1. Introduction
Gastric cancer is the fifth most common cancer and the fourth leading cause of cancer deaths worldwide [
1]. Approximately 17% of patients with gastric cancer have peritoneal carcinomatosis (PC) at the time of diagnosis, and this proportion is 40% among patients with advanced gastric cancer [
2]. Even after curative surgery, PC recurrence affects nearly 43% of patients with gastric cancer regardless of cancer stage [
3]. PC is considered a terminal illness, and its prognosis remains poor despite new chemotherapeutic drugs and agents with new molecular targets. Currently, systemic therapy or best supportive care is the standard recommended treatment for this condition. Since 2013, studies have reported a median survival of only 8–13.2 months [
4]. In the late 1990s, the Peritoneal Surface Oncology Group International (PSOGI) introduced a novel treatment, cytoreductive surgery (CRS) combined with intraoperative hyperthermic intraperitoneal (IP) chemotherapy (HIPEC), for peritoneal carcinomatosis originating from various cancer types [
5]. The aim of cytoreductive surgery is to eradicate macroscopic tumors, and the purpose of HIPEC is to eliminate tiny or microscopic seeding tumors. CRS-HIPEC has exhibited survival benefits in the treatment of PC from malignant mesothelioma and gynecological and gastrointestinal cancers [
6,
7,
8]. Yonemura et al. reported a median survival of 15.5 months in patients with gastric cancer with peritoneal carcinomatosis in which complete cytoreduction (CC) was achieved and 7.9 months in those in which cytoreduction was incomplete [
9]. In a French multi-institutional study, Glehen et al. reported 5-year survival rates of 23% for such patients after CC and 13% for those after incomplete cytoreduction [
10].
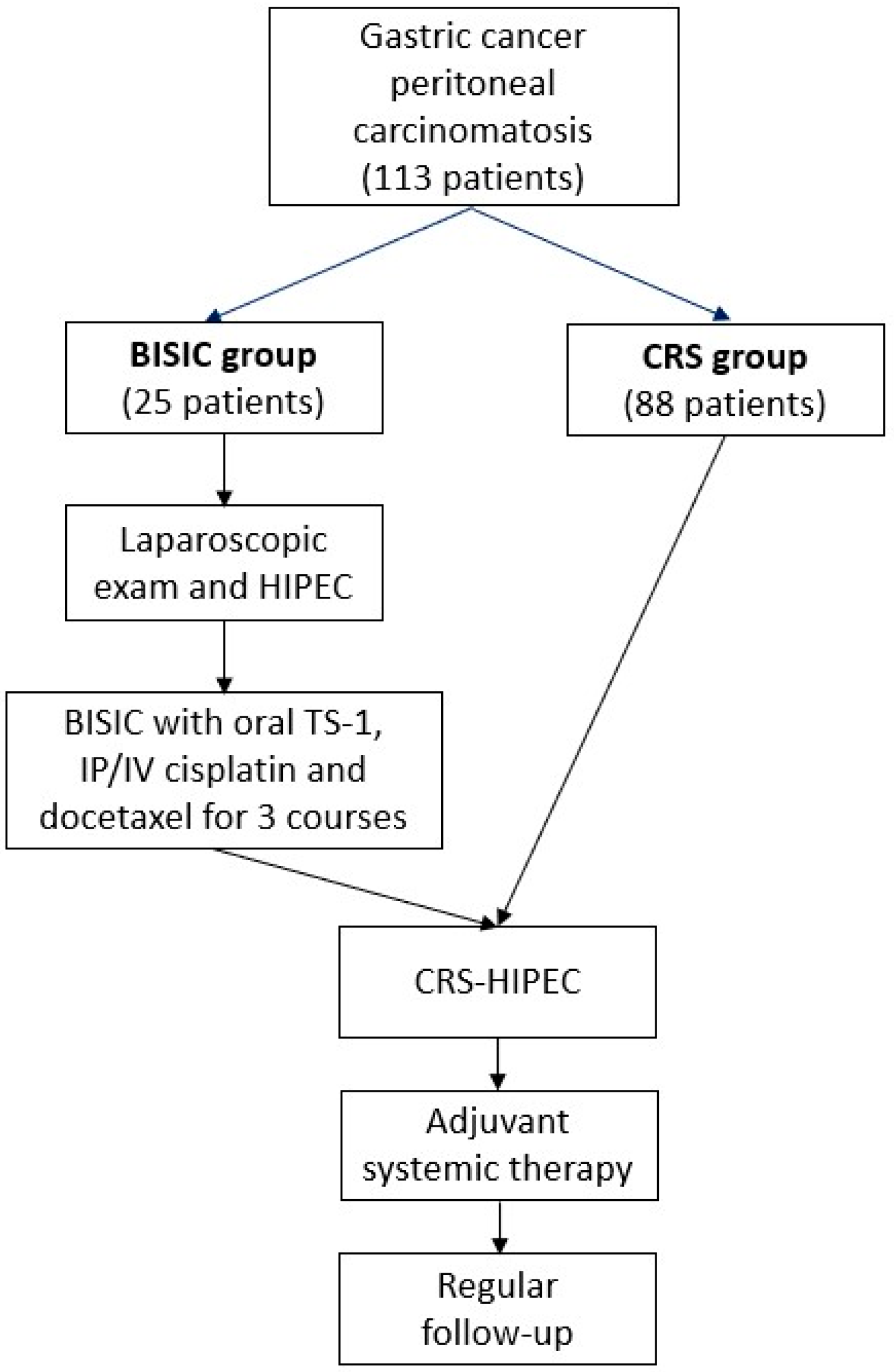
Although the clinical results of CRS-HIPEC for PC from gastric cancer have been promising, the prognosis of patients with gastric cancer with peritoneal carcinomatosis with a large tumor burden remains poor, and achievement of complete cytoreduction in these patients is difficult. In the mid-2010s, PSOGI developed a comprehensive gastric cancer with peritoneal carcinomatosis treatment strategy including CRS with chemotherapy for reducing large tumor burdens before surgery [
11]. The procedures include a first laparoscopic exam for a tissue biopsy, peritoneal cancer index (PCI) evaluation, and intraoperative HIPEC followed by neoadjuvant bidirectional chemotherapy and interval CRS-HIPEC with early postoperative IP chemotherapy and late systemic chemotherapy [
12]. Yonemura et al. reported a 5-year survival rate of 32.4% for patients who responded to neoadjuvant chemotherapy without malignant ascites and with a PCI lower than a cutoff of 7, and who had received complete CRS [
12].
However, few studies have reported the long-term results of this structured comprehensive strategy. The indications for this comprehensive treatment and the benefits of neoadjuvant laparoscopic HIPEC (L-HIPEC) and bidirectional chemotherapy before CRS have also not been clarified. Thus, this study assessed real-world survival data to investigate the effectiveness of this strategy. In addition, this study explored the indications for this treatment and whether an adequate cutoff level exists indicating that direct CRS should be avoided.
4. Discussion
Systemic chemotherapy is the standard treatment for stage IV gastric cancer. However, the prognosis of patients with peritoneal metastasis is worse than that of those with other distant metastases [
18]. CRS-HIPEC improves the outcomes of PC with several specific etiologies, including appendix and ovarian cancer and mesothelioma. A randomized phase III study by Yang et al. [
19]. demonstrated that CRS-HIPEC was associated with longer disease-free survival than CRS alone (11.0 vs. 6.5 months,
p < 0.05) in patients with gastric cancer with peritoneal carcinomatosis. A randomized phase III study, the GYMSSA trial, demonstrated that CRS-HIPEC was associated with longer overall survival than systemic therapy alone (11.3 vs. 4.3 months), but statistical significance was not reached because of limited patient numbers [
20]. A case–control propensity-score matching study, the Cytoreductive Surgery With or Without Hyperthermic Intraperitoneal Chemotherapy for Gastric Cancer With Peritoneal Metastases study, investigated 277 patients from 19 French centers and demonstrated that CRS-HIPEC was associated with longer overall survival that CRS alone (18.8 vs. 12.1 months,
p < 0.01) [
8]. The study demonstrated not only the effectiveness of HIPEC after CRS but also the crucial role in survival improvement of CC to a score of 0 or 1. Recently, the Italian Peritoneal Surface Malignancies Oncoteam (S.I.C.O), with 11 high-volume, specialized centers, has also demonstrated that the survival time of gastric cancer after CRS-HIPES was 20.2 months. In this Italian multi-center study, they revealed that the significantly better prognostic factors were PCI score ≤ 6 (44.3 vs. 13.4 months,
p = 0.005), CC score 0 (40.7 vs. 10.7 months,
p = 0.003), and receiving neoadjuvant chemotherapy (35.3 vs. 10.7 months,
p = 0.022) [
21]. In guidelines for the treatment of many advanced cancers, including ovarian, breast, pancreatic, rectal, and gastric cancer, neoadjuvant chemotherapy with or without radiotherapy is a standard strategy. Expectational downstaging or the conversion of inoperable conditions to operable conditions improves tumor clearance and long-term outcomes. Because of persistently poor results and only limited prognosis improvement in patients with gastric cancer with peritoneal carcinomatosis treated with CRS-HIPEC, in 2006, Yonemura introduced neoadjuvant IP and systemic chemotherapy (NIPS) using IP cisplatin and docetaxel on days 1 and 8 with systemic oral S-1 on days 1–14 every 3 weeks [
22]. The concept of IP chemotherapy with directly contacting peritoneal seeding tumors is based on consideration of the blood–peritoneum barrier, which is the stromal tissue between the mesothelium and submesothelial capillaries and impedes the transport of drugs from the systemic circulation to the IP cavity [
23]. In 2014, Yonemura modified this treatment to include BISIC using IP and IV cisplatin plus docetaxel on days 1 and 8 with systemic oral S-1 on days 1–14 every 3 weeks for 3 courses [
13]. Compared with NIPS, BISIC was associated with a higher histological response rate (60% vs. 83%) and conversion rate of malignant to nonmalignant ascites (70% vs. 79%) [
12]. In 2016, PSOGI developed a novel comprehensive treatment for gastric cancer with peritoneal carcinomatosis with curative intent involving laparoscopic examination with neoadjuvant HIPEC, neoadjuvant BISIC, CRS-HIPEC, and postoperative adjuvant chemotherapy [
11].
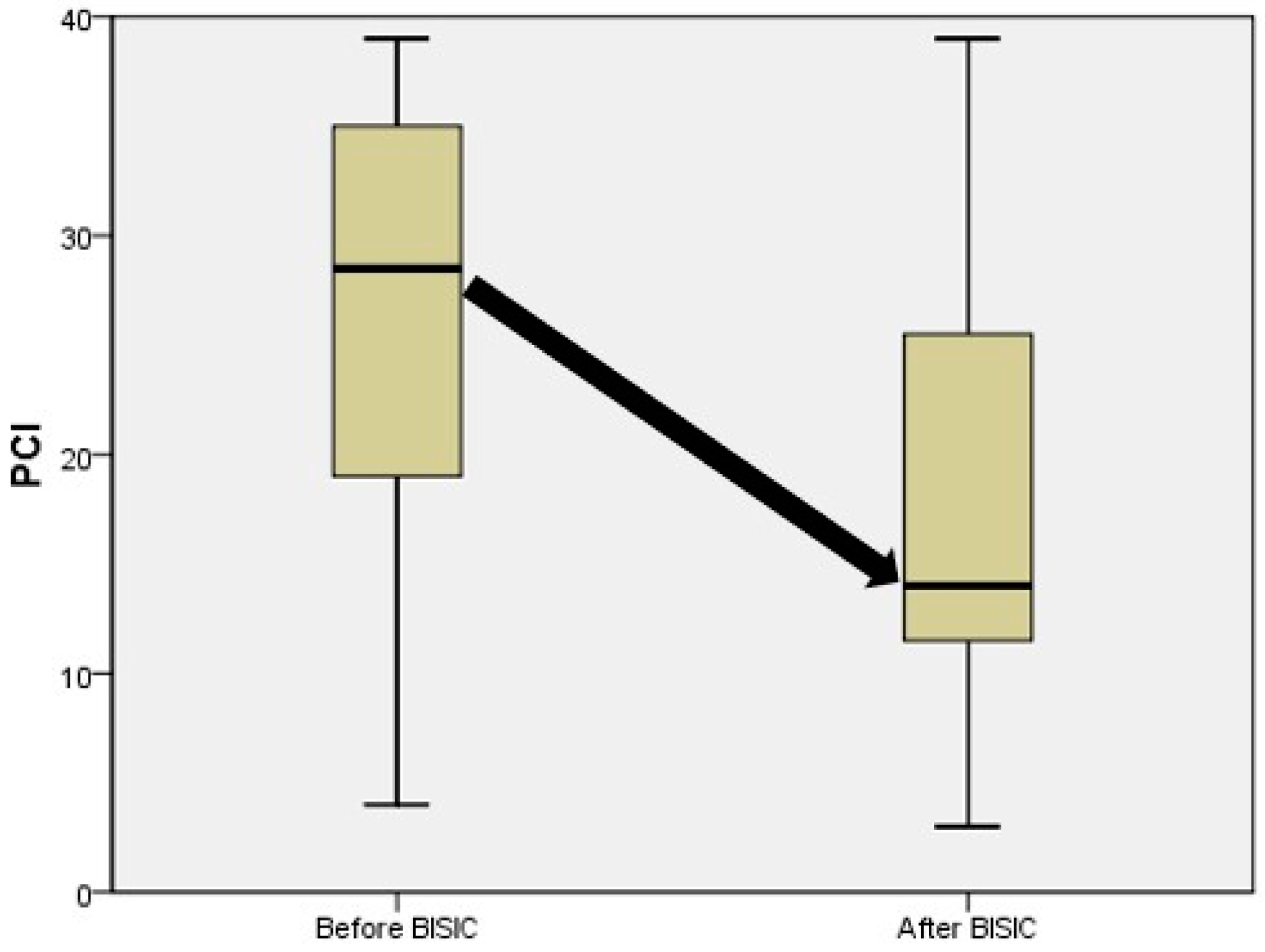
Real-world data on the use of this strategy are limited, and the benefits of neoadjuvant L-HIPEC and BISIC before CRS are unclear. In the present study, although the initial mean PCI was greater in the BISIC group than in the CRS group (27 vs. 21,
p = 0.045), the mean PCI was lower after BISIC treatment 16 vs. 21,
p = 0.017;
Table 2). In the BISIC group, the mean PCI significantly improved from 27 to 21 (
p = 0.003), and the rate of conversion from positive to negative cytology was 33% after the neoadjuvant treatment (
Table 6). The clearance rate measured in terms of postoperative PCI was also higher in the BISIC group than in the CRS group (6 vs. 14,
p = 0.002). The post-CRS PCI was also lower for the most difficult area for tumor clearance (the small-bowel mesentery) in the BISIC group than in the CRS group (3 vs. 5,
p = 0.025). This demonstrates the effectiveness of neoadjuvant L-HIPEC and BISIC treatment. The difference in overall survival indicated by the Cox multivariate proportional hazards regression suggested the benefit of BISIC (hazard ratio, 0.541; 95% confidence interval, 0.293–0.998,
p = 0.049). The safety of this treatment also warrants consideration. In a previous study, IP docetaxel achieved a high concentration over 12 h at a low plasma concentration, which resulted in less severe side effects than conventional systemic chemotherapies [
24,
25]. Yonemura et al. also demonstrated an acceptable side effect rate of 2.6% for grade 3 or 4 hematological events and 3.8% for renal toxicity [
26]. In our study, the groups’ pre-CRS conditions were identical except for the lower platelet count in the BISIC group (196.0 vs. 279.4/103/µL,
p = 0.001;
Table 1). Longer operation time, greater blood loss, and the transfusion of more units were observed in the BISIC group. However, these differences did not influence the morbidity rate after CRS (Clavien–Dindo class: 0–2, 68% vs. 67%; class 3 or 4, 28% vs. 23.9%,
p = 0.680;
Table 2). In addition, surgeons may be concerned about the formation of intra-abdominal adhesions, which may prevent future CRS, but many antiadhesive powders, fluids, and films can be used to reduce adhesions.
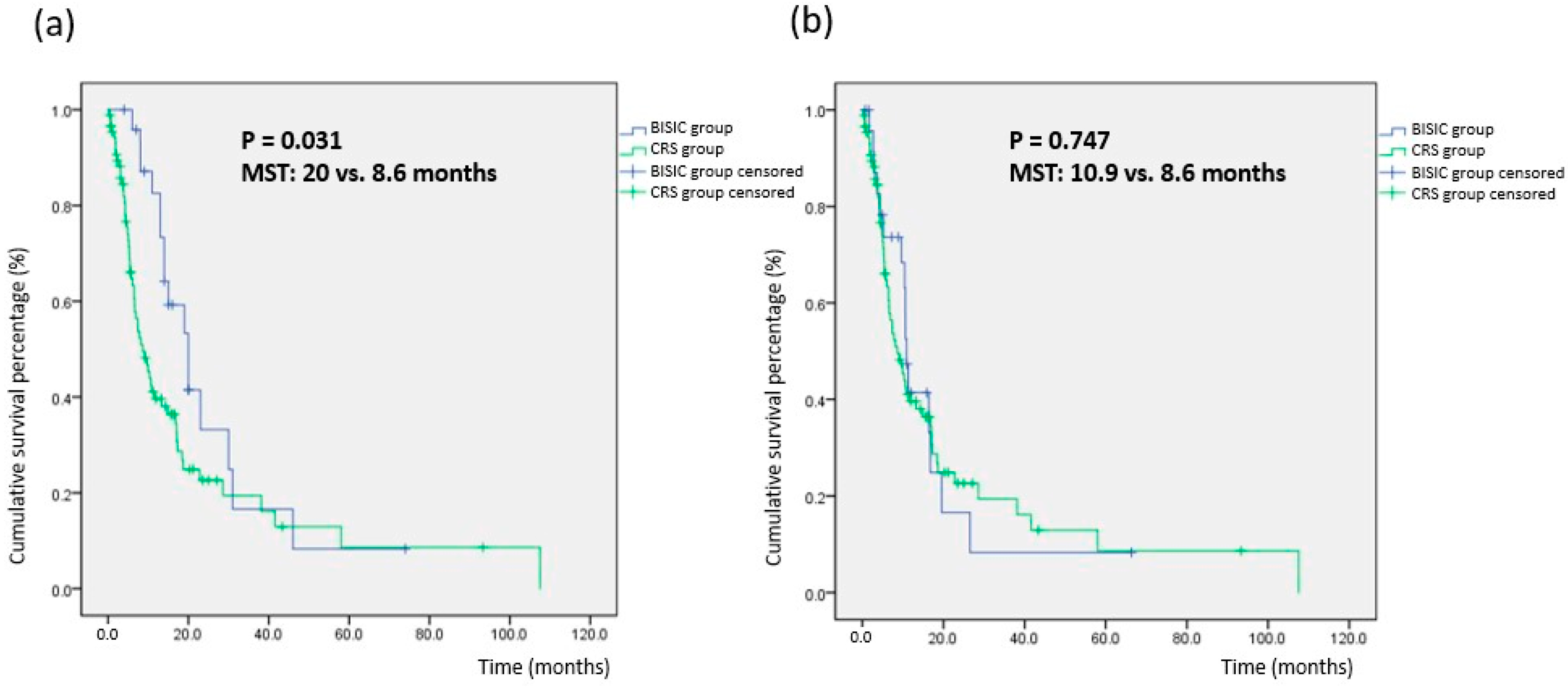
In our multivariable survival analysis, the key factors influencing survival were age, the use of BISIC, and the clearance rate of small-bowel mesentery seeding tumors (as indicated post-CRS PCI in regions 9 to 12; hazard ratio: 1.218, 95% confidence interval: 1.078–1.376,
p = 0.002;
Table 4). MST differed significantly between the BISIC group (20.0 months) and CRS group (8.6 months;
p = 0.031;
Table 3). However, MST after CRS did not differ significantly (BISIC vs. CRS: 10.9 vs. 8.6 months,
p = 0.747). This may indicate that L-HIPEC and BISIC can prolong survival. Furthermore, the 5-year survival rates of the groups were similar, at approximately 9%. Few studies have examined the factors affecting the long-term survival of patients with gastric cancer with peritoneal carcinomatosis treated with CRS-HIPEC. Brandl et al. and PSOGI conducted a multi-institutional cohort study using questionnaires and revealed that the factors affecting long-term survival were CC to a score of 0 and a PCI of <6 [
27]. Yonemura also demonstrated that the predictors of long-term survival were a PCI of <14 and ascites with nonmalignant ascites [
28].
The optimal criteria for selecting the L-HIPEC and BISIC protocol remain unclear. In the present study, the study population was stratified by initial PCI into mild (1–10), moderate (11–20), and severe (21–39) PC groups. The MSTs of the BISIC and CRS groups were 20.0 and 10.8 months (p = 0.061) for mild PC, 30.0 and 13.3 months (p = 0.029) for moderate PC, and 19.6 and 6.6 months (p = 0.015) for severe PC. This result suggests that direct CRS-HIPEC should be avoided for patients with gastric cancer with peritoneal carcinomatosis and a PCI of >10. To increase survival in gastric cancer with peritoneal carcinomatosis, we suggest L-HIPEC followed by 3 BISIC courses before CRS in patients with an initial PCI of >10.
The limitations of this study include the low patient number, use of a single specialized center, and nonrandomized retrospective cohort analysis. However, we standardized the treatment protocol, including the BISIC and HIPEC regimens, temperature, and the duration of HIPEC, and controlled for the quality of surgeon performance in CRS, which is not possible in multicenter studies. Furthermore, propensity-score matching was not performed between the BISIC and CRS groups, and the BISIC group had a higher mean PCI prior to treatment. However, although the patients in the BISIC group exhibited more severe PC, they nonetheless exhibited longer survival after enrollment. Finally, the indications for CRS, notably the cutoff PCI after BISIC, remain controversial. Further multi-institutional randomized-controlled studies with more patients are necessary to clarify these.