Improving Hepatocellular Carcinoma Surveillance Outcomes in Patients with Cirrhosis after Hepatitis C Cure: A Modelling Study
Abstract
:Simple Summary
Abstract
1. Introduction
Aims
2. Methods
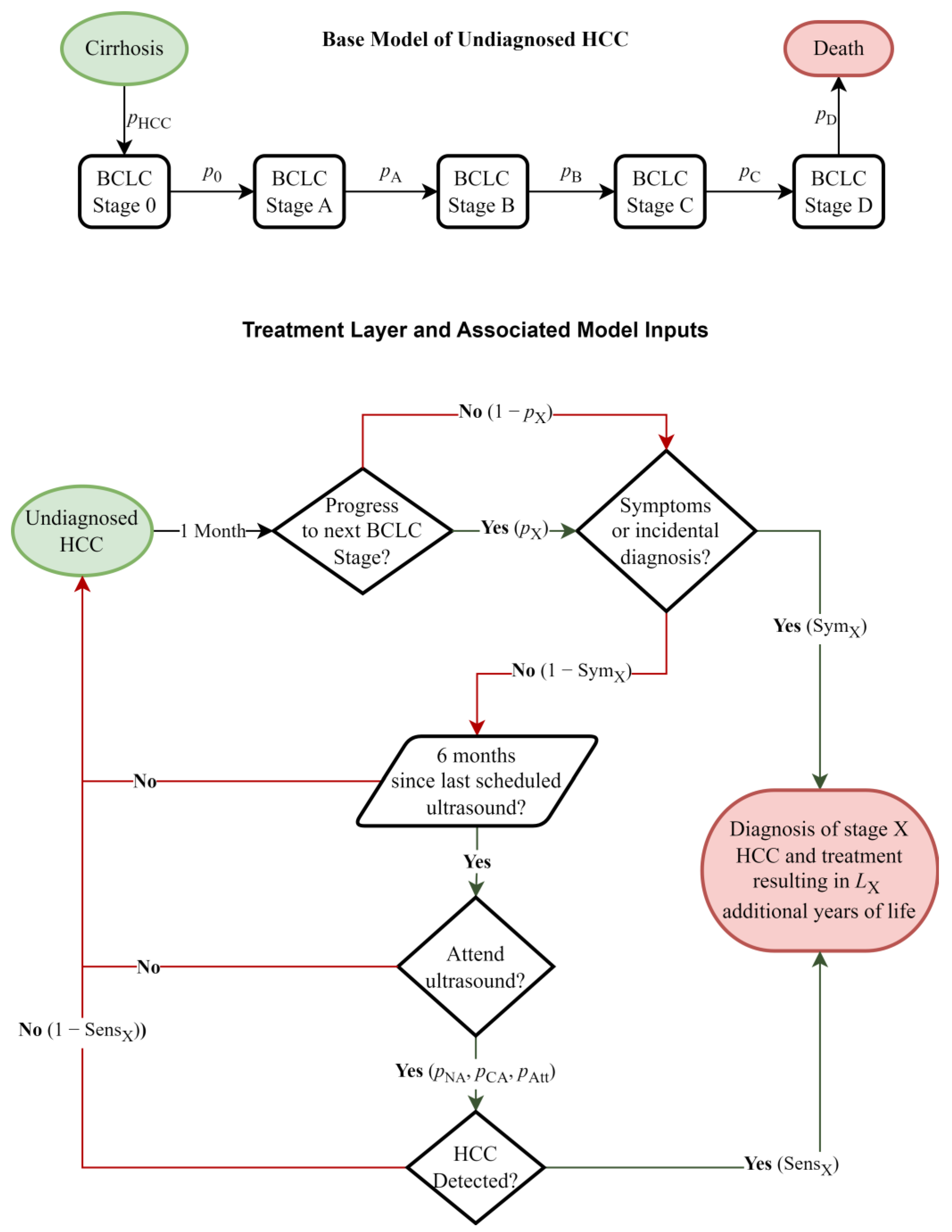
2.1. Model Structure
2.2. Data and Calibrations
2.2.1. Monthly Transition Probabilities, Ultrasound Sensitivity, Symptom Likelihood
2.2.2. Data Sources
2.3. Relative Impact of Changes to Modifiable Factors on Life Years Gained
2.4. Development of Specific Intervention Scenarios
2.5. Sensitivity and Uncertainty Analyses
3. Results
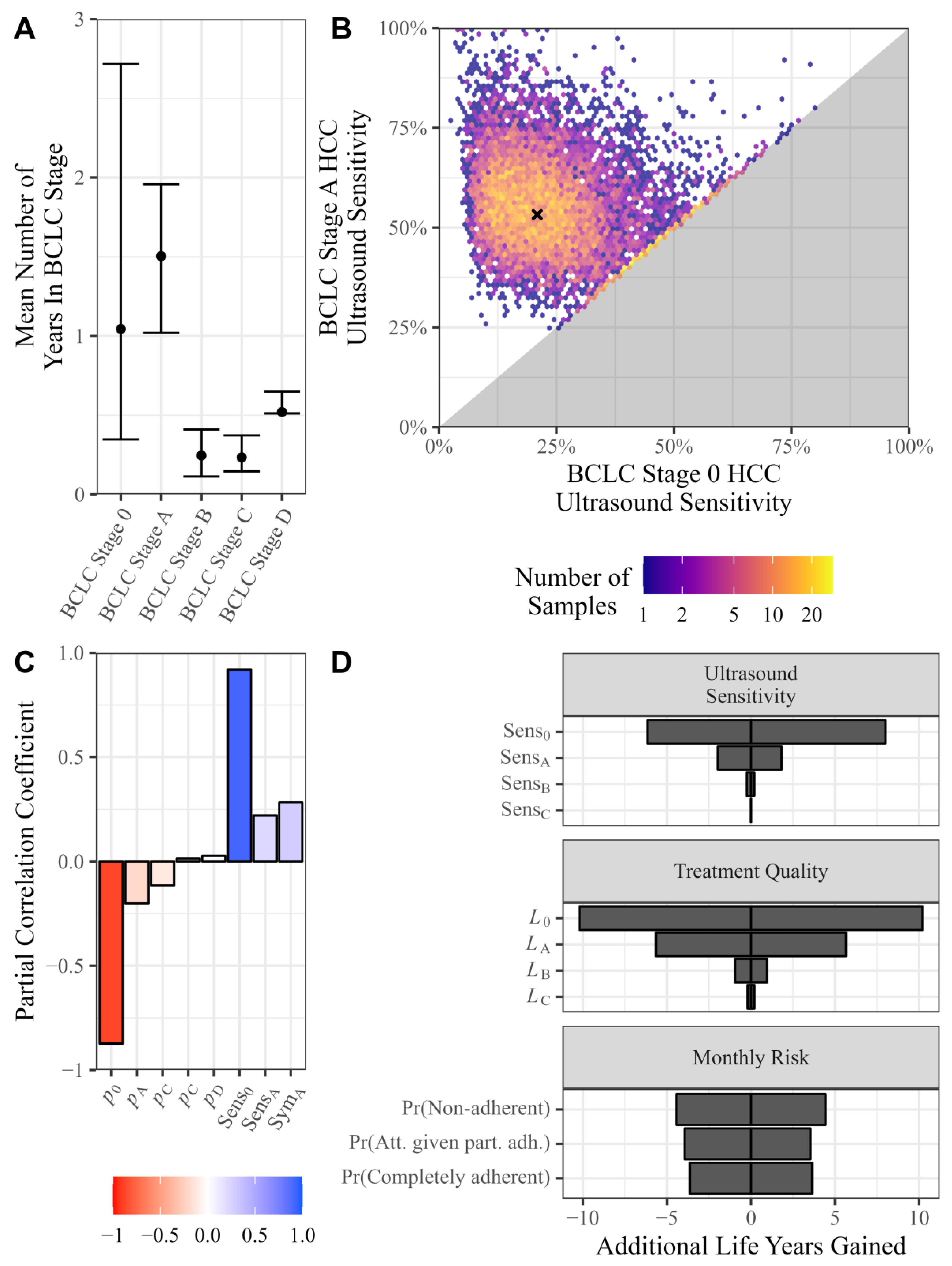
3.1. Data and Calibrations
Monthly Transition Probabilities, Ultrasound Sensitivity, Symptom Likelihood
3.2. Relative Impact of Changes to Modifiable Factors on Life Years Gained
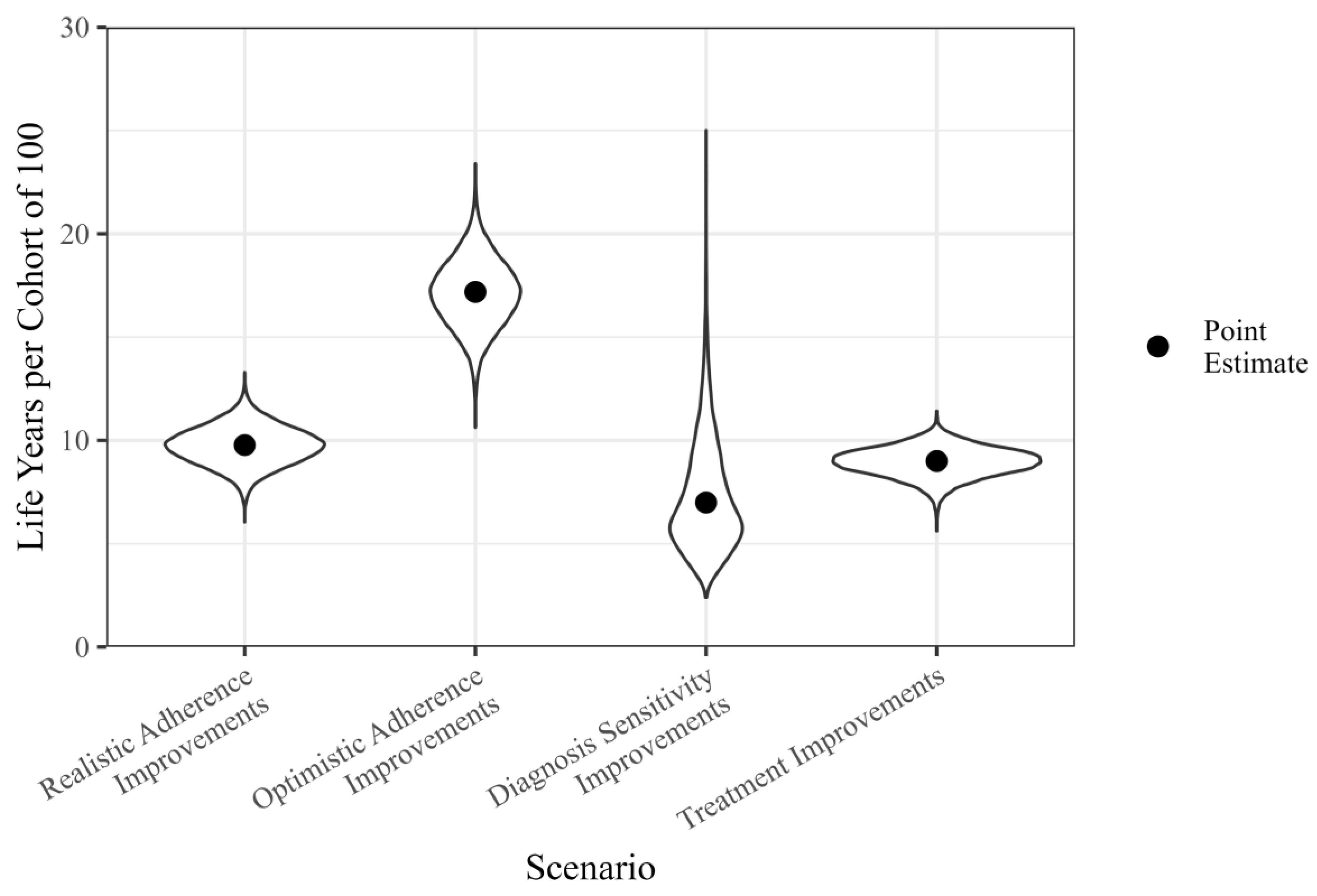
3.3. Life Years Gained by Specific Intervention Scenarios
3.4. Sensitivity and Uncertainty Analysis
4. Discussion
4.1. Implications for Future Work
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Llovet, J.M.; Kelley, R.K.; Villanueva, A.; Singal, A.G.; Pikarsky, E.; Roayaie, S.; Lencioni, R.; Koike, K.; Zucman-Rossi, J.; Finn, R.S. Hepatocellular carcinoma. Nat. Rev. Dis. Primers 2021, 7, 6. [Google Scholar] [CrossRef]
- Hong, T.P.; Gow, P.; Fink, M.; Dev, A.; Roberts, S.; Nicoll, A.; Lubel, J.; Kronborg, I.; Arachchi, N.; Ryan, M.; et al. Novel population-based study finding higher than reported hepatocellular carcinoma incidence suggests an updated approach is needed. Hepatology 2016, 63, 1205–1212. Available online: https://aasldpubs.onlinelibrary.wiley.com/doi/abs/10.1002/hep.28267 (accessed on 9 November 2023). [CrossRef]
- Galle, P.R.; Forner, A.; Llovet, J.M.; Mazzaferro, V.; Piscaglia, F.; Raoul, J.-L.; Schirmacher, P.; Vilgrain, V. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef]
- Howell, J.; Majumdar, A.; Fink, M.A.; Byrne, M.; McCaughan, G.; Strasser, S.I.; Crawford, M.; Hodgkinson, P.; Stuart, K.A.; Tallis, C.; et al. Turning the Tide on Hepatitis C Virus-Related Liver Transplantation: The Return on Investment in Hepatitis C Virus Treatment in Australia and New Zealand. Liver Transplant. 2022, 28, 236–246. Available online: https://journals.lww.com/10.1002/lt.26329 (accessed on 9 November 2023). [CrossRef]
- Kanda, T.; Goto, T.; Hirotsu, Y.; Moriyama, M.; Omata, M. Molecular Mechanisms Driving Progression of Liver Cirrhosis towards Hepatocellular Carcinoma in Chronic Hepatitis B and C Infections: A Review. Int. J. Mol. Sci. 2019, 20, 1358. Available online: https://www.ncbi.nlm.nih.gov/pubmed/30889843 (accessed on 2 October 2023). [CrossRef]
- Burnet Institute; Kirby Institute. Australia’s Progress towards Hepatitis C Elimination: Annual Report 2023; Burnet Institute: Melbourne, Australia, 2023. [Google Scholar]
- Pawlotsky, J.-M.; Negro, F.; Aghemo, A.; Berenguer, M.; Dalgard, O.; Dusheiko, G.; Marra, F.; Puoti, M.; Wedemeyer, H. EASL recommendations on treatment of hepatitis C: Final update of the series. J. Hepatol. 2020, 73, 1170–1218. Available online: https://www.sciencedirect.com/science/article/pii/S0168827820305481 (accessed on 2 October 2023). [CrossRef]
- Hong, T.P.; Gow, P.J.; Fink, M.; Dev, A.; Roberts, S.K.; Nicoll, A.; Lubel, J.S.; Kronborg, I.; Arachchi, N.; Ryan, M.; et al. Surveillance improves survival of patients with hepatocellular carcinoma: A prospective population-based study. Med. J. Aust. 2018, 209, 348–354. [Google Scholar] [CrossRef]
- Ghany, M.G.; Morgan, T.R.; Panel A-IHCG. Hepatitis C Guidance 2019 Update: American Association for the Study of Liver Diseases-Infectious Diseases Society of America Recommendations for Testing, Managing, and Treating Hepatitis C Virus Infection. Hepatology 2020, 71, 686–721. Available online: https://www.ncbi.nlm.nih.gov/pubmed/31816111 (accessed on 2 October 2023). [CrossRef]
- Omata, M.; Cheng, A.-L.; Kokudo, N.; Kudo, M.; Lee, J.M.; Jia, J.; Tateishi, R.; Han, K.-H.; Chawla, Y.K.; Shiina, S.; et al. Asia-pacific clinical practice guidelines on the management of hepatocellular carcinoma: A 2017 update. Hepatol. Int. 2017, 11, 317–370. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5491694/ (accessed on 8 December 2023). [CrossRef]
- Cancer Council Australia Hepatocellular Carcinoma Surveillance Working Group. Clinical Practice Guidelines for Hepatocellular Carcinoma Surveillance for People at High Risk in Australia; Cancer Council Australia: Sydney, Australia, 2023. [Google Scholar]
- Farhang Zangneh, H.; Wong, W.W.L.; Sander, B.; Bell, C.M.; Mumtaz, K.; Kowgier, M.; van der Meer, A.J.; Cleary, S.P.; Janssen, H.L.; Chan, K.K.; et al. Cost Effectiveness of Hepatocellular Carcinoma Surveillance After a Sustained Virologic Response to Therapy in Patients with Hepatitis C Virus Infection and Advanced Fibrosis. Clin. Gastroenterol. Hepatol. 2019, 17, 1840–1849.e16. [Google Scholar] [CrossRef]
- Mueller, P.P.; Chen, Q.; Ayer, T.; Nemutlu, G.S.; Hajjar, A.; Bethea, E.D.; Peters, M.L.B.; Lee, B.P.; Janjua, N.Z.; Kanwal, F.; et al. Duration and cost-effectiveness of hepatocellular carcinoma surveillance in hepatitis C patients after viral eradication. J. Hepatol. 2022, 77, 55–62. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0168827822000745 (accessed on 27 June 2024). [CrossRef] [PubMed]
- Chhatwal, J.; Hajjar, A.; Mueller, P.P.; Nemutlu, G.; Kulkarni, N.; Peters, M.L.B.; Kanwal, F. Hepatocellular Carcinoma Incidence Threshold for Surveillance in Virologically Cured Hepatitis C Individuals. Clin. Gastroenterol. Hepatol. 2024, 22, 91–101.e6. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1542356523004469 (accessed on 27 June 2024). [CrossRef]
- Singal, A.G.; Ioannou, G.N. For Whom is Hepatocellular Carcinoma Surveillance After Sustained Virologic Response Cost-Effective? Clin. Gastroenterol. Hepatol. 2019, 17, 1732–1735. [Google Scholar] [CrossRef]
- Mendiratta-Lala, M.; Fetzer, D.; Kamaya, A.; Parikh, N.D.; Singal, A.G. The Future Role of Abdominal US in Hepatocellular Carcinoma Surveillance. Radiology 2024, 311, e232624. Available online: http://pubs.rsna.org/doi/10.1148/radiol.232624 (accessed on 27 June 2024). [CrossRef]
- Lee, J.S.; Cho, I.R.; Lee, H.W.; Jeon, M.Y.; Lim, T.S.; Baatarkhuu, O.; Kim, D.Y.; Han, K.-H.; Park, J.Y. Conditional Survival Estimates Improve Over Time for Patients with Hepatocellular Carcinoma: An Analysis for Nationwide Korea Cancer Registry Database. Cancer Res. Treat. 2019, 51, 1347–1356. [Google Scholar] [CrossRef] [PubMed]
- Sangiovanni, A.; Colombo, M. Surveillance for hepatocellular carcinoma in patients with advanced liver fibrosis. Saudi J. Gastroenterol. 2021, 27, 64–72. [Google Scholar]
- Ioannou, G.N.; Beste, L.A.; Green, P.K.; Singal, A.G.; Tapper, E.B.; Waljee, A.K.; Sterling, R.K.; Feld, J.J.; Kaplan, D.E.; Taddei, T.H.; et al. Increased Risk for Hepatocellular Carcinoma Persists Up to 10 Years After HCV Eradication in Patients with Baseline Cirrhosis or High FIB-4 Scores. Gastroenterology 2019, 157, 1264–1278.e4. [Google Scholar] [CrossRef]
- Nahon, P.; Bourcier, V.; Layese, R.; Audureau, E.; Cagnot, C.; Marcellin, P.; Guyader, D.; Fontaine, H.; Larrey, D.; De Lédinghen, V.; et al. Eradication of Hepatitis C Virus Infection in Patients with Cirrhosis Reduces Risk of Liver and Non-Liver Complications. Gastroenterology 2017, 152, 142–156.e2. [Google Scholar] [CrossRef]
- El-Serag, H.B.; Kanwal, F.; Richardson, P.; Kramer, J. Risk of hepatocellular carcinoma after sustained virological response in Veterans with hepatitis C virus infection. Hepatology 2016, 64, 130–137. Available online: https://aasldpubs.onlinelibrary.wiley.com/doi/abs/10.1002/hep.28535 (accessed on 2 October 2023). [CrossRef]
- van der Meer, A.J.; Feld, J.J.; Hofer, H.; Almasio, P.L.; Calvaruso, V.; Fernández-Rodríguez, C.M.; Aleman, S.; Ganne-Carrié, N.; D’Ambrosio, R.; Pol, S.; et al. Risk of cirrhosis-related complications in patients with advanced fibrosis following hepatitis C virus eradication. J. Hepatol. 2017, 66, 485–493. Available online: https://www.sciencedirect.com/science/article/pii/S0168827816305827 (accessed on 2 October 2023). [CrossRef]
- Khalili, K.; Menezes, R.; Kim, T.K.; Yazdi, L.K.; Jang, H.-J.; Sharma, S.; Feld, J.; Sherman, M. The Effectiveness of Ultrasound Surveillance for Hepatocellular Carcinoma in a Canadian Centre and Determinants of Its Success. Can. J. Gastroenterol. Hepatol. 2015, 29, 563893. [Google Scholar] [CrossRef]
- Tzartzeva, K.; Obi, J.; Rich, N.E.; Parikh, N.D.; Marrero, J.A.; Yopp, A.; Waljee, A.K.; Singal, A.G. Surveillance Imaging and Alpha Fetoprotein for Early Detection of Hepatocellular Carcinoma in Patients with Cirrhosis: A Meta-analysis. Gastroenterology 2018, 154, 1706–1718.e1. [Google Scholar] [CrossRef]
- Giannini, E.G.; Farinati, F.; Ciccarese, F.; Pecorelli, A.; Rapaccini, G.L.; Di Marco, M.; Benvegnu, L.; Caturelli, E.; Zoli, M.; Borzio, F.; et al. Prognosis of untreated hepatocellular carcinoma. Hepatology 2015, 61, 184–190. Available online: https://aasldpubs.onlinelibrary.wiley.com/doi/abs/10.1002/hep.27443 (accessed on 10 October 2023). [CrossRef]
- Bruix, J.; Gores, G.J.; Mazzaferro, V. Hepatocellular carcinoma: Clinical frontiers and perspectives. Gut 2014, 63, 844–855. Available online: https://gut.bmj.com/content/gutjnl/63/5/844.full.pdf (accessed on 2 October 2023). [CrossRef]
- Singal, A.; Volk, M.L.; Waijee, A.; Salgia, R.; Higgins, P.; Rogers, M.A.M.; Marrero, J.A. Meta-analysis: Surveillance with ultrasound for early-stage hepatocellular carcinoma in patients with cirrhosis. Aliment. Pharmacol. Ther. 2009, 30, 37–47. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1365-2036.2009.04014.x (accessed on 2 October 2022). [CrossRef]
- Singal, A.G.; Tiro, J.A.; Murphy, C.C.; Marrero, J.A.; McCallister, K.; Fullington, H.; Mejias, C.; Waljee, A.K.; Bishop, W.P.; Santini, N.O.; et al. Mailed Outreach Invitations Significantly Improve HCC Surveillance Rates in Patients with Cirrhosis: A Randomized Clinical Trial. Hepatology 2019, 69, 121–130. Available online: https://aasldpubs.onlinelibrary.wiley.com/doi/abs/10.1002/hep.30129 (accessed on 2 October 2023). [CrossRef]
- Australian Institute of Health and Welfare. BreastScreen Australia Monitoring Report 2023; AIHW: Canberra, Australia, 2023.
- Australian Institute of Health and Welfare. National Cervical Screening Program Monitoring Report 2022; AIHW: Canberra, Australia, 2022. Available online: https://www.aihw.gov.au/reports/cancer-screening/national-cervical-screening-program-monitoring-rep/summary-1 (accessed on 10 November 2023).
- Wolf, E.; Rich, N.E.; Marrero, J.A.; Parikh, N.D.; Singal, A.G. Use of Hepatocellular Carcinoma Surveillance in Patients with Cirrhosis: A Systematic Review and Meta-Analysis. Hepatology 2021, 73, 713–725. Available online: https://www.ncbi.nlm.nih.gov/pubmed/32383272 (accessed on 2 October 2023). [CrossRef]
- Singal, A.G.; Yopp, A.; Skinner, C.S.; Packer, M.; Lee, W.M.; Tiro, J.A. Utilization of Hepatocellular Carcinoma Surveillance Among American Patients: A Systematic Review. J. Gen. Intern. Med. 2012, 27, 861–867. [Google Scholar] [CrossRef]
- Robinson, A.; Tavakoli, H.; Cheung, R.; Liu, B.; Bhuket, T.; Wong, R.J. Low Rates of Retention Into Hepatocellular Carcinoma (HCC) Surveillance Program After Initial HCC Screening. J. Clin. Gastroenterol. 2019, 53, 65–70. Available online: https://www.ncbi.nlm.nih.gov/pubmed/29629906 (accessed on 2 October 2023). [CrossRef]
- Kennedy, N.A.; Rodgers, A.; Altus, R.; McCormick, R.; Wundke, R.; Wigg, A.J. Optimisation of hepatocellular carcinoma surveillance in patients with viral hepatitis: A quality improvement study. Intern. Med. J. 2013, 43, 772–777. Available online: https://www.ncbi.nlm.nih.gov/pubmed/23611607 (accessed on 2 October 2023). [CrossRef]
- Flores, J.E.; Hong, T.; Thompson, A.J.; Ryan, M.; Valaydon, Z.; Roberts, S.; Nicoll, A.; Sood, S.; Gow, P.; Lewis, D.; et al. Metabolic-associated fatty liver disease and alcohol-related liver disease are leading causes of hepatocellular carcinoma: Interim analysis of the HOMER-2 study. J. Gastroenterol. Hepatol. 2022, 73, 56–57. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1111/jgh.15951 (accessed on 2 October 2023).
- Majeed, A.; Roberts, S.K.; Kemp, W. RE: No Association Between Screening for Hepatocellular Carcinoma and Reduced Cancer-Related Mortality in Patients with Cirrhosis. Gastroenterology 2019, 156, 1217. Available online: https://www.ncbi.nlm.nih.gov/pubmed/30543798 (accessed on 2 October 2023). [CrossRef]
- Del Poggio, P.; Mazzoleni, M.; Lazzaroni, S.; D’Alessio, A. Surveillance for hepatocellular carcinoma at the community level: Easier said than done. World J. Gastroenterol. 2021, 27, 6180–6190. [Google Scholar] [CrossRef]
- Rich, N.E.; John, B.V.; Parikh, N.D.; Rowe, I.; Mehta, N.; Khatri, G.; Thomas, S.M.; Anis, M.; Mendiratta-Lala, M.; Hernandez, C.; et al. Hepatocellular Carcinoma Demonstrates Heterogeneous Growth Patterns in a Multicenter Cohort of Patients with Cirrhosis. Hepatology 2020, 72, 1654–1665. [Google Scholar] [CrossRef]
- Singal, A.G.; Tayob, N.; Mehta, A.; Marrero, J.A.; El-Serag, H.; Jin, Q.; de Viteri, C.S.; Fobar, A.; Parikh, N.D. GALAD demonstrates high sensitivity for HCC surveillance in a cohort of patients with cirrhosis. Hepatology 2022, 75, 541–549. Available online: https://journals.lww.com/hep/abstract/2022/03000/galad_demonstrates_high_sensitivity_for_hcc.9.aspx (accessed on 13 November 2023). [CrossRef]
| BCLC Stage | Description |
|---|---|
| 0 (Very early stage) |
|
| A (Early stage) |
|
| B (Intermediate stage) |
|
| C (Advanced stage) |
|
| D (Terminal stage) |
|
| Parameter | Description | Value | References |
|---|---|---|---|
| Annual Probability of Developing HCC | 2.2% | [18,19,20,21,22] | |
| Ultrasound Sensitivity for BCLC B HCC | 84.0% | Informed by [8,23,24] | |
| Ultrasound Sensitivity for BCLC C HCC | 90.0% | Informed by [8,23,24] | |
| Ultrasound Sensitivity for BCLC D HCC | 95.0% | Informed by [8,23,24] | |
| Additional Life Expectancy after Stage 0 Diagnosis in Years | 12.5 | Derived from [17] | |
| Additional Life Expectancy after Stage A Diagnosis in Years | 3.6 | Derived from [17] | |
| Additional Life Expectancy after Stage B Diagnosis in Years | 1.7 | Derived from [17] | |
| Additional Life Expectancy after Stage C Diagnosis in Years | 0.25 | Derived from [17] | |
| Additional Life Expectancy after Stage D Diagnosis in Years | 0 | Derived from [17] | |
| Monthly Probability of Developing Symptoms at Stage 0 | 0.0% | Informed by [8] | |
| Monthly Probability of Developing Symptoms at Stage C | 31.9% | Informed by [8] | |
| Monthly Probability of Developing Symptoms at Stage D | 100.0% | Informed by [8] |
| Parameter | Description | Point Estimate (95% Confidence Interval) |
|---|---|---|
| Monthly Probability of Progressing from BCLC 0 to A | 8.0% (3.1%, 24.0%) | |
| Monthly Probability of Progressing from BCLC A to B | 5.5% (4.3%, 8.2%) | |
| Monthly Probability of Progressing from BCLC B to C | 34.4% (20.3%, 73.7%) | |
| Monthly Probability of Progressing from BCLC C to D | 35.2% (22.4%, 57.4%) | |
| Monthly Probability of Progressing from BCLC D to Death | 16.0% (12.8%, 16.3%) | |
| Ultrasound Sensitivity for BCLC 0 HCC | 20.9% (8.0%, 53.7%) | |
| Ultrasound Sensitivity for BCLC A HCC | 53.3% (36.1%, 82.0%) | |
| Monthly Probability of Developing Symptoms at Stage A | 1.0% (0.7%, 1.6%) | |
| Monthly Probability of Developing Symptoms at Stage B | 8.1% (5.4%, 12.5%) | |
| Probability of being completely non-adherent to surveillance | 31.5% (22.9%, 41.1%) | |
| Probability of being completely adherent to surveillance | 34.3% (25.4%, 44.0%) | |
| Probability of attending ultrasound if partially adherent | 44.9% (35.0%, 55.0%) |
| Scenario | Optimistic Adherence Improvements | Realistic Adherence Improvements | Treatment Improvements |
|---|---|---|---|
| Diagnostic sensitivity improvements | 98.4% | 80.1% | 74.7% |
| Treatment improvements | 100.0% | 73.6% | |
| Realistic adherence improvements | 100.0% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cumming, J.; Scott, N.; Howell, J.; Flores, J.E.; Pavlyshyn, D.; Hellard, M.E.; Winata, L.S.-h.; Ryan, M.; Sutherland, T.; Thompson, A.J.; et al. Improving Hepatocellular Carcinoma Surveillance Outcomes in Patients with Cirrhosis after Hepatitis C Cure: A Modelling Study. Cancers 2024, 16, 2745. https://doi.org/10.3390/cancers16152745
Cumming J, Scott N, Howell J, Flores JE, Pavlyshyn D, Hellard ME, Winata LS-h, Ryan M, Sutherland T, Thompson AJ, et al. Improving Hepatocellular Carcinoma Surveillance Outcomes in Patients with Cirrhosis after Hepatitis C Cure: A Modelling Study. Cancers. 2024; 16(15):2745. https://doi.org/10.3390/cancers16152745
Chicago/Turabian StyleCumming, Jacob, Nick Scott, Jessica Howell, Joan Ericka Flores, Damian Pavlyshyn, Margaret E. Hellard, Leon Shin-han Winata, Marno Ryan, Tom Sutherland, Alexander J. Thompson, and et al. 2024. "Improving Hepatocellular Carcinoma Surveillance Outcomes in Patients with Cirrhosis after Hepatitis C Cure: A Modelling Study" Cancers 16, no. 15: 2745. https://doi.org/10.3390/cancers16152745





