Sulfur Exafluoride Contrast-Enhanced Ultrasound Showing Early Wash-Out of Marked Degree Identifies Lymphoma Invasion of Spleen with Excellent Diagnostic Accuracy: A Monocentric Study of 260 Splenic Nodules
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
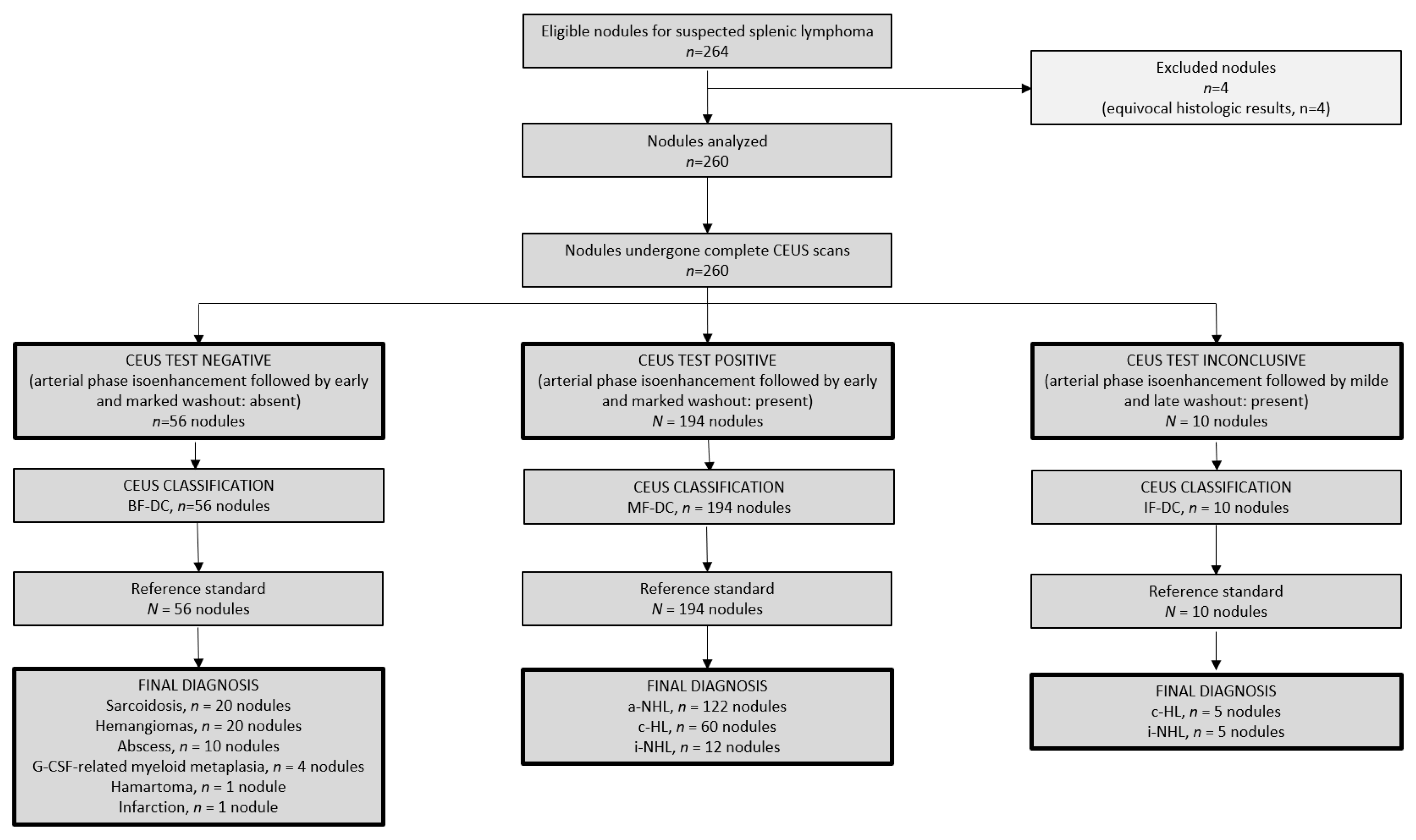
2.1. Study Design
2.2. Participants
2.2.1. Splenic CEUS Examination Methods
2.2.2. Splenic CEUS Patterns
2.2.3. Splenic CEUS Test Positivity
2.2.4. Immunohistochemical Study of the Tumoral Angiogenesis of Splenic Nodules
2.3. Statistical Analysis
3. Results
3.1. Participants
3.1.1. Spleen Status According to the Reference Standard
3.1.2. Spleen Status According to CEUS
3.2. Diagnostic Accuracy of CEUS
3.3. Distribution of CEUS Perfusional Patterns within the Three Clinical Categories of Lymphomas
3.4. Features of Tumoral Angiogenesis of Splenic Nodules
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bhatia, K.; Sahdev, A.; Reznek, R.H. Lymphoma of the Spleen. Semin. Ultrasound CT MRI 2007, 28, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Glatstein, E.; Kaplan, H.S.; Guernsey, J.M.; Rosenberg, S.A. The value of laparotomy and splenectomy in the staging of Hodgkin’s disease. Cancer 1969, 24, 709–718. [Google Scholar] [CrossRef]
- Zinzani, P.L.; Marchetti, M.; Billio, A.; Barosi, G.; Carella, A.M.; Lazzarino, M.; Martelli, M.; Rambaldi, A.; Rigacci, L.; Tarella, C.; et al. SIE, SIES, GITMO revised guidelines for the management of follicular lymphoma. Am. J. Hematol. 2013, 88, 185–192. [Google Scholar] [CrossRef] [Green Version]
- Tarella, C.; Arcaini, L.; Baldini, L.; Barosi, G.; Billio, A.; Marchetti, M.; Rambaldi, A.; Vitolo, U.; Zinzani, P.L.; Yura, S. Italian Society of Hematology, Italian Society of Experimental Hematology, and Italian Group for Bone Marrow Transplantation Guidelines for the Management of Indolent, Nonfollicular B-Cell Lymphoma (Marginal Zone, Lymphoplasmacytic, and Small Lymphocytic Lymphoma). Clin. Lymphoma Myeloma Leuk. 2015, 15, 75–85. [Google Scholar] [CrossRef]
- Picardi, M.; Soricelli, A.; Grimaldi, F.; Nicolai, E.; Gallamini, A.; Pane, F. Fused FDG-PET/contrast-enhanced CT detects occult subdiaphragmatic involvement of Hodgkin’s lymphoma thereby identifying patients requiring six cycles of anthracycline-containing chemotherapy and consolidation radiation of spleen. Ann. Oncol. 2011, 22, 671–680. [Google Scholar] [CrossRef] [PubMed]
- Picardi, M.; Soricelli, A.; Pane, F.; Zeppa, P.; Nicolai, E.; De Laurentis, M.; Grimaldi, F.; Rotoli, B. Contrast-enhanced harmonic compound US of the spleen to increase staging accuracy in patients with Hodgkin lymphoma: A prospective study. Radiology 2009, 251, 574–582. [Google Scholar] [CrossRef] [PubMed]
- Littooij, A.S.; Kwee, T.C.; Barber, I.; Granata, C.; de Keizer, B.; Beek, F.J.A.; Hobbelink, M.G.; Fijnheer, R.; Stoker, J.; Nievelstein, R.A.J. Accuracy of whole-body MRI in the assessment of splenic involvement in lymphoma. Acta Radiol. 2016, 57, 142–151. [Google Scholar] [CrossRef]
- Liver Reporting & Data System. American College of Radiology. Available online: https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS (accessed on 30 October 2021).
- Terzi, E.; Iavarone, M.; Pompili, M.; Veronese, L.; Cabibbo, G.; Fraquelli, M.; Riccardi, L.; De Bonis, L.; Sangiovanni, A.; Leoni, S.; et al. Contrast ultrasound LI-RADS LR-5 identifies hepatocellular carcinoma in cirrhosis ina multicenter restropective study of 1006 nodules. J. Hepatol. 2018, 68, 485–492. [Google Scholar] [CrossRef]
- Tedesco, G.; Sarno, A.; Rizzo, G.; Grecchi, A.; Testa, I.; Giannotti, G.; D’Onofrio, M. Clinical use of contrast-enhanced ultrasound beyond the liver: A focus on renal, splenic, and pancreatic applications. Ultrasonography 2019, 38, 278–288. [Google Scholar] [CrossRef]
- Johnson, S.A.; Kumar, A.; Matasar, M.J.; Schöder, H.; Rademaker, J. Imaging for staging and response assessment in lymphoma. Radiology 2015, 276, 323–338. [Google Scholar] [CrossRef] [Green Version]
- Iannitto, E.; Tripodo, C. How I diagnose and treat splenic lymphomas. Blood 2011, 117, 2585–2595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Picardi, M.; Pugliese, N.; Cirillo, M.; Zeppa, P.; Cozzolino, I.; Ciancia, G.; Pettinato, G.; Salvatore, C.; Quintarelli, C.; Pane, F. Advanced-stage Hodgkin lymphoma: US/chest radiography for detection of relapse in patients in first complete remission—A randomized trial of routine surveillance imaging procedures. Radiology 2014, 272, 262–274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Picardi, M.; Gennarelli, N.; Ciancia, R.; De Renzo, A.; Gargiulo, G.; Ciancia, G.; Sparano, L.; Zeppa, P.; Martinelli, V.; Pettinato, G.; et al. Randomized comparison of power Doppler ultrasound-directed excisional biopsy with standard excisional biopsy for the characterization of lymphadenopathies in patients with suspected lymphoma. J. Clin. Oncol. 2004, 22, 3733–3740. [Google Scholar] [CrossRef] [PubMed]
- Pugliese, N.; Di Perna, M.; Cozzolino, I.; Ciancia, G.; Pettinato, G.; Zeppa, P.; Varone, V.; Masone, S.; Cerchione, C.; Della Pepa, R.; et al. Randomized comparison of power Doppler ultrasonography-guided core-needle biopsy with open surgical biopsy for the characterization of lymphadenopathies in patients with suspected lymphoma. Ann. Hematol. 2017, 96, 627–637. [Google Scholar] [CrossRef] [Green Version]
- Picardi, M.; Giordano, C.; Della Pepa, R.; Pugliese, N.; Leone, A.; Mascolo, M.; Daniela, R.; Vigliar, E.; Troncone, G.; Salvatore, C.; et al. Correspondence in reference to the previously published Epub manuscript by Peter Hokland et al. ‘How I treat advanced Hodgkin lymphoma—A global view’. Br. J. Haematol. 2020, 190, 837–850. [Google Scholar] [CrossRef]
- Vardiman, J.W.; Swerdlow, S.H.; Campo, E.; Harris, N.L.; Jaffe, E.S.; Pileri, S.A.; Stein, H.; Theiele, J. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues; IARC Publications: Lyon, France, 2008. [Google Scholar] [CrossRef] [Green Version]
- Guidelines Detail. Available online: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1439 (accessed on 30 October 2021).
- Paes, F.M.; Kalkanis, D.G.; Sideras, P.A.; Serafini, A.N. FDG PET/CT of Extranodal Involvement in Non-Hodgkin Lymphoma and Hodgkin Disease. RadioGraphics 2010, 30, 269–291. [Google Scholar] [CrossRef]
- Sangiorgio, V.F.I.; Rizvi, H.; Padayatty, J.; Thayur, N.; Fujiwara, T.; Anyanwu, F.A.; Calaminici, M. Radiologically guided percutaneous core needle biopsy of the spleen: A reliable and safe diagnostic procedure for neoplastic and reactive conditions. Histopathology 2021, 78, 1051–1055. [Google Scholar] [CrossRef]
- Sidhu, P.S.; Cantisani, V.; Dietrich, C.F.; Gilja, O.H.; Saftoiu, A.; Bartels, E.; Bertolotto, M.; Calliada, F.; Clevert, D.-A.; Cosgrove, D.; et al. The EFSUMB guidelines and recommendations for the clinical practice of contrast-enhanced ultrasound (CEUS) in Non-Hepatic Applications: Update 2017 (Long Version). Ultraschall. Med. 2018, 39, e2–e44. [Google Scholar] [CrossRef] [Green Version]
- Tzankov, A.; Heiss, S.; Ebner, S.; Sterlacci, W.; Schaefer, G.; Augustin, F.; Fiegl, M.; Dirnhofer, S. Angiogenesis in nodal cell lymphomas: A high throughput study. J. Clin. Pathol. 2007, 60, 476–482. [Google Scholar] [CrossRef] [Green Version]
- Alberg, A.J.; Park, J.W.; Hager, B.W.; Brock, M.V.; Diener-West, M. The use of “overall accuracy” to evaluate the validity of screening or diagnostic tests. J. Gen. Intern. Med. 2004, 19, 460–465. [Google Scholar] [CrossRef] [Green Version]
- Menzel, L.; Höpken, U.E.; Rehm, A. Angiogenesis in Lymph Nodes Is a Critical Regulator of Immune Response and Lymphoma Growth. Front. Immunol. 2020, 11, 591741. [Google Scholar] [CrossRef] [PubMed]




| Variable | Patients (n = 77) | Nodules (n = 260) |
|---|---|---|
| Patients’ gender: male | 43 (57%) | |
| Patients’ age: median, years (range) | 48 (22–72) | |
| Lymph node biopsy-proven lymphoma | ||
| Non-Hodgkin lymphoma | 44 (57%) | |
| Hodgkin lymphoma | 33 (43%) | |
| Timing of the discovery of nodules | ||
| Staging for lymphoma | 56 (72%) | |
| Follow-up for lymphoma | 21 (28%) | |
| Anti-lymphomatous therapy administered * | ||
| R-CHOP-21 | 37 (48%) | |
| CHOP-21 | 4 (5%) | |
| R-Bendamustin | 3 (4%) | |
| ABVD | 33 (43%) | |
| Nodule size: median, cm (range) | 1.5 (0.5–7) | |
| Aetiology of nodules | ||
| Malignancy ** | 204 (78%) | |
| a-NHL | 122 (60%) | |
| DLBCL | 108 (53%) | |
| TRBCL | 10 (5%) | |
| ATCL | 4 (2%) | |
| c-HL | 65 (32%) | |
| NS-HL | 50 (25%) | |
| MC-HL | 11 (5%) | |
| LR-HL | 4 (2%) | |
| i-NHL | 17 (8%) | |
| FL | 17 (8%) | |
| Benignity *** | 56 (22%) | |
| Sarcoidosis | 20 (36%) | |
| Hemangiomas | 20 (36%) | |
| Abscess | 10 (17%) | |
| Myeloid metaplasia | 4 (7%) | |
| Hamartoma | 1 (1%) | |
| Splenic infarction | 1 (1%) | |
| Aetiology | Median Size, cm (Range) | CEUS Patterns | Reference Standard | |
|---|---|---|---|---|
| Arterial Phase | Portal Phase | |||
| Sarcoidosis, n = 20 | 1.5 (0.5–2.2) | Globally, hypoechoic compared to the surrounding parenchyma | Globally, hypoechoic (without later washout) compared to the surrounding parenchyma | Lymph nodes biopsy plus imaging follow-up after treatment, n = 20 |
| Hemangiomas, n = 20 | 1.5 (1.1–6) | Isoenhancement (n = 10), capillary hemangiomas Discontinuous globular peripheral enhancement (n = 10), cavernous hemangiomas | With or without later washout of mild degree as intensity (n = 10), capillary hemangiomas Progressive centripetal fill-in or very late (>3 min) washout of mild degree (n = 10), cavernous hemangiomas | Clinical and imaging follow-up, n = 20 |
| Abscesses, n = 10 | 1.1 (0.7–1.5) | Thin rim hyperenhancement pattern | Washout | Blood culture, n = 10 (positive test for fungus, n = 8; positive test for bacteria, n = 2) plus imaging monitoring after anti-microbial therapy |
| G-CSF-related myeloid metaplasia, n = 4 | 2.5 (2–3.2) | Relative hypoenhancement of the periphery of nodule and normal enhancement in the remaining portion of lesion compared to the surrounding parenchyma (reversed rim-enhancement) | Isoenhancement | Clinical and imaging follow-up, n = 4 |
| Hamartoma, n = 1 | 7 | Hyperenhancement | Hyperenhancement | Clinical and imaging follow-up, n = 1 |
| Infarction, n = 1 | 6 | Hypo-enhancement (triangular-shaped) | Hypo-enhancement (triangular-shaped) | Clinical and imaging follow-up, n = 1 |
| Accuracy Measurement | Results |
|---|---|
| Reference standard * | 100% (260/260 nodules) |
| Sensitivity | 95% (95% CI, 91–98) |
| Specificity | 100% (95% CI, 94–100) |
| Positive predictive value | 100% (95% CI, 98–100) |
| Negative predictive value | 85% (95% CI, 74–92) |
| False-negative finding | 5% (10/260 nodules) |
| False-positive finding | – |
| Overall diagnostic accuracy | 96% (95% CI, 93–98) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Picardi, M.; Giordano, C.; Trastulli, F.; Leone, A.; Della Pepa, R.; Pugliese, N.; Iula, R.; Delle Cave, G.; Rascato, M.G.; Esposito, M.; et al. Sulfur Exafluoride Contrast-Enhanced Ultrasound Showing Early Wash-Out of Marked Degree Identifies Lymphoma Invasion of Spleen with Excellent Diagnostic Accuracy: A Monocentric Study of 260 Splenic Nodules. Cancers 2022, 14, 1927. https://doi.org/10.3390/cancers14081927
Picardi M, Giordano C, Trastulli F, Leone A, Della Pepa R, Pugliese N, Iula R, Delle Cave G, Rascato MG, Esposito M, et al. Sulfur Exafluoride Contrast-Enhanced Ultrasound Showing Early Wash-Out of Marked Degree Identifies Lymphoma Invasion of Spleen with Excellent Diagnostic Accuracy: A Monocentric Study of 260 Splenic Nodules. Cancers. 2022; 14(8):1927. https://doi.org/10.3390/cancers14081927
Chicago/Turabian StylePicardi, Marco, Claudia Giordano, Fabio Trastulli, Aldo Leone, Roberta Della Pepa, Novella Pugliese, Rossella Iula, Giuseppe Delle Cave, Maria Gabriella Rascato, Maria Esposito, and et al. 2022. "Sulfur Exafluoride Contrast-Enhanced Ultrasound Showing Early Wash-Out of Marked Degree Identifies Lymphoma Invasion of Spleen with Excellent Diagnostic Accuracy: A Monocentric Study of 260 Splenic Nodules" Cancers 14, no. 8: 1927. https://doi.org/10.3390/cancers14081927
APA StylePicardi, M., Giordano, C., Trastulli, F., Leone, A., Della Pepa, R., Pugliese, N., Iula, R., Delle Cave, G., Rascato, M. G., Esposito, M., Vigliar, E., Troncone, G., Mascolo, M., Russo, D., Persico, M., & Pane, F. (2022). Sulfur Exafluoride Contrast-Enhanced Ultrasound Showing Early Wash-Out of Marked Degree Identifies Lymphoma Invasion of Spleen with Excellent Diagnostic Accuracy: A Monocentric Study of 260 Splenic Nodules. Cancers, 14(8), 1927. https://doi.org/10.3390/cancers14081927









