Phase I Clinical Trial on Pleural Mesothelioma Using Neoadjuvant Local Administration of Paclitaxel-Loaded Mesenchymal Stromal Cells (PACLIMES Trial): Study Rationale and Design
Abstract
Simple Summary
Abstract
1. Introduction
2. Pre-Clinical and Clinical Background
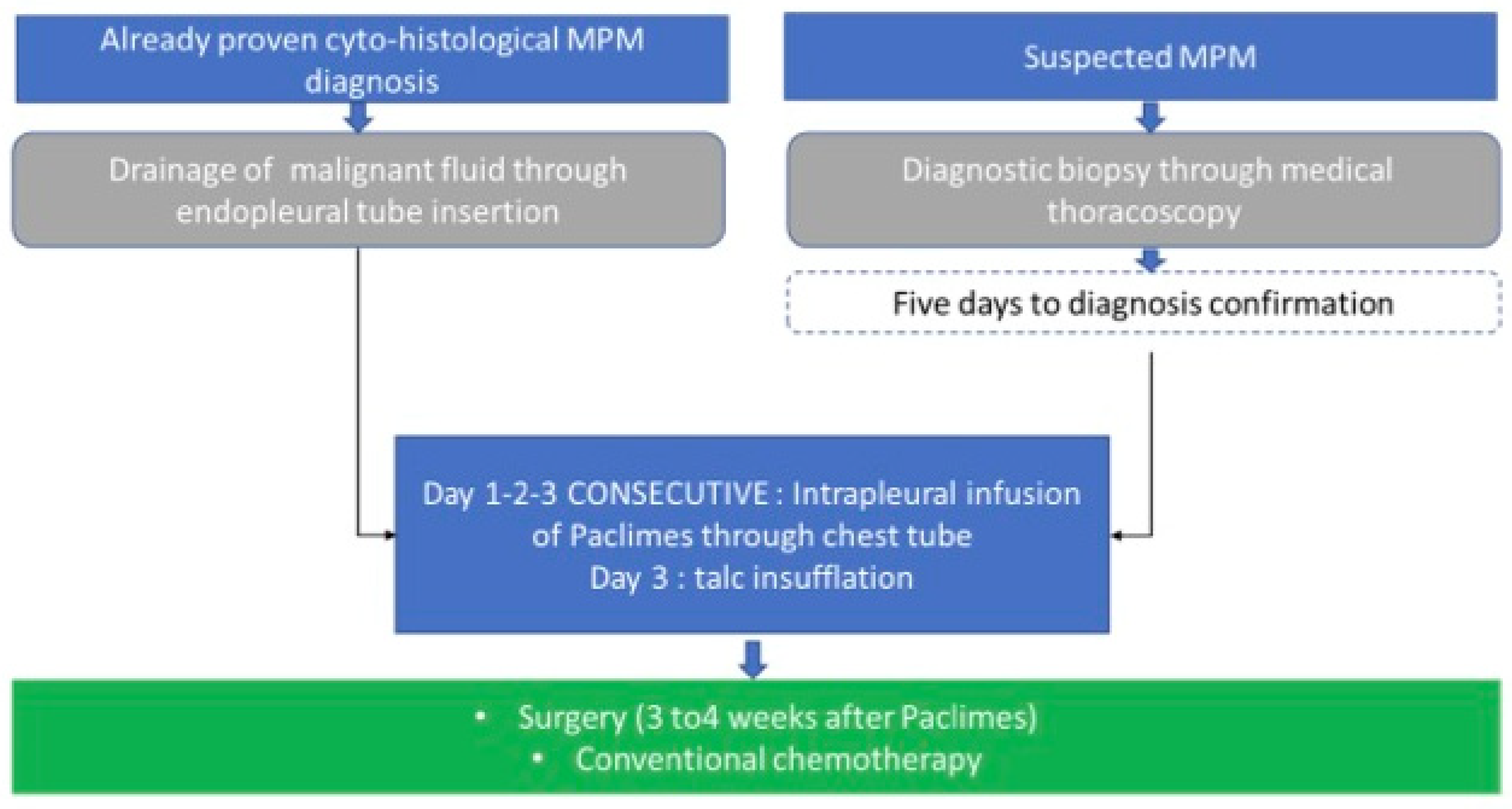
3. Trial Design
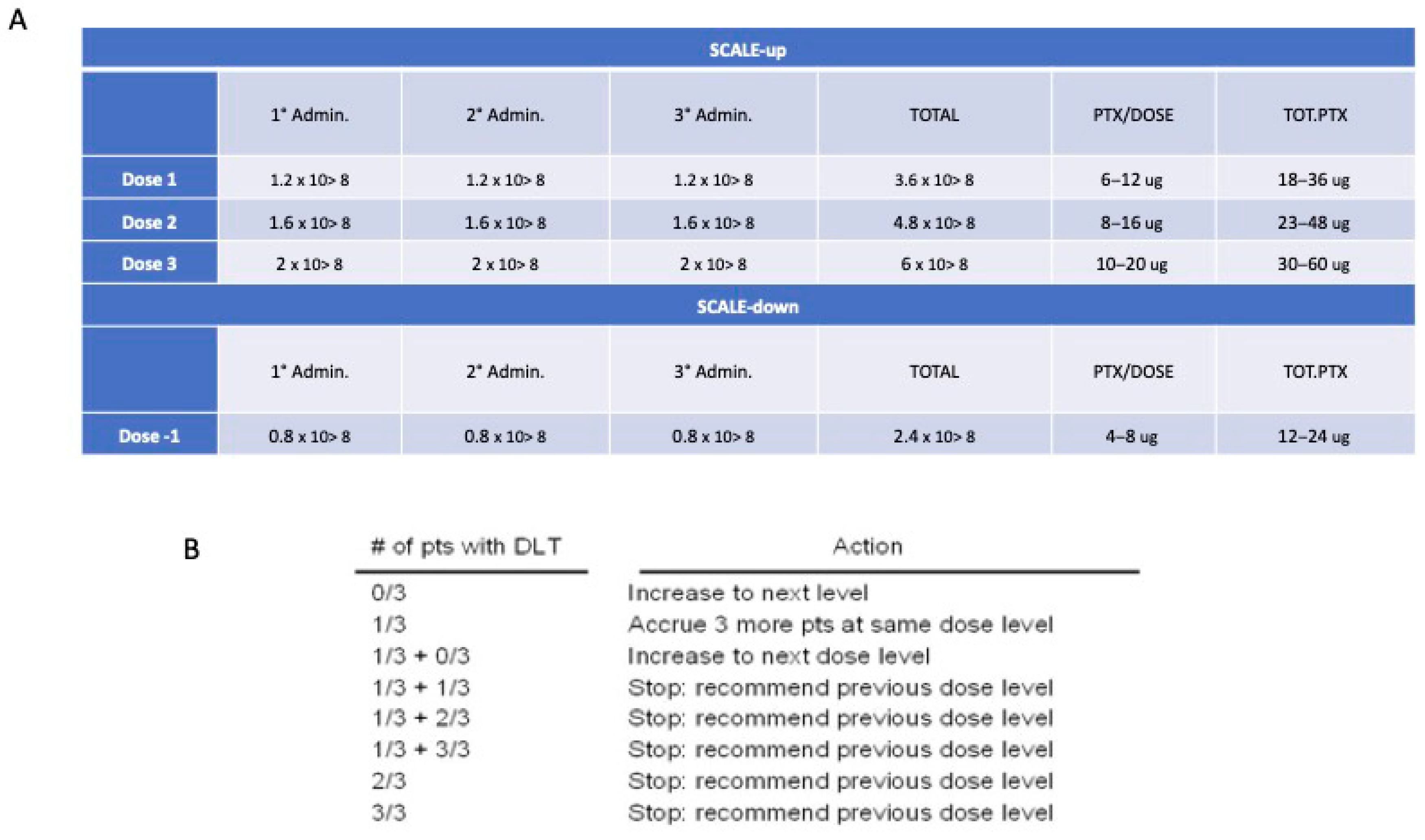
3.1. Route of Administration and Maximum Dosage Allowed
3.2. Statistical Methodology
4. Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nicolini, F.; Bocchini, M.; Bronte, G.; Delmonte, A.; Guidoboni, M.; Crinò, L.; Mazza, M. Malignant Pleural Mesothelioma: State-of-the-Art on Current Therapies and Promises for the Future. Front. Oncol. 2020, 9, 1519. [Google Scholar] [CrossRef] [PubMed]
- Alpert, N.; van Gerwen, M.; Taioli, E. Epidemiology of mesothelioma in the 21st century in Europe and the United States, 40 years after restricted/banned asbestos use. Transl. Lung Cancer Res. 2020, 9 (Suppl. S1), S28–S38. [Google Scholar] [CrossRef]
- Davis, A.P.; Kao, S.C.; Clarke, S.J.; Boyer, M.; Pavlakis, N. Emerging biological therapies for the treatment of malignant pleural mesothelioma. Expert Opin. Emerg. Drugs 2021, 26, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Yang, H.; Schmid, R.A.; Peng, R.W. Therapeutic Landscape of Malignant Pleural Mesothelioma: Collateral Vulnerabilities and Evolutionary Dependencies in the Spotlight. Front. Oncol. 2020, 10, 579464. [Google Scholar] [CrossRef]
- Cedres, S.; Valdivia, A.; Iranzo, P.; Callejo, A.; Pardo, N.; Navarro, A.; Martinez-Marti, A.; Assaf-Pastrana, J.D.; Felip, E.; Garrido, P. Current State-of-the-Art Therapy for Malignant Pleural Mesothelioma and Future Options Centered on Immunotherapy. Cancers 2023, 15, 5787. [Google Scholar] [CrossRef]
- de Gooijer, C.J.; Baas, P.; Burgers, J.A. Current chemotherapy strategies in malignant pleural mesothelioma. Transl. Lung Cancer Res. 2018, 7, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Mirra, L.; Beretta, G.L.; Lisini, D.; Marcianti, A.; Spampinato, E.; Corno, C.; Costantino, M.; Corsico, A.; Stella, G.M.; Perego, P. Therapeutic Strategies to Improve the Treatment of Pleural Mesothelioma. Curr. Med. Chem. 2024. [Google Scholar] [CrossRef]
- Scherpereel, A.; Opitz, I.; Berghmans, T.; Psallidas, I.; Glatzer, M.; Rigau, D.; Astoul, P.; Bölükbas, S.; Boyd, J.; Coolen, J.; et al. ERS/ESTS/EACTS/ESTRO guidelines for the management of malignant pleural mesothelioma. Eur. Respir. J. 2020, 55, 1900953. [Google Scholar] [CrossRef] [PubMed]
- Scherpereel, A.; Wallyn, F.; Albelda, S.M.; Munck, C. Novel therapies for malignant pleural mesothelioma. Lancet Oncol. 2018, 19, e161–e172. [Google Scholar] [CrossRef]
- Wei, X.; Yang, X.; Han, Z.P.; Qu, F.F.; Shao, L.; Shi, Y.F. Mesenchymal stem cells: A new trend for cell therapy. Acta Pharmacol. Sin. 2013, 34, 747–754. [Google Scholar] [CrossRef]
- Sasportas, L.S.; Kasmieh, R.; Wakimoto, H.; Hingtgen, S.; van de Water, J.A.; Mohapatra, G.; Figueiredo, J.L.; Martuza, R.L.; Weissleder, R. Shah Assessment of therapeutic efficacy and fate of engineered human mesenchymal stem cells for cancer therapy. Proc. Natl. Acad. Sci. USA 2009, 106, 4822–4827. [Google Scholar] [CrossRef] [PubMed]
- Studeny, M.; Marini, F.C.; Champlin, R.E.; Zompetta, C.; Fidler, I.J.; Andreeff, M. Bone marrow-derived mesenchymal stem cells as vehicles for interferon-beta delivery into tumors. Cancer Res. 2002, 62, 3603–3608. [Google Scholar] [PubMed]
- Loebinger, M.R.; Eddaoudi, A.; Davies, D.; Janes, S.M. Mesenchymal stem cell delivery of TRAIL can eliminate metastatic cancer. Cancer Res. 2009, 69, 4134–4142. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.G.; Liu, K.D.; Zhuo, H.; Caballero, L.; McMillan, M.; Fang, X.; Cosgrove, K.; Vojnik, R.; Calfee, C.S.; Lee, J.W.; et al. Mesenchymal stem (stromal) cells for treatment of ARDS: A phase 1 clinical trial. Lancet Respir. Med. 2014, 3, 24–32. [Google Scholar] [CrossRef]
- Freyman, T.; Polin, G.; Osman, H.; Crary, J.; Lu, M.; Cheng, L.; Palasis, M.; Wilensky, R.L. A quantitative, randomized study evaluating three methods of mesenchymal stem cell delivery following myocardial infarction. Eur. Heart J. 2006, 27, 1114–1122. [Google Scholar] [CrossRef]
- Sanchez-Diaz, M.; Quiñones-Vico, M.I.; Sanabria de la Torre, R.; Montero-Vílchez, T.; Sierra-Sánchez, A.; Molina-Leyva, A.; Arias-Santiago, S. Biodistribution of Mesenchymal Stromal Cells after Administration in Animal Models and Humans: A Systematic Review. J. Clin. Med. 2021, 10, 2925. [Google Scholar] [CrossRef]
- Pessina, A.; Coccè, V.; Pascucci, L.; Bonomi, A.; Cavicchini, L.; Sisto, F.; Ferrari, M.; Ciusani, E.; Crovace, A.; Falchetti, M.L.; et al. Mesenchymal stromal cells primed with Paclitaxel attract and kill leukaemia cells, inhibit angiogenesis and improve survival of leukaemia-bearing mice. Br. J. Haematol. 2013, 160, 766–778. [Google Scholar] [CrossRef] [PubMed]
- Petrella, F.; Coccè, V.; Masia, C.; Milani, M.; Salè, E.O.; Alessandri, G.; Parati, E.; Sisto, F.; Pentimalli, F.; Brini, A.T.; et al. Paclitaxel-releasing mesenchymal stromal cells inhibit in vitro proliferation of human mesothelioma cells. Biomed. Pharmacother. 2017, 87, 755–758. [Google Scholar] [CrossRef]
- Chulpanova, D.S.; Kitaeva, K.V.; Tazetdinova, L.G.; James, V.; Rizvanov, A.A.; Solovyeva, V.V. Application of Mesenchymal Stem Cells for Therapeutic Agent Delivery in Anti-tumor Treatment. Front. Pharmacol. 2018, 9, 259. [Google Scholar] [CrossRef]
- Kalimuthu, S.; Gangadaran, P.; Rajendran, R.L.; Zhu, L.; Oh, J.M.; Lee, H.W.; Gopal, A.; Baek, S.H.; Jeong, S.Y.; Lee, S.W.; et al. A New Approach for Loading Anticancer Drugs into Mesenchymal Stem Cell-Derived Exosome Mimetics for Cancer Therapy. Front. Pharmacol. 2018, 9, 1116. [Google Scholar] [CrossRef]
- Wang, X.; Omar, O.; Vazirisani, F.; Thomsen, P.; Ekström, K. Mesenchymal stem cell-derived exosomes have altered microRNA profiles and induce osteogenic differentiation depending on the stage of differentiation. PLoS ONE 2018, 13, e0193059. [Google Scholar] [CrossRef]
- Hmadcha, A.; Martin-Montalvo, A.; Gauthier, B.R.; Soria, B.; Capilla-Gonzalez, V. Therapeutic Potential of Mesenchymal Stem Cells for Cancer Therapy. Front. Bioeng. Biotechnol. 2020, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Coccè, V.; Bonelli, M.; La Monica, S.; Alfieri, R.; Doneda, L.; Martegani, E.; Alessandri, G.; Lagrasta, C.A.; Giannì, A.; Sordi, V.; et al. Mesenchymal stromal cells loaded with Paclitaxel (PacliMES) a potential new therapeutic approach on mesothelioma. Biochem. Pharmacol. 2023, 214, 115678. [Google Scholar] [CrossRef] [PubMed]
- Pascucci, L.; Coccè, V.; Bonomi, A.; Ami, D.; Ceccarelli, P.; Ciusani, E.; Viganò, L.; Locatelli, A.; Sisto, F.; Doglia, S.M.; et al. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: A new approach for drug delivery. J. Control. Release. 2014, 192, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Pessina, A.; Bonomi, A.; Coccè, V.; Invernici, G.; Navone, S.; Cavicchini, L.; Sisto, F.; Ferrari, M.; Viganò, L.; Locatelli, A.; et al. Mesenchymal stromal cells primed with paclitaxel provide a new approach for cancer therapy. PLoS ONE 2011, 6, e28321. [Google Scholar] [CrossRef]
- Pessina, A.; Leonetti, C.; Artuso, S.; Benetti, A.; Dessy, E.; Pascucci, L.; Passeri, D.; Orlandi, A.; Berenzi, A.; Bonomi, A.; et al. Drug-releasing mesenchymal cells strongly suppress B16 lung metastasis in a syngeneic murine model. J. Exp. Clin. Cancer Res. 2015, 34, 82. [Google Scholar] [CrossRef]
- Song, N.; Scholtemeijer, M.; Shah, K. Mesenchymal Stem Cell Immunomodulation: Mechanisms and Therapeutic Potential. Trends Pharmacol. Sci. 2020, 41, 653–664. [Google Scholar] [CrossRef]
- Liu, S.; Liu, F.; Zhou, Y.; Jin, B.; Sun, Q.; Guo, S. Immunosuppressive Property of MSCs Mediated by Cell Surface Receptors. Front. Immunol. 2020, 11, 1076. [Google Scholar] [CrossRef]
- Li, Y.; Hao, J.; Hu, Z.; Yang, Y.G.; Zhou, Q.; Sun, L.; Wu, J. Current status of clinical trials assessing mesenchymal stem cell therapy for graft versus host disease: A systematic review. Stem Cell Res. Ther. 2022, 13, 93. [Google Scholar] [CrossRef]
- Galderisi, U.; Peluso, G.; Di Bernardo, G. Clinical Trials Based on Mesenchymal Stromal Cells are Exponentially Increasing: Where are We in Recent Years? Stem Cell Rev. Rep. 2022, 18, 23–36. [Google Scholar] [CrossRef]
- Wang, Y.; Yi, H.; Song, Y. The safety of MSC therapy over the past 15 years: A meta-analysis. Stem Cell Res. Ther. 2021, 12, 545. [Google Scholar] [CrossRef] [PubMed]
- Bonomi, A.; Coccè, V.; Cavicchini, L.; Sisto, F.; Dossena, M.; Balzarini, P.; Portolani, N.; Ciusani, E.; Parati, E.; Alessandri, G.; et al. Adipose tissue-derived stromal cells primed in vitro with paclitaxel acquire anti-tumor activity. Int. J. Immunopathol. Pharmacol. 2013, 26 (Suppl. S1), 33–41. [Google Scholar] [CrossRef] [PubMed]
- Rimoldi, I.; Coccè, V.; Facchetti, G.; Alessandri, G.; Brini, A.T.; Sisto, F.; Parati, E.; Cavicchini, L.; Lucchini, G.; Petrella, F.; et al. Uptake-release by MSCs of a cationic platinum(II) complex active in vitro on human malignant cancer cell lines. Biomed. Pharmacother. 2018, 108, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Zeira, O.; Ghezzi, E.; Pettinari, L.; Re, V.; Lupi, D.M.; Benali, S.L.; Borgonovo, S.; Alessandri, G.; Petrella, F.; Paroni, R.; et al. Case Report: Microfragmented Adipose Tissue Drug Delivery in Canine Mesothelioma: A Case Report on Safety, Feasibility, and Clinical Findings. Front. Vet. Sci. 2021, 7, 585427. [Google Scholar] [CrossRef]
- Alessandri, G.; Coccè, V.; Pastorino, F.; Paroni, R.; Dei Cas, M.; Restelli, F.; Pollo, B.; Gatti, L.; Tremolada, C.; Berenzi, A.; et al. Microfragmented human fat tissue is a natural scaffold for drug delivery: Potential application in cancer chemotherapy. J. Control. Release 2019, 302, 2–18. [Google Scholar] [CrossRef]
- de Weger, V.A.; Beijnen, J.H.; Schellens, J.H. Cellular and clinical pharmacology of the taxanes docetaxel and paclitaxel—A review. Anticancer Drugs 2014, 25, 488–494. [Google Scholar] [CrossRef]
- Yardley, D.A. nab-Paclitaxel mechanisms of action and delivery. J Control. Release. 2013, 170, 365–372. [Google Scholar] [CrossRef]
- Sugarbaker, P.H.; Chang, D. Long-term regional chemotherapy for patients with epithelial malignant peritoneal mesothelioma results in improved survival. Eur. J. Surg. Oncol. 2017, 43, 1228–1235. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Q.; Zhao, W.; Duan, J.; Huang, T.P. Pleural resection and pleural infusion chemotherapy for therapy of malignant pleural mesothelioma. Genet. Mol. Res. 2014, 13, 483–489. [Google Scholar] [CrossRef]
- Lustgarten, D.E.; Deshpande, C.; Aggarwal, C.; Wang, L.C.; Saloura, V.; Vachani, A.; Wang, L.P.; Litzky, L.; Feldman, M.; Creaney, J.; et al. Thymidylate synthase and folyl-polyglutamate synthase are not clinically useful markers of response to pemetrexed in patients with malignant pleural mesothelioma. J. Thorac. Oncol. 2013, 8, 469–477. [Google Scholar] [CrossRef]
- Wahba, N.I.; Khorshid, O.; Abo Elkasem, F.; Bahnassy, A.; Gaafer, R. The Predictive Value of ERCC1, RRM1, and Thymidylate Synthase in Advanced Malignant Pleural Mesothelioma Patients Treated with Platinum-Based Chemotherapy. Asian Pac. J. Cancer Prev. 2023, 24, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Zucali, P.A.; Giovannetti, E.; Destro, A.; Mencoboni, M.; Ceresoli, G.L.; Gianoncelli, L.; Lorenzi, E.; De Vincenzo, F.; Simonelli, M.; Perrino, M.; et al. Thymidylate synthase and excision repair cross-complementing group-1 as predictors of responsiveness in mesothelioma patients treated with pemetrexed/carboplatin. Clin. Cancer Res. 2011, 17, 2581–2590. [Google Scholar] [CrossRef] [PubMed]
- Goldie, J.H.; Coldman, A.J. A mathematic model for relating the drug sensitivity of tumors to their spontaneous mutation rate. Cancer Treat. Rep. 1979, 63, 1727–1733. [Google Scholar] [PubMed]
- Matsumoto, Y.; Takano, H.; Fojo, T. Cellular adaptation to drug exposure: Evolution of the drug-resistant phenotype. Cancer Res. 1997, 57, 5086–5092. [Google Scholar]
- Balmativola, D.; Marchiò, C.; Maule, M.; Chiusa, L.; Annaratone, L.; Maletta, F.; Montemurro, F.; Kulka, J.; Figueiredo, P.; Varga, Z.; et al. Pathological non-response to chemotherapy in a neoadjuvant setting of breast cancer: An inter-institutional study. Breast Cancer Res. Treat. 2014, 148, 511–523. [Google Scholar] [CrossRef]
- Senetta, R.; Miracco, C.; Lanzafame, S.; Chiusa, L.; Caltabiano, R.; Galia, A.; Stella, G.; Cassoni, P. Epidermal growth factor receptor and caveolin-1 coexpression identifies adult supratentorial ependymomas with rapid unfavorable outcomes. Neuro Oncol. 2011, 13, 176–183. [Google Scholar] [CrossRef]
- Xu, L.L.; Yang, Y.; Wang, Z.; Wang, X.J.; Tong, Z.H.; Shi, H.Z. Malignant pleural mesothelioma: Diagnostic value of medical thoracoscopy and long-term prognostic analysis. BMC Pulm. Med. 2018, 18, 56. [Google Scholar] [CrossRef]
- Lisini, D.; Nava, S.; Frigerio, S.; Pogliani, S.; Maronati, G.; Marcianti, A.; Coccè, V.; Bondiolotti, G.; Cavicchini, L.; Paino, F.; et al. Automated Large-Scale Production of Paclitaxel Loaded Mesenchymal Stromal Cells for Cell Therapy Applications. Pharmaceutics 2020, 12, 411. [Google Scholar] [CrossRef]
- Ivy, S.P.; Siu, L.L.; Garrett-Mayer, E.; Rubinstein, L. Approaches to phase 1 clinical trial design focused on safety, efficiency, and selected patient populations: A report from the clinical trial design task force of the national cancer institute investigational drug steering committee. Clin. Cancer Res. 2010, 16, 1726–1736. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stella, G.M.; Lisini, D.; Pedrazzoli, P.; Galli, G.; Bortolotto, C.; Melloni, G.; D’Ambrosio, G.; Klersy, C.; Grosso, A.; Paino, F.; et al. Phase I Clinical Trial on Pleural Mesothelioma Using Neoadjuvant Local Administration of Paclitaxel-Loaded Mesenchymal Stromal Cells (PACLIMES Trial): Study Rationale and Design. Cancers 2024, 16, 3391. https://doi.org/10.3390/cancers16193391
Stella GM, Lisini D, Pedrazzoli P, Galli G, Bortolotto C, Melloni G, D’Ambrosio G, Klersy C, Grosso A, Paino F, et al. Phase I Clinical Trial on Pleural Mesothelioma Using Neoadjuvant Local Administration of Paclitaxel-Loaded Mesenchymal Stromal Cells (PACLIMES Trial): Study Rationale and Design. Cancers. 2024; 16(19):3391. https://doi.org/10.3390/cancers16193391
Chicago/Turabian StyleStella, Giulia Maria, Daniela Lisini, Paolo Pedrazzoli, Giulia Galli, Chandra Bortolotto, Giulio Melloni, Gioacchino D’Ambrosio, Catherine Klersy, Amelia Grosso, Francesca Paino, and et al. 2024. "Phase I Clinical Trial on Pleural Mesothelioma Using Neoadjuvant Local Administration of Paclitaxel-Loaded Mesenchymal Stromal Cells (PACLIMES Trial): Study Rationale and Design" Cancers 16, no. 19: 3391. https://doi.org/10.3390/cancers16193391
APA StyleStella, G. M., Lisini, D., Pedrazzoli, P., Galli, G., Bortolotto, C., Melloni, G., D’Ambrosio, G., Klersy, C., Grosso, A., Paino, F., Tomaselli, S., Saracino, L., Alessandri, G., Pessina, A., Grignani, E., Rosti, V., Corsico, A. G., Comoli, P., & Agustoni, F. (2024). Phase I Clinical Trial on Pleural Mesothelioma Using Neoadjuvant Local Administration of Paclitaxel-Loaded Mesenchymal Stromal Cells (PACLIMES Trial): Study Rationale and Design. Cancers, 16(19), 3391. https://doi.org/10.3390/cancers16193391







