Multi-Parametric Radiomic Model to Predict 1p/19q Co-Deletion in Patients with IDH-1 Mutant Glioma: Added Value to the T2-FLAIR Mismatch Sign
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
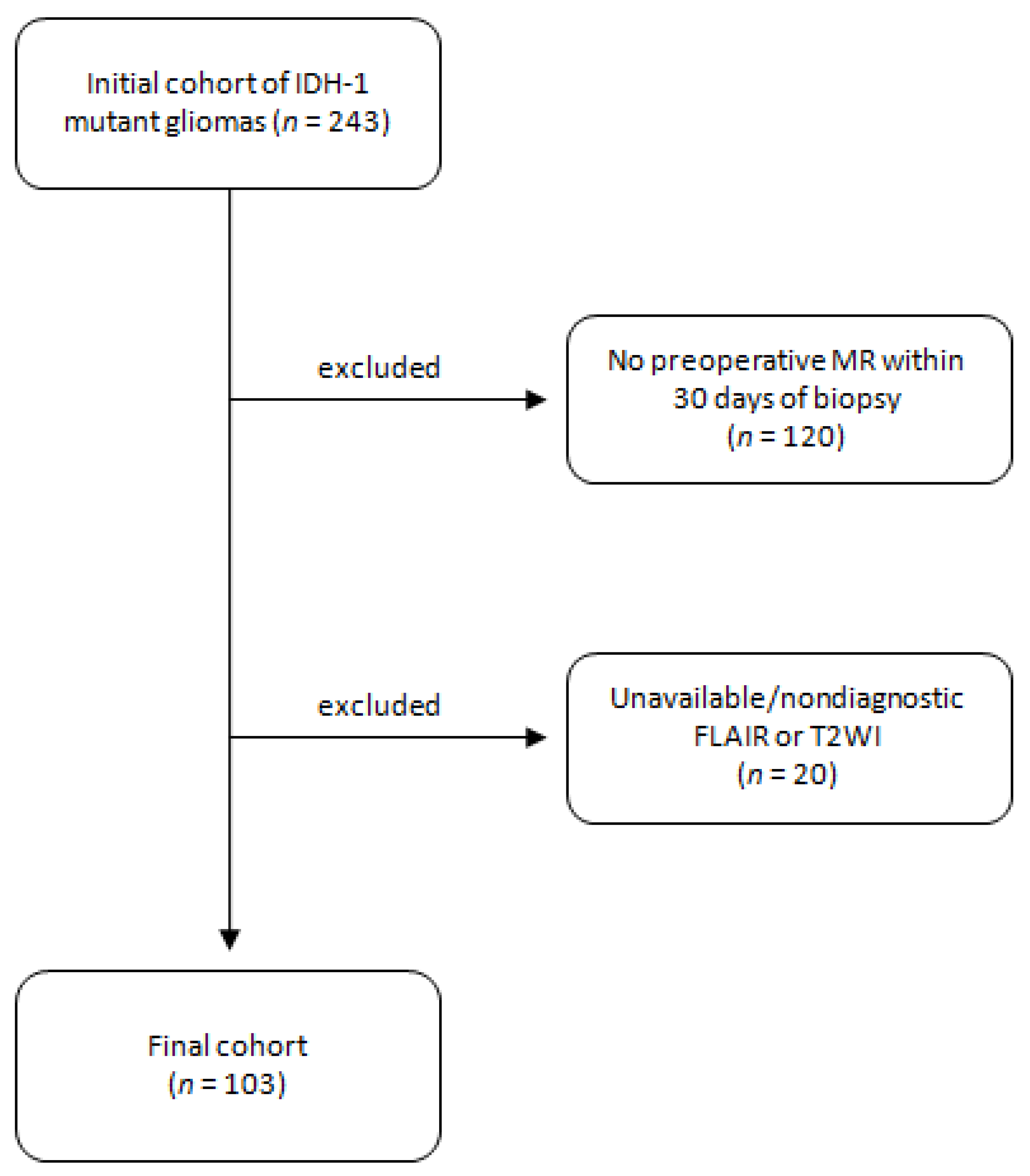
2.1. Patient Selection
2.2. Histopathologic Data
2.3. Image Analysis
2.4. Texture Analysis
2.5. Statistical Analysis
3. Results
3.1. Clinical Characteristics of Patient Population
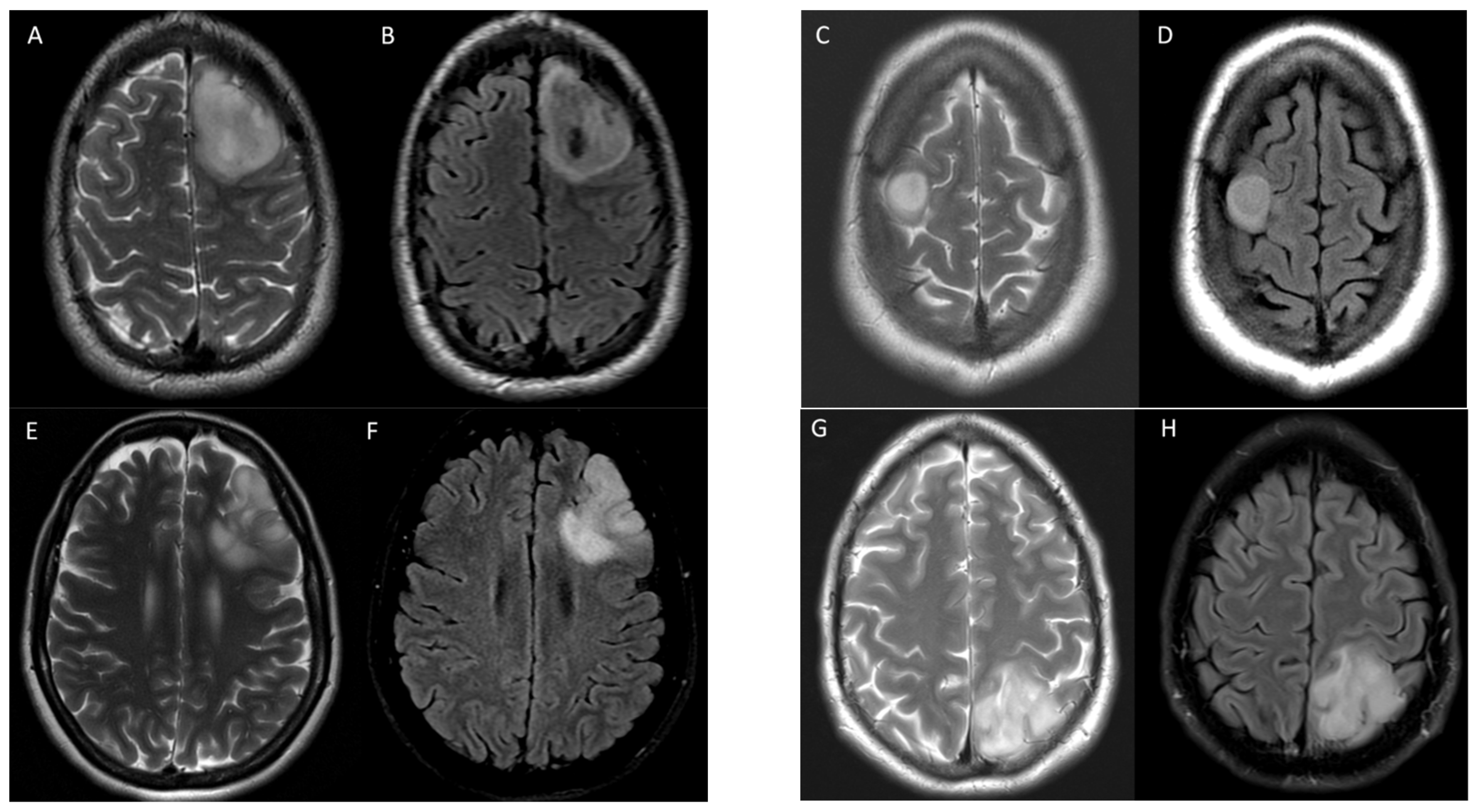
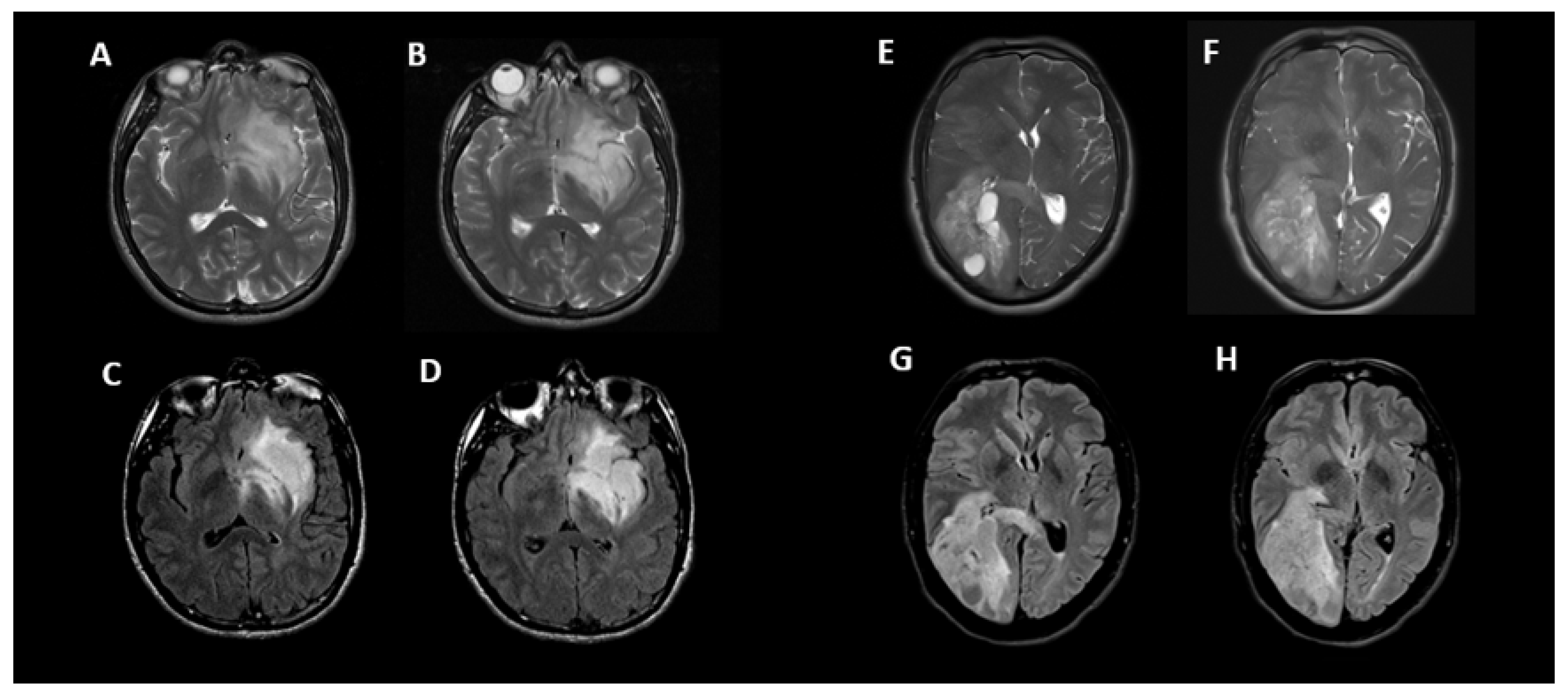
3.2. T2-FLAIR-Mismatch-Based Prediction of 1p/19q Co-Deletion Status
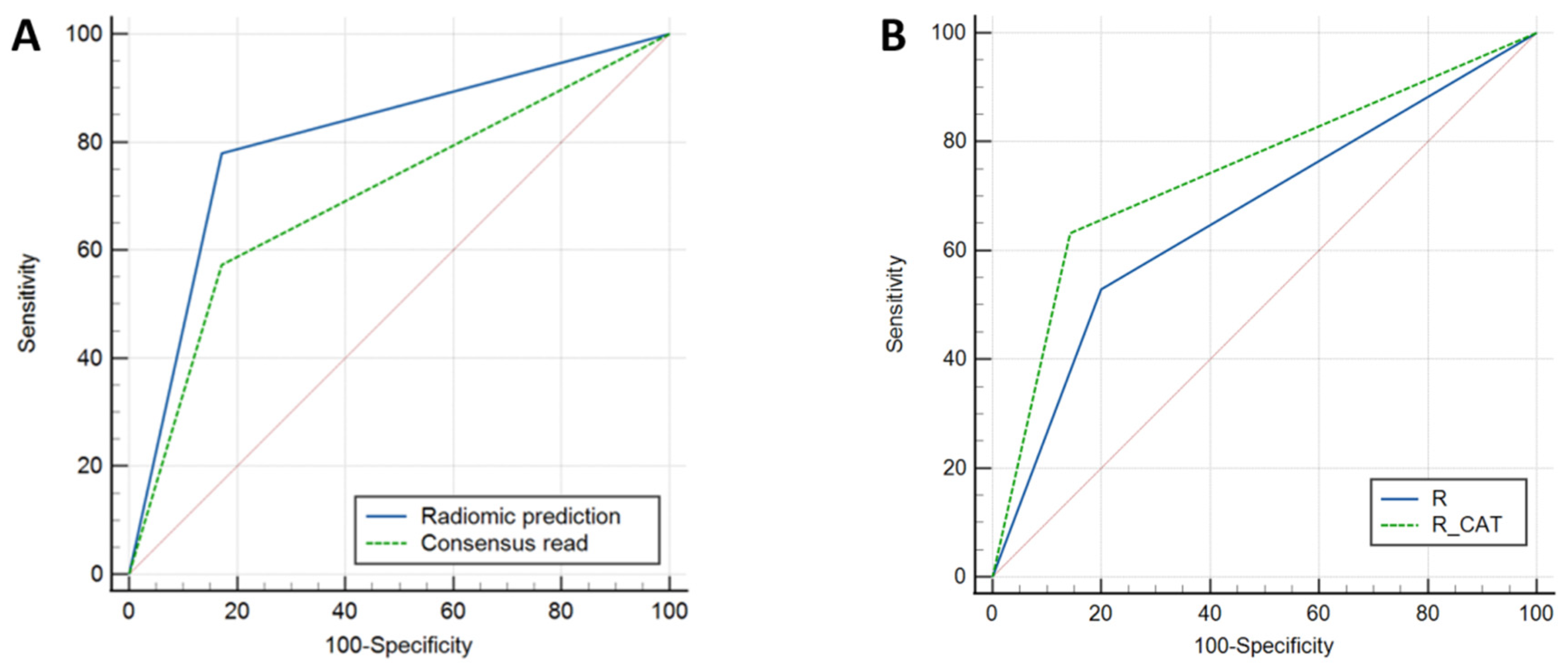
3.3. Radiomic-Based Prediction of 1p/19q Co-Deletion Status
3.4. External Validation Results
3.5. Neuroradiologist + Radiomics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A Summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Bigner, D.D.; Velculescu, V.; Parsons, D.W. Mutant Metabolic Enzymes Are at the Origin of GliomasIDH Mutations in Gliomas. Cancer Res. 2009, 69, 9157–9159. [Google Scholar] [CrossRef] [PubMed]
- Wen, P.Y.; Packer, R.J. The 2021 WHO Classification of Tumors of the Central Nervous System: Clinical Implications. Neuro Oncol. 2021, 23, 1215–1217. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A Summary. Neuro Oncol. 2021, 23, 1231–1251. [Google Scholar] [CrossRef]
- Cairncross, G.; Wang, M.; Shaw, E.; Jenkins, R.; Brachman, D.; Buckner, J.; Fink, K.; Souhami, L.; Laperriere, N.; Curran, W.; et al. Phase III Trial of Chemoradiotherapy for Anaplastic Oligodendroglioma: Long-Term Results of RTOG 9402. J. Clin. Oncol. 2013, 31, 337–343. [Google Scholar] [CrossRef]
- van den Bent, M.J.; Carpentier, A.F. Adjuvant Procarbazine, Lomustine, and Vincristine Improves Progression-Free Survival but Not Overall Survival in Newly Diagnosed Anaplastic Oligodendrogliomas and Oligoastrocytomas: A Randomized European Organisation for Research and Treatment of Cancer Phase III Trial. J. Clin. Oncol. 2006, 24, 2715–2722. [Google Scholar]
- Smits, M.; van den Bent, M.J. Imaging Correlates of Adult Glioma Genotypes. Radiology 2017, 284, 316–331. [Google Scholar] [CrossRef]
- Patel, S.H.; Poisson, L.M.; Brat, D.J.; Zhou, Y.; Cooper, L.; Snuderl, M.; Thomas, C.; Franceschi, A.M.; Griffith, B.; Flanders, A.E.; et al. T2–FLAIR Mismatch, an Imaging Biomarker for IDH and 1p/19q Status in Lower-Grade Gliomas: A TCGA/TCIA Project. Clin. Cancer Res. 2017, 23, 6078–6085. [Google Scholar] [CrossRef]
- Jain, R.; Johnson, D.R.; Patel, S.H.; Castillo, M.; Smits, M.; van den Bent, M.J.; Chi, A.S.; Cahill, D.P. “Real World” Use of a Highly Reliable Imaging Sign: “T2-FLAIR Mismatch” for Identification of IDH Mutant Astrocytomas. Neuro Oncol. 2020, 22, 936–943. [Google Scholar] [CrossRef]
- Corell, A.; Ferreyra Vega, S.; Hoefling, N.; Carstam, L.; Smits, A.; Olsson Bontell, T.; Björkman-Burtscher, I.M.; Carén, H.; Jakola, A.S. The Clinical Significance of the T2-FLAIR Mismatch Sign in Grade II and III Gliomas: A Population-Based Study. BMC Cancer 2020, 20, 450. [Google Scholar] [CrossRef]
- Lasocki, A.; Gaillard, F.; Gorelik, A.; Gonzales, M. MRI Features Can Predict 1p/19q Status in Intracranial Gliomas. AJNR Am. J. Neuroradiol. 2018, 39, 687–692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foltyn, M.; Nieto Taborda, K.N.; Neuberger, U.; Brugnara, G.; Reinhardt, A.; Stichel, D.; Heiland, S.; Herold-Mende, C.; Unterberg, A.; Debus, J.; et al. T2/FLAIR-Mismatch Sign for Noninvasive Detection of IDH-Mutant 1p/19q Non-Codeleted Gliomas: Validity and Pathophysiology. Neurooncol. Adv. 2020, 2, vdaa004. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Muragaki, Y.; Maruyama, T.; Komori, T.; Tamura, M.; Nitta, M.; Tsuzuki, S.; Kawamata, T. Calcification on CT Is a Simple and Valuable Preoperative Indicator of 1p/19q Loss of Heterozygosity in Supratentorial Brain Tumors That Are Suspected Grade II and III Gliomas. Brain Tumor Pathol. 2016, 33, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, M.A.; Lumenta, C.B. Oligodendrogliomas in the CT/MR-Era. Acta Neurochir. 2001, 143, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Vallières, M.; Bai, H.X.; Su, C.; Tang, H.; Oldridge, D.; Zhang, Z.; Xiao, B.; Liao, W.; Tao, Y.; et al. MRI Features Predict Survival and Molecular Markers in Diffuse Lower-Grade Gliomas. Neuro Oncol. 2017, 19, 862–870. [Google Scholar] [CrossRef]
- Fan, Z.; Sun, Z.; Fang, S.; Li, Y.; Liu, X.; Liang, Y.; Liu, Y.; Zhou, C.; Zhu, Q.; Zhang, H.; et al. Preoperative Radiomics Analysis of 1p/19q Status in WHO Grade II Gliomas. Front. Oncol. 2021, 11, 616740. [Google Scholar] [CrossRef] [PubMed]
- Shofty, B.; Artzi, M.; Ben Bashat, D.; Liberman, G.; Haim, O.; Kashanian, A.; Bokstein, F.; Blumenthal, D.T.; Ram, Z.; Shahar, T. MRI Radiomics Analysis of Molecular Alterations in Low-Grade Gliomas. Int. J. Comput. Assist. Radiol. Surg. 2018, 13, 563–571. [Google Scholar] [CrossRef]
- Ren, X.; Cui, X.; Lin, S.; Wang, J.; Jiang, Z.; Sui, D.; Li, J.; Wang, Z. Co-Deletion of Chromosome 1p/19q and IDH1/2 Mutation in Glioma Subsets of Brain Tumors in Chinese Patients. PLoS ONE 2012, 7, e32764. [Google Scholar] [CrossRef]
- Kocak, B.; Durmaz, E.S.; Ates, E.; Sel, I.; Turgut Gunes, S.; Kaya, O.K.; Zeynalova, A.; Kilickesmez, O. Radiogenomics of Lower-Grade Gliomas: Machine Learning-Based MRI Texture Analysis for Predicting 1p/19q Codeletion Status. Eur. Radiol. 2020, 30, 877–886. [Google Scholar] [CrossRef]
- Han, Y.; Xie, Z.; Zang, Y.; Zhang, S.; Gu, D.; Zhou, M.; Gevaert, O.; Wei, J.; Li, C.; Chen, H.; et al. Non-Invasive Genotype Prediction of Chromosome 1p/19q Co-Deletion by Development and Validation of an MRI-Based Radiomics Signature in Lower-Grade Gliomas. J. Neurooncol. 2018, 140, 297–306. [Google Scholar] [CrossRef]
- Batchala, P.P.; Muttikkal, T.J.E.; Donahue, J.H.; Patrie, J.T.; Schiff, D.; Fadul, C.E.; Mrachek, E.K.; Lopes, M.-B.; Jain, R.; Patel, S.H. Neuroimaging-Based Classification Algorithm for Predicting 1p/19q-Codeletion Status in IDH-Mutant Lower Grade Gliomas. AJNR Am. J. Neuroradiol. 2019, 40, 426–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, S.H.; Batchala, P.P.; Mrachek, E.K.S.; Lopes, M.-B.S.; Schiff, D.; Fadul, C.E.; Patrie, J.T.; Jain, R.; Druzgal, T.J.; Williams, E.S. MRI and CT Identify Isocitrate Dehydrogenase (IDH)-Mutant Lower-Grade Gliomas Misclassified to 1p/19q Codeletion Status with Fluorescence in Situ Hybridization. Radiology 2020, 294, 160–167. [Google Scholar] [CrossRef]
- Ellingson, B.M.; Bendszus, M.; Boxerman, J.; Barboriak, D.; Erickson, B.J.; Smits, M.; Nelson, S.J.; Gerstner, E.; Alexander, B.; Goldmacher, G.; et al. Consensus Recommendations for a Standardized Brain Tumor Imaging Protocol in Clinical Trials. Neuro. Oncol. 2015, 17, 1188–1198. [Google Scholar] [PubMed]
- Chicklore, S.; Goh, V.; Siddique, M.; Roy, A.; Marsden, P.K.; Cook, G.J.R. Quantifying Tumour Heterogeneity in 18F-FDG PET/CT Imaging by Texture Analysis. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 133–140. [Google Scholar] [CrossRef]
- Kickingereder, P.; Burth, S.; Wick, A.; Götz, M.; Eidel, O.; Schlemmer, H.-P.; Maier-Hein, K.H.; Wick, W.; Bendszus, M.; Radbruch, A.; et al. Radiomic Profiling of Glioblastoma: Identifying an Imaging Predictor of Patient Survival with Improved Performance over Established Clinical and Radiologic Risk Models. Radiology 2016, 280, 880–889. [Google Scholar] [CrossRef]
- Sakai, Y.; Yang, C.; Kihira, S.; Tsankova, N.; Khan, F.; Hormigo, A.; Lai, A.; Cloughesy, T.; Nael, K. MRI Radiomic Features to Predict IDH1 Mutation Status in Gliomas: A Machine Learning Approach Using Gradient Tree Boosting. Int. J. Mol. Sci. 2020, 21, 8004. [Google Scholar] [CrossRef]
- Chen, T.; Guestrin, C. XGBoost: A Scalable Tree Boosting System. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, New York, NY, USA, 13 August 2016; Association for Computing Machinery: New York, NY, USA, 2016; pp. 785–794. [Google Scholar]
- Lasocki, A.; Rosenthal, M.A.; Roberts-Thomson, S.J.; Neal, A.; Drummond, K.J. Neuro-Oncology and Radiogenomics: Time to Integrate? AJNR Am. J. Neuroradiol. 2020, 41, 1982–1988. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Polson, J.; Nael, K.; Salamon, N.; Yoo, B.; Speier, W.; Arnold, C. A Machine Learning Approach to Predict Acute Ischemic Stroke Thrombectomy Reperfusion Using Discriminative MR Image Features. In Proceedings of the 2021 IEEE EMBS International Conference on Biomedical and Health Informatics (BHI), Athens, Greece, 27–30 July 2021. [Google Scholar] [CrossRef]
- Polson, J.S.; Zhang, H.; Nael, K.; Salamon, N.; Yoo, B.Y.; El-Saden, S.; Starkman, S.; Kim, N.; Kang, D.-W.; Speier, W.F., 4th; et al. Identifying Acute Ischemic Stroke Patients within the Thrombolytic Treatment Window Using Deep Learning. J. Neuroimaging 2022, 32, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Pasquini, L.; Napolitano, A.; Lucignani, M.; Tagliente, E.; Dellepiane, F.; Rossi-Espagnet, M.C.; Ritrovato, M.; Vidiri, A.; Villani, V.; Ranazzi, G.; et al. AI and High-Grade Glioma for Diagnosis and Outcome Prediction: Do All Machine Learning Models Perform Equally Well? Front. Oncol. 2021, 11, 601425. [Google Scholar] [CrossRef]
- Parmar, C.; Grossmann, P.; Bussink, J.; Lambin, P.; Aerts, H.J.W.L. Machine Learning Methods for Quantitative Radiomic Biomarkers. Sci. Rep. 2015, 5, 13087. [Google Scholar] [CrossRef] [Green Version]
| 1p19q Co-Deleted (n = 35) | 1p19q Non-Co-Deleted (n = 68) | p Value | |
|---|---|---|---|
| Age (mean/SD) | 43/13 | 40/13 | 0.91 |
| Sex (M/F) | 12/23 | 23/45 | 0.93 |
| Location (F/P/T/O/C) * | 23/7/4/0/1 | 28/11/18/0/4 | 0.81 |
| T2-FLAIR mismatch (Y/N) | 6/29 | 39/29 | <0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kihira, S.; Derakhshani, A.; Leung, M.; Mahmoudi, K.; Bauer, A.; Zhang, H.; Polson, J.; Arnold, C.; Tsankova, N.M.; Hormigo, A.; et al. Multi-Parametric Radiomic Model to Predict 1p/19q Co-Deletion in Patients with IDH-1 Mutant Glioma: Added Value to the T2-FLAIR Mismatch Sign. Cancers 2023, 15, 1037. https://doi.org/10.3390/cancers15041037
Kihira S, Derakhshani A, Leung M, Mahmoudi K, Bauer A, Zhang H, Polson J, Arnold C, Tsankova NM, Hormigo A, et al. Multi-Parametric Radiomic Model to Predict 1p/19q Co-Deletion in Patients with IDH-1 Mutant Glioma: Added Value to the T2-FLAIR Mismatch Sign. Cancers. 2023; 15(4):1037. https://doi.org/10.3390/cancers15041037
Chicago/Turabian StyleKihira, Shingo, Ahrya Derakhshani, Michael Leung, Keon Mahmoudi, Adam Bauer, Haoyue Zhang, Jennifer Polson, Corey Arnold, Nadejda M. Tsankova, Adilia Hormigo, and et al. 2023. "Multi-Parametric Radiomic Model to Predict 1p/19q Co-Deletion in Patients with IDH-1 Mutant Glioma: Added Value to the T2-FLAIR Mismatch Sign" Cancers 15, no. 4: 1037. https://doi.org/10.3390/cancers15041037
APA StyleKihira, S., Derakhshani, A., Leung, M., Mahmoudi, K., Bauer, A., Zhang, H., Polson, J., Arnold, C., Tsankova, N. M., Hormigo, A., Salehi, B., Pham, N., Ellingson, B. M., Cloughesy, T. F., & Nael, K. (2023). Multi-Parametric Radiomic Model to Predict 1p/19q Co-Deletion in Patients with IDH-1 Mutant Glioma: Added Value to the T2-FLAIR Mismatch Sign. Cancers, 15(4), 1037. https://doi.org/10.3390/cancers15041037







