Tumor Necrosis Factor Alpha: Implications of Anesthesia on Cancers
Abstract
:Simple Summary
Abstract
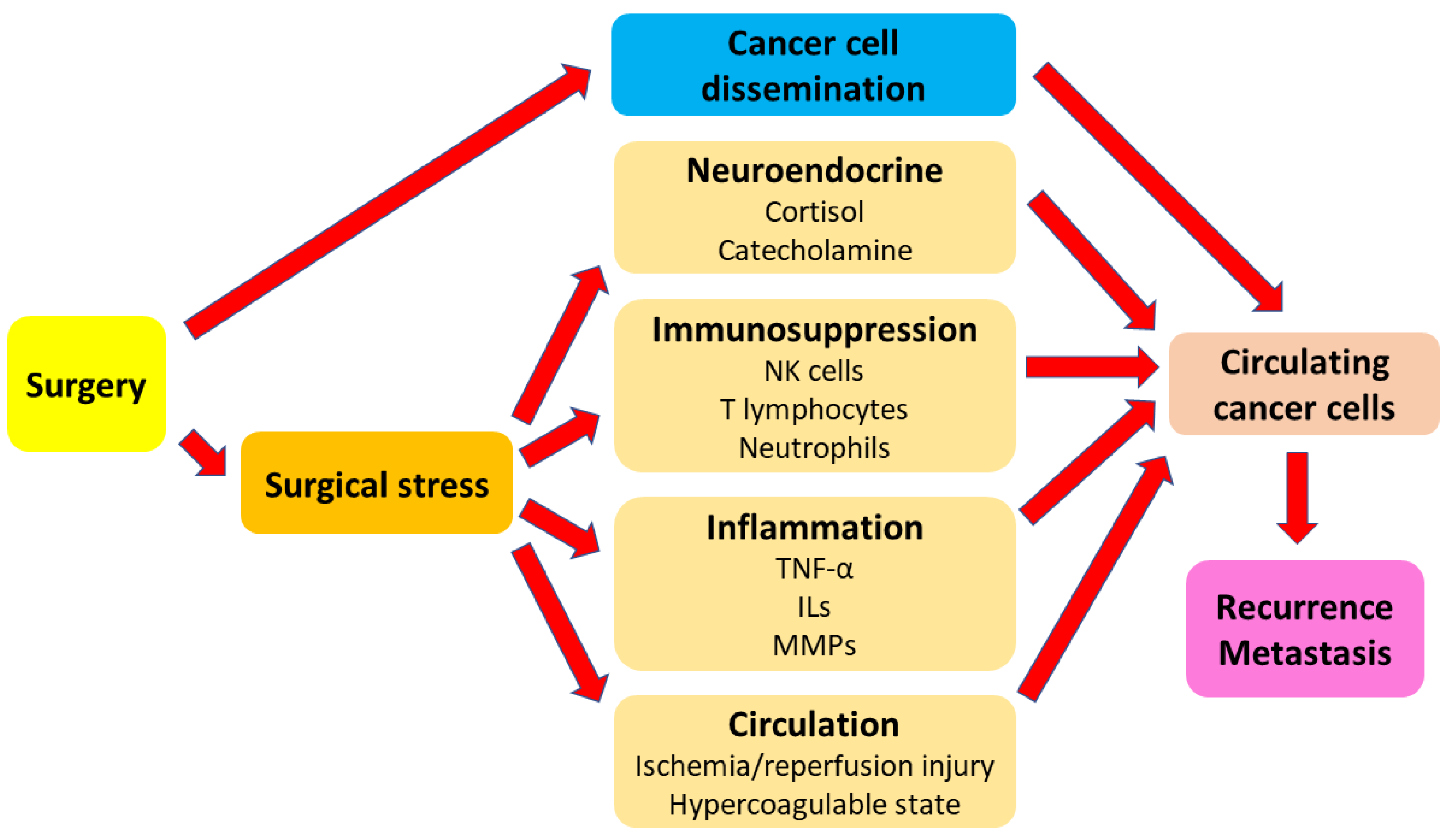
1. Introduction
2. General Anesthesia
2.1. Volatile Anesthetics
2.1.1. Laboratory Research
2.1.2. Clinical Studies
2.2. Propofol
2.2.1. Laboratory Research
2.2.2. Clinical Studies
2.3. Ketamine
2.3.1. Laboratory Research
2.3.2. Clinical Studies
2.4. Dexmedetomidine
2.4.1. Laboratory Research
2.4.2. Clinical Studies
2.5. Systemic Lidocaine
2.5.1. Laboratory Research
2.5.2. Clinical Studies
2.6. Midazolam
2.6.1. Laboratory Research
2.6.2. Clinical Studies
2.7. Thiopental
2.7.1. Laboratory Research
2.7.2. Clinical Studies
2.8. Opioids
2.8.1. Laboratory Research
2.8.2. Clinical Studies
2.9. Nonsteroidal Anti-Inflammatory Drugs
2.9.1. Laboratory Research
2.9.2. Clinical Studies
3. Regional Anesthesia/Analgesia
3.1. Regional Anesthesia/Analgesia
3.1.1. Laboratory Research
3.1.2. Clinical Studies
4. Perioperative Care
4.1. Body Temperature
4.1.1. Laboratory Research
4.1.2. Clinical Studies
4.2. Hyperglycemia
4.2.1. Laboratory Research
| Anesthetics/Analgesics and Perioperative Management | TNF-α Release | Effects |
|---|---|---|
| VAs | Not applicable | There is no relevant experimental study. |
| Propofol | Decreased | Propofol ameliorated oxygen-glucose deprivation/reperfusion-induced inflammation in mouse neuroblastoma cell lines [67]. |
| Increased | Propofol suppressed the proliferation and killing activity of anti-tumor immune cells during tumor resection in murine models of breast cancer [68]. | |
| Ketamine | Decreased | Ketamine attenuated LPS-induced inflammatory responses in mouse neuroblastoma cell lines [79]. |
| DEX | Decreased | DEX improved the immune function and decreased the tumor invasion and migration in rat models of ovarian cancer [92]; DEX enhanced the immune function and suppressed the tumor growth in rat models of ovarian cancer [93]; DEX suppressed surgical stress responses, sped up the recovery of NK cell activity, and lowered the tumor burden after surgery in mouse models of ovarian cancer [94]; DEX attenuated oxygen-glucose deprivation/reperfusion-induced inflammation and apoptosis in rat pheochromocytoma cell lines [95]. |
| Lidocaine | No difference | Lidocaine contributed to tumor reduction but had no influence on tumor-induced inflammatory responses [117]. |
| Midazolam | Decreased | Midazolam reduced the progression of hepatocellular carcinoma cell lines and influenced the immune microenvironment in mouse models [124]. |
| Increased | Midazolam inhibited the growth and induced the differentiation of murine myeloid leukemia cell lines [126]. | |
| Thiopental | Decreased | Thiopental exerted anti-inflammatory effects on LPS-treated human glioma cell lines and murine brain inflammation models [129]. |
| Opioids | Decreased | Morphine dose-dependently suppressed tumor cell growth in human cancer cell lines [144]; morphine and its derivatives induced the apoptosis of human cancer cell lines [145]; tramadol and morphine inhibited cytokine release in LPS-treated human histiocytic lymphoma cell lines [147]. |
| Increased | Naltrexone decreased proliferation and promoted apoptosis in mouse models of colorectal cancer [149]. | |
| No difference | Fentanyl had no effect on the production of cytokines in LPS-treated human histiocytic lymphoma cell lines [147]. | |
| NSAIDs | Decreased | Celecoxib downregulated inflammation and prevented against colitis-mediated colon carcinogenesis in mouse models [170]. |
| Increased | Sulindac and celecoxib exerted anti-inflammatory and anti-neoplastic actions in rat models of colorectal cancer [172]. | |
| No difference | NSAIDs had no influence on cytokine release in dog models of osteosarcoma [173]. | |
| RA | Decreased | The combination of GA and SA reduced stress responses to surgery and attenuated the suppression of innate tumor immunity compared with GA alone in mouse models of prostate cancer [185]. |
| Body temperature | Increased | Hypothermia stimulated lung cancer boost [201]; mild hyperthermia enhanced the efficacy of immunotherapy and reduced the risk of cancer metastasis in mouse models of pancreatic cancer [202]. |
| Hyperglycemia | Increased | The interaction between hyperglycemia and macrophages promoted malignancy-associated alterations in pancreatic ductal epithelial cells [210]. |
| Blood transfusion | Not applicable | There is no relevant experimental study. |
4.2.2. Clinical Studies
4.3. Blood Transfusion
4.3.1. Laboratory Research
4.3.2. Clinical Studies
| Anesthetics/Analgesics and Perioperative Management | TNF-α Release | Effects |
|---|---|---|
| VAs | Decreased | Sevoflurane improved hemodynamics and inflammatory responses in lung lobectomies but increased the incidence of postoperative complications compared with propofol [43]; isoflurane decreased inflammatory responses associated with OLV during open thoracic cancer surgery and had better postoperative outcomes compared with propofol [44]. |
| Increased | Sevoflurane exacerbated the injury to pulmonary function in lung cancer surgery compared with propofol [45]; sevoflurane increased the incidence of POCD after esophageal cancer surgery compared with propofol [46]. | |
| No difference | Sevoflurane had no effect on complement activation and cytokine release in major colorectal cancer surgery compared with propofol [47]; sevoflurane had no effect on NK cell and cytotoxic T lymphocyte counts or the apoptosis rate in breast cancer surgery compared with propofol [48]. | |
| Propofol | Decreased | Propofol attenuated the injury to pulmonary function in lung cancer surgery compared with sevoflurane [45]; propofol decreased the incidence of POCD after esophageal cancer surgery compared with sevoflurane [46]. |
| Increased | Propofol enhanced unstable hemodynamics and inflammatory responses in lung lobectomies but decreased the incidence of postoperative complications compared with sevoflurane [43]; propofol increased inflammatory responses associated with OLV during open thoracic cancer surgery and had worse postoperative outcomes compared with isoflurane [44]. | |
| No difference | Propofol had no effect on complement activation and cytokine release in major colorectal surgery compared with sevoflurane [47]; propofol had no effect on NK cell and cytotoxic T lymphocyte counts or the apoptosis rate in breast cancer surgery compared with sevoflurane [48]. | |
| Ketamine | Decreased | Ketamine suppressed pro-inflammatory cytokine production in radical prostatectomies [80]; ketamine prevented brain injury and improved postoperative fatigue syndrome after laparoscopic colorectal cancer surgery [81]; ketamine reduced inflammatory responses and improved postoperative anxiety and depression after colorectal cancer surgery [82]. |
| No difference | Ketamine in colorectal cancer surgery had no impact on postoperative NK cell activity, inflammatory responses, or long-term prognosis [77]; ketamine had no direct immunomodulation during minimally invasive robotic radical prostatectomies [78]. | |
| DEX | Decreased | DEX reduced the oxidative stress and inflammation in lung lobectomies with OLV [97]; DEX in GA provided better hemodynamic control, dismissed inflammatory responses, protected intestinal function, and shortened the length of hospital stay after laparoscopic radical gastrectomies compared with GA plus EA [98]; DEX attenuated systemic inflammation and ameliorated the impaired immune function in radical gastric cancer surgery [99]; DEX improved postoperative cognitive function after laparoscopic colon cancer surgery [100]; DEX reduced adverse responses and the occurrence of POCD after laparoscopic total hysterectomies [101]; DEX reduced postoperative inflammation and promoted the recovery of postoperative cognitive function after gastric cancer surgery [102]; DEX, during esophageal cancer surgery, alleviated the incidence of POCD [103]; DEX provided better cardiocerebral protection in colorectal cancer surgery [104]; DEX stabilized hemodynamics, reduced inflammation, and inhibited free radical generation in glioma surgery [105]; DEX stabilized hemodynamics and alleviated stress responses in ovarian cancer surgery compared with midazolam [106]. |
| Lidocaine | Decreased | Lidocaine, during supratentorial tumor resections, improved early recovery quality and had brain-injury alleviation effects [118]; lidocaine improved postoperative recovery, alleviated inflammation and immunosuppression, and accelerated the return of bowel function after laparoscopic radical gastrectomies [119]. |
| No difference | Lidocaine accelerated the return of bowel function and shortened the length of hospital stay after colorectal surgery [120]. | |
| Midazolam | Decreased | Midazolam, during thoracoscopic lung cancer surgery, inhibited inflammatory responses, decreased the requirement of anesthetics, stabilized perioperative hemodynamics, and mitigated postoperative pain [127]. |
| Increased | Midazolam enhanced unstable hemodynamics and stress responses in ovarian cancer surgery compared with DEX [106]. | |
| Thiopental | Not applicable | There is no relevant clinical study. |
| Opioids | Decreased | Nalbuphine enhanced analgesic effects, reduced the incidence of adverse reactions, and alleviated postoperative inflammatory responses in thoracoscopic lung lobectomies [152]. |
| No difference | Opioid-free anesthesia influenced acute inflammation in the perioperative period of oncological surgery [153]. | |
| NSAIDs | Decreased | Flurbiprofen axetil plus fentanyl provided similar postoperative analgesia in breast cancer surgery compared with fentanyl alone [174]. |
| RA | Decreased | Thoracic paravertebral blocks decreased the incidence of postoperative delirium and enhanced pain control and recovery quality compared with IVPCA in thoracoscopic lung lobectomies [186]; continuous wound analgesia alleviated systemic inflammation, decreased pain scores and opioid intake, and accelerated the recovery of respiratory function in lung cancer surgery [187]; EA, during gastric cancer surgery, mitigated inflammatory responses, preserved innate tumor immunity, and decreased the incidence of postoperative adverse reactions [188]; EA, during gastric cancer surgery, reduced the stress reactions and maintained the integrity of immune function [189]; EA improved postoperative recovery and cognitive function and relieved inflammatory responses in liver cancer surgery [190]. |
| No difference | Thoracic EA attenuated local inflammation but did not affect systemic inflammation during lung cancer surgery [192]; thoracic paravertebral blocks or EA decreased pain scores, reduced the use of opioids and vasoactive agents, and improved perioperative immune function and postoperative survival after esophageal cancer surgery [193]; GA plus EA and patient-controlled epidural analgesia provided lower opioid consumption and shorter hospital stay in radical cystectomies compared with GA plus IVPCA [194]; thoracic EA blunted early postoperative stress responses and reduced postoperative pain after radical prostatectomies [195]; surgical rather than analgesic techniques had a great impact on postoperative inflammation in colorectal cancer surgery [196]. | |
| Body temperature | Decreased | Preoperative hyperthermia improved immune responses to surgical stress but did not enhance the quality of recovery after colorectal cancer surgery [203]. |
| Increased | Cancer patients treated with hyperthermia had a redistribution of innate immune cells [204]. | |
| No difference | The cytokine levels paralleled hemodynamic and metabolic derangements in the hyperthermic phase of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy [205]; warmed and humidified CO2 insufflation had no benefit in terms of pain scores and cytokine levels during robotic radical prostatectomies [206]; moderate hypothermia during malignant melanoma resection affected the release of circulating cytokines and adhesion molecules [207]. | |
| Hyperglycemia | Decreased | Perioperative hyperglycemia attenuated postoperative immune activation during esophageal or pancreatic cancer resections [212]. |
| Increased | The degree of perioperative glucose fluctuation was positively related to postoperative cytokine levels and poor short-term prognosis after supratentorial tumor resection [211]. | |
| Blood transfusion | Increased | Allogenic RBC transfusion increased the serum levels of cytokines in colorectal cancer surgery [217]; RBC transfusion in advanced colorectal cancer surgery exacerbated the antioxidative and immune systems and increased serum cytokine levels [218]. |
5. TNF-α in Cancer
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Yuki, K. The Role of General Anesthetic Drug Selection in Cancer Outcome. Biomed Res. Int. 2021, 2021, 2563093. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.; Tie, Y.; Tu, C.; Wei, X. Surgical trauma-induced immunosuppression in cancer: Recent advances and the potential therapies. Clin. Transl. Med. 2020, 10, 199–223. [Google Scholar] [CrossRef]
- Dubowitz, J.A.; Sloan, E.K.; Riedel, B.J. Implicating anaesthesia and the perioperative period in cancer recurrence and metastasis. Clin. Exp. Metastasis 2018, 35, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, P.; Xu, Y.; Yan, J.; Liu, Z.; Lau, W.B.; Lau, B.; Li, Y.; Zhao, X.; Wei, Y.; et al. Surgical stress and cancer progression: The twisted tango. Mol. Cancer 2019, 18, 132. [Google Scholar] [CrossRef] [Green Version]
- Kim, R. Effects of surgery and anesthetic choice on immunosuppression and cancer recurrence. J. Transl. Med. 2018, 16, 8. [Google Scholar] [CrossRef] [Green Version]
- Anderson, N.M.; Simon, M.C. The tumor microenvironment. Curr. Biol. 2020, 30, R921–R925. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Yu, D. Tumor microenvironment as a therapeutic target in cancer. Pharmacol. Ther. 2021, 221, 107753. [Google Scholar] [CrossRef]
- Arneth, B. Tumor Microenvironment. Medicina 2019, 56, 15. [Google Scholar] [CrossRef] [Green Version]
- Cheng, X.; Zhang, H.; Hamad, A.; Huang, H.; Tsung, A. Surgery-mediated tumor-promoting effects on the immune microenvironment. Semin. Cancer Biol. 2022, 86, 408–419. [Google Scholar] [CrossRef]
- Onuma, A.E.; Zhang, H.; Gil, L.; Huang, H.; Tsung, A. Surgical Stress Promotes Tumor Progression: A Focus on the Impact of the Immune Response. J. Clin. Med. 2020, 9, 4096. [Google Scholar] [CrossRef]
- Chu, W.M. Tumor necrosis factor. Cancer Lett. 2013, 328, 222–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laha, D.; Grant, R.; Mishra, P.; Nilubol, N. The Role of Tumor Necrosis Factor in Manipulating the Immunological Response of Tumor Microenvironment. Front. Immunol. 2021, 12, 656908. [Google Scholar] [CrossRef] [PubMed]
- Mercogliano, M.F.; Bruni, S.; Mauro, F.; Elizalde, P.V.; Schillaci, R. Harnessing Tumor Necrosis Factor Alpha to Achieve Effective Cancer Immunotherapy. Cancers 2021, 13, 564. [Google Scholar] [CrossRef] [PubMed]
- Sha, J.; Du, J.; Yang, J.; Hu, X.; Li, L. Changes of serum levels of tumor necrosis factor (TNF-α) and soluble interleukin-2 receptor (SIL 2R) in patients with cervical cancer and their clinical significance. Am. J. Transl. Res. 2021, 13, 6599–6604. [Google Scholar] [PubMed]
- Pawlik, W.; Pawlik, J.; Kozłowski, M.; Łuczkowska, K.; Kwiatkowski, S.; Kwiatkowska, E.; Machaliński, B.; Cymbaluk-Płoska, A. The Clinical Importance of IL-6, IL-8, and TNF-α in Patients with Ovarian Carcinoma and Benign Cystic Lesions. Diagnostics 2021, 11, 1625. [Google Scholar] [CrossRef] [PubMed]
- Wieser, V.; Abdel Azim, S.; Sprung, S.; Knoll, K.; Kögl, J.; Hackl, H.; Marth, C.; Zeimet, A.G.; Fiegl, H. TNFα signalling predicts poor prognosis of patients with endometrial cancer. Carcinogenesis 2020, 41, 1065–1073. [Google Scholar] [CrossRef]
- Erturk, K.; Tastekin, D.; Serilmez, M.; Bilgin, E.; Bozbey, H.U.; Vatansever, S. Clinical significance of serum interleukin-29, interleukin-32, and tumor necrosis factor alpha levels in patients with gastric cancer. Tumor Biol. 2016, 37, 405–412. [Google Scholar] [CrossRef]
- Stanilov, N.; Miteva, L.; Dobreva, Z.; Stanilova, S. Colorectal cancer severity and survival in correlation with tumour necrosis factor-alpha. Biotechnol. Biotechnol. Equip. 2014, 28, 911–917. [Google Scholar] [CrossRef]
- Wang, H.; Liu, J.; Hu, X.; Liu, S.; He, B. Prognostic and Therapeutic Values of Tumor Necrosis Factor-Alpha in Hepatocellular Carcinoma. Med. Sci. Monit. 2016, 22, 3694–3704. [Google Scholar] [CrossRef] [Green Version]
- Sun, T.Y.; Hsu, C.L.; Lee, M.S.; Yeh, T.T.; Lai, H.C.; Wu, K.L.; Wu, Z.F.; Tseng, W.C. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in limb-salvage surgery for osteosarcoma: A retrospective analysis. Medicine 2022, 101, e30840. [Google Scholar] [CrossRef] [PubMed]
- Tseng, W.C.; Lee, M.S.; Lin, Y.C.; Lai, H.C.; Yu, M.H.; Wu, K.L.; Wu, Z.F. Propofol-Based Total Intravenous Anesthesia is Associated with Better Survival than Desflurane Anesthesia in Epithelial Ovarian Cancer Surgery: A Retrospective Cohort Study. Front. Pharmacol. 2021, 12, 685265. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.H.; Wu, Z.F.; Lee, M.S.; Lou, Y.S.; Wu, K.L.; Cheng, K.I.; Lai, H.C. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in glioblastoma surgery. PLoS ONE 2021, 16, e0255627. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.C.; Lee, M.S.; Lin, K.T.; Huang, Y.H.; Chen, J.Y.; Lin, Y.T.; Hung, K.C.; Wu, Z.F. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in robot-assisted radical prostatectomy. PLoS ONE 2020, 15, e0230290. [Google Scholar] [CrossRef]
- Lai, H.C.; Lee, M.S.; Liu, Y.T.; Lin, K.T.; Hung, K.C.; Chen, J.Y.; Wu, Z.F. Propofol-based intravenous anesthesia is associated with better survival than desflurane anesthesia in pancreatic cancer surgery. PLoS ONE 2020, 15, e0233598. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.C.; Lee, M.S.; Lin, C.; Lin, K.T.; Huang, Y.H.; Wong, C.S.; Chan, S.M.; Wu, Z.F. Propofol-based total intravenous anaesthesia is associated with better survival than desflurane anaesthesia in hepatectomy for hepatocellular carcinoma: A retrospective cohort study. Br. J. Anaesth. 2019, 123, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.F.; Lee, M.S.; Wong, C.S.; Lu, C.H.; Huang, Y.S.; Lin, K.T.; Lou, Y.S.; Lin, C.; Chang, Y.C.; Lai, H.C. Propofol-based Total Intravenous Anesthesia Is Associated with Better Survival Than Desflurane Anesthesia in Colon Cancer Surgery. Anesthesiology 2018, 129, 932–941. [Google Scholar] [CrossRef]
- Chang, C.Y.; Wu, M.Y.; Chien, Y.J.; Su, I.M.; Wang, S.C.; Kao, M.C. Anesthesia and Long-term Oncological Outcomes: A Systematic Review and Meta-analysis. Anesth. Analg. 2021, 132, 623–634. [Google Scholar] [CrossRef]
- Lu, N.; Piao, M.H.; Feng, C.S.; Yuan, Y. Isoflurane promotes epithelial-to-mesenchymal transition and metastasis of bladder cancer cells through HIF-1α-β-catenin/Notch1 pathways. Life Sci. 2020, 258, 118154. [Google Scholar] [CrossRef]
- Lai, R.C.; Shan, W.R.; Zhou, D.; Zeng, X.Q.; Zuo, K.; Pan, D.F.; Zeng, W.A.; Zuo, Z.Y. Sevoflurane promotes migration, invasion, and colony-forming ability of human glioblastoma cells possibly via increasing the expression of cell surface protein 44. Acta Pharmacol. Sin. 2019, 40, 1424–1435. [Google Scholar] [CrossRef]
- Zhang, W.; Shao, X. Isoflurane Promotes Non-Small Cell Lung Cancer Malignancy by Activating the Akt-Mammalian Target of Rapamycin (mTOR) Signaling Pathway. Med. Sci. Monit. 2016, 22, 4644–4650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, X.; Zhao, H.; Hennah, L.; Ning, J.; Liu, J.; Tu, H.; Ma, D. Impact of isoflurane on malignant capability of ovarian cancer in vitro. Br. J. Anaesth. 2015, 114, 831–839. [Google Scholar] [CrossRef] [Green Version]
- Ecimovic, P.; McHugh, B.; Murray, D.; Doran, P.; Buggy, D.J. Effects of sevoflurane on breast cancer cell function in vitro. Anticancer Res. 2013, 33, 4255–4260. [Google Scholar]
- Benzonana, L.L.; Perry, N.J.; Watts, H.R.; Yang, B.; Perry, I.A.; Coombes, C.; Takata, M.; Ma, D. Isoflurane, a commonly used volatile anesthetic, enhances renal cancer growth and malignant potential via the hypoxia-inducible factor cellular signaling pathway in vitro. Anesthesiology 2013, 119, 593–605. [Google Scholar] [CrossRef]
- Zhao, H.; Xing, F.; Yuan, J.; Li, Z.; Zhang, W. Sevoflurane inhibits migration and invasion of glioma cells via regulating miR-34a-5p/MMP-2 axis. Life Sci. 2020, 256, 117897. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, B.; Wang, X.; Sheng, X.; Cui, Y. Sevoflurane inhibits the progression of ovarian cancer through down-regulating stanniocalcin 1 (STC1). Cancer Cell Int. 2019, 19, 339. [Google Scholar] [CrossRef] [Green Version]
- Bundscherer, A.C.; Ullrich, V.; Malsy, M.; Gruber, M.A.; Graf, B.M.; Brockhoff, G.; Sinner, B. Effects of Volatile Anesthetics on Proliferation and Viability of SW480 Colon Cancer Cells In Vitro. Anticancer Res. 2019, 39, 6049–6055. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Yang, C.X.; Zhang, B.; Wang, H.B.; Liu, H.Z.; Lai, X.H.; Liao, M.J.; Zhang, T. Sevoflurane suppresses hypoxia-induced growth and metastasis of lung cancer cells via inhibiting hypoxia-inducible factor-1α. J. Anesth. 2015, 29, 821–830. [Google Scholar] [CrossRef]
- Müller-Edenborn, B.; Roth-Zʼgraggen, B.; Bartnicka, K.; Borgeat, A.; Hoos, A.; Borsig, L.; Beck-Schimmer, B. Volatile anesthetics reduce invasion of colorectal cancer cells through down-regulation of matrix metalloproteinase-9. Anesthesiology 2012, 117, 293–301. [Google Scholar] [CrossRef] [Green Version]
- Stollings, L.M.; Jia, L.J.; Tang, P.; Dou, H.; Lu, B.; Xu, Y. Immune Modulation by Volatile Anesthetics. Anesthesiology 2016, 125, 399–411. [Google Scholar] [CrossRef] [Green Version]
- Yuki, K.; Eckenhoff, R.G. Mechanisms of the Immunological Effects of Volatile Anesthetics: A Review. Anesth. Analg. 2016, 123, 326–335. [Google Scholar] [CrossRef] [Green Version]
- Kawaraguchi, Y.; Horikawa, Y.T.; Murphy, A.N.; Murray, F.; Miyanohara, A.; Ali, S.S.; Head, B.P.; Patel, P.M.; Roth, D.M.; Patel, H.H. Volatile anesthetics protect cancer cells against tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis via caveolins. Anesthesiology 2011, 115, 499–508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.; Li, M.; Zheng, R.; Huang, Q.; Li, Y.; Zhu, Y.; Chen, Z.; Lin, J. Effects of sevoflurane inhalation anesthesia on IL-6, TNF-α and MMP-9 expression and hemodynamics in elderly patients undergoing lobectomy for lung cancer. Cell. Mol. Biol. 2020, 66, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, K.; Ammar, A. Immunomodulatory Effects of Anesthetics during Thoracic Surgery. Anesthesiol. Res. Pract. 2011, 2011, 317410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, Y.; Zhao, X.; Li, H.; Wang, Z.; Wang, D. Effects of sevoflurane and propofol on the inflammatory response and pulmonary function of perioperative patients with one-lung ventilation. Exp. Ther. Med. 2013, 6, 781–785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiao, Y.; Feng, H.; Zhao, T.; Yan, H.; Zhang, H.; Zhao, X. Postoperative cognitive dysfunction after inhalational anesthesia in elderly patients undergoing major surgery: The influence of anesthetic technique, cerebral injury and systemic inflammation. BMC Anesthesiol. 2015, 15, 154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kvarnström, A.L.; Sarbinowski, R.T.; Bengtson, J.P.; Jacobsson, L.M.; Bengtsson, A.L. Complement activation and interleukin response in major abdominal surgery. Scand. J. Immunol. 2012, 75, 510–516. [Google Scholar] [CrossRef]
- Lim, J.A.; Oh, C.S.; Yoon, T.G.; Lee, J.Y.; Lee, S.H.; Yoo, Y.B.; Yang, J.H.; Kim, S.H. The effect of propofol and sevoflurane on cancer cell, natural killer cell, and cytotoxic T lymphocyte function in patients undergoing breast cancer surgery: An in vitro analysis. BMC Cancer 2018, 18, 159. [Google Scholar] [CrossRef]
- Jiang, S.; Liu, Y.; Huang, L.; Zhang, F.; Kang, R. Effects of propofol on cancer development and chemotherapy: Potential mechanisms. Eur. J. Pharmacol. 2018, 831, 46–51. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, H.; Shi, Z.A.; Li, Y. Propofol Prevents the Growth, Migration, Invasion, and Glycolysis of Colorectal Cancer Cells by Downregulating Lactate Dehydrogenase Both In Vitro and In Vivo. J. Oncol. 2022, 2022, 8317466. [Google Scholar] [CrossRef]
- Sun, Y.; Peng, Y.B.; Ye, L.L.; Ma, L.X.; Zou, M.Y.; Cheng, Z.G. Propofol inhibits proliferation and cisplatin resistance in ovarian cancer cells through regulating the microRNA-374a/forkhead box O1 signaling axis. Mol. Med. Rep. 2020, 21, 1471–1480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, W.Z.; Liu, N. Propofol Inhibits Lung Cancer A549 Cell Growth and Epithelial-Mesenchymal Transition Process by Upregulation of MicroRNA-1284. Oncol. Res. 2018, 27, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Zhang, Y. Propofol inhibits proliferation and accelerates apoptosis of human gastric cancer cells by regulation of microRNA-451 and MMP-2 expression. Genet. Mol. Res. 2016, 15, 10.4238. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.B.; Jiang, W.; Zhao, F.R.; Li, G.; Du, Q.H.; Zhang, M.Y.; Guo, X.G. Propofol suppresses invasion and induces apoptosis of osteosarcoma cell in vitro via downregulation of TGF-β1 expression. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1430–1435. [Google Scholar]
- Wang, Z.T.; Gong, H.Y.; Zheng, F.; Liu, D.J.; Dong, T.L. Propofol suppresses proliferation and invasion of pancreatic cancer cells by upregulating microRNA-133a expression. Genet. Mol. Res. 2015, 14, 7529–7537. [Google Scholar] [CrossRef]
- Xu, J.; Xu, W.; Zhu, J. Propofol suppresses proliferation and invasion of glioma cells by upregulating microRNA-218 expression. Mol. Med. Rep. 2015, 12, 4815–4820. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.B.; Du, Q.H.; Zhang, M.Y.; Yun, P.; He, C.Y. Propofol suppresses proliferation, invasion and angiogenesis by down-regulating ERK-VEGF/MMP-9 signaling in Eca-109 esophageal squamous cell carcinoma cells. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 2486–2494. [Google Scholar]
- Ling, Q.; Wu, S.; Liao, X.; Liu, C.; Chen, Y. Anesthetic propofol enhances cisplatin-sensitivity of non-small cell lung cancer cells through N6-methyladenosine-dependently regulating the miR-486-5p/RAP1-NF-κB axis. BMC Cancer 2022, 22, 765. [Google Scholar] [CrossRef]
- Qian, J.; Shen, S.; Chen, W.; Chen, N. Propofol Reversed Hypoxia-Induced Docetaxel Resistance in Prostate Cancer Cells by Preventing Epithelial-Mesenchymal Transition by Inhibiting Hypoxia-Inducible Factor 1α. Biomed Res. Int. 2018, 2018, 4174232. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Lu, Y.; Pang, Y.; Li, M.; Cheng, X.; Chen, J. Propofol enhances the cisplatin-induced apoptosis on cervical cancer cells via EGFR/JAK2/STAT3 pathway. Biomed. Pharmacother. 2017, 86, 324–333. [Google Scholar] [CrossRef]
- Du, Q.H.; Xu, Y.B.; Zhang, M.Y.; Yun, P.; He, C.Y. Propofol induces apoptosis and increases gemcitabine sensitivity in pancreatic cancer cells in vitro by inhibition of nuclear factor-κB activity. World J. Gastroenterol. 2013, 19, 5485–5492. [Google Scholar] [CrossRef]
- Cui, W.Y.; Liu, Y.; Zhu, Y.Q.; Song, T.; Wang, Q.S. Propofol induces endoplasmic reticulum (ER) stress and apoptosis in lung cancer cell H460. Tumor Biol. 2014, 35, 5213–5217. [Google Scholar] [CrossRef]
- Kushida, A.; Inada, T.; Shingu, K. Enhancement of antitumor immunity after propofol treatment in mice. Immunopharmacol. Immunotoxicol. 2007, 29, 477–486. [Google Scholar] [CrossRef]
- Melamed, R.; Bar-Yosef, S.; Shakhar, G.; Shakhar, K.; Ben-Eliyahu, S. Suppression of natural killer cell activity and promotion of tumor metastasis by ketamine, thiopental, and halothane, but not by propofol: Mediating mechanisms and prophylactic measures. Anesth. Analg. 2003, 97, 1331–1339. [Google Scholar] [CrossRef] [PubMed]
- Meng, C.; Song, L.; Wang, J.; Li, D.; Liu, Y.; Cui, X. Propofol induces proliferation partially via downregulation of p53 protein and promotes migration via activation of the Nrf2 pathway in human breast cancer cell line MDA-MB-231. Oncol. Rep. 2017, 37, 841–848. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Wang, N.; Zhou, S.; Ye, W.; Jing, G.; Zhang, M. Propofol induces proliferation and invasion of gallbladder cancer cells through activation of Nrf2. J. Exp. Clin. Cancer Res. 2012, 31, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, F.; Jiang, J.; Yu, G.; Zang, H.; Sun, H. Propofol Pretreatment Prevents Oxygen-Glucose Deprivation/Reoxygenation (OGD/R)-induced Inflammation Through Nuclear Transcription Factor κB (NF-κB) Pathway in Neuroblastoma Cells. Curr. Neurovasc. Res. 2020, 17, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.; Song, T.; Wang, W.; Tian, J.; Ma, X. Immunomodulatory roles of propofol and sevoflurane in murine models of breast cancer. Immunopharmacol. Immunotoxicol. 2022, 1–7. [Google Scholar] [CrossRef]
- Li, H.; Liu, W.; Zhang, X.; Wu, F.; Sun, D.; Wang, Z. Ketamine suppresses proliferation and induces ferroptosis and apoptosis of breast cancer cells by targeting KAT5/GPX4 axis. Biochem. Biophys. Res. Commun. 2021, 585, 111–116. [Google Scholar] [CrossRef]
- He, G.N.; Bao, N.R.; Wang, S.; Xi, M.; Zhang, T.H.; Chen, F.S. Ketamine Induces Ferroptosis of Liver Cancer Cells by Targeting lncRNA PVT1/miR-214-3p/GPX4. Drug Des. Dev. Ther. 2021, 15, 3965–3978. [Google Scholar] [CrossRef]
- Li, T.; Yang, J.; Yang, B.; Zhao, G.; Lin, H.; Liu, Q.; Wang, L.; Wan, Y.; Jiang, H. Ketamine Inhibits Ovarian Cancer Cell Growth by Regulating the lncRNA-PVT1/EZH2/p57 Axis. Front. Genet. 2020, 11, 597467. [Google Scholar] [CrossRef]
- Zhao, S.; Shao, L.; Wang, Y.; Meng, Q.; Yu, J. Ketamine exhibits anti-gastric cancer activity via induction of apoptosis and attenuation of PI3K/Akt/mTOR. Arch. Med. Sci. 2020, 16, 1140–1149. [Google Scholar] [CrossRef]
- Duan, W.; Hu, J.; Liu, Y. Ketamine inhibits colorectal cancer cells malignant potential via blockage of NMDA receptor. Exp. Mol. Pathol. 2019, 107, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhang, P.; Luo, W.; Zhang, L.; Hu, R.; Sun, Y.; Jiang, H. Ketamine induces apoptosis in lung adenocarcinoma cells by regulating the expression of CD69. Cancer Med. 2018, 7, 788–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shapiro, J.; Jersky, J.; Katzav, S.; Feldman, M.; Segal, S. Anesthetic drugs accelerate the progression of postoperative metastases of mouse tumors. J. Clin. Investig. 1981, 68, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Forget, P.; Collet, V.; Lavand′homme, P.; De Kock, M. Does analgesia and condition influence immunity after surgery? Effects of fentanyl, ketamine and clonidine on natural killer activity at different ages. Eur. J. Anaesthesiol. 2010, 27, 233–240. [Google Scholar] [CrossRef]
- Cho, J.S.; Kim, N.Y.; Shim, J.K.; Jun, J.H.; Lee, S.; Kwak, Y.L. The immunomodulatory effect of ketamine in colorectal cancer surgery: A randomized-controlled trial. Can. J. Anaesth. 2021, 68, 683–692. [Google Scholar] [CrossRef]
- Kawaguchi, J.; Ota, D.; Niwa, H.; Sugo, Y.; Kushikata, T.; Hirota, K. Immunomodulation by ketamine as an adjunct to total intravenous anesthesia in patients undergoing minimally invasive radical prostatectomy: A randomized pilot trial. Mol. Clin. Oncol. 2020, 13, 203–208. [Google Scholar] [CrossRef]
- Yang, C.; Jiang, R.Y.; Shen, J.; Hong, T.; Liu, N.; Ding, L.C.; Wang, D.M.; Chen, L.J.; Xu, B.; Zhu, B. Ketamine attenuates the lipopolysaccharide-induced inflammatory response in cultured N2a cells. Mol. Med. Rep. 2013, 8, 217–220. [Google Scholar] [CrossRef] [Green Version]
- Ali, H.M.; Mokhtar, A.M. Effect of Single Compared to Repeated Doses of Intravenous S(+) Ketamine on the Release of Pro-inflammatory Cytokines in Patients Undergoing Radical Prostatectomy. Anesth. Essays Res. 2017, 11, 282–286. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Zhang, H.; Cheng, H. Effect of a single sub-dose of ketamine on postoperative fatigue syndrome in colorectal cancer patients undergoing radical laparoscopic surgery: A double-blind, pilot study. J. Affect. Disord. 2022, 312, 146–151. [Google Scholar] [CrossRef]
- Ren, Q.; Hua, L.; Zhou, X.; Cheng, Y.; Lu, M.; Zhang, C.; Guo, J.; Xu, H. Effects of a Single Sub-Anesthetic Dose of Ketamine on Postoperative Emotional Responses and Inflammatory Factors in Colorectal Cancer Patients. Front. Pharmacol. 2022, 13, 818822. [Google Scholar] [CrossRef]
- Tian, H.; Hou, L.; Xiong, Y.; Cheng, Q. Dexmedetomidine upregulates microRNA-185 to suppress ovarian cancer growth via inhibiting the SOX9/Wnt/β-catenin signaling pathway. Cell Cycle 2021, 20, 765–780. [Google Scholar] [CrossRef]
- Xu, B.; Qian, Y.; Hu, C.; Wang, Y.; Gao, H.; Yang, J. Dexmedetomidine upregulates the expression of miR-493-5p, inhibiting growth and inducing the apoptosis of lung adenocarcinoma cells by targeting RASL11B. Biochem. Cell Biol. 2021, 99, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.; Jin, S.; Liu, H.; Le, C.; Gao, J.; Cheng, J.; Chen, L.; Li, N. Dexmedetomidine inhibits cell malignancy in osteosarcoma cells via miR-520a-3p-YOD1 interactome. Biochem. Biophys. Res. Commun. 2021, 543, 56–64. [Google Scholar] [CrossRef]
- Zhang, P.; He, H.; Bai, Y.; Liu, W.; Huang, L. Dexmedetomidine suppresses the progression of esophageal cancer via miR-143-3p/epidermal growth factor receptor pathway substrate 8 axis. Anticancer Drugs 2020, 31, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Chi, M.; Shi, X.; Huo, X.; Wu, X.; Zhang, P.; Wang, G. Dexmedetomidine promotes breast cancer cell migration through Rab11-mediated secretion of exosomal TMPRSS2. Ann. Transl. Med. 2020, 8, 531. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Li, G.H.; Tan, G.C.; Liang, H.; Lai, X.H.; Huang, Q.; Zhong, J.Y. Dexmedetomidine enhances hypoxia-induced cancer cell progression. Exp. Ther. Med. 2019, 18, 4820–4828. [Google Scholar] [CrossRef] [Green Version]
- Lavon, H.; Matzner, P.; Benbenishty, A.; Sorski, L.; Rossene, E.; Haldar, R.; Elbaz, E.; Cata, J.P.; Gottumukkala, V.; Ben-Eliyahu, S. Dexmedetomidine promotes metastasis in rodent models of breast, lung, and colon cancers. Br. J. Anaesth. 2018, 120, 188–196. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Datoo, T.; Zhao, H.; Wu, L.; Date, A.; Jiang, C.; Sanders, R.D.; Wang, G.; Bevan, C.; Ma, D. Midazolam and Dexmedetomidine Affect Neuroglioma and Lung Carcinoma Cell Biology In Vitro and In Vivo. Anesthesiology 2018, 129, 1000–1014. [Google Scholar] [CrossRef]
- Yuki, K. The immunomodulatory mechanism of dexmedetomidine. Int. Immunopharmacol. 2021, 97, 107709. [Google Scholar] [CrossRef]
- Tian, H.; Hou, L.; Xiong, Y.; Cheng, Q.; Huang, J. Effect of Dexmedetomidine-Mediated Insulin-Like Growth Factor 2 (IGF2) Signal Pathway on Immune Function and Invasion and Migration of Cancer Cells in Rats with Ovarian Cancer. Med. Sci. Monit. 2019, 25, 4655–4664. [Google Scholar] [CrossRef]
- Cai, Q.H.; Tang, Y.; Fan, S.H.; Zhang, Z.F.; Li, H.; Huang, S.Q.; Wu, D.M.; Lu, J.; Zheng, Y.L. In vivo effects of dexmedetomidine on immune function and tumor growth in rats with ovarian cancer through inhibiting the p38MAPK/NF-κB signaling pathway. Biomed. Pharmacother. 2017, 95, 1830–1837. [Google Scholar] [CrossRef]
- Shin, S.; Kim, K.J.; Hwang, H.J.; Noh, S.; Oh, J.E.; Yoo, Y.C. Immunomodulatory Effects of Perioperative Dexmedetomidine in Ovarian Cancer: An In Vitro and Xenograft Mouse Model Study. Front. Oncol. 2021, 11, 722743. [Google Scholar] [CrossRef] [PubMed]
- Suo, L.; Wang, M. Dexmedetomidine attenuates oxygen-glucose deprivation/ reperfusion-induced inflammation through the miR-17-5p/ TLR4/ NF-κB axis. BMC Anesthesiol. 2022, 22, 126. [Google Scholar] [CrossRef]
- Wang, K.; Wu, M.; Xu, J.; Wu, C.; Zhang, B.; Wang, G.; Ma, D. Effects of dexmedetomidine on perioperative stress, inflammation, and immune function: Systematic review and meta-analysis. Br. J. Anaesth. 2019, 123, 777–794. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Wang, Y.; Zhao, J.; Su, A. Effects of dexmedetomidine pretreatment on heme oxygenase-1 expression and oxidative stress during one-lung ventilation. Int. J. Clin. Exp. Pathol. 2015, 8, 3144–3149. [Google Scholar] [PubMed]
- Guo, L.; Liu, Y.; Wang, M. Effect of Perioperative Dexmedetomidine Anesthesia on Prognosis of Elderly Patients with Gastrointestinal Tumor Surgery. Comput. Math. Methods Med. 2022, 2022, 7889372. [Google Scholar] [CrossRef]
- Dong, W.; Chen, M.H.; Yang, Y.H.; Zhang, X.; Huang, M.J.; Yang, X.J.; Wang, H.Z. The effect of dexmedetomidine on expressions of inflammatory factors in patients with radical resection of gastric cancer. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 3510–3515. [Google Scholar] [PubMed]
- Ding, M.; Xu, X.; Xia, L.; Cao, Y. Magnetic Resonance Imaging Characteristic Evaluation of Dexmedetomidine on Neurocognitive Dysfunction in Elderly Patients with Colorectal Tumors after Laparoscopic Operation. Comput. Math. Methods Med. 2022, 2022, 1345695. [Google Scholar] [CrossRef]
- Huang, H.; Xu, X.; Xiao, Y.; Jia, J. The Influence of Different Dexmedetomidine Doses on Cognitive Function at Early Period of Patients Undergoing Laparoscopic Extensive Total Hysterectomy. J. Healthc. Eng. 2021, 2021, 3531199. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Shen, Z.; Wang, H.; Zhang, L.; Dong, R. Effect of dexmedetomidine on the cognitive function of patients undergoing gastric cancer surgery by regulating the PI3K/AKT signaling pathway. Oncol. Lett. 2020, 19, 1151–1156. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Wu, Z.; Zhao, X.; Qiao, Y. Role of dexmedetomidine in reducing the incidence of postoperative cognitive dysfunction caused by sevoflurane inhalation anesthesia in elderly patients with esophageal carcinoma. J. Cancer Res. Ther. 2018, 14, 1497–1502. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.L.; Wang, J.T.; Chu, C.Q.; Li, Y.X.; Yin, J.H.; Liu, S.L. Cardiocerebral protective effects of dexmedetomidine as anesthetic in colorectal cancer surgery. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 3570–3576. [Google Scholar] [CrossRef]
- Luo, X.; Zheng, X.; Huang, H. Protective effects of dexmedetomidine on brain function of glioma patients undergoing craniotomy resection and its underlying mechanism. Clin. Neurol. Neurosurg. 2016, 146, 105–108. [Google Scholar] [CrossRef]
- Liu, M.; Yi, Y.; Zhao, M. Effect of dexmedetomidine anesthesia on perioperative levels of TNF-α and IL-6 in patients with ovarian cancer. Oncol. Lett. 2019, 17, 5517–5522. [Google Scholar] [CrossRef] [Green Version]
- Tang, C.; Hu, Y.; Zhang, Z.; Wei, Z.; Wang, H.; Geng, Q.; Shi, S.; Wang, S.; Wang, J.; Chai, X. Dexmedetomidine with sufentanil in intravenous patient-controlled analgesia for relief from postoperative pain, inflammation and delirium after esophageal cancer surgery. Biosci. Rep. 2020, 40, BSR20193410. [Google Scholar] [CrossRef]
- Miao, Z.; Wu, P.; Wang, J.; Zhou, F.C.; Lin, Y.; Lu, X.Y.; Lv, R.; Hou, Q.H.; Wen, Q.P. Whole-Course Application of Dexmedetomidine Combined with Ketorolac in Nonnarcotic Postoperative Analgesia for Patients with Lung Cancer Undergoing Thoracoscopic Surgery: A Randomized Control Trial. Pain Physician 2020, 23, E185–E193. [Google Scholar] [PubMed]
- Song, J.; Wei, N.; Zhang, J.; Wang, G. Effect of dexmedetomidine combined with oxycodone patient-controlled intravenous analgesia on the levels of inflammatory cytokine in patients with rectal cancer. Pak. J. Pharm. Sci. 2019, 32, 1381–1385. [Google Scholar]
- Beaussier, M.; Delbos, A.; Maurice-Szamburski, A.; Ecoffey, C.; Mercadal, L. Perioperative Use of Intravenous Lidocaine. Drugs 2018, 78, 1229–1246. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, X.; Li, Y.; Li, Y.; Pang, T. Lidocaine hampers colorectal cancer process via circITFG2/miR-1204/SOCS2 axis. Anticancer Drugs 2022, 33, 235–244. [Google Scholar] [CrossRef]
- Guan, E.; Liu, H.; Xu, N. Lidocaine Suppresses Gastric Cancer Development Through Circ_ANO5/miR-21-5p/LIFR Axis. Dig. Dis. Sci. 2022, 67, 2244–2256. [Google Scholar] [CrossRef]
- Li, B.; Xu, H.; He, C.; Zou, W.; Tu, Y. Lidocaine prevents breast cancer growth by targeting neuronatin to inhibit nerve fibers formation. J. Toxicol. Sci. 2021, 46, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Ma, N.; Liu, G.; Mao, N.; Chen, F.; Li, J. Lidocaine Inhibits Hepatocellular Carcinoma Development by Modulating circ_ITCH/miR-421/CPEB3 Axis. Dig. Dis. Sci. 2021, 66, 4384–4397. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Sun, Y. Lidocaine inhibits proliferation and metastasis of lung cancer cell via regulation of miR-539/EGFR axis. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2866–2874. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wall, T.P.; Buggy, D.J. Perioperative Intravenous Lidocaine and Metastatic Cancer Recurrence—A Narrative Review. Front. Oncol. 2021, 11, 688896. [Google Scholar] [CrossRef]
- Ferreira, L.E.N.; Abdalla, H.B.; da Costa, J.P.; de Freitas Domingues, J.S.; Burga-Sánchez, J.; Groppo, F.C.; Volpato, M.C. Effects of 2-Hydroxypropil-Β-Cyclodextrin-Lidocaine on Tumor Growth and Inflammatory Response. Curr. Drug Deliv. 2020, 17, 588–598. [Google Scholar] [CrossRef]
- Zhao, K.; Dong, Y.; Su, G.; Wang, Y.; Ji, T.; Wu, N.; Cui, X.; Li, W.; Yang, Y.; Chen, X. Effect of Systemic Lidocaine on Postoperative Early Recovery Quality in Patients Undergoing Supratentorial Tumor Resection. Drug Des. Dev. Ther. 2022, 16, 1171–1181. [Google Scholar] [CrossRef]
- Lv, X.; Li, X.; Guo, K.; Li, T.; Yang, Y.; Lu, W.; Wang, S.; Liu, S. Effects of Systemic Lidocaine on Postoperative Recovery Quality and Immune Function in Patients Undergoing Laparoscopic Radical Gastrectomy. Drug Des. Dev. Ther. 2021, 15, 1861–1872. [Google Scholar] [CrossRef]
- Herroeder, S.; Pecher, S.; Schönherr, M.E.; Kaulitz, G.; Hahnenkamp, K.; Friess, H.; Böttiger, B.W.; Bauer, H.; Dijkgraaf, M.G.; Durieux, M.E.; et al. Systemic lidocaine shortens length of hospital stay after colorectal surgery: A double-blinded, randomized, placebo-controlled trial. Ann. Surg. 2007, 246, 192–200. [Google Scholar] [CrossRef]
- Mishra, S.K.; Kang, J.H.; Lee, C.W.; Oh, S.H.; Ryu, J.S.; Bae, Y.S.; Kim, H.M. Midazolam induces cellular apoptosis in human cancer cells and inhibits tumor growth in xenograft mice. Mol. Cells 2013, 36, 219–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, Y.; Yao, X.; Du, X. Midazolam inhibits proliferation and accelerates apoptosis of hepatocellular carcinoma cells by elevating microRNA-124-3p and suppressing PIM-1. IUBMB Life 2020, 72, 452–464. [Google Scholar] [CrossRef] [PubMed]
- Oshima, Y.; Sano, M.; Kajiwara, I.; Ichimaru, Y.; Itaya, T.; Kuramochi, T.; Hayashi, E.; Kim, J.; Kitajima, O.; Masugi, Y.; et al. Midazolam exhibits antitumour and anti-inflammatory effects in a mouse model of pancreatic ductal adenocarcinoma. Br. J. Anaesth. 2022, 128, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Zheng, Z.; Li, X.; Huang, T.; Rong, D.; Liu, X.; Qin, M.; Wang, Y.; Kong, X.; Song, J.; et al. Midazolam exhibits antitumour and enhances the efficiency of Anti-PD-1 immunotherapy in hepatocellular carcinoma. Cancer Cell Int. 2022, 22, 312. [Google Scholar] [CrossRef]
- Nishina, K.; Akamatsu, H.; Mikawa, K.; Shiga, M.; Maekawa, N.; Obara, H.; Niwa, Y. The inhibitory effects of thiopental, midazolam, and ketamine on human neutrophil functions. Anesth. Analg. 1998, 86, 159–165. [Google Scholar] [CrossRef] [Green Version]
- Mak, N.K.; Szeto, Y.Y.; Fung, M.C.; Leung, K.N.; Kwan, S.K. Effects of midazolam on the differentiation of murine myeloid leukemia cells. Chemotherapy 1997, 43, 272–281. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, G.; Gan, J.; Dou, Z.; Bai, L. Analgesic effect of the midazolam-induced anesthesia in different doses on the patients after the thoracoscopic resection of lung cancer. Saudi J. Biol. Sci. 2019, 26, 2064–2067. [Google Scholar] [CrossRef]
- Roesslein, M.; Schibilsky, D.; Muller, L.; Goebel, U.; Schwer, C.; Humar, M.; Schmidt, R.; Geiger, K.K.; Pahl, H.L.; Pannen, B.H.; et al. Thiopental protects human T lymphocytes from apoptosis in vitro via the expression of heat shock protein 70. J. Pharmacol. Exp. Ther. 2008, 325, 217–225. [Google Scholar] [CrossRef]
- Ichiyama, T.; Nishikawa, M.; Lipton, J.M.; Matsubara, T.; Takashi, H.; Furukawa, S. Thiopental inhibits NF-kappaB activation in human glioma cells and experimental brain inflammation. Brain Res. 2001, 911, 56–61. [Google Scholar] [CrossRef]
- Huang, Y.H.; Sue, S.H.; Wu, Z.S.; Huang, S.M.; Lee, S.Y.; Wu, Z.F. Antitumorigenic Effect of Tramadol and Synergistic Effect with Doxorubicin in Human Breast Cancer Cells. Front. Oncol. 2022, 12, 811716. [Google Scholar] [CrossRef]
- Liu, X.; Yang, J.; Yang, C.; Huang, X.; Han, M.; Kang, F.; Li, J. Morphine promotes the malignant biological behavior of non-small cell lung cancer cells through the MOR/Src/mTOR pathway. Cancer Cell Int. 2021, 21, 622. [Google Scholar] [CrossRef]
- Li, C.; Qin, Y.; Zhong, Y.; Qin, Y.; Wei, Y.; Li, L.; Xie, Y. Fentanyl inhibits the progression of gastric cancer through the suppression of MMP-9 via the PI3K/Akt signaling pathway. Ann. Transl. Med. 2020, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Gong, S.; Ying, L.; Fan, Y.; Sun, Z. Fentanyl Inhibits Lung Cancer Viability and Invasion via Upregulation of miR-331-3p and Repression of HDAC5. OncoTargets Ther. 2020, 13, 13131–13141. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Qin, Y.; Li, L.; Chen, J.; Zhang, X.; Xie, Y. Morphine Can Inhibit the Growth of Breast Cancer MCF-7 Cells by Arresting the Cell Cycle and Inducing Apoptosis. Biol. Pharm. Bull. 2017, 40, 1686–1692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.Y.; Ahn, H.J.; Kim, J.K.; Kim, J.; Lee, S.H.; Chae, H.B. Morphine Suppresses Lung Cancer Cell Proliferation Through the Interaction with Opioid Growth Factor Receptor: An In Vitro and Human Lung Tissue Study. Anesth. Analg. 2016, 123, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Bimonte, S.; Barbieri, A.; Rea, D.; Palma, G.; Luciano, A.; Cuomo, A.; Arra, C.; Izzo, F. Morphine Promotes Tumor Angiogenesis and Increases Breast Cancer Progression. Biomed Res. Int. 2015, 2015, 161508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sekandarzad, M.W.; van Zundert, A.A.J.; Lirk, P.B.; Doornebal, C.W.; Hollmann, M.W. Perioperative Anesthesia Care and Tumor Progression. Anesth. Analg. 2017, 124, 1697–1708. [Google Scholar] [CrossRef]
- Falk, W.; Magnuson, A.; Eintrei, C.; Henningsson, R.; Myrelid, P.; Matthiessen, P.; Gupta, A. Comparison between epidural and intravenous analgesia effects on disease-free survival after colorectal cancer surgery: A randomised multicentre controlled trial. Br. J. Anaesth. 2021, 127, 65–74. [Google Scholar] [CrossRef]
- Sessler, D.I.; Pei, L.; Huang, Y.; Fleischmann, E.; Marhofer, P.; Kurz, A.; Mayers, D.B.; Meyer-Treschan, T.A.; Grady, M.; Tan, E.Y.; et al. Recurrence of breast cancer after regional or general anaesthesia: A randomised controlled trial. Lancet 2019, 394, 1807–1815. [Google Scholar] [CrossRef]
- Connolly, J.G.; Tan, K.S.; Mastrogiacomo, B.; Dycoco, J.; Caso, R.; Jones, G.D.; McCormick, P.J.; Sanchez-Vega, F.; Irie, T.; Scarpa, J.R.; et al. Intraoperative opioid exposure, tumour genomic alterations, and survival differences in people with lung adenocarcinoma. Br. J. Anaesth. 2021, 127, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Cata, J.P.; Keerty, V.; Keerty, D.; Feng, L.; Norman, P.H.; Gottumukkala, V.; Mehran, J.R.; Engle, M. A retrospective analysis of the effect of intraoperative opioid dose on cancer recurrence after non-small cell lung cancer resection. Cancer Med. 2014, 3, 900–908. [Google Scholar] [CrossRef] [PubMed]
- Zylla, D.; Gourley, B.L.; Vang, D.; Jackson, S.; Boatman, S.; Lindgren, B.; Kuskowski, M.A.; Le, C.; Gupta, K.; Gupta, P. Opioid requirement, opioid receptor expression, and clinical outcomes in patients with advanced prostate cancer. Cancer 2013, 119, 4103–4110. [Google Scholar] [CrossRef]
- Ramirez, M.F.; Gorur, A.; Cata, J.P. Opioids and cancer prognosis: A summary of the clinical evidence. Neurosci. Lett. 2021, 746, 135661. [Google Scholar] [CrossRef]
- Sueoka, N.; Sueoka, E.; Okabe, S.; Fujiki, H. Anti-cancer effects of morphine through inhibition of tumour necrosis factor-alpha release and mRNA expression. Carcinogenesis 1996, 17, 2337–2341. [Google Scholar] [CrossRef] [PubMed]
- Sueoka, E.; Sueoka, N.; Kai, Y.; Okabe, S.; Suganuma, M.; Kanematsu, K.; Yamamoto, T.; Fujiki, H. Anticancer activity of morphine and its synthetic derivative, KT-90, mediated through apoptosis and inhibition of NF-kappaB activation. Biochem. Biophys. Res. Commun. 1998, 252, 566–570. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.L.; Buck, D.J.; Saffarian, N.; Stevens, C.W. The opioid antagonist, beta-funaltrexamine, inhibits chemokine expression in human astroglial cells. J. Neuroimmunol. 2007, 186, 141–149. [Google Scholar] [CrossRef] [Green Version]
- Bastami, S.; Norling, C.; Trinks, C.; Holmlund, B.; Walz, T.M.; Ahlner, J.; Uppugunduri, S. Inhibitory effect of opiates on LPS mediated release of TNF and IL-8. Acta Oncol. 2013, 52, 1022–1033. [Google Scholar] [CrossRef]
- Kim, M.H.; Lee, J.R.; Kim, K.J.; Jun, J.H.; Hwang, H.J.; Lee, W.; Nam, S.H.; Oh, J.E.; Yoo, Y.C. Identification for antitumor effects of tramadol in a xenograft mouse model using orthotopic breast cancer cells. Sci. Rep. 2021, 11, 22113. [Google Scholar] [CrossRef]
- Ma, M.; Wang, X.; Liu, N.; Shan, F.; Feng, Y. Low-dose naltrexone inhibits colorectal cancer progression and promotes apoptosis by increasing M1-type macrophages and activating the Bax/Bcl-2/caspase-3/PARP pathway. Int. Immunopharmacol. 2020, 83, 106388. [Google Scholar] [CrossRef]
- Ding, S.; Ma, H.; Wang, G.; Yu, Z.; Li, K.; Huang, A. Effect of Remifentanil Combined Anesthesia on Cytokines and Oxidative Stress in Patients undergoing Laparoscopic Surgery for Colon Cancer. J. Coll. Physicians Surg. Pak. 2019, 29, 8–11. [Google Scholar] [CrossRef]
- Wan, Z.; Chu, C.; Zhou, R.; Que, B. Effects of Oxycodone Combined with Flurbiprofen Axetil on Postoperative Analgesia and Immune Function in Patients Undergoing Radical Resection of Colorectal Cancer. Clin. Pharmacol. Drug Dev. 2021, 10, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiang, Q.; Li, T. Nalbuphine analgesic and anti-inflammatory effects on patients undergoing thoracoscopic lobectomy during the perioperative period. Exp. Ther. Med. 2017, 14, 3117–3121. [Google Scholar] [CrossRef] [Green Version]
- Titon, O.J.; Titon, J.P.; Silva, J.C.D.; Ferreira, M.O.; Garbim, M.R.; Rech, D.; Souza, J.A.; Panis, C. Influence of exogenous opioids on the acute inflammatory response in the perioperative period of oncological surgery: A clinical study. Braz. J. Anesthesiol. 2021; online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya, I.; Nagai, N.; Oyama, K.; Hayashi, H.; Tajima, H.; Kitagawa, H.; Fushida, S.; Fujimura, T.; Ohta, T. Antitumor and anti-metastatic effects of cyclooxygenase-2 inhibition by celecoxib on human colorectal carcinoma xenografts in nude mouse rectum. Oncol. Rep. 2012, 28, 777–784. [Google Scholar] [CrossRef] [Green Version]
- Valle, B.L.; D′Souza, T.; Becker, K.G.; Wood, W.H., 3rd; Zhang, Y.; Wersto, R.P.; Morin, P.J. Non-steroidal anti-inflammatory drugs decrease E2F1 expression and inhibit cell growth in ovarian cancer cells. PLoS ONE 2013, 8, e61836. [Google Scholar] [CrossRef] [Green Version]
- Garcia, M.; Velez, R.; Romagosa, C.; Majem, B.; Pedrola, N.; Olivan, M.; Rigau, M.; Guiu, M.; Gomis, R.R.; Morote, J.; et al. Cyclooxygenase-2 inhibitor suppresses tumour progression of prostate cancer bone metastases in nude mice. BJU Int. 2014, 113, E164–E177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henry, W.S.; Laszewski, T.; Tsang, T.; Beca, F.; Beck, A.H.; McAllister, S.S.; Toker, A. Aspirin Suppresses Growth in PI3K-Mutant Breast Cancer by Activating AMPK and Inhibiting mTORC1 Signaling. Cancer Res. 2017, 77, 790–801. [Google Scholar] [CrossRef] [Green Version]
- Zappavigna, S.; Cossu, A.M.; Grimaldi, A.; Bocchetti, M.; Ferraro, G.A.; Nicoletti, G.F.; Filosa, R.; Caraglia, M. Anti-Inflammatory Drugs as Anticancer Agents. Int. J. Mol. Sci. 2020, 21, 2605. [Google Scholar] [CrossRef] [Green Version]
- Forget, P.; Vandenhende, J.; Berliere, M.; Machiels, J.P.; Nussbaum, B.; Legrand, C.; De Kock, M. Do intraoperative analgesics influence breast cancer recurrence after mastectomy? A retrospective analysis. Anesth. Analg. 2010, 110, 1630–1635. [Google Scholar] [CrossRef] [Green Version]
- Forget, P.; Tombal, B.; Scholtès, J.L.; Nzimbala, J.; Meulders, C.; Legrand, C.; Van Cangh, P.; Cosyns, J.P.; De Kock, M. Do intraoperative analgesics influence oncological outcomes after radical prostatectomy for prostate cancer? Eur. J. Anaesthesiol. 2011, 28, 830–835. [Google Scholar] [CrossRef]
- Choi, J.E.; Villarreal, J.; Lasala, J.; Gottumukkala, V.; Mehran, R.J.; Rice, D.; Yu, J.; Feng, L.; Cata, J.P. Perioperative neutrophil:lymphocyte ratio and postoperative NSAID use as predictors of survival after lung cancer surgery: A retrospective study. Cancer Med. 2015, 4, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Yu, M.Y.; Huang, X.; Zhu, D.; Cheng, S.; Ma, R.; Gu, G. Synergistic effect of celecoxib in tumor necrosis factor-related apoptosis-inducing ligand treatment in osteosarcoma cells. Mol. Med. Rep. 2014, 10, 2198–2202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Todo, M.; Horinaka, M.; Tomosugi, M.; Tanaka, R.; Ikawa, H.; Sowa, Y.; Ishikawa, H.; Fujiwara, H.; Otsuji, E.; Sakai, T. Ibuprofen enhances TRAIL-induced apoptosis through DR5 upregulation. Oncol. Rep. 2013, 30, 2379–2384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sareddy, G.R.; Geeviman, K.; Ramulu, C.; Babu, P.P. The nonsteroidal anti-inflammatory drug celecoxib suppresses the growth and induces apoptosis of human glioblastoma cells via the NF-κB pathway. J. Neurooncol. 2012, 106, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Strohecker, A.; Chen, F.; Kwan, T.; Bosman, J.; Jordan, V.C.; Cryns, V.L. Aspirin sensitizes cancer cells to TRAIL-induced apoptosis by reducing survivin levels. Clin. Cancer Res. 2008, 14, 3168–3176. [Google Scholar] [CrossRef] [Green Version]
- Katerinaki, E.; Haycock, J.W.; Lalla, R.; Carlson, K.E.; Yang, Y.; Hill, R.P.; Lorigan, P.C.; MacNeil, S. Sodium salicylate inhibits TNF-alpha-induced NF-kappaB activation, cell migration, invasion and ICAM-1 expression in human melanoma cells. Melanoma Res. 2006, 16, 11–22. [Google Scholar] [CrossRef]
- Yasui, H.; Adachi, M.; Imai, K. Combination of tumor necrosis factor-alpha with sulindac augments its apoptotic potential and suppresses tumor growth of human carcinoma cells in nude mice. Cancer 2003, 97, 1412–1420. [Google Scholar] [CrossRef]
- Takada, Y.; Bhardwaj, A.; Potdar, P.; Aggarwal, B.B. Nonsteroidal anti-inflammatory agents differ in their ability to suppress NF-kappaB activation, inhibition of expression of cyclooxygenase-2 and cyclin D1, and abrogation of tumor cell proliferation. Oncogene 2004, 23, 9247–9258. [Google Scholar] [CrossRef] [Green Version]
- Redpath, M.; Marques, C.M.; Dibden, C.; Waddon, A.; Lalla, R.; Macneil, S. Ibuprofen and hydrogel-released ibuprofen in the reduction of inflammation-induced migration in melanoma cells. Br. J. Dermatol. 2009, 161, 25–33. [Google Scholar] [CrossRef]
- Setia, S.; Nehru, B.; Sanyal, S.N. Activation of NF-κB: Bridging the gap between inflammation and cancer in colitis-mediated colon carcinogenesis. Biomed. Pharmacother. 2014, 68, 119–128. [Google Scholar] [CrossRef]
- Aboelella, N.S.; Brandle, C.; Okoko, O.; Gazi, M.Y.; Ding, Z.C.; Xu, H.; Gorman, G.; Bollag, R.; Davila, M.L.; Bryan, L.J.; et al. Indomethacin-induced oxidative stress enhances death receptor 5 signaling and sensitizes tumor cells to adoptive T-cell therapy. J. Immunother. Cancer 2022, 10, e004938. [Google Scholar] [CrossRef] [PubMed]
- Vaish, V.; Sanyal, S.N. Chemopreventive effects of NSAIDs on cytokines and transcription factors during the early stages of colorectal cancer. Pharmacol. Rep. 2011, 63, 1210–1221. [Google Scholar] [CrossRef] [PubMed]
- Axiak-Bechtel, S.M.; Mathew, L.M.; Amorim, J.R.; DeClue, A.E. Dogs with osteosarcoma have altered pro- and anti-inflammatory cytokine profiles. Vet. Med. Sci. 2019, 5, 485–493. [Google Scholar] [CrossRef] [Green Version]
- Wen, Y.; Wang, M.; Yang, J.; Wang, Y.; Sun, H.; Zhao, J.; Liu, W.; Zhou, Z.; Deng, H.; Castillo-Pedraza, C.; et al. A Comparison of Fentanyl and Flurbiprofen Axetil on Serum VEGF-C, TNF-α, and IL-1ß Concentrations in Women Undergoing Surgery for Breast Cancer. Pain Pract. 2015, 15, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Pandazi, A.; Kapota, E.; Matsota, P.; Paraskevopoulou, P.; Dervenis, C.; Kostopanagiotou, G. Preincisional versus postincisional administration of parecoxib in colorectal surgery: Effect on postoperative pain control and cytokine response. A randomized clinical trial. World J. Surg. 2010, 34, 2463–2469. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.J.; Li, S.Y.; Cheng, Q.; Chen, W.K.; Wang, S.L.; Ren, Y.; Miao, C.H. Effects of anaesthesia on proliferation, invasion and apoptosis of LoVo colon cancer cells in vitro. Anaesthesia 2016, 71, 147–154. [Google Scholar] [CrossRef]
- Buckley, A.; McQuaid, S.; Johnson, P.; Buggy, D.J. Effect of anaesthetic technique on the natural killer cell anti-tumour activity of serum from women undergoing breast cancer surgery: A pilot study. Br. J. Anaesth. 2014, 113 (Suppl. 1), i56–i62. [Google Scholar] [CrossRef] [Green Version]
- Hirano, Y.; Kaneko, H.; Konishi, T.; Itoh, H.; Matsuda, S.; Kawakubo, H.; Uda, K.; Matsui, H.; Fushimi, K.; Daiko, H.; et al. Short-Term Outcomes of Epidural Analgesia in Minimally Invasive Esophagectomy for Esophageal Cancer: Nationwide Inpatient Data Study in Japan. Ann. Surg. Oncol. 2022, 29, 8225–8234. [Google Scholar] [CrossRef]
- Yen, F.Y.; Chang, W.K.; Lin, S.P.; Lin, T.P.; Chang, K.Y. Association Between Epidural Analgesia and Cancer Recurrence or Survival After Surgery for Renal Cell Carcinoma: A Propensity Weighted Analysis. Front. Med. 2021, 8, 782336. [Google Scholar] [CrossRef]
- Xu, Z.Z.; Li, H.J.; Li, M.H.; Huang, S.M.; Li, X.; Liu, Q.H.; Li, J.; Li, X.Y.; Wang, D.X.; Sessler, D.I. Epidural Anesthesia-Analgesia and Recurrence-free Survival after Lung Cancer Surgery: A Randomized Trial. Anesthesiology 2021, 135, 419–432. [Google Scholar] [CrossRef]
- Karmakar, M.K.; Samy, W.; Lee, A.; Li, J.W.; Chan, W.C.; Chen, P.P.; Tsui, B.C.H. Survival Analysis of Patients with Breast Cancer Undergoing a Modified Radical Mastectomy with or Without a Thoracic Paravertebral Block: A 5-Year Follow-up of a Randomized Controlled Trial. Anticancer Res. 2017, 37, 5813–5820. [Google Scholar] [CrossRef] [PubMed]
- Myles, P.S.; Peyton, P.; Silbert, B.; Hunt, J.; Rigg, J.R.; Sessler, D.I. Perioperative epidural analgesia for major abdominal surgery for cancer and recurrence-free survival: Randomised trial. Bmj 2011, 342, d1491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grandhi, R.K.; Lee, S.; Abd-Elsayed, A. The Relationship Between Regional Anesthesia and Cancer: A Metaanalysis. Ochsner J. 2017, 17, 345–361. [Google Scholar]
- Cakmakkaya, O.S.; Kolodzie, K.; Apfel, C.C.; Pace, N.L. Anaesthetic techniques for risk of malignant tumour recurrence. Cochrane Database Syst Rev. 2014, 11, Cd008877. [Google Scholar] [CrossRef] [PubMed]
- Inoue, G.N.C.; Pimenta, R.; Camargo, J.A.; Viana, N.I.; Guimarães, V.R.; Srougi, M.; Nahas, W.C.; Leite, K.R.M.; Reis, S.T. Combined spinal and general anesthesia attenuate tumor promoting effects of surgery. An experimental animal study. Ann. Med. Surg. 2022, 75, 103398. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Zheng, X.; Gu, Y.; Fu, W.; Tang, C.; Yao, Y. Effect of general anesthesia with thoracic paravertebral block on postoperative delirium in elderly patients undergoing thoracoscopic lobectomy: A randomized-controlled trial. BMC Anesthesiol. 2022, 22, 1. [Google Scholar] [CrossRef] [PubMed]
- Fiorelli, A.; Izzo, A.C.; Frongillo, E.M.; Del Prete, A.; Liguori, G.; Di Costanzo, E.; Vicidomini, G.; Santini, M. Efficacy of wound analgesia for controlling post-thoracotomy pain: A randomized double-blind study. Eur. J. Cardiothorac. Surg. 2016, 49, 339–347. [Google Scholar] [CrossRef] [Green Version]
- Zhou, M.; Liu, W.; Peng, J.; Wang, Y. Impact of propofol epidural anesthesia on immune function and inflammatory factors in patients undergoing gastric cancer surgery. Am. J. Transl. Res. 2021, 13, 3064–3073. [Google Scholar] [PubMed]
- Zhao, J.; Mo, H. The Impact of Different Anesthesia Methods on Stress Reaction and Immune Function of the Patients with Gastric Cancer during Peri-Operative Period. J. Med. Assoc. Thail. 2015, 98, 568–573. [Google Scholar]
- Su, Y.; Pu, Y.; Zhao, Z.; Yang, X. Influence of combined epidural anesthesia on cognitive function, inflammation and stress response in elderly liver cancer patients undergoing surgery. Oncol. Lett. 2020, 19, 2733–2738. [Google Scholar] [CrossRef] [Green Version]
- Geng, J.; Wang, J.; Zhang, Y.; Song, W.; Zhu, J.; Chen, J.; Wu, Z. The Effect of a Combined Modified Pectoral and Stellate Ganglion Block on Stress and Inflammatory Response in Patients Undergoing Modified Radical Mastectomy. Int. J. Breast Cancer 2022, 2022, 3359130. [Google Scholar] [CrossRef] [PubMed]
- Okuda, J.; Suzuki, T.; Wakaizumi, K.; Kato, J.; Yamada, T.; Morisaki, H. Effects of Thoracic Epidural Anesthesia on Systemic and Local Inflammatory Responses in Patients Undergoing Lung Cancer Surgery: A Randomized Controlled Trial. J. Cardiothorac. Vasc. Anesth. 2022, 36, 1380–1386. [Google Scholar] [CrossRef] [PubMed]
- Cong, X.; Huang, Z.; Zhang, L.; Sun, M.; Chang, E.; Zhang, W.; Zhang, J. Effect of different anaesthesia methods on perioperative cellular immune function and long-term outcome in patients undergoing radical resection of esophageal cancer: A prospective cohort study. Am. J. Transl. Res. 2021, 13, 11427–11438. [Google Scholar] [PubMed]
- Karadeniz, M.S.; Mammadov, O.; Çiftci, H.; Usta, S.A.; Pembeci, K. Comparing the Effects of Combined General/Epidural Anaesthesia and General Anaesthesia on Serum Cytokine Levels in Radical Cystectomy. Turk. J. Anaesthesiol. Reanim. 2017, 45, 203–209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fant, F.; Tina, E.; Sandblom, D.; Andersson, S.O.; Magnuson, A.; Hultgren-Hörnkvist, E.; Axelsson, K.; Gupta, A. Thoracic epidural analgesia inhibits the neuro-hormonal but not the acute inflammatory stress response after radical retropubic prostatectomy. Br. J. Anaesth. 2013, 110, 747–757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siekmann, W.; Eintrei, C.; Magnuson, A.; Sjölander, A.; Matthiessen, P.; Myrelid, P.; Gupta, A. Surgical and not analgesic technique affects postoperative inflammation following colorectal cancer surgery: A prospective, randomized study. Color. Dis. 2017, 19, O186–O195. [Google Scholar] [CrossRef] [PubMed]
- Rauch, S.; Miller, C.; Bräuer, A.; Wallner, B.; Bock, M.; Paal, P. Perioperative Hypothermia—A Narrative Review. Int. J. Environ. Res. Public Health 2021, 18, 8749. [Google Scholar] [CrossRef] [PubMed]
- Snyder, G.L.; Greenberg, S. Effect of anaesthetic technique and other perioperative factors on cancer recurrence. Br. J. Anaesth. 2010, 105, 106–115. [Google Scholar] [CrossRef] [Green Version]
- Kurosawa, S. Anesthesia in patients with cancer disorders. Curr. Opin. Anaesthesiol. 2012, 25, 376–384. [Google Scholar] [CrossRef]
- Horowitz, M.; Neeman, E.; Sharon, E.; Ben-Eliyahu, S. Exploiting the critical perioperative period to improve long-term cancer outcomes. Nat. Rev. Clin. Oncol. 2015, 12, 213–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, G.; Zhao, B.; Zhang, Y.; Sun, T.; Liu, W.; Li, J.; Liu, Y.; Wang, Y.; Li, H.; Hou, X. Hypothermia activates adipose tissue to promote malignant lung cancer progression. PLoS ONE 2013, 8, e72044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, Q.; Tang, X.; Zhao, W.; Qiu, Y.; He, J.; Wan, D.; Li, J.; Wang, X.; He, X.; Liu, Y.; et al. Mild hyperthermia promotes immune checkpoint blockade-based immunotherapy against metastatic pancreatic cancer using size-adjustable nanoparticles. Acta Biomater. 2021, 133, 244–256. [Google Scholar] [CrossRef] [PubMed]
- Sulyok, I.; Fleischmann, E.; Stift, A.; Roth, G.; Lebherz-Eichinger, D.; Kasper, D.; Spittler, A.; Kimberger, O. Effect of preoperative fever-range whole-body hyperthermia on immunological markers in patients undergoing colorectal cancer surgery. Br. J. Anaesth. 2012, 109, 754–761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atanackovic, D.; Pollok, K.; Faltz, C.; Boeters, I.; Jung, R.; Nierhaus, A.; Braumann, K.M.; Hossfeld, D.K.; Hegewisch-Becker, S. Patients with solid tumors treated with high-temperature whole body hyperthermia show a redistribution of naive/memory T-cell subtypes. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R585–R594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coccolini, F.; Corbella, D.; Finazzi, P.; Brambillasca, P.; Benigni, A.; Prussiani, V.; Ceresoli, M.; Manfredi, R.; Poiasina, E.; Bertoli, P.; et al. Time course of cytokines, hemodynamic and metabolic parameters during hyperthermic intraperitoneal chemotherapy. Minerva Anestesiol. 2016, 82, 310–319. [Google Scholar]
- Oderda, M.; Cerutti, E.; Gontero, P.; Manetta, T.; Mengozzi, G.; Meyer, N.; Munegato, S.; Noll, E.; Rampa, P.; Piéchaud, T.; et al. The impact of warmed and humidified CO(2) insufflation during robotic radical prostatectomy: Results of a randomized controlled trial. Urologia 2019, 86, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Hansen, D.; Bogatzki, S.; Syben, R.; Bechrakis, N.E.; Dopjans, D.; Spies, C.; Welte, M.; Schaffartzik, W. Effect of profound normovolemic hypotension and moderate hypothermia on circulating cytokines and adhesion molecules. Shock 1999, 12, 335–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, S.C.; Yang, W.V. Hyperglycemia, tumorigenesis, and chronic inflammation. Crit. Rev. Oncol. Hematol. 2016, 108, 146–153. [Google Scholar] [CrossRef]
- Duan, W.; Shen, X.; Lei, J.; Xu, Q.; Yu, Y.; Li, R.; Wu, E.; Ma, Q. Hyperglycemia, a neglected factor during cancer progression. Biomed Res. Int. 2014, 2014, 461917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otto, L.; Rahn, S.; Daunke, T.; Walter, F.; Winter, E.; Möller, J.L.; Rose-John, S.; Wesch, D.; Schäfer, H.; Sebens, S. Initiation of Pancreatic Cancer: The Interplay of Hyperglycemia and Macrophages Promotes the Acquisition of Malignancy-Associated Properties in Pancreatic Ductal Epithelial Cells. Int. J. Mol. Sci. 2021, 22, 5086. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.C.; Liu, Q.R.; Huang, Y. Effect of intraoperative glucose fluctuation and postoperative IL-6, TNF-α, CRP levels on the short-term prognosis of patients with intracranial supratentorial neoplasms. Asian Pac. J. Cancer Prev. 2014, 15, 10879–10882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lachmann, G.; von Haefen, C.; Wollersheim, T.; Spies, C. Severe perioperative hyperglycemia attenuates postoperative monocytic function, basophil count and T cell activation. Minerva Anestesiol. 2017, 83, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Amato, A.; Pescatori, M. Perioperative blood transfusions for the recurrence of colorectal cancer. Cochrane Database Syst Rev. 2006, 2006, Cd005033. [Google Scholar] [CrossRef] [PubMed]
- Cata, J.P.; Lasala, J.; Pratt, G.; Feng, L.; Shah, J.B. Association between Perioperative Blood Transfusions and Clinical Outcomes in Patients Undergoing Bladder Cancer Surgery: A Systematic Review and Meta-Analysis Study. J. Blood Transfus. 2016, 2016, 9876394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.L.; Ye, Y.; Yuan, X.H. Association between Allogeneic or Autologous Blood Transfusion and Survival in Patients after Radical Prostatectomy: A Systematic Review and Meta-Analysis. PLoS ONE 2017, 12, e0171081. [Google Scholar] [CrossRef]
- Agnes, A.; Lirosi, M.C.; Panunzi, S.; Santocchi, P.; Persiani, R.; D′Ugo, D. The prognostic role of perioperative allogeneic blood transfusions in gastric cancer patients undergoing curative resection: A systematic review and meta-analysis of non-randomized, adjusted studies. Eur. J. Surg. Oncol. 2018, 44, 404–419. [Google Scholar] [CrossRef] [PubMed]
- Milasiene, V.; Stratilatovas, E.; Characiejus, D.; Kazbariene, B.; Norkiene, V. TGF-beta1 and TNF-alpha after red blood cell transfusion in colorectal cancer patients. Exp. Oncol. 2007, 29, 67–70. [Google Scholar]
- Surinenaite, B.; Prasmickiene, G.; Milasiene, V.; Stratilatovas, E.; Didziapetriene, J. The influence of surgical treatment and red blood cell transfusion on changes in antioxidative and immune system parameters in colorectal cancer patients. Medicina 2009, 45, 785–791. [Google Scholar] [CrossRef] [Green Version]
- Xing, Y.L.; Wang, Y.C. Influence of autologous and homologous blood transfusion on interleukins and tumor necrosis factor-α in peri-operative patients with esophageal cancer. Asian Pac. J. Cancer Prev. 2014, 15, 7831–7834. [Google Scholar] [CrossRef] [Green Version]
- Heiss, M.M.; Fraunberger, P.; Delanoff, C.; Stets, R.; Allgayer, H.; Ströhlein, M.A.; Tarabichi, A.; Faist, E.; Jauch, K.W.; Schildberg, F.W. Modulation of immune response by blood transfusion: Evidence for a differential effect of allogeneic and autologous blood in colorectal cancer surgery. Shock 1997, 8, 402–408. [Google Scholar] [CrossRef] [Green Version]
- Geng, Y.; Liu, L. Impact of Allogeneic Leukocyte-Depleted Red Blood Cell Transfusion on Inflammatory Response and Blood Coagulation in Patients with Recurrence of Colon Cancer after Operation. Evid. Based Complement. Alternat. Med. 2021, 2021, 6957569. [Google Scholar] [CrossRef] [PubMed]
- Benson, D.D.; Beck, A.W.; Burdine, M.S.; Brekken, R.; Silliman, C.C.; Barnett, C.C., Jr. Accumulation of pro-cancer cytokines in the plasma fraction of stored packed red cells. J. Gastrointest. Surg. 2012, 16, 460–468. [Google Scholar] [CrossRef] [Green Version]
- Beck-Schimmer, B.; Romero, B.; Booy, C.; Joch, H.; Hallers, U.; Pasch, T.; Spahn, D.R. Release of inflammatory mediators in irradiated cell salvage blood and their biological consequences in human beings following transfusion. Eur. J. Anaesthesiol. 2004, 21, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wu, H.; Zhang, J.; Qi, Q.; Zhang, W.; Xia, R. Preoperative Immune Response is Associated with Perioperative Transfusion Requirements in Glioma Surgery. J. Cancer 2019, 10, 3526–3532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Lin, Y. Tumor necrosis factor and cancer, buddies or foes? Acta Pharm. Sin. 2008, 29, 1275–1288. [Google Scholar] [CrossRef] [Green Version]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tseng, W.-C.; Lai, H.-C.; Huang, Y.-H.; Chan, S.-M.; Wu, Z.-F. Tumor Necrosis Factor Alpha: Implications of Anesthesia on Cancers. Cancers 2023, 15, 739. https://doi.org/10.3390/cancers15030739
Tseng W-C, Lai H-C, Huang Y-H, Chan S-M, Wu Z-F. Tumor Necrosis Factor Alpha: Implications of Anesthesia on Cancers. Cancers. 2023; 15(3):739. https://doi.org/10.3390/cancers15030739
Chicago/Turabian StyleTseng, Wei-Cheng, Hou-Chuan Lai, Yi-Hsuan Huang, Shun-Ming Chan, and Zhi-Fu Wu. 2023. "Tumor Necrosis Factor Alpha: Implications of Anesthesia on Cancers" Cancers 15, no. 3: 739. https://doi.org/10.3390/cancers15030739
APA StyleTseng, W.-C., Lai, H.-C., Huang, Y.-H., Chan, S.-M., & Wu, Z.-F. (2023). Tumor Necrosis Factor Alpha: Implications of Anesthesia on Cancers. Cancers, 15(3), 739. https://doi.org/10.3390/cancers15030739






