Physical Activity, Insulin Resistance and Cancer: A Systematic Review
Abstract
:Simple Summary
Abstract
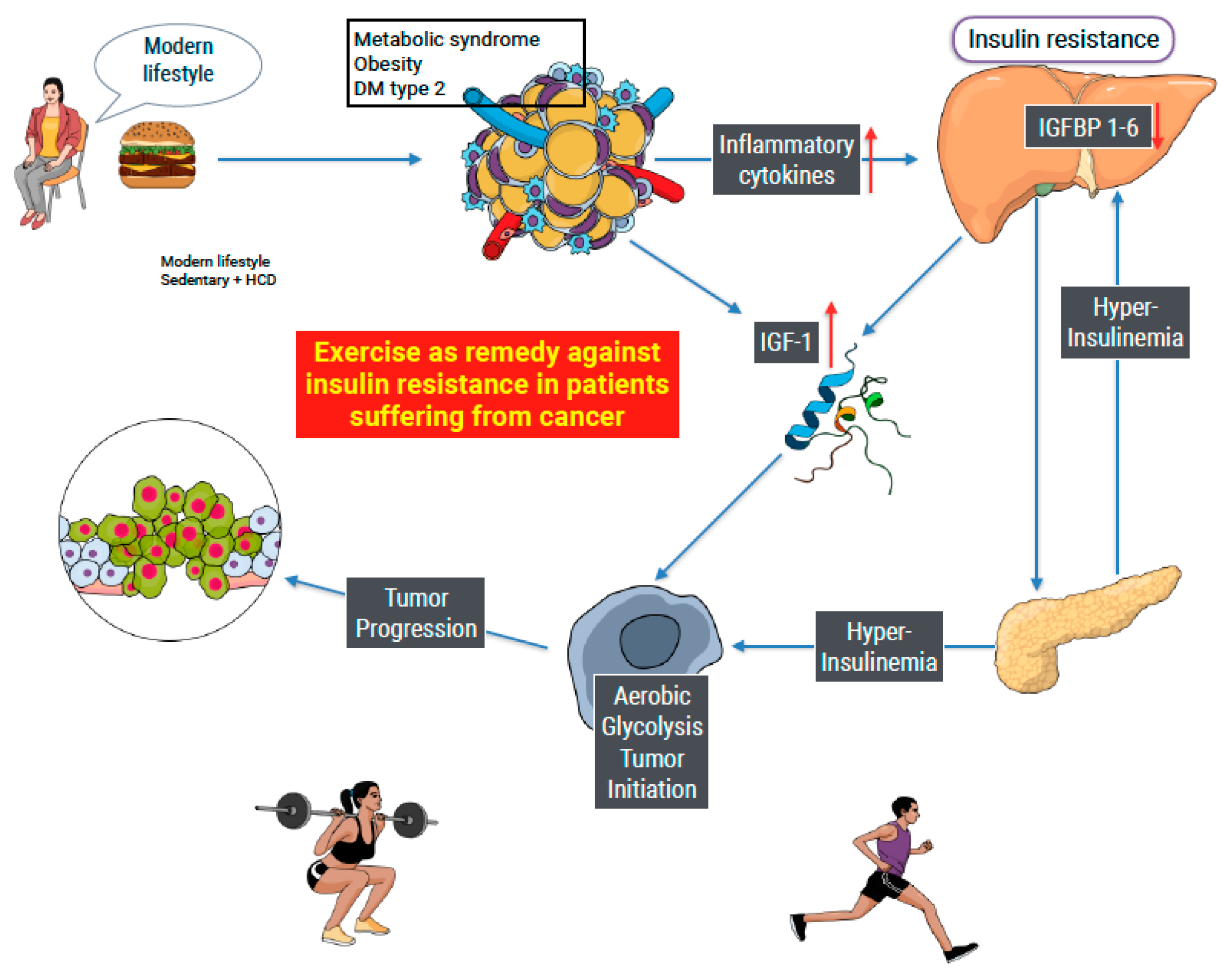
1. Introduction
2. Material and Method
2.1. Study Design
2.2. Sources Reviewed
2.3. Search Approach
2.4. Inclusion Criteria
- RCT published within the last 13 years;
- Publications are available in either English or Spanish.
2.5. Exclusion Criteria
2.6. Study Selection Process
2.7. Data Extraction
2.8. Risk of Bias Measurement Tool
2.9. Quality of the Evidence
3. Results
3.1. Study Identification and Selection Process
3.2. General Characteristics of the Selected Studies
3.3. Risk of Bias
3.4. Intervention Characteristics
3.4.1. Breast Cancer
3.4.2. Prostate Cancer
3.4.3. Liver Cancer
3.5. Results from each study
3.5.1. Breast Cancer
3.5.2. Prostate Cancer
3.5.3. Liver Cancer
3.6. Grade System
4. Discussion
4.1. Practical Application
4.2. Study Limitation
4.3. Prospective
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chiefari, E.; Mirabelli, M.; La Vignera, S.; Tanyolaç, S.; Foti, D.P.; Aversa, A.; Brunetti, A. Insulin Resistance and Cancer: In Search for a Causal Link. Int. J. Mol. Sci. 2021, 22, 11137. [Google Scholar] [CrossRef] [PubMed]
- Casanova, A.; Wevers, A.; Navarro-Ledesma, S.; Pruimboom, L. Mitochondria: It is all about energy. Front. Physiol. 2023, 14, 1114231. [Google Scholar] [CrossRef] [PubMed]
- Karlstad, O.; Linde, J.S.; Vestergaard, P.; Hjellvik, V.; Bazelier, M.T.; Schmidt, M.K.; Andersen, M.; Auvinen, A.; Haukka, J.; Furu, K.; et al. Use of Insulin and Insulin Analogs and Risk of Cancer—Systematic Review and Meta-Analysis of Observational Studies. Curr. Drug Saf. 2013, 8, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.W.; Azoulay, L.; Majdan, A.; Boivin, J.-F.; Pollak, M.; Suissa, S. Long-Term Use of Long-Acting Insulin Analogs and Breast Cancer Incidence in Women with Type 2 Diabetes. J. Clin. Oncol. 2017, 35, 3647–3653. [Google Scholar] [CrossRef]
- García-Jiménez, C.; Gutiérrez-Salmerón, M.; Chocarro-Calvo, A.; García-Martinez, J.M.; Castaño, A.; De la Vieja, A. From obesity to diabetes and cancer: Epidemiological links and role of therapies. Br. J. Cancer 2016, 114, 716–722. [Google Scholar] [CrossRef]
- Vicentini, M.; Ballotari, P.; Venturelli, F.; Ottone, M.; Manicardi, V.; Gallo, M.; Greci, M.; Pinotti, M.; Pezzarossi, A.; Rossi, P.G. Impact of Insulin Therapies on Cancer Incidence in Type 1 and Type 2 Diabetes: A Population-Based Cohort Study in Reggio Emilia, Italy. Cancers 2022, 14, 2719. [Google Scholar] [CrossRef]
- Pearson-Stuttard, J.; Zhou, B.; Kontis, V.; Bentham, J.; Gunter, M.J.; Ezzati, M. Worldwide burden of cancer attributable to diabetes and high body-mass index: A comparative risk assessment. Lancet Diabetes Endocrinol. 2018, 6, e6–e15. [Google Scholar] [CrossRef]
- Tsujimoto, T.; Kajio, H.; Sugiyama, T. Association between hyperinsulinemia and increased risk of cancer death in nonobese and obese people: A population-based observational study. Int. J. Cancer 2017, 141, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Kabat, G.C.; Kim, M.Y.; Lee, J.S.; Ho, G.Y.; Going, S.B.; Beebe-Dimmer, J.; Manson, J.E.; Chlebowski, R.T.; Rohan, T.E. Metabolic Obesity Phenotypes and Risk of Breast Cancer in Postmenopausal Women. Cancer Epidemiol. Biomark. Prev. 2017, 26, 1730–1735. [Google Scholar] [CrossRef]
- Walraven, I.; van T’Riet, E.; Stehouwer, C.D.A.; Polak, B.C.P.; Moll, A.C.; Dekker, J.M.; Nijpels, G. Fasting proinsulin levels are significantly associated with 20 year cancer mortality rates. The Hoorn Study. Diabetologia 2013, 56, 1148–1154. [Google Scholar] [CrossRef]
- Zhang, A.M.Y.; Chu, K.H.; Daly, B.F.; Ruiter, T.; Dou, Y.; Yang, J.C.C.; de Winter, T.J.J.; Chhuor, J.; Wang, S.; Flibotte, S.; et al. Effects of hyperinsulinemia on pancreatic cancer development and the immune microenvironment revealed through single-cell transcriptomics. Cancer Metab. 2022, 10, 5. [Google Scholar] [CrossRef]
- Sanaki, Y.; Nagata, R.; Kizawa, D.; Léopold, P.; Igaki, T. Hyperinsulinemia Drives Epithelial Tumorigenesis by Abrogating Cell Competition. Dev. Cell 2020, 53, 379–389.e5. [Google Scholar] [CrossRef]
- Schmidt, M.; de Mattos, S.F.; van der Horst, A.; Klompmaker, R.; Kops, G.J.P.L.; Lam, E.W.-F.; Burgering, B.M.T.; Medema, R.H. Cell Cycle Inhibition by FoxO Forkhead Transcription Factors Involves Downregulation of Cyclin D. Mol. Cell. Biol. 2002, 22, 7842–7852. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Jiang, Y.; Zhu, M.; Chen, L.; Zhou, X.; Zhou, C.; Ye, P.; Chen, X.; Wang, B.; Xu, Z.; et al. Clinical study using mesenchymal stem cells for the treatment of patients with severe COVID-19. Front. Med. 2020, 14, 664–673. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Diabetes. Available online: http://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 29 August 2023).
- Pearson-Stuttard, J.; Bennett, J.; Cheng, Y.J.; Vamos, E.P.; Cross, A.J.; Ezzati, M.; Gregg, E.W. Trends in predominant causes of death in individuals with and without diabetes in England from 2001 to 2018: An epidemiological analysis of linked primary care records. Lancet Diabetes Endocrinol. 2021, 9, 165–173. [Google Scholar] [CrossRef]
- Sona, M.F.; Myung, S.-K.; Park, K.; Jargalsaikhan, G. Type 1 diabetes mellitus and risk of cancer: A meta-analysis of observational studies. Jpn. J. Clin. Oncol. 2018, 48, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Callegari, I.O.M.; Rocha, G.Z.; Oliveira, A.G. Physical exercise, health, and disease treatment: The role of macrophages. Front. Physiol. 2023, 14, 1061353. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Higgins, J.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.; Welch, V. Cochrane Handbook for Systematic Reviews of Interventions; The Cochrane Collaboration: London, UK, 2019. [Google Scholar] [CrossRef]
- Aguayo-Albasini, J.L.; Flores-Pastor, B.; Soria-Aledo, V. GRADE system: Classification of quality of evidence and strength of recommendation. Cirugía Española 2014, 92, 82–88. [Google Scholar] [CrossRef]
- Galvão, D.A.; Taaffe, D.R.; Spry, N.; Joseph, D.; Newton, R.U. Combined Resistance and Aerobic Exercise Program Reverses Muscle Loss in Men Undergoing Androgen Suppression Therapy for Prostate Cancer Without Bone Metastases: A Randomized Controlled Trial. J. Clin. Oncol. 2010, 28, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Nobes, J.P.; Langley, S.E.; Klopper, T.; Russell-Jones, D.; Laing, R.W. A prospective, randomized pilot study evaluating the effects of metformin and lifestyle intervention on patients with prostate cancer receiving androgen deprivation therapy. BJU Int. 2011, 109, 1495–1502. [Google Scholar] [CrossRef] [PubMed]
- Elme, A.; Utriainen, M.; Kellokumpu-Lehtinen, P.; Palva, T.; Luoto, R.; Nikander, R.; Huovinen, R.; Kautiainen, H.; Järvenpää, S.; Penttinen, H.M.; et al. Obesity and Physical Inactivity Are Related to Impaired Physical Health of Breast Cancer Survivors. Anticancer Res. 2013, 33, 1595–1602. Available online: https://researchportal.helsinki.fi/en/publications/obesity-and-physical-inactivity-are-related-to-impaired-physical- (accessed on 29 August 2023). [PubMed]
- Kaibori, M.; Ishizaki, M.; Matsui, K.; Nakatake, R.; Yoshiuchi, S.; Kimura, Y.; Kwon, A.-H. Perioperative exercise for chronic liver injury patients with hepatocellular carcinoma undergoing hepatectomy. Am. J. Surg. 2013, 206, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Hvid, T.; Winding, K.; Rinnov, A.; Dejgaard, T.; Thomsen, C.; Iversen, P.; Brasso, K.; Mikines, K.J.; van Hall, G.; Lindegaard, B.; et al. Endurance training improves insulin sensitivity and body composition in prostate cancer patients treated with androgen deprivation therapy. Endocr. -Relat. Cancer 2013, 20, 621–632. [Google Scholar] [CrossRef]
- Hvid, T.; Lindegaard, B.; Winding, K.; Iversen, P.; Brasso, K.; Solomon, T.P.J.; Pedersen, B.K.; Hojman, P. Effect of a 2-year home-based endurance training intervention on physiological function and PSA doubling time in prostate cancer patients. Cancer Causes Control. 2015, 27, 165–174. [Google Scholar] [CrossRef]
- Bruno, E.; Roveda, E.; Vitale, J.; Montaruli, A.; Berrino, F.; Villarini, A.; Venturelli, E.; Gargano, G.; Galasso, L.; Caumo, A.; et al. Effect of aerobic exercise intervention on markers of insulin resistance in breast cancer women. Eur. J. Cancer Care 2016, 27, e12617. [Google Scholar] [CrossRef]
- Dieli-Conwright, C.M.; Courneya, K.S.; Demark-Wahnefried, W.; Sami, N.; Lee, K.; Buchanan, T.A.; Spicer, D.V.; Tripathy, D.; Bernstein, L.; Mortimer, J.E. Effects of Aerobic and Resistance Exercise on Metabolic Syndrome, Sarcopenic Obesity, and Circulating Biomarkers in Overweight or Obese Survivors of Breast Cancer: A Randomized Controlled Trial. J. Clin. Oncol. 2018, 36, 875–883. [Google Scholar] [CrossRef]
- Freedland, S.J.; Howard, L.; Allen, J.; Smith, J.; Stout, J.; Aronson, W.; Inman, B.A.; Armstrong, A.J.; George, D.; Westman, E.; et al. A lifestyle intervention of weight loss via a low-carbohydrate diet plus walking to reduce metabolic disturbances caused by androgen deprivation therapy among prostate cancer patients: Carbohydrate and prostate study 1 (CAPS1) randomized controlled trial. Prostate Cancer Prostatic Dis. 2019, 22, 428–437. [Google Scholar] [CrossRef]
- Viskochil, R.; Blankenship, J.M.; Makari-Judson, G.; Staudenmayer, J.; Freedson, P.S.; E Hankinson, S.; Braun, B. Metrics of Diabetes Risk Are Only Minimally Improved by Exercise Training in Postmenopausal Breast Cancer Survivors. J. Clin. Endocrinol. Metab. 2019, 105, e1958–e1966. [Google Scholar] [CrossRef] [PubMed]
- Pistelli, M.; Natalucci, V.; Scortichini, L.; Agostinelli, V.; Lenci, E.; Crocetti, S.; Merloni, F.; Bastianelli, L.; Taus, M.; Fumelli, D.; et al. The Impact of Lifestyle Interventions in High-Risk Early Breast Cancer Patients: A Modeling Approach from a Single Institution Experience. Cancers 2021, 13, 5539. [Google Scholar] [CrossRef]
- Dieli-Conwright, C.M.; Courneya, K.S.; Demark-Wahnefried, W.; Sami, N.; Norris, M.K.; Fox, F.S.; A Buchanan, T.; Spicer, D.; Bernstein, L.; Tripathy, D. Aerobic and resistance exercise improve patient-reported sleep quality and is associated with cardiometabolic biomarkers in Hispanic and non-Hispanic breast cancer survivors who are overweight or obese: Results from a secondary analysis. Sleep 2021, 44, zsab111. [Google Scholar] [CrossRef]
- Ryan, B.J.; Schleh, M.W.; Ahn, C.; Ludzki, A.C.; Gillen, J.B.; Varshney, P.; Van Pelt, D.W.; Pitchford, L.M.; Chenevert, T.L.; A Gioscia-Ryan, R.; et al. Moderate-Intensity Exercise and High-Intensity Interval Training Affect Insulin Sensitivity Similarly in Obese Adults. J. Clin. Endocrinol. Metab. 2020, 105, e2941–e2959. [Google Scholar] [CrossRef]
- Bird, S.R.; Hawley, J.A. Update on the effects of physical activity on insulin sensitivity in humans. BMJ Open Sport Exerc. Med. 2017, 2, 143. [Google Scholar] [CrossRef]
- D’alonzo, N.J.; Qiu, L.; Sears, D.D.; Chinchilli, V.; Brown, J.C.; Sarwer, D.B.; Schmitz, K.H.; Sturgeon, K.M. WISER Survivor Trial: Combined Effect of Exercise and Weight Loss Interventions on Insulin and Insulin Resistance in Breast Cancer Survivors. Nutrients 2021, 13, 3108. [Google Scholar] [CrossRef] [PubMed]
- Kullmann, S.; Goj, T.; Veit, R.; Fritsche, L.; Wagner, L.; Schneeweiss, P.; Hoene, M.; Hoffmann, C.; Machann, J.; Niess, A.; et al. Exercise restores brain insulin sensitivity in sedentary adults who are overweight and obese. J. Clin. Investig. 2022, 7, e161498. [Google Scholar] [CrossRef] [PubMed]
- Ruffino, J.S.; Songsorn, P.; Haggett, M.; Edmonds, D.; Robinson, A.M.; Thompson, D.; Vollaard, N.B. A comparison of the health benefits of reduced-exertion high-intensity interval training (REHIT) and moderate-intensity walking in type 2 diabetes patients. Appl. Physiol. Nutr. Metab. 2017, 42, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Arcidiacono, B.; Iiritano, S.; Nocera, A.; Possidente, K.; Nevolo, M.T.; Ventura, V.; Foti, D.; Chiefari, E.; Brunetti, A. Insulin Resistance and Cancer Risk: An Overview of the Pathogenetic Mechanisms. Exp. Diabetes Res. 2012, 2012, 789174. [Google Scholar] [CrossRef] [PubMed]
- Murphy, N.; Knuppel, A.; Papadimitriou, N.; Martin, R.; Tsilidis, K.; Smith-Byrne, K.; Fensom, G.; Perez-Cornago, A.; Travis, R.; Key, T.; et al. Insulin-like growth factor-1, insulin-like growth factor-binding protein-3, and breast cancer risk: Observational and Mendelian randomization analyses with ∼430 000 women. Ann. Oncol. 2020, 31, 641–649. [Google Scholar] [CrossRef]
- Thomas, R.J.; A Kenfield, S.; Jimenez, A. Exercise-induced biochemical changes and their potential influence on cancer: A scientific review. Br. J. Sports Med. 2016, 51, 640–644. [Google Scholar] [CrossRef]
- Pérez-López, F.R.; Chedraui, P.; Haya, J.; Cuadros, J.L. Effects of the Mediterranean diet on longevity and age-related morbid conditions. Maturitas 2009, 64, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Ledesma, S.; Hamed-Hamed, D.; González-Muñoz, A.; Pruimboom, L. Effectiveness of Treatments That Alter Metabolomics in Cancer Patients—A Systematic Review. Cancers 2023, 15, 4297. [Google Scholar] [CrossRef] [PubMed]

| Platform | Search Strategies |
|---|---|
| PubMed | ((“Neoplasms”[Mesh]) AND (“Diet”[Mesh] OR “Fasting”[Mesh] OR “Caloric Restriction”[Mesh] OR “Diet, Ketogenic”[Mesh] OR “exercise”[Mesh]) AND (“insulin resistance”[Mesh] OR “metabolic syndrome”[Mesh]) AND (“Healthy Lifestyle”[Mesh] OR “Quality of Life/psychology”[Mesh])) OR ((“Cancer”[tw] OR “neoplasms”[tw]) AND (“Fasting”[tw] OR “Caloric Restriction”[tw] OR “Diet, Ketogenic”[tw] OR “Nutritional strategies” [tw] OR “intermittent fasting”[tw] OR “time restriction” [tw] OR “exercise” [tw]) AND (“insulin resistance”[tw] OR “insulin resistance index”[tw] OR “metabolic syndrome”[tw] OR “homa-ir”[tw]) AND (“Healthy Lifestyle”[tw] OR “Quality of Life/psychology”[tw])) |
| Author | Year | Study Type | Country | Cancer | Sample | Gender | Age (Years) |
|---|---|---|---|---|---|---|---|
| Galvao et al. [23] | 2010 | RCC | Australia | Prostate Cancer | n = 57 | Male | 69.5 ± 7.3 |
| Nobes J et al. [24] | 2011 | PE | UK | Prostate Cancer | n = 40 | Male | 69.5 |
| Elme et al. [25] | 2013 | RCC | Finlandia | Breast Cancer | n = 537 | Female | 52 |
| Kaibori et al. [26] | 2013 | RCC | Osaka | Liver Cancer | n = 51 | 36 male | 69.7 |
| 15 female | |||||||
| Hvid et al. [27] | 2013 | RCC | Denmark | Prostate Cancer | n = 21 | Male | 67.8 |
| Hvid 2 et al. [28] | 2015 | PE | Denmark | Prostate Cancer | n = 17 | Male | 69.8 |
| Bruno E et al. [29] | 2016 | RCC | Italy | Breast Cancer | n = 38 | Female | 56.5 ± 6.8 |
| Dieli-Conwright C et al. [30] | 2018 | RCC | EEUU | Breast Cancer | n = 100 | Female | 53 ± 10.4 |
| Freedland et al. [31] | 2019 | RCC | EEUU | Prostate Cancer | n = 42 | Male | 66 |
| Viskochil et al. [32] | 2020 | E | EEUU | Breast Cancer | n = 15 | Female | 59.9 ± 9.2 |
| Pistelli et al. [33] | 2021 | E | Italy | Breast Cancer | n = 98 | Female | >18 |
| Dieli-Conwright C 2 et al. [34] | 2022 | ECC | EEUU | Breast Cancer | n = 418 | Female | 52 ± 10.4 |
| Galvao et al. [23] | .Nobes J et al. [24] | Elme et al. [25] | Kaibori et al. [26] | Hvid et al. [27] | Hvid 2 et al. [28] | Bruno E et al. [29] | Dieli-Conwright C et al. [30] | Freedland et al. [31] | Viskochil et al. [32] | Pistelli et al. [33] | Dieli-Conwright C 2 et al. [34] | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Proper sequence generation (selection risk) | + | + | + | + | + | + | + | + | + | - | - | + |
| Selection hiding (selection bias) | + | + | + | + | + | + | + | + | + | - | - | + |
| Blinding of participants and staff (implementation bias) | + | + | + | + | + | + | + | + | + | - | - | + |
| Blinding of outcome evaluators (detection bias) | + | - | + | + | + | - | + | + | + | - | - | + |
| Incomplete results data (wear bias) | + | - | + | + | + | - | + | + | + | - | - | ? |
| Selective reporting of results (notification bias) | + | + | + | + | + | + | + | + | + | - | - | + |
| Other sources of bias | ? | ? | ? | ? | ? | ? | ? | ? | + | ? | ? |
| Author | Year | Type of Intervention | Groups | Duratión Intervention | Frequency Intervention | Variable | Measuring Instrument | ||
|---|---|---|---|---|---|---|---|---|---|
| Galvao et al. [23] | 2010 | Aerobic and resistance exercises | GE = 29 GC = 28 | 12 weeks | 2 times/week | Insulin | |||
| Nobes J et al. [24] | 2011 | Exercise program | GE = 20 | GC = 20 | 24 weeks | Not shown | Insulin | HOMA-IR | |
| Elme et al. [25] | 2013 | Exercise program | G1:IMC < 25 | G2:IMC 25–30 | G3:IMCC ≥ 30 | 48 weeks | Not shown | Insulin | HOMA-IR |
| Kaibori et al. [26] | 2013 | Aerobic and resistance exercises | GE = 26 | GC = 25 | 24 weeks | 3 times/week | Insulin | HOMA-IR | |
| Hvid et al. [27] | 2013 | Resistance exercises | GE = 9 | GC = 10 | 12 weeks | 3 times/week | Insulin | HOMA-IR | |
| Hvid2 et al. [28] | 2015 | Resistance Training at home | GE = 12 | GC = 7 | 96 weeks | 3 times/week | Insulin | HOMA-IR | |
| Bruno E et al. [29] | 2016 | Aerobic exercises | GE = 28 | GC = 20 | 12 weeks | 2 times/week | Insulin | HOMA-IR | |
| Dieli-Conwright C et al. [30] | 2018 | Aerobic and resistance exercises | GE = 50 | GC = 50 | 16 weeks | 3 times/week | Insulin | HOMA-IR | |
| Freendlan et al. [31] | 2019 | Walk | GE = 20 | GC = 22 | 24 weeks | 5 times/week | Insulin | HOMA-IR | |
| Viskochil et al. [32] | 2020 | Supervised aerobic exercise program | G1 = 15 | 12 weeks | 3–4 times/week | Insulin | Oral glucose tolerance test. | ||
| Pistelli et al. [33] | 2021 | Aerobic and resistance exercises | G1 = 98 | 48 weeks | 3–5 times/week | Insulin | HOMA IR | ||
| Dieli-Conwright C 2 et al. [34] | 2022 | Aerobic and resistance exercises | GE = 50 | GC = 50 | 16 weeks | 3 times/week | Insulin | HOMA-IR | |
| Author | Variable | Before Treatment | |
|---|---|---|---|
| Galvao et al. [23] | Insulin | GE:9.4 (7.2) GC:10.0 (5.7) | |
| Nobes J et al. [24] | Insulin | GE:107.0 (160.0) | GC = 61.69 (40.0) |
| Etme et al. [25] | |||
| Kaibori et al. [26] | Insulin | GE = 8.56, 5.8 | GC = 9.16, 4.5 |
| Hvid et al. [27] | Insulin | GE93.4 | GC = 58.6 |
| Hvid2 et al. [28] | Insulin | GE = 48.0 ± 28.5 | GC = 42.0 ± 23.0 |
| Bruno E et al. [29] | Insulin | GE = 7.9 ± 4.2 | GC = 6.60 ± 3.1 |
| Dieli-Conwright C et al. [30] | Insulin | GE = 11.1 (8.9) | GC = 12.3 (7.1) |
| Freedland et al. [31] | Insulin | GE = −16.9 (−42.6, −3.2) | GC = 7.0 (−9.6, 49.7) |
| Viskochil et al. [32] | Insulin | G1 = 11.4 ± 5.4 | |
| Pistelli et al. [33] | Insulin | G1 = 3.78 (0.06) | |
| Dieli-Conwright C 2 et al. [34] | Insulin | − | − |
| Author | Variable | After Treatment | p | |
|---|---|---|---|---|
| Galvao et al. [23] | Insulin | GE: 10.2 (7.4) GC: 10.9 (7.1) | p = 0.435 | |
| Nobes J et al. [24] | Insulin | GE = 92.4 (153.3) | GC = 66.6 (53.7) | p > 0.5 |
| Etme et al. [25] | p = 0.0098 | |||
| Kaibori et al. [26] | Insulin | GE = 5.86, 2.8 | GC = 10.76, 4.6 | p = 0.193 |
| Hvid et al. [27] | Insulin | GE = 78.3 | GC = 56.0 | p = 0.030 |
| Hvid2 et al. [28] | Insulin | GE = 44.5 ± 23.5 | GC = 42.3 ± 21.3 | p = 0.05 |
| Bruno E et al. [29] | Insulin | GE = 6.7 ± 3.1 (p = 0.8) | GC = 7.43 ± (4.5) (p = 0.17) | p = 0.4 |
| Dieli-Conwright C et al. [30] | Insulin | GE = 6.8 (1.3) | GC = 15.1 (6.4) | p = 0.002 |
| Freedland et al. [31] | Insulin | GE = 7.8 (−35.2, 29.1) | GC = 26.2 (−1.2, 80.7) | p = 0.11 |
| Viskochil et al. [32] | Insulin | G1 = 11.6 ± 5.1 | p = 0.79 | |
| Pistelli et al. [33] | Insulin | G1 = 10.51 (0.17) | p = 0.923 | |
| Dieli-Conwright C 2 et al. [34] | Insulin | - | - | p < 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro-Ledesma, S.; Hamed-Hamed, D.; González-Muñoz, A.; Pruimboom, L. Physical Activity, Insulin Resistance and Cancer: A Systematic Review. Cancers 2024, 16, 656. https://doi.org/10.3390/cancers16030656
Navarro-Ledesma S, Hamed-Hamed D, González-Muñoz A, Pruimboom L. Physical Activity, Insulin Resistance and Cancer: A Systematic Review. Cancers. 2024; 16(3):656. https://doi.org/10.3390/cancers16030656
Chicago/Turabian StyleNavarro-Ledesma, Santiago, Dina Hamed-Hamed, Ana González-Muñoz, and Leo Pruimboom. 2024. "Physical Activity, Insulin Resistance and Cancer: A Systematic Review" Cancers 16, no. 3: 656. https://doi.org/10.3390/cancers16030656
APA StyleNavarro-Ledesma, S., Hamed-Hamed, D., González-Muñoz, A., & Pruimboom, L. (2024). Physical Activity, Insulin Resistance and Cancer: A Systematic Review. Cancers, 16(3), 656. https://doi.org/10.3390/cancers16030656











