [Google Scholar] [CrossRef]
- Molina, R.; Auge, J.M.; Farrus, B.; Zanon, G.; Pahisa, J.; Munoz, M.; Torne, A.; Filella, X.; Escudero, J.M.; Fernandez, P.; et al. Prospective evaluation of carcinoembryonic antigen (CEA) and carbohydrate antigen 15.3 (CA 15.3) in patients with primary locoregional breast cancer. Clin. Chem. 2010, 56, 1148–1157. [Google Scholar] [CrossRef] [Green Version]
- Lumachi, F.; Basso, S.M.; Bonamini, M.; Marzano, B.; Milan, E.; Waclaw, B.U.; Chiara, G.B. Relationship between preoperative serum markers CA 15-3 and CEA and relapse of the disease in elderly (>65 years) women with breast cancer. Anticancer Res. 2010, 30, 2331–2334. [Google Scholar] [PubMed]
- Duffy, M.J.; Duggan, C.; Keane, R.; Hill, A.D.; McDermott, E.; Crown, J.; O’Higgins, N. High preoperative CA 15-3 concentrations predict adverse outcome in node-negative and node-positive breast cancer: Study of 600 patients with histologically confirmed breast cancer. Clin. Chem. 2004, 50, 559–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumpulainen, E.J.; Keskikuru, R.J.; Johansson, R.T. Serum tumor marker CA 15.3 and stage are the two most powerful predictors of survival in primary breast cancer. Breast Cancer Res. Treat. 2002, 76, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Gion, M.; Boracchi, P.; Dittadi, R.; Biganzoli, E.; Peloso, L.; Mione, R.; Gatti, C.; Paccagnella, A.; Marubini, E. Prognostic role of serum CA15.3 in 362 node-negative breast cancers. An old player for a new game. Eur. J. Cancer 2002, 38, 1181–1188. [Google Scholar] [CrossRef]
- Ebeling, F.G.; Stieber, P.; Untch, M.; Nagel, D.; Konecny, G.E.; Schmitt, U.M.; Fateh-Moghadam, A.; Seidel, D. Serum CEA and CA 15-3 as prognostic factors in primary breast cancer. Br. J. Cancer 2002, 86, 1217–1222. [Google Scholar] [CrossRef] [PubMed]
- Canizares, F.; Sola, J.; Perez, M.; Tovar, I.; De Las Heras, M.; Salinas, J.; Penafiel, R.; Martinez, P. Preoperative values of CA 15-3 and CEA as prognostic factors in breast cancer: A multivariate analysis. Tumour Biol. 2001, 22, 273–281. [Google Scholar]
- Shering, S.G.; Sherry, F.; McDermott, E.W.; O’Higgins, N.J.; Duffy, M.J. Preoperative CA 15-3 concentrations predict outcome of patients with breast carcinoma. Cancer 1998, 83, 2521–2527. [Google Scholar] [CrossRef]
- Molina, R.; Jo, J.; Filella, X.; Zanon, G.; Pahisa, J.; Munoz, M.; Farrus, B.; Latre, M.L.; Escriche, C.; Estape, J.; et al. c-erbB-2 oncoprotein, CEA, and CA 15.3 in patients with breast cancer: Prognostic value. Breast Cancer Res. Treat. 1998, 51, 109–119. [Google Scholar] [CrossRef]
- Di Gioia, D.; Heinemann, V.; Nagel, D.; Untch, M.; Kahlert, S.; Bauerfeind, I.; Koehnke, T.; Stieber, P. Kinetics of CEA and CA15-3 correlate with treatment response in patients undergoing chemotherapy for metastatic breast cancer (MBC). Tumour Biol. 2011, 32, 777–785. [Google Scholar] [CrossRef]
- Laessig, D.; Nagel, D.; Heinemann, V.; Untch, M.; Kahlert, S.; Bauerfeind, I.; Stieber, P. Importance of CEA and CA 15-3 during disease progression in metastatic breast cancer patients. Anticancer Res. 2007, 27, 1963–1968. [Google Scholar]
- de Gregorio, A.; Haberle, L.; Fasching, P.A.; Muller, V.; Schrader, I.; Lorenz, R.; Forstbauer, H.; Friedl, T.W.P.; Bauer, E.; de Gregorio, N.; et al. Gemcitabine as adjuvant chemotherapy in patients with high-risk early breast cancer-results from the randomized phase III SUCCESS-A trial. Breast Cancer Res. 2020, 22, 111. [Google Scholar] [CrossRef] [PubMed]
- von Minckwitz, G.; Arnold, N.; Denkert, C.; Dittmer, J.; Fehm, T.; Harbeck, N.; Janni, W.; Meinhold-Heerlein, I.; Muller, V.; Pantel, K.; et al. Preservation of Biomaterials for translational research in current breast cancer studies: Review and recommendations. Geburtshilfe Frauenheilkd 2007, 67, 451–459. [Google Scholar] [CrossRef]
- Salmen, J.; Neugebauer, J.; Fasching, P.A.; Haeberle, L.; Huober, J.; Wockel, A.; Rauh, C.; Schuetz, F.; Weissenbacher, T.; Kost, B.; et al. Pooled analysis of the prognostic relevance of progesterone receptor status in five German cohort studies. Breast Cancer Res. Treat. 2014, 148, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Hastie, T.; Tibshirani, R. Generalized additive models for medical research. Stat. Methods Med. Res. 1995, 4, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Ripatti, S.; Palmgren, J. Estimation of multivariate frailty models using penalized partial likelihood. Biometrics 2000, 56, 1016–1022. [Google Scholar] [CrossRef]
- Grambsch, P.M.; Therneau, T.M. Proportional Hazards Tests and Diagnostics Based on Weighted Residuals. Biometrika 1994, 81, 515–526. [Google Scholar] [CrossRef]
- Ware, J.H. The limitations of risk factors as prognostic tools. N. Engl. J. Med. 2006, 355, 2615–2617. [Google Scholar] [CrossRef] [Green Version]
- Uno, H.; Cai, T.X.; Tian, L.; Wei, L.J. Evaluating prediction rules for t-year survivors with censored regression models. J. Am. Stat. Assoc. 2007, 102, 527–537. [Google Scholar] [CrossRef]
- McShane, L.M.; Altman, D.G.; Sauerbrei, W.; Taube, S.E.; Gion, M.; Clark, G.M.; Statistics Subcommittee of the NCI-EORTC Working Group on Cancer Diagnostics. REporting recommendations for tumour MARKer prognostic studies (REMARK). Br. J. Cancer 2005, 93, 387–391. [Google Scholar] [CrossRef] [Green Version]
- Stieber, P.; Nagel, D.; Blankenburg, I.; Heinemann, V.; Untch, M.; Bauerfeind, I.; Di Gioia, D. Diagnostic efficacy of CA 15-3 and CEA in the early detection of metastatic breast cancer-A retrospective analysis of kinetics on 743 breast cancer patients. Clin. Chim. Acta 2015, 448, 228–231. [Google Scholar] [CrossRef]
- Pedersen, A.C.; Sorensen, P.D.; Jacobsen, E.H.; Madsen, J.S.; Brandslund, I. Sensitivity of CA 15-3, CEA and serum HER2 in the early detection of recurrence of breast cancer. Clin. Chem. Lab. Med. 2013, 51, 1511–1519. [Google Scholar] [CrossRef] [PubMed]
- Bahrami-Ahmadi, A.; Makarian, F.; Mortazavizadeh, M.R.; Yazdi, M.F.; Chamani, M. Symptomatic metastasis prediction with serial measurements of CA 15.3 in primary breast cancer patients. J. Res. Med. Sci. 2012, 17, 850–854. [Google Scholar] [PubMed]
- Bahrami, A.; Mortazavizadeh, M.R.; Yazdi, M.F.; Chamani, M. Serial tumour markers serum carcinoembryonic antigen and cancer antigen 15-3 assays in detecting symptomatic metastasis in breast cancer patients. East. Mediterr. Health J. 2012, 18, 1055–1059. [Google Scholar] [CrossRef] [PubMed]
- Evangelista, L.; Baretta, Z.; Vinante, L.; Cervino, A.R.; Gregianin, M.; Ghiotto, C.; Bozza, F.; Saladini, G. Could the serial determination of Ca15.3 serum improve the diagnostic accuracy of PET/CT? Results from small population with previous breast cancer. Ann. Nucl. Med. 2011, 25, 469–477. [Google Scholar] [CrossRef]
- Wu, S.C.; Chou, F.F.; Rau, K.M. Clinical significance of a serum CA 15-3 surge and the usefulness of CA 15-3 kinetics in monitoring chemotherapy response in patients with metastatic breast cancer. Breast Cancer Res. Treat. 2010, 124, 879–882. [Google Scholar] [CrossRef]
- Mariani, L.; Miceli, R.; Michilin, S.; Gion, M. Serial determination of CEA and CA 15.3 in breast cancer follow-up: An assessment of their diagnostic accuracy for the detection of tumour recurrences. Biomarkers 2009, 14, 130–136. [Google Scholar] [CrossRef]
- Kim, H.S.; Park, Y.H.; Park, M.J.; Chang, M.H.; Jun, H.J.; Kim, K.H.; Ahn, J.S.; Kang, W.K.; Park, K.; Im, Y.H. Clinical significance of a serum CA15-3 surge and the usefulness of CA15-3 kinetics in monitoring chemotherapy response in patients with metastatic breast cancer. Breast Cancer Res. Treat. 2009, 118, 89–97. [Google Scholar] [CrossRef]
- De La Lande, B.; Hacene, K.; Floiras, J.L.; Alatrakchi, N.; Pichon, M.F. Prognostic value of CA 15.3 kinetics for metastatic breast cancer. Int. J. Biol. Markers 2002, 17, 231–238. [Google Scholar] [CrossRef]
- Jager, W.; Eibner, K.; Loffler, B.; Gleixner, S.; Kramer, S. Serial CEA and CA 15-3 measurements during follow-up of breast cancer patients. Anticancer Res. 2000, 20, 5179–5182. [Google Scholar]
- Gion, M.; Cappelli, G.; Mione, R.; Pistorello, M.; Meo, S.; Vignati, G.; Fortunato, A.; Saracchini, S.; Biasioli, R.; Giulisano, M. Evaluation of critical differences of CEA and CA 15.3 levels in serial samples from patients operated for breast cancer. Int. J. Biol. Markers 1994, 9, 135–139. [Google Scholar] [CrossRef]
- Tang, C.; Wang, P.; Li, X.; Zhao, B.; Yang, H.; Yu, H.; Li, C. Lymph node status have a prognostic impact in breast cancer patients with distant metastasis. PLoS ONE 2017, 12, e0182953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hung, M.; Xu, J.; Nielson, D.; Bounsanga, J.; Gu, Y.; Hansen, A.R.; Voss, M.W. Evaluating the Prediction of Breast Cancer Survival Using Lymph Node Ratio. J. Breast Cancer 2018, 21, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Rack, B.; Schindlbeck, C.; Juckstock, J.; Genss, E.M.; Hepp, P.; Lorenz, R.; Tesch, H.; Schneeweiss, A.; Beckmann, M.W.; Lichtenegger, W.; et al. Prevalence of CA 27.29 in primary breast cancer patients before the start of systemic treatment. Anticancer Res. 2010, 30, 1837–1841. [Google Scholar] [PubMed]
- Pillai, K.; Pourgholami, M.H.; Chua, T.C.; Morris, D.L. MUC1 as a potential target in anticancer therapies. Am. J. Clin. Oncol. 2015, 38, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Acres, B.; Limacher, J.M. MUC1 as a target antigen for cancer immunotherapy. Exp. Rev. Vaccines 2005, 4, 493–502. [Google Scholar] [CrossRef]
- Dhar, P.; McAuley, J. The Role of the Cell Surface Mucin MUC1 as a Barrier to Infection and Regulator of Inflammation. Front. Cell Infect. Microbiol. 2019, 9, 117. [Google Scholar] [CrossRef]
- Konowalchuk, J.D.; Agrawal, B. MUC1 mucin is expressed on human T-regulatory cells: Function in both co-stimulation and co-inhibition. Cell Immunol. 2012, 272, 193–199. [Google Scholar] [CrossRef]
- Konowalchuk, J.D.; Agrawal, B. MUC1 is a novel costimulatory molecule of human T cells and functions in an AP-1-dependent manner. Hum. Immunol. 2012, 73, 448–455. [Google Scholar] [CrossRef]
- Agrawal, B.; Gupta, N.; Konowalchuk, J.D. MUC1 Mucin: A Putative Regulatory (Checkpoint) Molecule of T Cells. Front. Immunol. 2018, 9, 2391. [Google Scholar] [CrossRef] [Green Version]
- Hossain, M.K.; Wall, K.A. Immunological Evaluation of Recent MUC1 Glycopeptide Cancer Vaccines. Vaccines 2016, 4, 25. [Google Scholar] [CrossRef]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).