Cancer Stem Cells in Sarcomas: In Vitro Isolation and Role as Prognostic Markers: A Systematic Review
Abstract
:Simple Summary
Abstract
1. Introduction
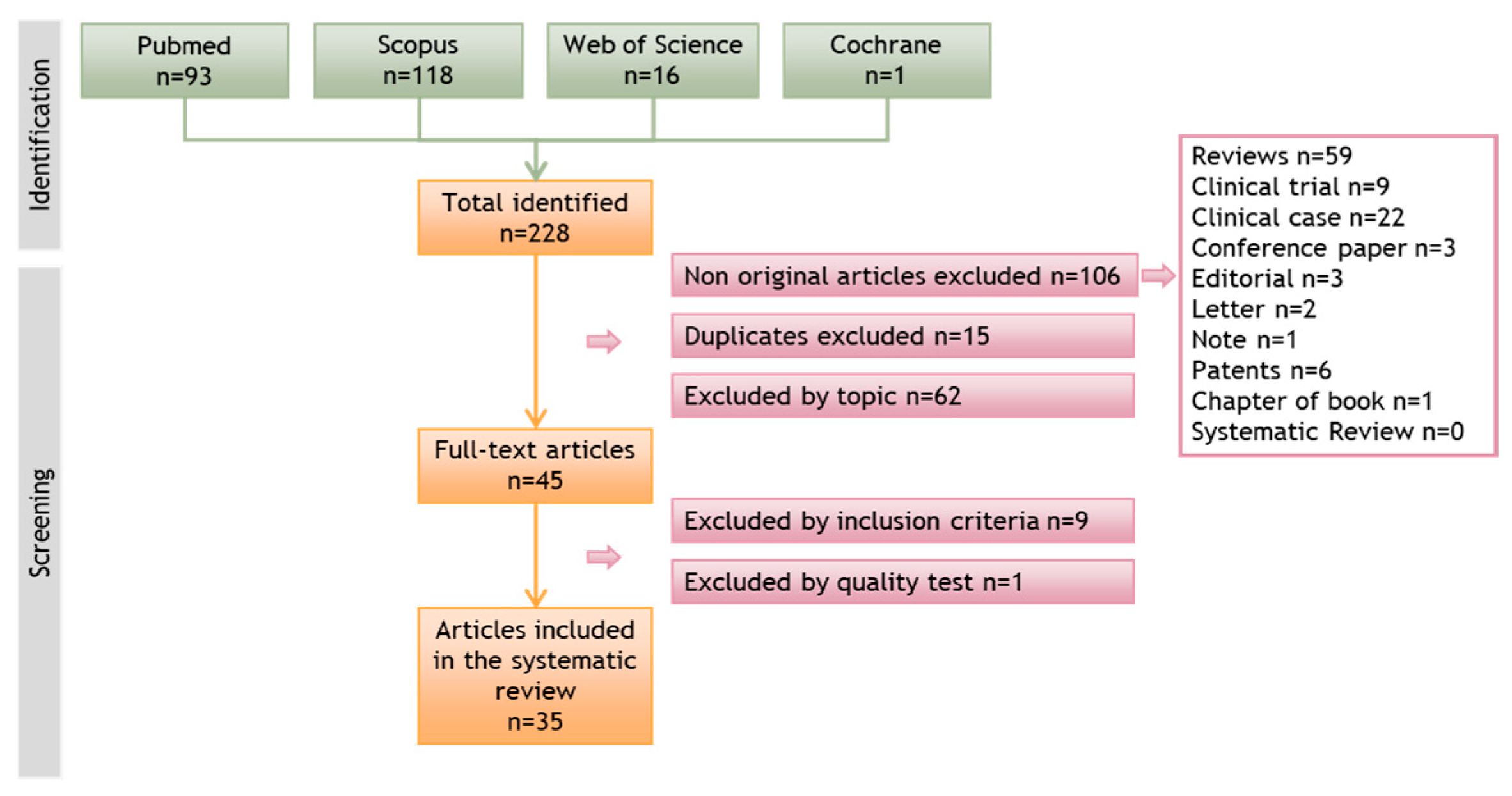
2. Materials and Methods
2.1. Study Eligibility
2.2. Inclusion Criteria
2.3. Exclusion Criteria
2.4. Data Sources
2.5. Study Selection
2.6. Data Extraction
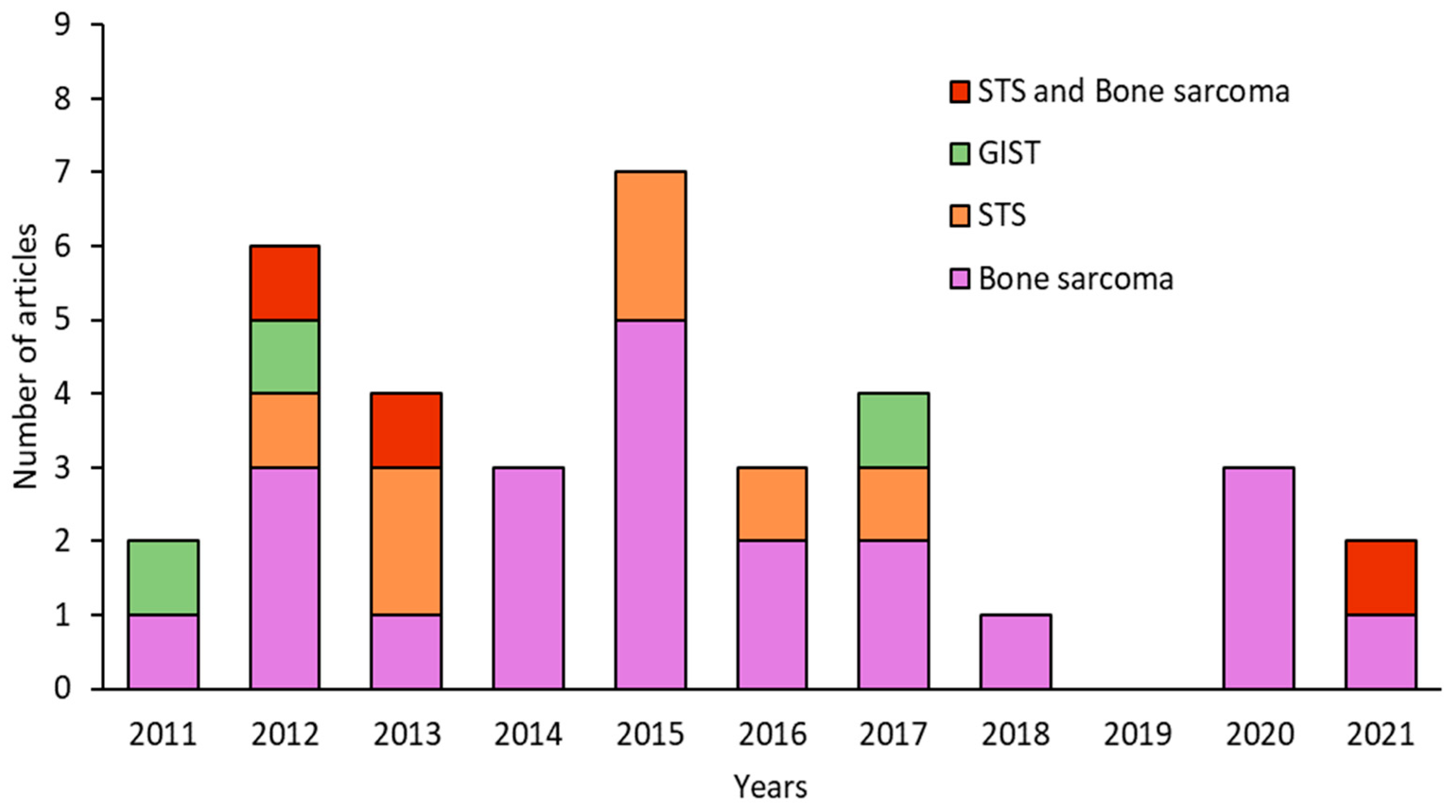
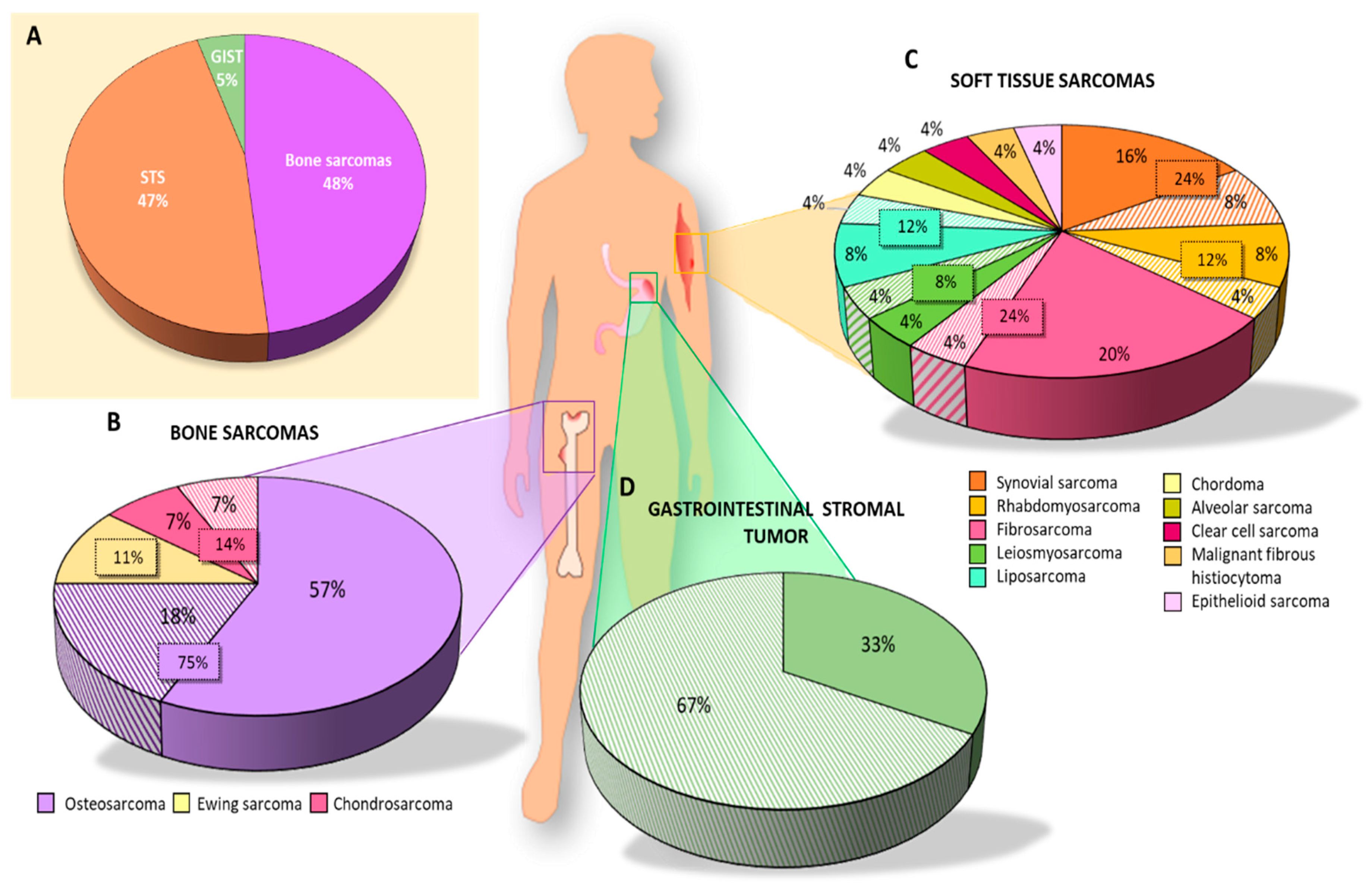
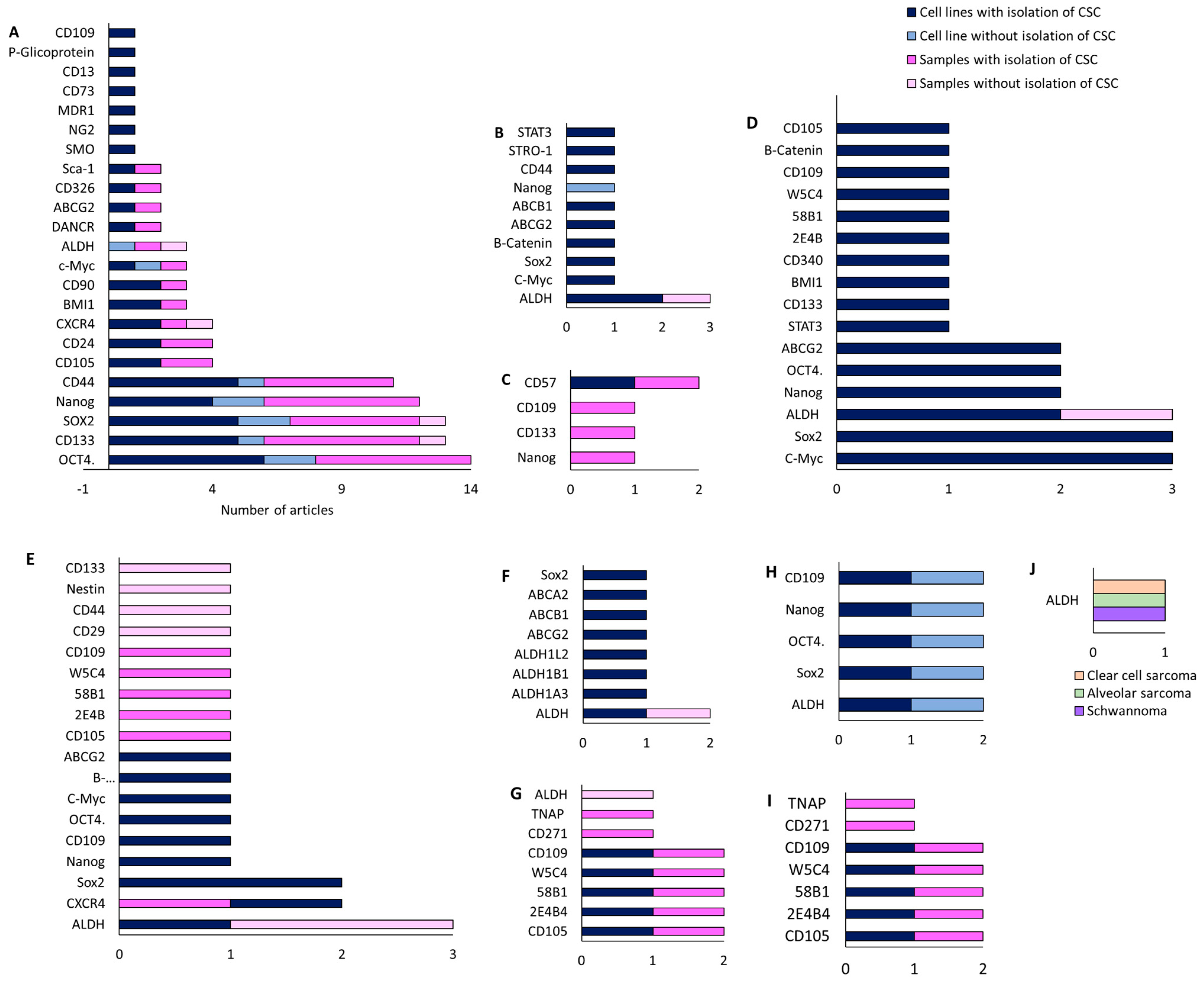
3. Results
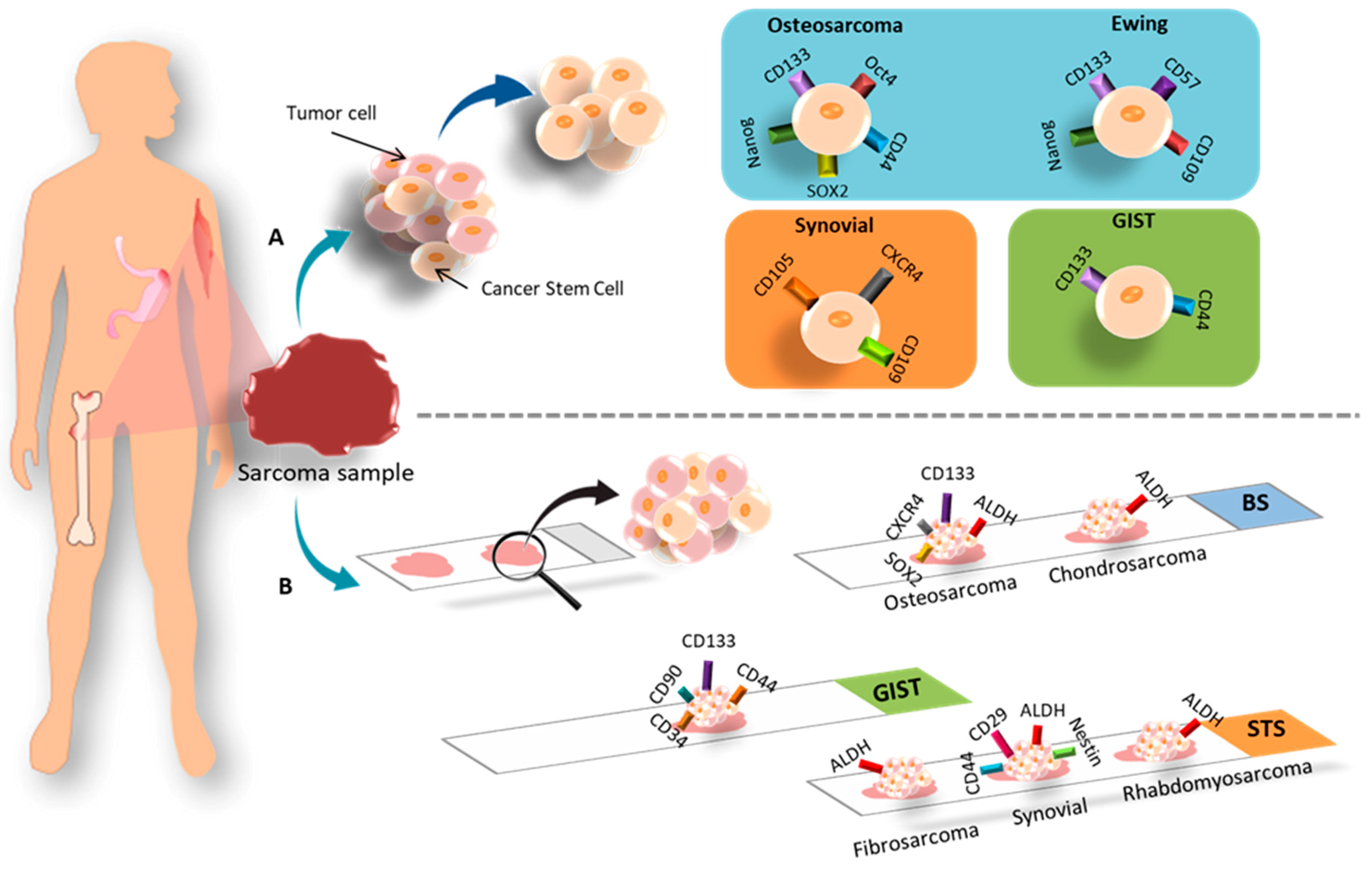
3.1. Different In Vitro Techniques for Isolating CSCs
3.1.1. Bone Sarcoma
3.1.2. Soft Tissue Sarcomas (STS)
3.1.3. GIST
3.2. CSC Markers in Histological Samples
3.2.1. Bone Sarcoma
3.2.2. Soft Tissue Sarcoma
3.2.3. GIST
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fletcher, C.; Bridge, J.A.; Hogendoorn, P.C.W.; Mertens, F. WHO Classification of Tumours of Soft Tissue and Bone; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Occupational Cancers; Anttila, S.; Boffetta, P. (Eds.) Springer International Publishing: Cham, Switzerland, 2020. [Google Scholar] [CrossRef]
- Zhao, X.; Yue, C. Gastrointestinal stromal tumor. J. Gastrointest. Oncol. 2012, 3, 189–208. [Google Scholar] [CrossRef] [PubMed]
- Arifi, S.; Belbaraka, R.; Rahhali, R.; Ismaili, N. Treatment of Adult Soft Tissue Sarcomas: An Overview. Rare Cancers Ther. 2015, 3, 69–87. [Google Scholar] [CrossRef] [PubMed]
- Harting, M.T.; Blakely, M.L. Management of osteosarcoma pulmonary metastases. Semin. Pediatr. Surg. 2006, 15, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Heymann, M.-F.; Brown, H.K.; Heymann, D. Drugs in early clinical development for the treatment of osteosarcoma. Expert Opin. Investig. Drugs 2016, 25, 1265–1280. [Google Scholar] [CrossRef]
- Mialou, V.; Philip, T.; Kalifa, C.; Perol, D.; Gentet, J.-C.; Marec-Berard, P.; Pacquement, H.; Chastagner, P.; Defaschelles, A.-S.; Hartmann, O. Metastatic osteosarcoma at diagnosis: Prognostic Factors and Long-Term Outcome--the French Pediatric Experience. Cancer 2005, 104, 1100–1109. [Google Scholar] [CrossRef]
- Tellez-Gabriel, M.; Heymann, M.-F.; Heymann, D. Circulating Tumor Cells as a Tool for Assessing Tumor Heterogeneity. Theranostics 2019, 9, 4580–4594. [Google Scholar] [CrossRef]
- Fourneaux, B.; Bourdon, A.; Dadone, B.; Lucchesi, C.; Daigle, S.R.; Richard, E.; Laroche-Clary, A.; Le Loarer, F.; Italiano, A. Identifying and targeting cancer stem cells in leiomyosarcoma: Prognostic impact and role to overcome secondary resistance to PI3K/mTOR inhibition. J. Hematol. Oncol. 2019, 12, 11. [Google Scholar] [CrossRef]
- Hatina, J.; Kripnerova, M.; Houfkova, K.; Pesta, M.; Kuncova, J.; Sana, J.; Slaby, O.; Rodríguez, R. Sarcoma Stem Cell Heterogeneity. Adv. Exp. Med. Biol. 2019, 1123, 95–118. [Google Scholar] [CrossRef]
- Vallette, F.M.; Olivier, C.; Lézot, F.; Oliver, L.; Cochonneau, D.; Lalier, L.; Cartron, P.-F.; Heymann, D. Dormant, quiescent, tolerant and persister cells: Four synonyms for the same target in cancer. Biochem. Pharmacol. 2019, 162, 169–176. [Google Scholar] [CrossRef]
- Yang, M.; Yan, M.; Zhang, R.; Li, J.; Luo, Z. Side population cells isolated from human osteosarcoma are enriched with tumor-initiating cells. Cancer Sci. 2011, 102, 1774–1781. [Google Scholar] [CrossRef] [PubMed]
- Damerell, V.; Pepper, M.S.; Prince, S. Molecular mechanisms underpinning sarcomas and implications for current and future therapy. Signal Transduct. Target. Ther. 2021, 6, 246. [Google Scholar] [CrossRef]
- Adhikari, A.S.; Agarwal, N.; Wood, B.M.; Porretta, C.; Ruiz, B.; Pochampally, R.R.; Iwakuma, T. CD117 and Stro-1 Identify Osteosarcoma Tumor-Initiating Cells Associated with Metastasis and Drug Resistance. Cancer Res. 2010, 70, 4602–4612. [Google Scholar] [CrossRef]
- Basu-Roy, U.; Bayin, N.S.; Rattanakorn, K.; Han, E.; Placantonakis, D.G.; Mansukhani, A.; Basilico, C. Sox2 antagonizes the Hippo pathway to maintain stemness in cancer cells. Nat. Commun. 2015, 6, 6411. [Google Scholar] [CrossRef] [PubMed]
- Greco, N.; Schott, T.; Mu, X.; Rothenberg, A.; Voigt, C.; III, R.L.M.; Goodman, M.; Huard, J.; Weiss, K.R. ALDH Activity Correlates with Metastatic Potential in Primary Sarcomas of Bone. J. Cancer Ther. 2014, 5, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Tirino, V.; Desiderio, V.; Paino, F.; De Rosa, A.; Papaccio, F.; Fazioli, F.; Pirozzi, G.; Papaccio, G. Human primary bone sarcomas contain CD133+ cancer stem cells displaying high tumorigenicity in vivo. FASEB J. 2011, 25, 2022–2030. [Google Scholar] [CrossRef]
- Schiavone, K.; Garnier, D.; Heymann, M.-F.; Heymann, D. The Heterogeneity of Osteosarcoma: The Role Played by Cancer Stem Cells. Adv. Exp. Med. Biol. 2019, 1139, 187–200. [Google Scholar] [CrossRef]
- Salerno, M.; Avnet, S.; Bonuccelli, G.; Eramo, A.; DE Maria, R.; Gambarotti, M.; Gamberi, G.; Baldini, N. Sphere-forming cell subsets with cancer stem cell properties in human musculoskeletal sarcomas. Int. J. Oncol. 2013, 43, 95–102. [Google Scholar] [CrossRef]
- Muka, T.; Glisic, M.; Milic, J.; Verhoog, S.; Bohlius, J.; Bramer, W.; Chowdhury, R.; Franco, O.H. A 24-step guide on how to design, conduct, and successfully publish a systematic review and meta-analysis in medical research. Eur. J. Epidemiol. 2020, 35, 49–60. [Google Scholar] [CrossRef]
- Száva-Kováts, E. Unfounded attribution of the “half-life” index-number of literature obsolescence to Burton and Kebler: A literature science study. J. Am. Soc. Inf. Sci. Technol. 2002, 53, 1098–1105. [Google Scholar] [CrossRef]
- Cohen, J. Weighted kappa: Nominal scale agreement provision for scaled disagreement or partial credit. Psychol. Bull. 1968, 70, 213–220. [Google Scholar] [CrossRef]
- Palmini, G.; Romagnoli, C.; Donati, S.; Zonefrati, R.; Galli, G.; Marini, F.; Iantomasi, T.; Aldinucci, A.; Leoncini, G.; Franchi, A.; et al. Analysis of a Preliminary microRNA Expression Signature in a Human Telangiectatic Osteogenic Sarcoma Cancer Cell Line. Int. J. Mol. Sci. 2021, 22, 1163. [Google Scholar] [CrossRef]
- Zhou, Z.; Li, Y.; Kuang, M.; Wang, X.; Jia, Q.; Cao, J.; Hu, J.; Wu, S.; Wang, Z.; Xiao, J. The CD24+ cell subset promotes invasion and metastasis in human osteosarcoma. Ebiomedicine 2020, 51, 102598. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chen, D.; Zhu, K. SOX2OT variant 7 contributes to the synergistic interaction between EGCG and Doxorubicin to kill osteosarcoma via autophagy and stemness inhibition. J. Exp. Clin. Cancer Res. 2018, 37, 37. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Wang, X.; Xie, X.; Liao, Y.; Liu, N.; Liu, J.; Miao, N.; Shen, J.; Peng, T. lncRNA DANCR promotes tumor progression and cancer stemness features in osteosarcoma by upregulating AXL via miR-33a-5p inhibition. Cancer Lett. 2017, 405, 46–55. [Google Scholar] [CrossRef]
- Cortini, M.; Massa, A.; Avnet, S.; Bonuccelli, G.; Baldini, N. Tumor-Activated Mesenchymal Stromal Cells Promote Osteosarcoma Stemness and Migratory Potential via IL-6 Secretion. PLoS ONE 2016, 11, e0166500. [Google Scholar] [CrossRef] [PubMed]
- He, A.; Yang, X.; Huang, Y.; Feng, T.; Wang, Y.; Sun, Y.; Shen, Z.; Yao, Y. CD133+CD44+Cells Mediate in the Lung Metastasis of Osteosarcoma. J. Cell. Biochem. 2015, 116, 1719–1729. [Google Scholar] [CrossRef]
- Sun, D.-X.; Liao, G.-J.; Liu, K.-G.; Jian, H. Endosialin-expressing bone sarcoma stem-like cells are highly tumor-initiating and invasive. Mol. Med. Rep. 2015, 12, 5665–5670. [Google Scholar] [CrossRef]
- Ni, M.; Xiong, M.; Zhang, X.; Cai, G.; Chen, H.; Zeng, Q.; Yu, Z. Poly(lactic-co-glycolic acid) nanoparticles conjugated with CD133 aptamers for targeted salinomycin delivery to CD133+ osteosarcoma cancer stem cells. Int. J. Nanomed. 2015, 10, 2537–2554. [Google Scholar] [CrossRef]
- Yi, X.-J.; Zhao, Y.-H.; Qiao, L.-X.; Jin, C.-L.; Tian, J.; Li, Q.-S. Aberrant Wnt/β-catenin signaling and elevated expression of stem cell proteins are associated with osteosarcoma side population cells of high tumorigenicity. Mol. Med. Rep. 2015, 12, 5042–5048. [Google Scholar] [CrossRef]
- Penfornis, P.; Cai, D.Z.; Harris, M.R.; Walker, R.; Licini, D.; Fernandes, J.D.A.; Orr, G.; Koganti, T.; Hicks, C.; Induru, S.; et al. High CD49f expression is associated with osteosarcoma tumor progression: A study using patient-derived primary cell cultures. Cancer Med. 2014, 3, 796–811. [Google Scholar] [CrossRef]
- Li, J.; Zhong, X.-Y.; Li, Z.-Y.; Cai, J.-F.; Zou, L.; Li, J.-M.; Yang, T.; Liu, W. CD133 expression in osteosarcoma and derivation of CD133+ cells. Mol. Med. Rep. 2012, 7, 577–584. [Google Scholar] [CrossRef]
- Saini, V.; Hose, C.D.; Monks, A.; Nagashima, K.; Han, B.; Newton, D.L.; Millione, A.; Shah, J.; Hollingshead, M.G.; Hite, K.M.; et al. Identification of CBX3 and ABCA5 as Putative Biomarkers for Tumor Stem Cells in Osteosarcoma. PLoS ONE 2012, 7, e41401. [Google Scholar] [CrossRef]
- Martins-Neves, S.R.; Lopes, O.; Carmo, A.D.; Paiva, A.A.; Simões, P.C.P.S.S.; Abrunhosa, A.J.; Gomes, C.M.F. Therapeutic implications of an enriched cancer stem-like cell population in a human osteosarcoma cell line. BMC Cancer 2012, 12, 139. [Google Scholar] [CrossRef] [PubMed]
- Basu-Roy, U.; Seo, E.; Ramanathapuram, L.; Rapp, T.B.; Perry, J.A.; Orkin, S.H.; Mansukhani, A.; Basilico, C. Sox2 maintains self renewal of tumor-initiating cells in osteosarcomas. Oncogene 2012, 31, 2270–2282. [Google Scholar] [CrossRef] [PubMed]
- Cornaz-Buros, S.; Riggi, N.; DeVito, C.; Sarre, A.; Letovanec, I.; Provero, P.; Stamenkovic, I. Targeting Cancer Stem–like Cells as an Approach to Defeating Cellular Heterogeneity in Ewing Sarcoma. Cancer Res. 2014, 74, 6610–6622. [Google Scholar] [CrossRef]
- Leuchte, K.; Altvater, B.; Hoffschlag, S.; Potratz, J.; Meltzer, J.; Clemens, D.; Luecke, A.; Hardes, J.; Dirksen, U.; Juergens, H.; et al. Anchorage-independent growth of Ewing sarcoma cells under serum-free conditions is not associated with stem-cell like phenotype and function. Oncol. Rep. 2014, 32, 845–852. [Google Scholar] [CrossRef]
- Granger, C.J.; Hoyt, A.K.; Moran, A.; Becker, B.; Saigh, S.; Conway, S.A.; Brown, J.; Galoian, K. Cancer stem cells as a therapeutic target in 3D tumor models of human chondrosarcoma: An encouraging future for proline rich polypeptide-1. Mol. Med. Rep. 2020, 22, 3747–3758. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Wang, L.; Tabu, K.; Tsuda, M.; Tanino, M.; Maekawa, A.; Nishihara, H.; Hiraga, H.; Taga, T.; Oda, Y.; et al. Identification and analysis of CXCR4-positive synovial sarcoma-initiating cells. Oncogene 2015, 35, 3932–3943. [Google Scholar] [CrossRef]
- Nakahata, K.; Uehara, S.; Nishikawa, S.; Kawatsu, M.; Zenitani, M.; Oue, T.; Okuyama, H. Aldehyde Dehydrogenase 1 (ALDH1) Is a Potential Marker for Cancer Stem Cells in Embryonal Rhabdomyosarcoma. PLoS ONE 2015, 10, e0125454. [Google Scholar] [CrossRef]
- Li, D.; Zhang, T.; Gu, W.; Li, P.; Cheng, X.; Tong, T.; Wang, W. The ALDH1+ subpopulation of the human NMFH-1 cell line exhibits cancer stem-like characteristics. Oncol. Rep. 2015, 33, 2291–2298. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.-H.; Liu, A.-G.; Gu, W.-G.; Deng, L.; Cheng, X.-G.; Tong, T.-J.; Zhang, H.-Z. CD133+ subpopulation of the HT1080 human fibrosarcoma cell line exhibits cancer stem-like characteristics. Oncol. Rep. 2013, 30, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Guo, T.; Zhang, L.; Qin, L.-X.; Singer, S.; Maki, R.G.; Taguchi, T.; DeMatteo, R.; Besmer, P.; Antonescu, C.R. CD133 and CD44 are universally overexpressed in GIST and do not represent cancer stem cell markers. Genes Chromosom. Cancer 2011, 51, 186–195. [Google Scholar] [CrossRef]
- Emori, M.; Tsukahara, T.; Murase, M.; Kano, M.; Murata, K.; Takahashi, A.; Kubo, T.; Asanuma, H.; Yasuda, K.; Kochin, V.; et al. High Expression of CD109 Antigen Regulates the Phenotype of Cancer Stem-Like Cells/Cancer-Initiating Cells in the Novel Epithelioid Sarcoma Cell Line ESX and Is Related to Poor Prognosis of Soft Tissue Sarcoma. PLoS ONE 2013, 8, e84187. [Google Scholar] [CrossRef]
- Wirths, S.; Malenke, E.; Kluba, T.; Rieger, S.; Müller, M.R.; Schleicher, S.; von Weyhern, C.H.; Nagl, F.; Fend, F.; Vogel, W.; et al. Shared Cell Surface Marker Expression in Mesenchymal Stem Cells and Adult Sarcomas. STEM CELLS Transl. Med. 2012, 2, 53–60. [Google Scholar] [CrossRef]
- Lohberger, B.; Rinner, B.; Stuendl, N.; Absenger, M.; Liegl-Atzwanger, B.; Walzer, S.M.; Windhager, R.; Leithner, A. Aldehyde Dehydrogenase 1, a Potential Marker for Cancer Stem Cells in Human Sarcoma. PLoS ONE 2012, 7, e43664. [Google Scholar] [CrossRef] [PubMed]
- Mardani, A.; Gheytanchi, E.; Mousavie, S.H.; Jabari, Z.M.; Shooshtarizadeh, T. Clinical Significance of Cancer Stem Cell Markers CD133 and CXCR4 in Osteosarcomas. Asian Pac. J. Cancer Prev. 2020, 21, 67–73. [Google Scholar] [CrossRef]
- Wang, Z.; Tan, M.; Chen, G.; Li, Z.; Lu, X. LncRNA SOX2-OT is a novel prognostic biomarker for osteosarcoma patients and regulates osteosarcoma cells proliferation and motility through modulating SOX2. IUBMB Life 2017, 69, 867–876. [Google Scholar] [CrossRef]
- Yoshiyama, A.; Morii, T.; Ohtsuka, K.; Ohnishi, H.; Tajima, T.; Aoyagi, T.; Mochizuki, K.; Satomi, K.; Ichimura, S. Development of Stemness in Cancer Cell Lines Resistant to the Anticancer Effects of Zoledronic Acid. Anticancer. Res. 2016, 36, 625–631. [Google Scholar] [PubMed]
- Galoian, K.; Qureshi, A.; D’ippolito, G.; Schiller, P.C.; Molinari, M.; Johnstone, A.L.; Brothers, S.P.; Paz, A.C.; Temple, H.T. Epigenetic regulation of embryonic stem cell marker miR302C in human chondrosarcoma as determinant of antiproliferative activity of proline-rich polypeptide 1. Int. J. Oncol. 2015, 47, 465–472. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, D.; Qi, Y.; Liu, R.; Li, S.; Zou, H.; Lan, J.; Ju, X.; Jiang, J.; Liang, W.; et al. Evaluation of expression of cancer stem cell markers and fusion gene in synovial sarcoma: Insights into histogenesis and pathogenesis. Oncol. Rep. 2017, 37, 3351–3360. [Google Scholar] [CrossRef] [PubMed]
- Hegyi, L.; Thway, K.; Fisher, C.; Sheppard, M.N. Primary cardiac sarcomas may develop from resident or bone marrow-derived mesenchymal stem cells: Use of immunohistochemistry including CD44 and octamer binding protein 3/4. Histopathology 2012, 61, 966–973. [Google Scholar] [CrossRef]
- Geddert, H.; Braun, A.; Kayser, C.; Dimmler, A.; Faller, G.; Agaimy, A.; Haller, F.; Moskalev, E.A. Epigenetic Regulation of CD133 in Gastrointestinal Stromal Tumors. Am. J. Clin. Pathol. 2017, 147, 515–524. [Google Scholar] [CrossRef]
- Bozzi, F.; Conca, E.; Manenti, G.; Negri, T.; Brich, S.; Gronchi, A.; Pierotti, M.A.; Tamborini, E.; Pilotti, S. High CD133 expression levels in gastrointestinal stromal tumors. Cytom. Part B Clin. Cytom. 2011, 80, 238–247. [Google Scholar] [CrossRef]
- Avdonkina, N.; Danilova, A.; Misyurin, V.; Prosekina, E.; Girdyuk, D.; Emelyanova, N.; Nekhaeva, T.; Gafton, G.; Baldueva, I. Biological features of tissue and bone sarcomas investigated using an in vitro model of clonal selection. Pathol.-Res. Pract. 2020, 217, 153214. [Google Scholar] [CrossRef] [PubMed]
- Skapek, S.X.; Ferrari, A.; Gupta, A.A.; Lupo, P.J.; Butler, E.; Shipley, J.; Barr, F.G.; Hawkins, D.S. Rhabdomyosarcoma. Nat. Rev. Dis. Prim. 2019, 5, 1. [Google Scholar] [CrossRef] [PubMed]
- Saab, R.; Spunt, S.L.; Skapek, S.X. Chapter 7—Myogenesis and Rhabdomyosarcoma: The Jekyll and Hyde of Skeletal Muscle. In Current Topics in Developmental Biology; Dyer, M.A., Ed.; Cancer and Development; Academic Press: Cambridge, MA, USA, 2011; Volume 94, pp. 197–234. [Google Scholar] [CrossRef]
- Kashi, V.P.; Hatley, M.E.; Galindo, R.L. Probing for a deeper understanding of rhabdomyosarcoma: Insights from complementary model systems. Nat. Rev. Cancer 2015, 15, 426–439. [Google Scholar] [CrossRef]
- Amer, K.M.; Thomson, J.E.; Congiusta, D.; Dobitsch, A.; Chaudhry, A.; Li, M.; Chaudhry, A.; Bozzo, A.; Siracuse, B.; Aytekin, M.N.; et al. Epidemiology, Incidence, and Survival of Rhabdomyosarcoma Subtypes: SEER and ICES Database Analysis. J. Orthop. Res. 2019, 37, 2226–2230. [Google Scholar] [CrossRef]
- Heymann, M.; Schiavone, K.; Heymann, D. Bone sarcomas in the immunotherapy era. Br. J. Pharmacol. 2020, 178, 1955–1972. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.D.; Lizardo, M.M.; Reed, D.R.; Hingorani, P.; Glover, J.; Allen-Rhoades, W.; Fan, T.; Khanna, C.; Sweet-Cordero, E.A.; Cash, T.; et al. Provocative questions in osteosarcoma basic and translational biology: A report from the Children’s Oncology Group. Cancer 2019, 125, 3514–3525. [Google Scholar] [CrossRef]
- Lytle, N.K.; Barber, A.G.; Reya, T. Stem cell fate in cancer growth, progression and therapy resistance. Nat. Rev. Cancer 2018, 18, 669–680. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Norgard, R.J.; Stanger, B.Z. Cellular Plasticity in Cancer. Cancer Discov. 2019, 9, 837–851. [Google Scholar] [CrossRef] [PubMed]
- Muff, R.; Botter, S.M.; Husmann, K.; Tchinda, J.; Selvam, P.; Seeli-Maduz, F.; Fuchs, B. Explant culture of sarcoma patients’ tissue. Lab. Investig. 2016, 96, 752–762. [Google Scholar] [CrossRef]
- Tellez-Gabriel, M.; Brown, H.K.; Young, R.; Heymann, M.-F.; Heymann, D. The Challenges of Detecting Circulating Tumor Cells in Sarcoma. Front. Oncol. 2016, 6, 202. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Seebacher, N.A.; Hornicek, F.J.; Xiao, T.; Duan, Z. Application of liquid biopsy in bone and soft tissue sarcomas: Present and future. Cancer Lett. 2018, 439, 66–77. [Google Scholar] [CrossRef] [PubMed]
- van der Laan, P.; van Houdt, W.J.; Broek, D.v.D.; Steeghs, N.; van der Graaf, W.T.A. Liquid Biopsies in Sarcoma Clinical Practice: Where Do We Stand? Biomedicines 2021, 9, 1315. [Google Scholar] [CrossRef]
- Chang, L.; Asatrian, G.; Dry, S.M.; James, A.W. Circulating tumor cells in sarcomas: A brief review. Med. Oncol. Northwood Lond. Engl. 2015, 32, 430. [Google Scholar] [CrossRef]
- Nicolazzo, C.; Gradilone, A. Significance of Circulating Tumor Cells in Soft Tissue Sarcoma. Anal. Cell. Pathol. 2015, 2015, 697395. [Google Scholar] [CrossRef]
- Agnoletto, C.; Caruso, C.; Garofalo, C. Heterogeneous Circulating Tumor Cells in Sarcoma: Implication for Clinical Practice. Cancers 2021, 13, 2189. [Google Scholar] [CrossRef]

| Type of Sarcoma (Ref.) | Sample | CSC Isolation Technique and Induction Medium | Methods for CSCs Characterization | Biomarkers | Treatment | Summary of Findings |
|---|---|---|---|---|---|---|
| Osteosarcoma [24] | Cell line: - Primary tumor: 1 patient | Spheres formation (Serum free 2× Ham’s F12 Coon’s medium supplemented with progesterone (20 nM), putrescine 100 µM, sodium selenite (30 nM), transferrin (25 µg/mL), insulin (20 µg/mL), EGF (10 ng/mL), b-FGF (10 ng/mL) were mixed with equal volume of 2% sterile methylcellulose). |
| ALDH CD44 CD45 CD90 CD105 Nanog POU5F1 LIN28A SOX2 SATB2 KLF4 Nestin c-Kit PROM1 EZR AXL MYC | - |
|
| Osteosarcoma [25] | Cell line: MG63 MNNG/HOS U2OS Primary tumor: 30 patients |
|
| OCT4 NANOG SOX2 BMI1 CD24 CD14 CD117 CD133 | Cisplatin Epirubicin Hydrochloride |
|
| Osteosarcoma [26] | Cell line: SaoS2 U2OS Primary tumor: - |
|
| SOX-2 OCT-4 c-Myc Nanog CD44 CD133 | EGCG |
|
| Osteosarcoma [27] | Cell line: MG63 U2OS SaOS2 HOS 143B Primary tumor: 34 patients |
|
| CD133 CD44 SOX2 CD90 | DANCR gene |
|
| Osteosarcoma [28] | Cell line: HOS MG63 MSC | Spheres formation with serum-free DMEM-F12 medium with progesterone (20 nM), putrescine (10 mg/mL), sodium selenite (30 nM), apo-transferrin (100 mg/mL), and insulin (25 mg/mL), EGF (20 ng/mL), and FGF (10 ng/mL). | RT-PCR | Nanog Oct4 SOX2 CXCR4 | Co-culture with MSC |
|
| Osteosarcoma [29] | Cell line: MG-63 Saos-2 U2OS Primary tumor: 15 samples of patients with sarcoma lung metastasis |
|
| CD133 CD44 Oct4 NANOG CXCR4 | - |
|
| Osteosarcoma [30] | Cell line: - Primary tumor: 10 patients |
|
| CD248 CD133 Oct3/4A Nestin Nanog ABCG2 ABCB2 ABCA1 ABCB1 | Doxorubicin Cisplatin Methotrexate |
|
| Osteosarcoma [31] | Cell line: Saos-2 U-2 OS MG-63 Primary tumor: - |
|
| CD133 BMI-1 c-Myc Oct-4 SMO NG2 | AP-SAL-NP |
|
| Osteosarcoma [32] | Cell line: - Primary tumor: 10 patients |
|
| CD133 Oct-4 Sox2 Nanog Nestin ABCG2 | - |
|
| Osteosarcoma [33] | Cell line: KHOS Primary tumor: 4 patients | FACS (CD49f+) |
| CD44 CD90 CD49b CD105 CD117 CD49f | Doxorubicin Cisplatin |
|
| Osteosarcoma [34] | Cell line: Saos-2 Primary tumor: 55 patients: 4 parosteal 13 parosteal 12 chondroblastic 26 osteoblastic | MACS (CD133+) | RT-PCR | CD133 SOX2 MDR1 | - |
|
| Osteosarcoma [35] | Cell line: Saos-2 HuO9 Primary tumor: 1 patient (CHA59) | Spheres formation (serum free RPMI-1640 containing 15% KnockOut Serum Replacement and 2 mM L-glutamine). |
| PPARG ETS1 WNT1 WNT5B SOX2 NANOG POU55F1 Nestin ALDH CD24 CD44 CD133 CD166 ABCB1 ABCC1 ABCG2 | Cisplatin 5-fluorouracil Imatinib |
|
| Osteosarcoma [36] | Cell line: MNNG/HOS SAR-OS (culture of adherent sphere-derived cells) Primary tumor: - | Spheres formation (serum-free DMEM/F12 medium supplemented with 1% of methylcellulose supplemented with 1% penicillin/streptomycin, 20 nM progesterone, 100 μM putrescine, 1% ITS, 10 ng/mL FGF, and 10 ng/mL EGF). |
| CD105 CD73 CD13 CD90 CD34 CD44 CD11b CD19 HLA-DR Oct4 Nanog P-Glycoprotein BCRP | Doxorubicin Cisplatin Methotrexate Verapamil |
|
| Osteosarcoma [37] | Cell line: U-2OS MG63 HOS OS-187 OS-99-1 Saos-2 Saos-2-LM7 Primary tumor: 18 patients |
|
| Sox2 Sca-1 | - |
|
| Osteosarcoma [13] | Cell line: - Primary tumor: 6 Primary tumor |
|
| ABCA2 ABCB1/MDR1 ABCC1/MRP1 ABCG2 Oct-4 Nanog CD44 CD117 CD133 | Doxorubicin Cisplatin Methotrexate |
|
| Ewing’s sarcoma [38] | Cell line: A673 TC252 Primary tumor: 4 patients | Spheres formation (IMDM supplemented with 20% KO serum, 10 mg/mL LIF, 10 ng/mL recombinant human EGF, and 10 ng/mL recombinant human FGF) |
| CD133 Nanog Oct4 | Doxorubicin Enoxacin |
|
| Ewing’s sarcoma [39] | Cell line: VH-64 WE-68 TC-71 A-4573 TC-32 Primary tumor: 4 patients | Spheres formation (serum-free DMEM/F12 (1:1) supplemented with 4% B27, 20 ng/mL rhEGF, 20 ng/mL LIF, and 10 IE/mL (5 μg/mL) heparin). |
| CD99 CD117 CD133 CD57 | _ |
|
| Chondrosarcoma [40] | Cell line: JJ012 Primary tumor: - |
|
| ALDH CD44 STRO-1 STAT3 | PRP-1 |
|
| Synovial sarcoma [41] | Cell line: SYO-1 Fuji HS-SYII Primary tumor: 39 patients | Spheres formation (Serum free Medium/F12 supplement with 10 ng/mL FGF and 20 ng/mL EGF). |
| NANOG OCT4 SOX2 CXCR4 | - |
|
| Rhabdomyosarcoma [42] | Cell line: RD KYM-1 Primary tumor: - | FACS (ALDH) |
| ALDH1 ALDH1A1 ALDH1A2 ALDH1A3 ALDH1B1 ALDH1L1 ALDH1L2 c-Myc Sox2 ABCG2/BCRP ABCB1/MDR1 ABCA2 | Vincristine Cyclophosphamide Etoposide |
|
| Fibrosarcoma [43] | Cell line: NMFH-1 Primary tumor: - |
|
| ALDH1 c-Myc Bmi-1 Sox2 Nanog OCT3/4 STAT3 ABCG2 | Doxorubicin Cisplatin |
|
| Fibrosarcoma [44] | Cell line: HT1080 Primary tumor: - |
|
| CD133 Nanog Oct3/4 SOX2 ABCG2 c-Myc Bmi-1 | Cisplatin Doxorubicin |
|
| GIST [45] | Cell line: GIST882 GIST48 GIST62 GIST-T1 Primary tumor: 131 GIST primary tumor 25 soft tissue sarcoma (microarrays) | FACS (CD133+/CD44+) |
| CD133 CD44 | Imatinib |
|
| Osteosarcoma Ewing’s sarcoma Synovial sarcoma Epithelioid sarcoma Malignant fibrous histiocytoma [46] | Cell line: NY U2OS HOS OS2000 KIKU SKES WES RDES FUJI YaFuSS FU-EPS-1 VA-ES-BJ MFH2003 MFH2004 Primary tumor: 81 patients |
|
| ALDH1 CD109 Sox2 Oct3/4 Nanog Twist 1 Snail 1 | - |
|
| Fibrosarcoma Leiomyosarcoma Liposarcoma Synovial sarcoma [47] | Cell line: HT1080 SK-LMS1l SW872 SW982 Primary tumor: 12 patients: LMS (n = 4), rhabdomyosarcoma (n = 4), and liposarcoma (n = 4) |
| IHC | CD105 2E4B4 58B1 W5C4 CD109 CD340 CD164 CD56 (W1C3) W4A5 W7C6 CD271 CD140b CD56 NPC CD10 CD318 CD344 F9-3C2F1 HEK3D6 CD172a CD349 W3D5A9 W5C5 TNAP CD117 CD133 CD326 CD34 CD324 W3C3 | Doxorubicin |
|
| Fibrosarcoma Liposarcoma Synovial sarcoma Chondrosarcoma Rhabdomyosarcoma Chordoma [48] | Cell line: SW-684 SW-872 SW-982 SW-1353 TE-671 MUG-Chor1 Primary tumor: - | FACS (ALDH) |
| ALDH1 c-Myc β-catenin SOX2 ABCG2/BCRP1 ABCA2 ABCB1/MDR1 | Doxorubicin Epirubicin Cisplatin |
|
| Type of Sarcoma (Ref.) | Sample | Biomarkers | Technique | Treatment | Summary of Findings |
|---|---|---|---|---|---|
| Osteosarcoma [49] | Cell line: - Primary tumor 50 patients | CXCR4 CD133 | IHC | - |
|
| Osteosarcoma [50] | Cell line: Saos-2 MG-63 U-2 OS MNNG/HOS Primary tumor: 138 patients | SOX2 OCT4 NANOG ALDH1 CD44 CD133 lncRNA SOX2-OT |
| - |
|
| Osteosarcoma [51] | Cell line: MG63 Primary tumor - | NANOG c-MYC OCT-4 SOX2 |
| Zoledronate |
|
| Chondrosarcoma [52] | Cell line: JJ012 Primary tumor - | Nanog |
| PRP-1 |
|
| Synovial sarcoma [53] | Cell line: - Primary tumor 20 patients | CD133 CD29 CD44 Nestin ALDH1 |
| - |
|
| Undifferentiated cardiac sarcoma [54] | Cell line: - Primary tumor 5 patients | CD44 Oct ¾ |
| - |
|
| GIST [55] | Cell line: GIST882 GIST48b Primary tumor 95 patients | CD133 |
| 5-aza-dC |
|
| GIST [56] | Cell line: - Primary tumor 27 patients | CD133 CD90 CD44 CD34 |
| Imatinib |
|
| Liposarcoma Osteosarcoma Synovial sarcoma Fibrosarcoma Leiomyosarcoma Rhabdomyosarcoma Schwannoma Alveolar sarcoma Clear cell sarcoma Chondrosarcoma [57] | Cell line: - Primary tumor: 38 STS 16 osteosarcoma 43 metastasis 7 local recurrence 4 primary tumor | ALDH CD133 |
| Doxorubicin Ifosfamide Gemcitabine Docetaxel Cisplatin Etoposide |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chico, M.A.; Mesas, C.; Doello, K.; Quiñonero, F.; Perazzoli, G.; Ortiz, R.; Prados, J.; Melguizo, C. Cancer Stem Cells in Sarcomas: In Vitro Isolation and Role as Prognostic Markers: A Systematic Review. Cancers 2023, 15, 2449. https://doi.org/10.3390/cancers15092449
Chico MA, Mesas C, Doello K, Quiñonero F, Perazzoli G, Ortiz R, Prados J, Melguizo C. Cancer Stem Cells in Sarcomas: In Vitro Isolation and Role as Prognostic Markers: A Systematic Review. Cancers. 2023; 15(9):2449. https://doi.org/10.3390/cancers15092449
Chicago/Turabian StyleChico, Maria Angeles, Cristina Mesas, Kevin Doello, Francisco Quiñonero, Gloria Perazzoli, Raul Ortiz, Jose Prados, and Consolacion Melguizo. 2023. "Cancer Stem Cells in Sarcomas: In Vitro Isolation and Role as Prognostic Markers: A Systematic Review" Cancers 15, no. 9: 2449. https://doi.org/10.3390/cancers15092449
APA StyleChico, M. A., Mesas, C., Doello, K., Quiñonero, F., Perazzoli, G., Ortiz, R., Prados, J., & Melguizo, C. (2023). Cancer Stem Cells in Sarcomas: In Vitro Isolation and Role as Prognostic Markers: A Systematic Review. Cancers, 15(9), 2449. https://doi.org/10.3390/cancers15092449







