Serum β-hCG as a Biomarker in Pancreatic Neuroendocrine Tumors: Rethinking Single-Analyte Approach
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Design
2.2. Data Collection and Assays
2.3. Statistical Analysis
3. Results
3.1. Baseline Characteristics
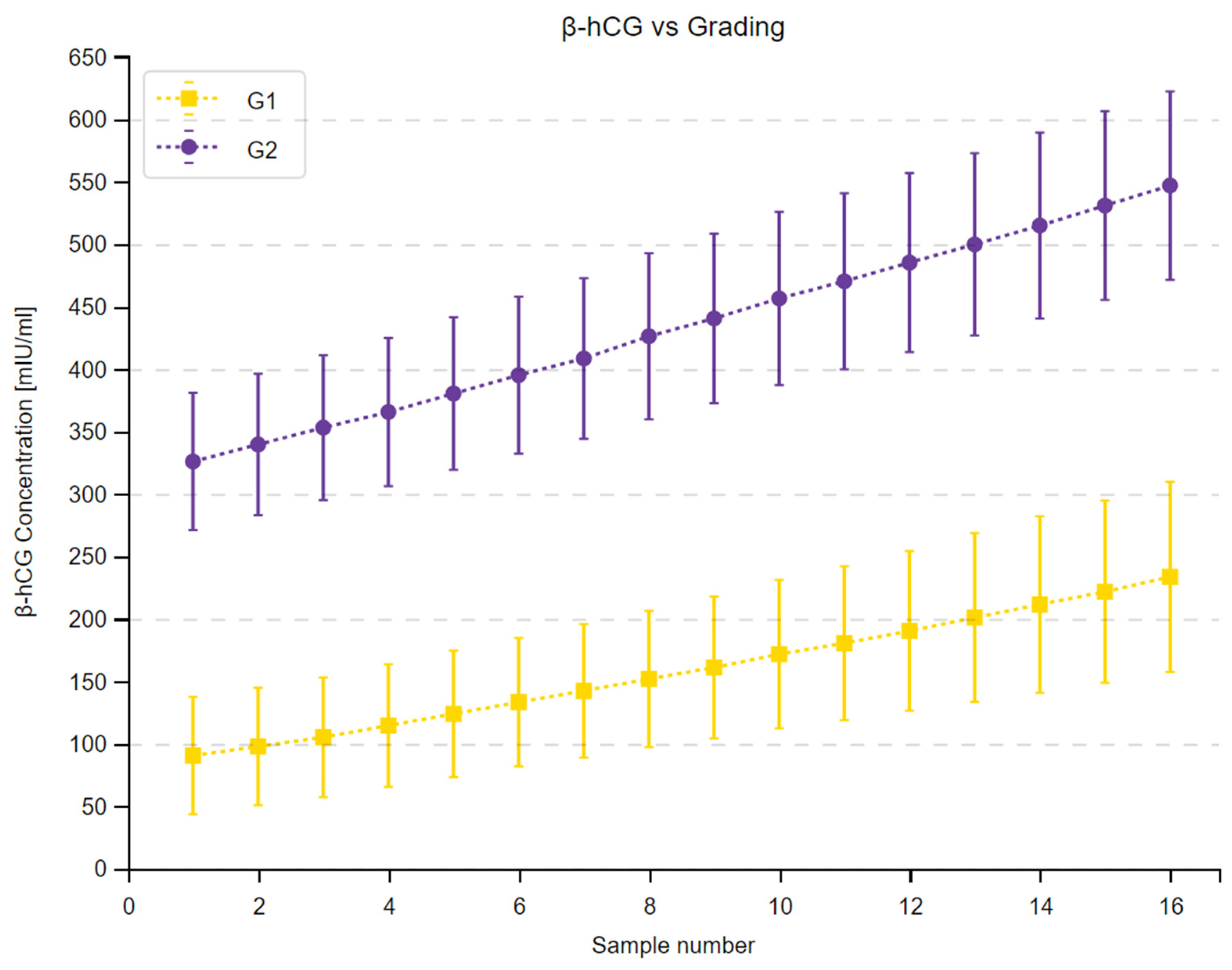
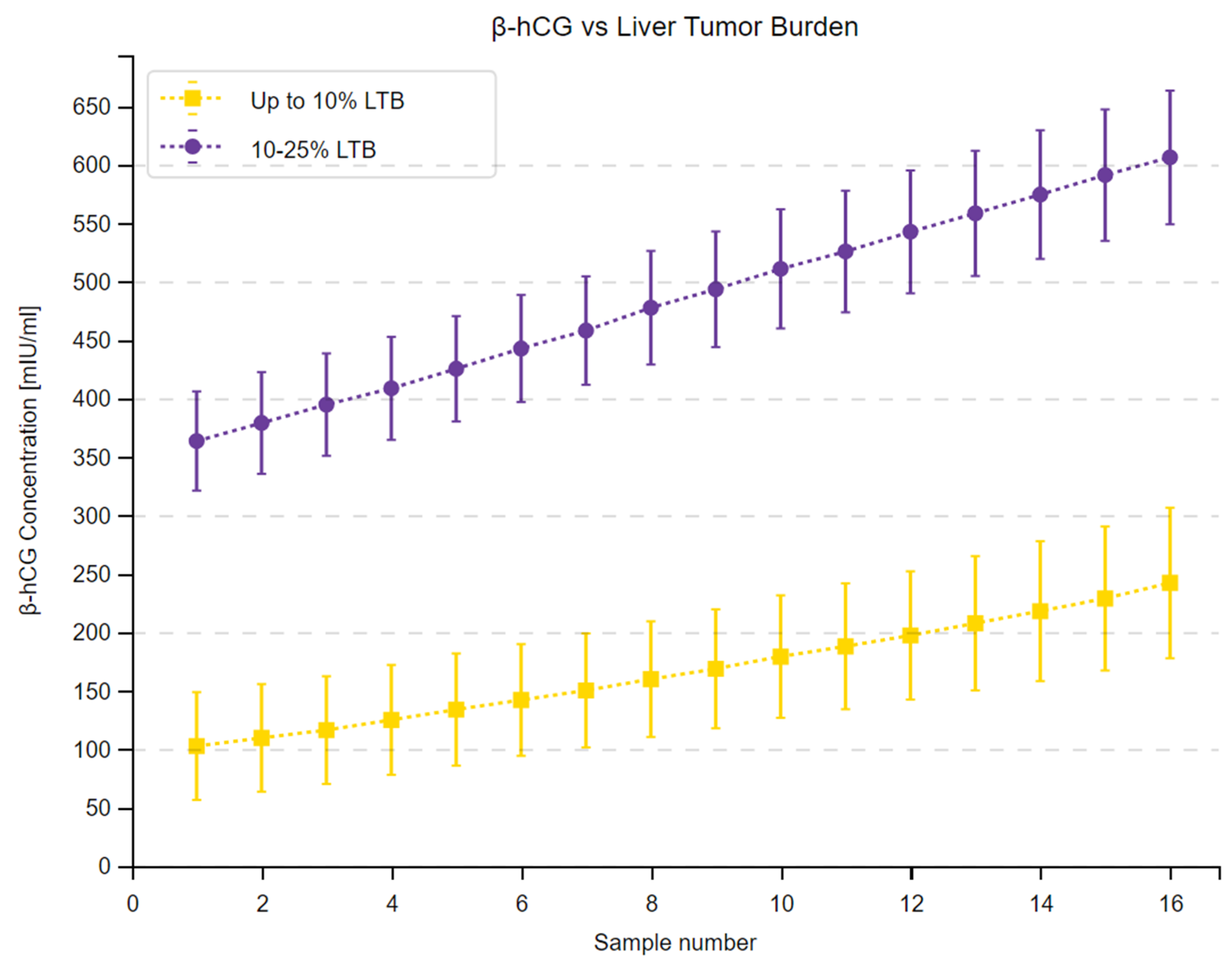
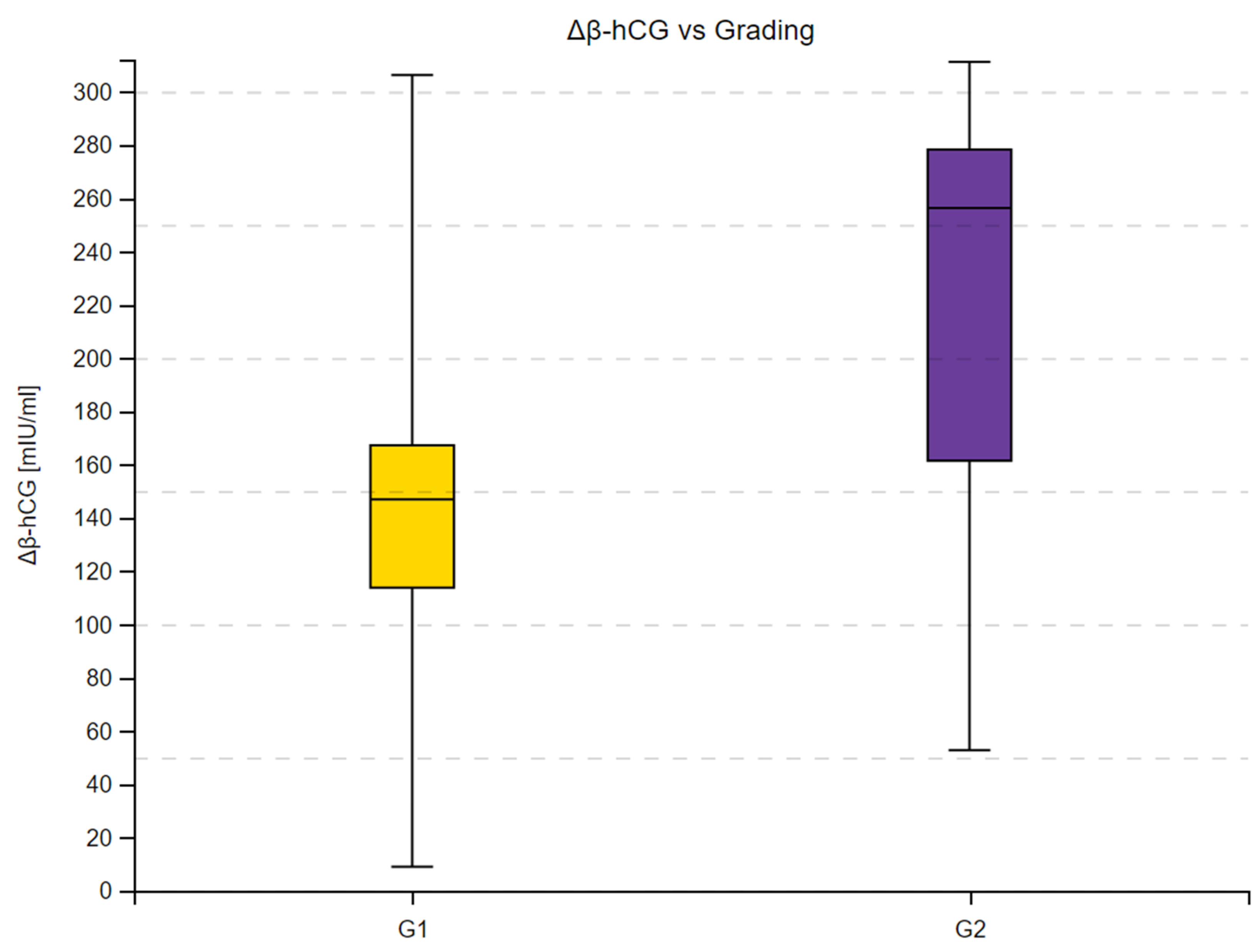
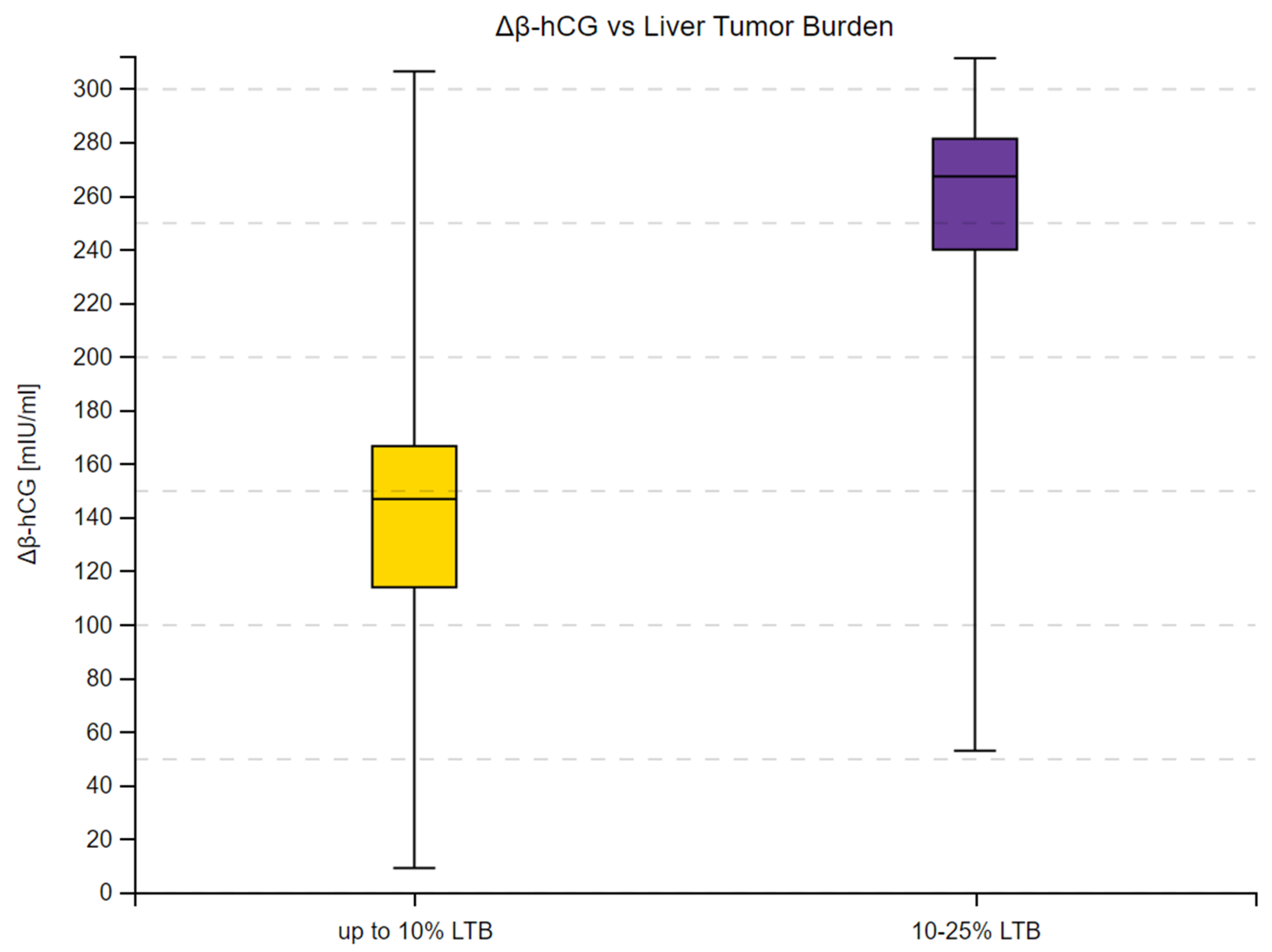
3.2. β-hCG Concentration Depending on Tumor Malignancy
3.3. β-hCG Changes over Time
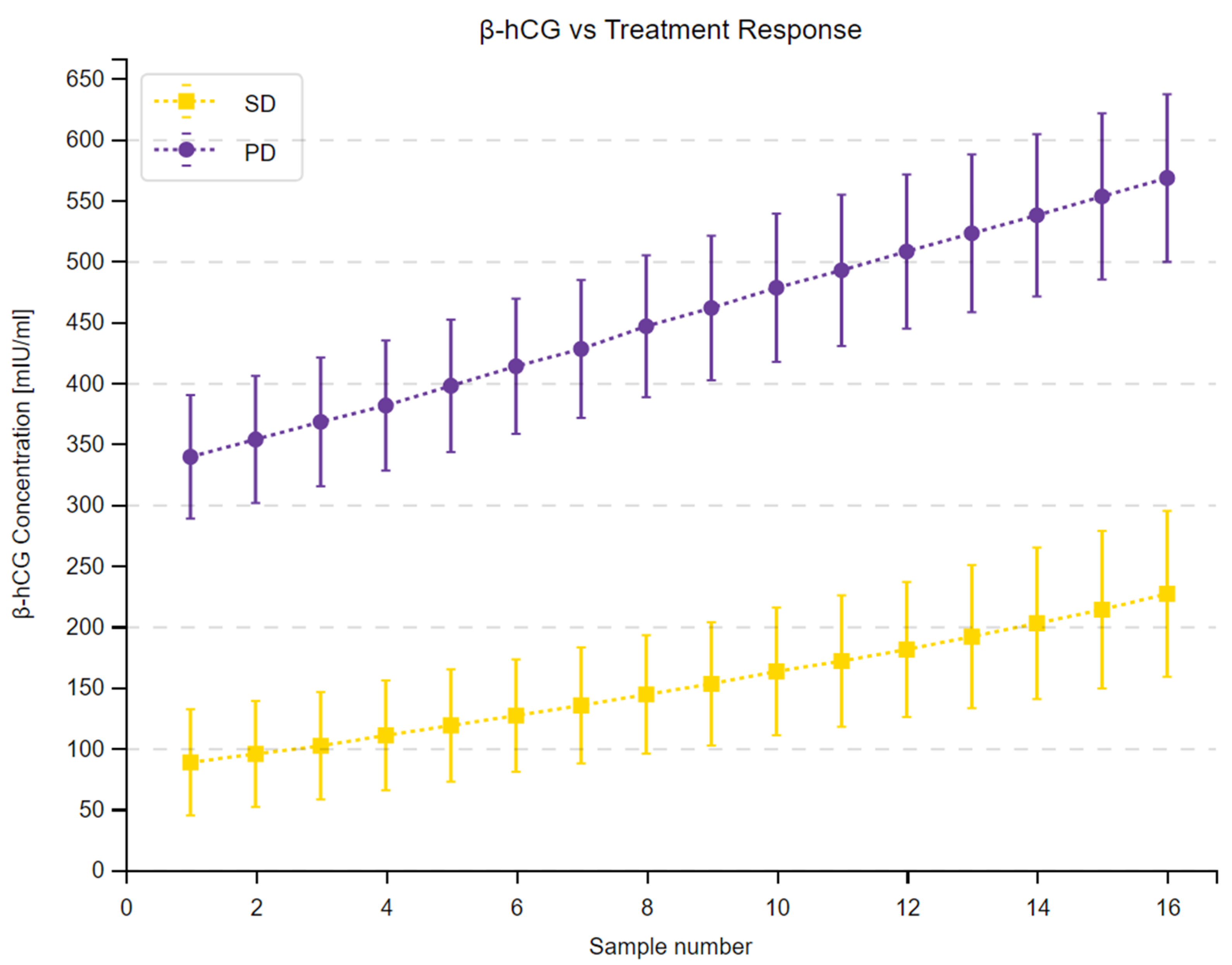
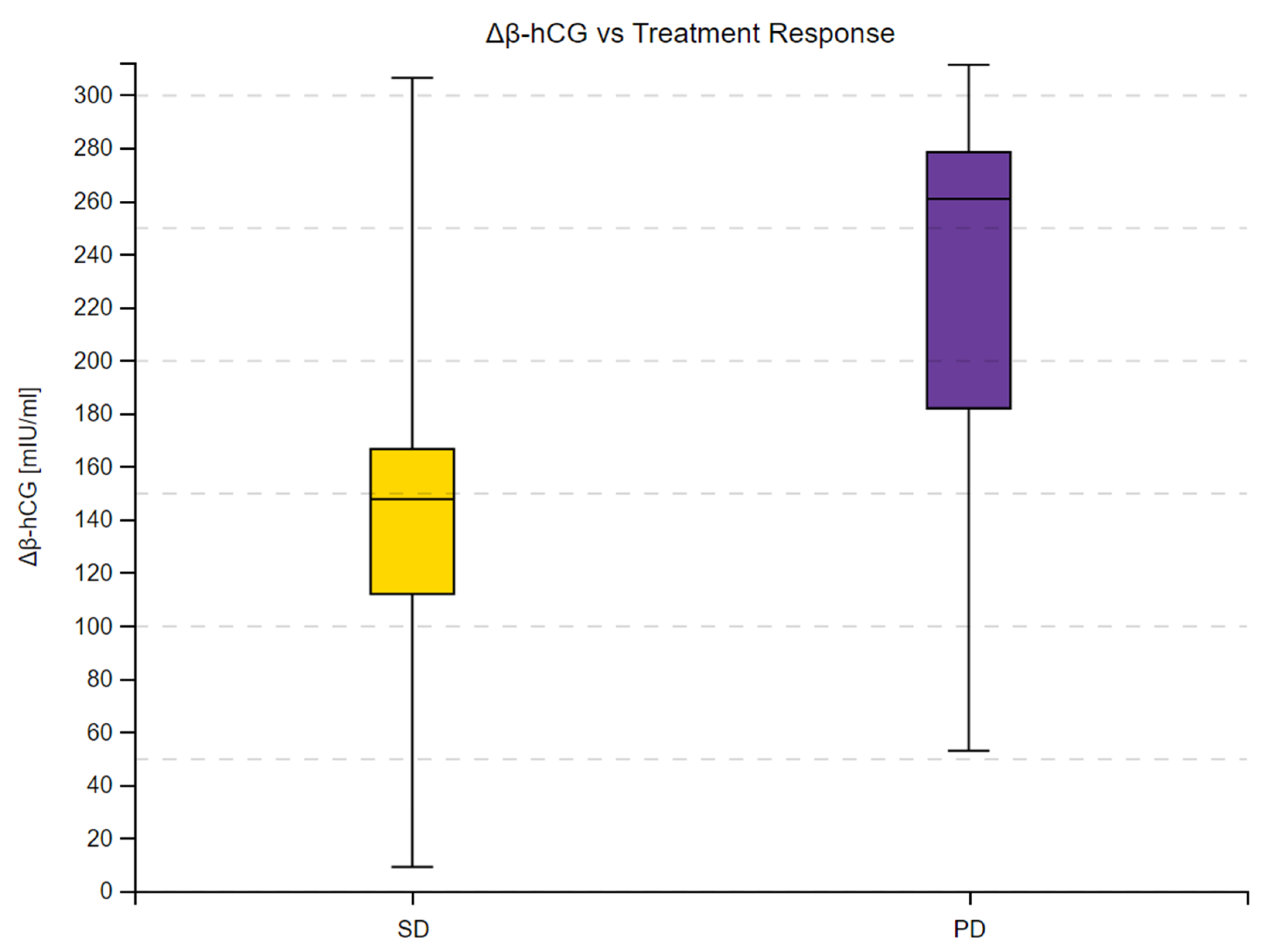
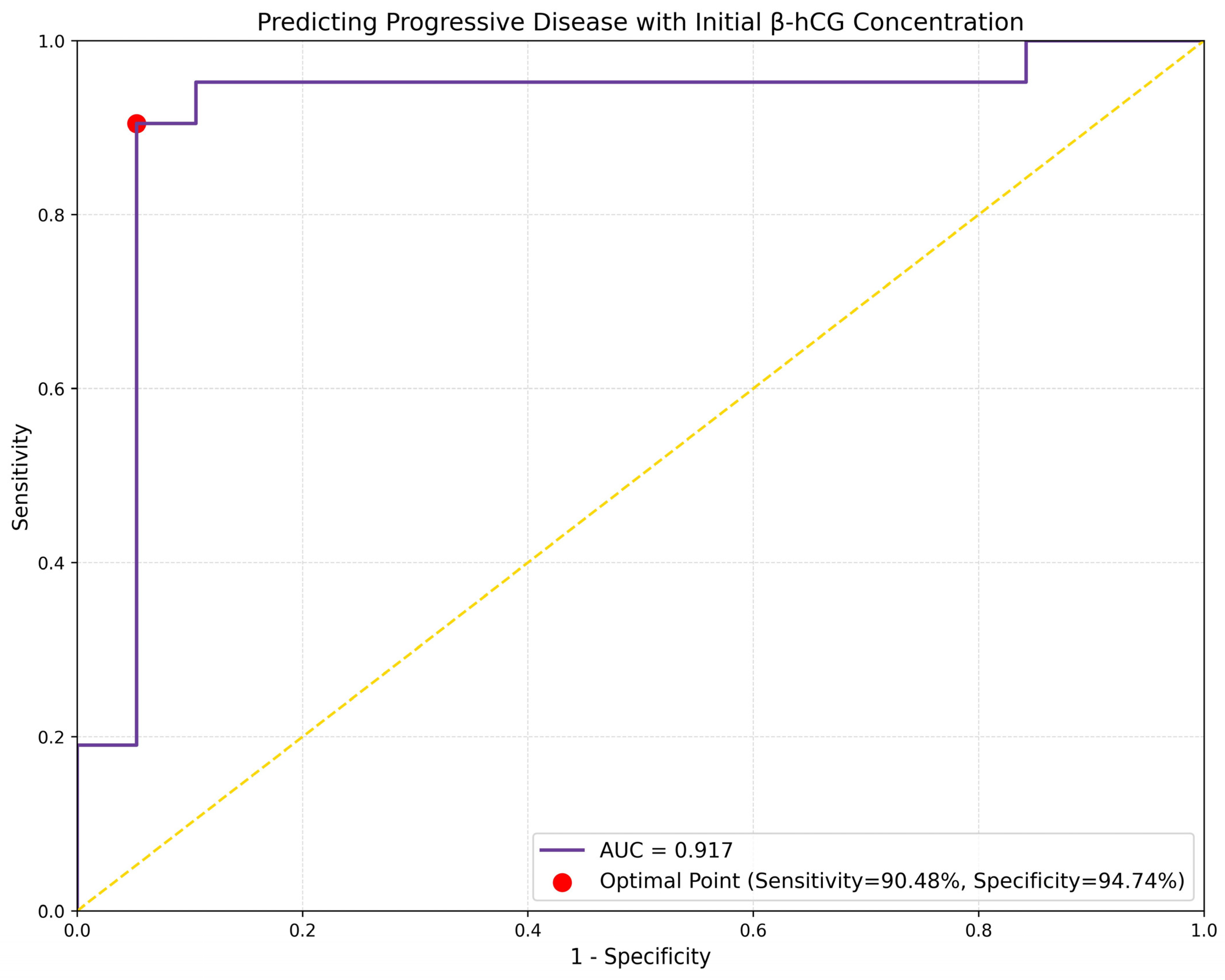
3.4. β-hCG and Predicting Treatment Response
4. Discussion
4.1. History of β-hCG Research in NETs
4.2. Commentary on the Results of the Study
4.3. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taal, B.G.; Visser, O. Epidemiology of Neuroendocrine Tumours. Neuroendocrinology 2004, 80, 3–7. [Google Scholar] [CrossRef]
- Oberg, K.; Modlin, I.M.; De Herder, W.; Pavel, M.; Klimstra, D.; Frilling, A.; Metz, D.C.; Heaney, A.; Kwekkeboom, D.; Strosberg, J.; et al. Consensus on Biomarkers for Neuroendocrine Tumour Disease. Lancet Oncol. 2015, 16, e435–e446. [Google Scholar] [CrossRef] [PubMed]
- Gut, P.; Czarnywojtek, A.; Sawicka-Gutaj, N.; Woliński, K.; Maciejewski, A.; Komarnicki, P.; Ruchała, M. Determination of Neuron-Specific Enolase in Patients with Midgut-Type Tumour Treated with Somatostatin Analogues. Endokrynol. Pol. 2021, 72, 308–318. [Google Scholar] [CrossRef]
- Prinz, R.A.; Bermes, E.W.; Kimmel, J.R.; Marangos, P.J. Serum Markers for Pancreatic Islet Cell and Intestinal Carcinoid Tumors: A Comparison of Neuron-Specific Enolase, Beta-Human Chorionic Gonadotropin and Pancreatic Polypeptide. Surgery 1983, 94, 1019–1023. [Google Scholar] [PubMed]
- Komarnicki, P.; Gut, P.; Musiałkiewicz, J.; Cieślewicz, M.; Maciejewski, A.; Patel, P.; Mastorakos, G.; Ruchała, M. NT-ProBNP as a Neuroendocrine Tumor Biomarker: Beyond Heart Failure. Endocr. Connect. 2023, 12, e230249. [Google Scholar] [CrossRef] [PubMed]
- Oberg, K. Circulating Biomarkers in Gastroenteropancreatic Neuroendocrine Tumours. Endocr. Relat. Cancer 2011, 18, 17–25. [Google Scholar] [CrossRef]
- Komarnicki, P.; Musiałkiewicz, J.; Stańska, A.; Maciejewski, A.; Gut, P.; Mastorakos, G.; Ruchała, M. Circulating Neuroendocrine Tumor Biomarkers: Past, Present and Future. J. Clin. Med. 2022, 11, 5542. [Google Scholar] [CrossRef] [PubMed]
- Modlin, I.M.; Drozdov, I.; Alaimo, D.; Callahan, S.; Teixiera, N.; Bodei, L.; Kidd, M. A Multianalyte PCR Blood Test Outperforms Single Analyte ELISAs (Chromogranin A, Pancreastatin, Neurokinin A) for Neuroendocrine Tumor Detection. Endocr. Relat. Cancer 2014, 21, 615–628. [Google Scholar] [CrossRef]
- Malczewska, A.; Kos-Kudła, B.; Kidd, M.; Drozdov, I.; Bodei, L.; Matar, S.; Oberg, K.; Modlin, I.M. The Clinical Applications of a Multigene Liquid Biopsy (NETest) in Neuroendocrine Tumors. Adv. Med. Sci. 2020, 65, 18–29. [Google Scholar] [CrossRef]
- Marinoni, I.; Kurrer, A.S.; Vassella, E.; Dettmer, M.; Rudolph, T.; Banz, V.; Hunger, F.; Pasquinelli, S.; Speel, E.J.; Perren, A. Loss of DAXX and ATRX Are Associated with Chromosome Instability and Reduced Survival of Patients with Pancreatic Neuroendocrine Tumors. Gastroenterology 2014, 146, 453–460.e5. [Google Scholar] [CrossRef]
- Hackeng, W.M.; Brosens, L.A.A.; Kim, J.Y.; O’Sullivan, R.; Sung, Y.N.; Liu, T.C.; Cao, D.; Heayn, M.; Brosnan-Cashman, J.; An, S.; et al. Non-Functional Pancreatic Neuroendocrine Tumours: ATRX/DAXX and Alternative Lengthening of Telomeres (ALT) Are Prognostically Independent from ARX/PDX1 Expression and Tumour Size. Gut 2022, 71, 961–973. [Google Scholar] [CrossRef]
- Dunn, C.; Gately, L.; Gibbs, P. Drinking from the Firehose—A Clinician’s Perspective on the Challenges of Delivering Biomarker-Driven Care in Routine Practice. Eur. J. Cancer 2021, 157, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Cole, L.A. HCG, the Wonder of Today’s Science. Reprod. Biol. Endocrinol. 2012, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Cole, L.A. HCG, Five Independent Molecules. Clin. Chim. Acta 2012, 413, 48–65. [Google Scholar] [CrossRef]
- Seidel, C.; Daugaard, G.; Nestler, T.; Tryakin, A.; Fedyanin, M.; Fankhauser, C.; Hermanns, T.; Aparicio, J.; Heinzelbecker, J.; Paffenholz, P.; et al. Human Chorionic Gonadotropin–Positive Seminoma Patients: A Registry Compiled by the Global Germ Cell Tumor Collaborative Group (G3). Eur. J. Cancer 2020, 132, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.; Scott-Mackie, P.; Cunningham, D.; Norman, A.; Andreyev, J.; O’Brien, M.; Bensted, J. The Prognostic Value of Serum and Immunohistochemical Tumour Markers in Advanced Gastric Cancer. Eur. J. Cancer 1996, 32, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Doi, F.; Chi, D.D.J.; Charuworn, B.B.; Conrad, A.J.; Russell, J.; Morton, D.L.; Hoon, D.S.B. Detection of β-Human Chorionic Gonadotropin MRNA as a Marker for Cutaneous Malignant Melanoma. Int. J. Cancer 1996, 65, 454–459. [Google Scholar] [CrossRef]
- Heitz, P.U.; Von Herbay, G.; Klöppel, G.; Komminoth, P.; Kasper, M.; Höfler, H.; Müller, K.-M.; Oberholzer, M. The Expression of Subunits of Human Chorionic Gonadotropin (HCG) by Nontrophoblastic, Nonendocrine, and Endocrine Tumors. Am. J. Clin. Pathol. 1987, 88, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Dirnhofer, S.; Freund, M.; Rogatsch, H.; Krabichler, S.; Berger, P. Selective Expression of Trophoblastic Hormones by Lung Carcinoma: Neurendocrine Tumors Exclusively Produce Human Chorionic Gonadotropin α-Subunit (HCGα). Hum. Pathol. 2000, 31, 966–972. [Google Scholar] [CrossRef]
- Stenman, U.H.; Alfthan, H.; Hotakainen, K. Human Chorionic Gonadotropin in Cancer. Clin. Biochem. 2004, 37, 549–561. [Google Scholar] [CrossRef]
- Shah, T.; Srirajaskanthan, R.; Bhogal, M.; Toubanakis, C.; Meyer, T.; Noonan, A.; Witney-Smith, C.; Amin, T.; Bhogal, P.; Sivathasan, N.; et al. α-Fetoprotein and Human Chorionic Gonadotrophin-β as Prognostic Markers in Neuroendocrine Tumour Patients. Br. J. Cancer 2008, 99, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, B.; Ouberg, K.; Skogseid, B. Neuroendocrine Pancreatic Tumors Clinical Findings in a Prospective Study of 84 Patients. Acta Oncol. 1989, 28, 373–377. [Google Scholar] [CrossRef]
- Oberg, K.; Wide, L. HCG and HCG Subunits as Tumour Markers in Patients with Endocrine Pancreatic Tumours and Carcinoids. Acta Endocrinol. 1981, 98, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Rindi, G.; Mete, O.; Uccella, S.; Basturk, O.; La Rosa, S.; Brosens, L.A.A.; Ezzat, S.; de Herder, W.W.; Klimstra, D.S.; Papotti, M.; et al. Overview of the 2022 WHO Classification of Neuroendocrine Neoplasms. Endocr. Pathol. 2022, 33, 115–154. [Google Scholar] [CrossRef] [PubMed]
- Kahn, C.R.; Rosen, S.W.; Weintraub, B.D.; Fajans, S.S.; Gorden, P. Ectopic Production of Chorionic Gonadotropin and Its Subunits by Islet-Cell Tumors. N. Engl. J. Med. 1977, 297, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, M.; Trautmann, M.E.; Poertl, S.; Hoermann, R.; Berger, P.; Arnold, R.; Mann, K. Alpha-subunit and Human Chorionic Gonadotropin-β Immunoreactivity in Patients with Malignant Endocrine Gastroenteropancreatic Tumours. Eur. J. Clin. Investig. 1994, 24, 131–136. [Google Scholar] [CrossRef]
- Baudin, E.; Bidart, J.M.; Rougier, P.; Lazar, V.; Ruffié, P.; Ropers, J.; Ducreux, M.; Troalen, F.; Sabourin, J.C.; Comoy, E.; et al. Screening for Multiple Endocrine Neoplasia Type I and Hormonal Production in Apparently Sporadic Neuroendocrine Tumors. J. Clin. Endocrinol. Metab. 1999, 84, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Ramage, J.K.; Davies, A.H.G.; Ardill, J.; Bax, N.; Caplin, M.; Grossman, A.; Hawkins, R.; McNicol, A.M.; Reed, N.; Sutton, R.; et al. Guidelines for the Management of Gastroenteropancreatic Neuroendocrine (Including Carcinoid) Tumours. Gut 2005, 54, iv1–iv16. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, F.; Fazio, N.; Attanasio, R.; Frasoldati, A.; Papini, E.; Angelini, F.; Baldelli, R.; Berretti, D.; Bianchetti, S.; Bizzarri, G.; et al. Italian Association of Clinical Endocrinologists (AME) Position Statement: A Stepwise Clinical Approach to the Diagnosis of Gastroenteropancreatic Neuroendocrine Neoplasms. J. Endocrinol. Investig. 2014, 37, 875–909. [Google Scholar] [CrossRef]
- Sansone, A.; Lauretta, R.; Vottari, S.; Chiefari, A.; Barnabei, A.; Romanelli, F.; Appetecchia, M. Specific and Non-Specific Biomarkers in Neuroendocrine Gastroenteropancreatic Tumors. Cancers 2019, 11, 1113. [Google Scholar] [CrossRef]
- Cole, L.A.; Butler, S. Hyperglycosylated HCG, HCGβ and Hyperglycosylated HCGβ: Interchangeable Cancer Promoters. Mol. Cell. Endocrinol. 2012, 349, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Kanakis, G.; Kaltsas, G. Biochemical Markers for Gastroenteropancreatic Neuroendocrine Tumours (GEP-NETs). Best Pract. Res. Clin. Gastroenterol. 2012, 26, 791–802. [Google Scholar] [CrossRef] [PubMed]
- Cole, L.A.; Khanlian, S.A. The Need for a Quantitative Urine HCG Assay. Clin. Biochem. 2009, 42, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Merola, E.; Alonso Gordoa, T.; Zhang, P.; Al-Toubah, T.; Pellè, E.; Kolasińska-Ćwikła, A.; Zandee, W.; Laskaratos, F.; Mestier, L.; Lamarca, A.; et al. Somatostatin Analogs for Pancreatic Neuroendocrine Tumors: Any Benefit When Ki-67 Is ≥10%? Oncologist 2021, 26, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Dasari, A. Epidemiology, Incidence, and Prevalence of Neuroendocrine Neoplasms: Are There Global Differences? Curr. Oncol. Rep. 2021, 23, 10–20. [Google Scholar] [CrossRef]
- Dasari, A.; Shen, C.; Halperin, D.; Zhao, B.; Zhou, S.; Xu, Y. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients with Neuroendocrine Tumors in the United States. JAMA Oncol. 2017, 3, 1335–1342. [Google Scholar] [CrossRef]
| Variable | All Patients (n = 40) |
|---|---|
| Demographics | |
| Age, years | 63 (8) * |
| Sex | |
| Male | 12 (30%) |
| Female | 28 (70%) |
| Clinical characteristics | |
| Primary tumor location | |
| Pancreas | 40 (100%) |
| Liver Tumor Burden | |
| Up to 10% | 22 (55%) |
| 10–25% | 18 (45%) |
| Grading by WHO | |
| G1 | 18 (45%) |
| G2 | 22 (55%) |
| Treatment response by RECIST 1.1. | |
| Stable Disease (SD) | 19 (47.5%) |
| Progressive Disease (PD) | 21 (52.5%) |
| Ki 67 | |
| 1% | 2 (5%) |
| 2% | 16 (40%) |
| 4% | 2 (5%) |
| 5% | 8 (20%) |
| 10% | 12 (30%) |
| Measuring Time Point | Median β-hCG—SD [mIU/mL] | Median β-hCG—PD [mIU/mL] | Sum of Ranks SD | Sum of Ranks PD | U | Z Corrected | p |
|---|---|---|---|---|---|---|---|
| 1. | 67.80 | 356.40 | 223 | 597 | 33 | −4.4964 | <0.001 |
| 2. | 76.50 | 376.30 | 220 | 600 | 30 | −4.5783 | <0.001 |
| 3. | 86.30 | 389.50 | 219 | 601 | 29 | −4.6047 | <0.001 |
| 4. | 93.40 | 402.60 | 220 | 600 | 30 | −4.5776 | <0.001 |
| 5. | 102.30 | 420.40 | 219 | 601 | 29 | −4.6047 | <0.001 |
| 6. | 113.40 | 438.40 | 218 | 602 | 28 | −4.6314 | <0.001 |
| 7. | 123.40 | 451.20 | 219 | 601 | 29 | −4.6045 | <0.001 |
| 8. | 134.20 | 462.40 | 217 | 603 | 27 | −4.6587 | <0.001 |
| 9. | 143.50 | 476.50 | 218 | 602 | 28 | −4.6314 | <0.001 |
| 10. | 153.40 | 487.60 | 219 | 601 | 29 | −4.6043 | <0.001 |
| 11. | 159.40 | 496.70 | 219 | 601 | 29 | −4.6043 | <0.001 |
| 12. | 167.80 | 515.80 | 220 | 600 | 30 | −4.5772 | <0.001 |
| 13. | 178.50 | 527.80 | 222 | 598 | 32 | −4.5230 | <0.001 |
| 14. | 189.70 | 546.70 | 223 | 597 | 33 | −4.4964 | <0.001 |
| 15. | 199.60 | 576.30 | 223 | 597 | 33 | −4.4962 | <0.001 |
| 16. | 212.80 | 598.70 | 223 | 597 | 33 | −4.4959 | <0.001 |
| Measuring Time Point | Median β-hCG—G1 [mIU/mL] | Median β-hCG—G2 [mIU/mL] | Sum of Ranks G1 | Sum of Ranks G2 | U | Z Corrected | p |
|---|---|---|---|---|---|---|---|
| 1. | 67.70 | 354.35 | 211 | 609 | 40 | −4.2822 | <0.001 |
| 2. | 76.50 | 368.75 | 211 | 609 | 40 | −4.2829 | <0.001 |
| 3. | 85.40 | 375.70 | 210 | 610 | 39 | −4.3094 | <0.001 |
| 4. | 95.60 | 390.60 | 213.5 | 606.5 | 42 | −4.2143 | <0.001 |
| 5. | 102.40 | 409.55 | 214 | 606 | 43 | −4.2007 | <0.001 |
| 6. | 114.40 | 427.60 | 213 | 607 | 42 | −4.2275 | <0.001 |
| 7. | 123.45 | 442.00 | 216 | 604 | 45 | −4.1461 | <0.001 |
| 8. | 135.45 | 457.80 | 215 | 605 | 44 | −4.1733 | <0.001 |
| 9. | 143.60 | 473.15 | 217 | 603 | 46 | −4.1187 | <0.001 |
| 10. | 154.90 | 480.40 | 216 | 604 | 45 | −4.1459 | <0.001 |
| 11. | 162.55 | 488.95 | 216 | 604 | 45 | −4.1459 | <0.001 |
| 12. | 171.05 | 507.25 | 215 | 605 | 44 | −4.1731 | <0.001 |
| 13. | 182.95 | 522.15 | 216 | 604 | 45 | −4.1459 | <0.001 |
| 14. | 192.55 | 545.20 | 217 | 603 | 46 | −4.1191 | <0.001 |
| 15. | 200.95 | 570.35 | 218 | 602 | 47 | −4.0917 | <0.001 |
| 16. | 213.10 | 593.15 | 216 | 604 | 45 | −4.1459 | <0.001 |
| Measuring Time Point | Median β-hCG—up to 10% [mIU/mL] | Median β-hCG—10–25% [mIU/mL] | Sum of Ranks up to 10% | Sum of Ranks 10–25% | U | Z Corrected | p |
|---|---|---|---|---|---|---|---|
| 1. | 72.15 | 382.20 | 275 | 545 | 22 | −4.7717 | <0.001 |
| 2. | 79.40 | 397.15 | 272 | 548 | 19 | −4.8539 | <0.001 |
| 3. | 87.40 | 413.05 | 271 | 549 | 18 | −4.8804 | <0.001 |
| 4. | 95.60 | 429.95 | 271 | 549 | 18 | −4.8804 | <0.001 |
| 5. | 102.40 | 442.80 | 271 | 549 | 18 | −4.8804 | <0.001 |
| 6. | 114.40 | 457.90 | 270 | 550 | 17 | −4.9071 | <0.001 |
| 7. | 123.45 | 472.40 | 271 | 549 | 18 | −4.8802 | <0.001 |
| 8. | 135.45 | 490.95 | 268 | 552 | 15 | −4.9617 | <0.001 |
| 9. | 143.60 | 510.25 | 269 | 551 | 16 | −4.9343 | <0.001 |
| 10. | 154.90 | 523.00 | 269 | 551 | 16 | −4.9343 | <0.001 |
| 11. | 162.55 | 535.05 | 269 | 551 | 16 | −4.9343 | <0.001 |
| 12. | 171.05 | 550.30 | 269 | 551 | 16 | −4.9343 | <0.001 |
| 13. | 182.95 | 562.30 | 271 | 549 | 18 | −4.8800 | <0.001 |
| 14. | 192.55 | 578.30 | 271 | 549 | 18 | −4.8804 | <0.001 |
| 15. | 200.95 | 594.70 | 272 | 548 | 19 | −4.8530 | <0.001 |
| 16. | 213.10 | 612.15 | 272 | 548 | 19 | −4.8530 | <0.001 |
| Variable | Median β-hCG [mIU/mL] | Sum of Ranks | U | Z Corrected | p | |
|---|---|---|---|---|---|---|
| Treatment Response | ||||||
| SD | 147.50 | 263 | 73 | −3.4127 | <0.001 | |
| PD | 260.70 | 557 | ||||
| Grading | ||||||
| G1 | 143.23 | 261 | 90 | −2.9227 | 0.003 | |
| G2 | 256.25 | 559 | ||||
| Liver Tumor Burden | ||||||
| up to 10% | 146.65 | 307 | 54 | −3.9014 | <0.001 | |
| 10–25% | 267.00 | 513 | ||||
| Patient Age | R Spearman | T (N-2) | 0.97 | |||
| −0.0069 | −0.0423 | |||||
| Variable | Beta Coefficient | 95% CI for Beta | p-Value | Odds Ratio | 95% CI for OR |
|---|---|---|---|---|---|
| Intercept | −3.2841 | (−5.2093, −1.359) | 0.0008 | 0.0375 | (0.0055, 0.2569) |
| Initial β-hCG | 0.0171 | (0.0079, 0.0264) | 0.0003 | 1.0173 | (1.0079, 1.0268) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Komarnicki, P.; Gut, P.; Cieślewicz, M.; Musiałkiewicz, J.; Maciejewski, A.; Czupińska, M.; Mastorakos, G.; Ruchała, M. Serum β-hCG as a Biomarker in Pancreatic Neuroendocrine Tumors: Rethinking Single-Analyte Approach. Cancers 2024, 16, 2060. https://doi.org/10.3390/cancers16112060
Komarnicki P, Gut P, Cieślewicz M, Musiałkiewicz J, Maciejewski A, Czupińska M, Mastorakos G, Ruchała M. Serum β-hCG as a Biomarker in Pancreatic Neuroendocrine Tumors: Rethinking Single-Analyte Approach. Cancers. 2024; 16(11):2060. https://doi.org/10.3390/cancers16112060
Chicago/Turabian StyleKomarnicki, Paweł, Paweł Gut, Maja Cieślewicz, Jan Musiałkiewicz, Adam Maciejewski, Michalina Czupińska, George Mastorakos, and Marek Ruchała. 2024. "Serum β-hCG as a Biomarker in Pancreatic Neuroendocrine Tumors: Rethinking Single-Analyte Approach" Cancers 16, no. 11: 2060. https://doi.org/10.3390/cancers16112060
APA StyleKomarnicki, P., Gut, P., Cieślewicz, M., Musiałkiewicz, J., Maciejewski, A., Czupińska, M., Mastorakos, G., & Ruchała, M. (2024). Serum β-hCG as a Biomarker in Pancreatic Neuroendocrine Tumors: Rethinking Single-Analyte Approach. Cancers, 16(11), 2060. https://doi.org/10.3390/cancers16112060






