Removal of Hydrogen Sulfide From Various Industrial Gases: A Review of The Most Promising Adsorbing Materials
Abstract
1. Introduction
2. Adsorption
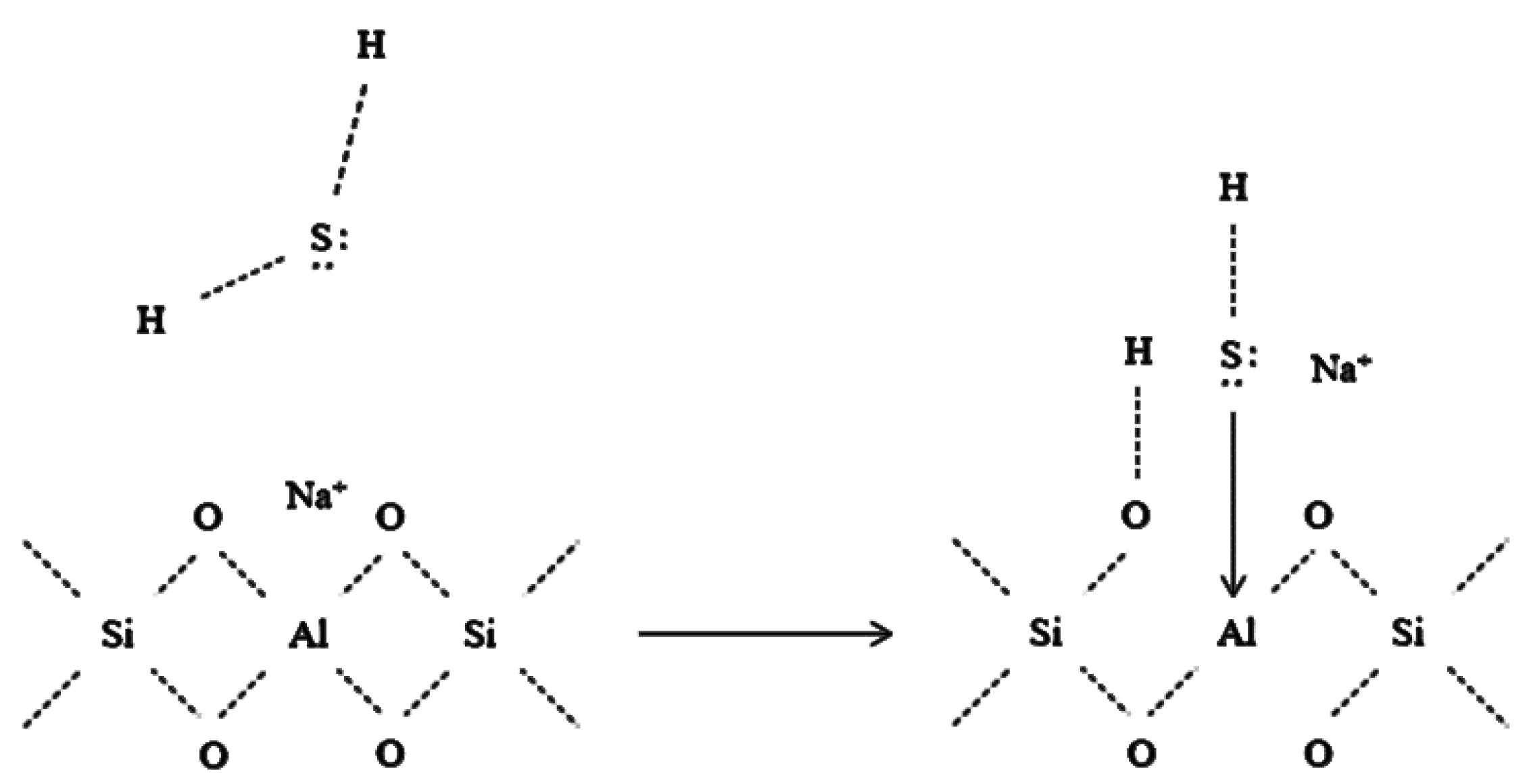
2.1. Zeolites
2.2. Activated Carbons
2.3. Metal Oxides
2.4. Composite Materials
2.4.1. Metal Oxide/Mesoporous Silica
2.4.2. Metal Oxide/Activated Carbon
2.4.3. Metal Hydroxide/Activated Carbon
2.4.4. Supported Amines
2.5. MOFs (Metal–Organic Frameworks)
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- De Crisci, A.G.; Moniri, A.; Xu, Y. Hydrogen from hydrogen sulfide: Towards a more sustainable hydrogen economy. Int. J. Hydrog. Energy 2019, 44, 1299–1327. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Siakavelas, G.; Papageridis, K.N.; Baklavaridis, A.; Tzounis, L.; Avraam, D.G.; Goula, M.A. Syngas production via the biogas dry reforming reaction over nickel supported on modified with CeO2 and/or La2O3 alumina catalysts. J. Nat. Gas Sci. Eng. 2016, 31, 164–183. [Google Scholar] [CrossRef]
- Roberts, E.S.; Wong, V.A.; McManus, B.E.; Marshall, M.W.; Lancianese, S.; Dorman, D.C. Changes in intracellular pH play a secondary role in hydrogen sulfide-induced nasal cytotoxicity. Inhal. Toxicol. 2006, 15, 159–167. [Google Scholar] [CrossRef]
- Lindenmann, J.; Matzi, V.; Neuboeck, N.; Ratzenhofer-Komenda, B.; Maier, A.; Smolle-Juettner, F.M. Severe hydrogen sulphide poisoning treated with 4-dimethylaminophenol and hyperbaric oxygen. Diving Hyperb. Med. 2010, 40, 213–217. [Google Scholar]
- Burnett, W.W.; King, E.G.; Grace, M.; Hall, W.F. Hydrogen sulfide poisoning: Review of 5 years’ experience. Can. Med. Assoc. J. 1977, 177, 1277–1280. [Google Scholar]
- National Library of Medicine, National Center for Biotechnology Information. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Hydrogen-sulfide (accessed on 14 March 2020).
- Chen, Q.J.; Fan, F.C.; Long, D.H.; Liu, X.J.; Liang, X.Y.; Qiao, W.M.; Ling, L.C. Poly (ethyleneimine)-loaded silica monolith with a hierarchical pore structurefor H2S adsorptive removal. Ind. Eng. Chem. Res. 2010, 49, 11408–11414. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Baklavaridis, A.; Papadakis, V.G.; Goula, M.A. Synthesis gas production via the biogas reforming reaction over Ni/MgO-Al2O3 and Ni/CaO-Al2O3 catalysts. Waste Biomass Valorization 2016, 7, 725–736. [Google Scholar] [CrossRef]
- Barelli, L.; Bidini, G.; Micoli, L.; Sisani, E.; Turco, M. 13X Ex-Cu zeolite performance characterization towards H2S removal for biogas use in molten carbonate fuel cells. Energy 2018, 160, 44–53. [Google Scholar] [CrossRef]
- Hervya, M.; Minha, D.P.; Gérenteb, C.; Weiss-Hortalaa, E.; Nzihoua, A.; Villotb, A.; Le Coq, L. H2S removal from syngas using wastes pyrolysis chars. Chem. Eng. J. 2018, 334, 2179–2189. [Google Scholar] [CrossRef]
- Vikrant, K.; Kailasa, S.K.; Tsang, D.C.W.; Lee, S.S.; Kumar, P.; Giri, B.S.; Singh, R.S.; Kim, K.H. Biofiltration of hydrogen sulfide: Trends and challenges. J. Clean. Prod. 2018, 187, 131–147. [Google Scholar] [CrossRef]
- Carrera, L.; Springer, F.; Lipeme-Kouyi, G.; Buffiere, P. A review of sulfide emissions in sewer networks: Overall approach and systemic modelling. Water Sci. Technol. 2015, 73, 1231–1242. [Google Scholar] [CrossRef]
- Bandosz, T.J. On the adsorption/oxidation of hydrogen sulfide on activated carbons at ambient temperatures. J. Colloid Interface Sci. 2002, 246, 1–20. [Google Scholar] [CrossRef]
- Sisani, E.; Cinti, G.; Discepoli, G.; Penchini, D.; Desideri, U.; Marmottini, F. Adsorptive removal of H2S in biogas conditions for high temperature fuel cell systems. Int. J. Hydrog. Energy 2014, 39, 21753–21766. [Google Scholar] [CrossRef]
- Mescia, D.; Hernandez, S.P.; Conoci, A.; Russo, N. MSW landfill biogas desulfurization. Int. J. Hydrog. Energy 2011, 36, 7884–7890. [Google Scholar] [CrossRef]
- Bamdad, H.; Hawboldt, K.; MacQuarrie, S. A review on common adsorbents for acid gases removal: Focus on biochar. Renew. Sustain. Energy Rev. 2018, 81, 1705–1720. [Google Scholar] [CrossRef]
- Khabazipour, M.; Mansoor, A. Removal of hydrogen sulfide from gas streams using porous materials: A review. Ind. Eng. Chem. Res. 2019, 58, 22133–22164. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Rouquerol, J.; Rouquerol, F. Adsorption by Powders and Porous Solids; Academic Press: San Diego, CA, USA, 1998. [Google Scholar]
- Dabrowski, A. Adsorption-from theory to practice. Adv. Colloid Interface Sci. 2001, 93, 135–224. [Google Scholar] [CrossRef]
- Ozekmekci, M.; Salkic, G.; Fellah, M.F. Use of zeolites for the removal of H2S: A mini-review. Fuel Process. Technol. 2015, 139, 49–60. [Google Scholar] [CrossRef]
- Kerr, G.T.; Johnson, G.C. Catalytic oxidation of hydrogen sulfide to sulfur over a crystalline aluminosilicate. J. Phys. Chem. 1960, 64, 381–382. [Google Scholar] [CrossRef]
- Steijns, M.; Derks, F.; Verloop, A.; Mars, P. The mechanism of the catalytic oxidation of hydrogen sulfide. II. Kinetics and mechanism of hydrogen sulfide oxidation catalyzed by sulfur. J. Catal. 1976, 42, 87–95. [Google Scholar] [CrossRef]
- Micoli, L.; Bagnasco, G.; Turco, M. H2S removal from biogas for fuelling MCFCs: New adsorbing materials. Int. J. Hydrog. Energy 2014, 39, 1783–1787. [Google Scholar] [CrossRef]
- Deo, A.V.; Dalla Lana, I.G.; Habgood, H.W. Infrared studies of the adsorption and surface reactions of hydrogen sulfide and sulfur dioxide on some aluminas and zeolites. J. Catal. 1971, 21, 270–281. [Google Scholar] [CrossRef]
- Sigot, L.; Ducom, G.; Germain, P. Adsorption of hydrogen sulfide (H2S) on zeolite (Z): Retention Mechanism. Chem. Eng. J. 2016, 287, 47–53. [Google Scholar] [CrossRef]
- Kumar, P.; Sung, C.; Muraza, O.; Cococcioni, M.; Al Hashimi, S.; McCormick, A.; Tsapatsis, M. H2S adsorption by Ag and Cu ion exchanged faujasites. Microporous Mesoporous Mater. 2011, 146, 127–133. [Google Scholar] [CrossRef]
- Mohammed, A.; Nassrullah, Z.K. Preparation and formation of zeolite 5A from local kaolin clay for drying and desulphurization of liquefied petroleum gas. IJCPE 2013, 14, 1–13. [Google Scholar]
- Kikkinides, E.S.; Sikavitsas, V.I.; Yang, R.T. Natural gas desulfurization by adsorption: Feasibility and multiplicity of cyclic steady states. Ind. Eng. Chem. Res. 1995, 34, 255–262. [Google Scholar] [CrossRef]
- Bülow, M.; Lutz, W.; Suckow, M. The mutual transformation of hydrogen sulphide and carbonyl sulphide and its role for gas desulphurization processes with zeolitic molecular sieve sorbents. Stud. Surf. Sci. Catal. 1999, 120, 301–345. [Google Scholar]
- Tomadakis, M.M.; Heck, H.H.; Jubran, M.J.; Al-Harthi, K. Pressure swing adsorption separation of H2S from CO2 with molecular sieves 4A, 5A and 13X. Sep. Sci. Technol. 2011, 46, 428–433. [Google Scholar] [CrossRef]
- Wynnyk, K.G.; Hojjati, B.; Pirzadeh, P.; Marriott, R.A. High-pressure sour gas adsorption on zeolite 4A. Adsorption 2017, 23, 149–162. [Google Scholar] [CrossRef]
- Sigot, L.; Obis, M.F.; Benbelkacem, H.; Germain, P.; Ducom, G. Comparing the performance of a 13X zeolite and an impregnated activated carbon for H2S removal from biogas to fuel an SOFC: Influence of water. Int. J. Hydrog. Energy 2016, 41, 18533–18541. [Google Scholar] [CrossRef]
- Xiao, Y.; Wang, S.; Wu, D.; Yuan, Q. Experimental and simulation study of hydrogen sulfide adsorption on impregnated activated carbon under anaerobic conditions. J. Hazard Mater. 2008, 153, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Vicario, A.; Ochoa-Gomez, J.R.; Gil-Rıo, S.; Gomez-Jimenez-Aberasturi, O.; Ramırez-Lopez, C.A.; Torrecilla-Soria, J.; Domínguez, A. Purification and upgrading of biogas by pressure swing adsorption on synthetic and natural zeolites. Microporous Mesoporous Mater. 2010, 134, 100–107. [Google Scholar] [CrossRef]
- Melo, D.M.A.; De Souza, J.R.; Melo, M.A.F.; Martinelli, A.E.; Cachima, G.H.B.; Cunha, J.D. Evaluation of the zinox and zeolite materials as adsorbents to remove H2S from natural gas. Colloids Surf. A 2006, 272, 32–36. [Google Scholar] [CrossRef]
- Lee, S.K.; Jang, Y.N.; Bae, I.K.; Chae, S.C.; Ryu, K.W.; Kim, J.K. Adsorption of toxic gases iron-incorporated Na-A zeolites synthesized from melting slag. Mater. Trans. 2009, 50, 2476–2483. [Google Scholar] [CrossRef]
- Abdullah, A.H.; Mat, R.; Somderam, S.; Aziz, A.S.A.; Mohamed, A. Hydrogen sulfide adsorption by zinc oxide-impregnated zeolite (synthesized from Malaysian kaolin) for biogas desulfurization. J. Ind. Eng. Chem. 2018, 65, 334–342. [Google Scholar] [CrossRef]
- Liu, X.; Wang, R. Effective removal of hydrogen sulfide using 4A molecular sieve zeolites synthesized from attapulgite. J. Hazard Mater. 2017, 326, 157–164. [Google Scholar] [CrossRef]
- Yokogawa, Y.; Sakanishi, M.; Morikawa, N.; Nakamura, A.; Kishida, I.; Varma, H.K. VSC adsorptive properties in ion exchanged zeolite materials in gaseous and aqueous medium. Procedia Eng. 2012, 36, 168–172. [Google Scholar] [CrossRef][Green Version]
- Karge, H.G.; Raskó, J. Hydrogen sulfide adsorption on faujasite-type zeolites with systematically caried Si-Al ratios. J. Colloid Interface Sci. 1978, 64, 522–532. [Google Scholar] [CrossRef]
- De Oliveira, L.H.; Meneguin, J.G.; Pereira, M.V.; Da Silva, E.A.; Grava, W.M.; Do Nascimento, J.F.; Arroyo, P.A. H2S adsorption on NaY zeolite. Microporous Mesoporous Mater. 2019, 284, 247–257. [Google Scholar] [CrossRef]
- Ratnasamy, C.; Wagner, J.P.; Spivey, S.; Weston, E. Removal of sulfur compounds from natural gas for fuel cell applications using a sequential bed system. Catal. Today 2012, 198, 233–238. [Google Scholar] [CrossRef]
- Rong, C.; Chu, D.; Hopkins, J. Test and Characterization of Some Zeolite Supported Gas Phase Desulphurization Sorbents; ARL-TR-4859; Army Research Laboratory: Adelphi, MD, USA, 2009. [Google Scholar]
- Crespo, D.; Qi, G.; Wang, Y.; Yang, F.H.; Yang, R.T. Superior sorbent for natural gas desulfurization. Ind. Eng. Chem. Res. 2008, 47, 1238–1244. [Google Scholar] [CrossRef]
- Hernández-Maldonado, A.J.; Yang, R.T.; Chinn, D.; Munson, C.L. Partially calcined gismondine type silicoaluminophosphate SAPO-43: Isopropylamine elimination and separation of carbon dioxide, hydrogen sulfide, and water. Langmuir 2003, 19, 2193–2200. [Google Scholar] [CrossRef]
- Yasyerli, S.; Ar, I.; Dogu, G.; Dogu, T. Removal of hydrogen sulfide by clinoptilolite in a fixed bed adsorber. Chem. Eng. Process. 2002, 41, 785–792. [Google Scholar] [CrossRef]
- Pourzolfaghar, H.; Ismail, M.H.S.; Izhar, S.; Maghareh Esfahan, Z. Review of H2S sorbents at low-temperature desulfurization of biogas. Int. J. Chem. Environ. Eng. 2014, 5, 22–28. [Google Scholar]
- Bashkova, S.; Baker, F.S.; Wu, X.; Armstrong, T.R.; Schwartz, V. Activated carbon catalyst for selective oxidation of hydrogen sulphide: On the influence of pore structure, surface characteristics, and catalytically-active nitrogen. Carbon 2007, 45, 1354–1363. [Google Scholar] [CrossRef]
- Shanmugam, S.R.; Adhikari, S.; Wang, Z.; Shakya, R. Treatment of aqueous phase of bio-oil by granular activated carbon and evaluation of biogas production. Bioresour. Technol. 2017, 223, 115–120. [Google Scholar] [CrossRef]
- Singhal, S.; Agarwal, S.; Arora, S.; Sharma, P.; Singhal, N. Upgrading techniques for transformation of biogas to bio-CNG: A review. Int. J. Energy Res. 2017, 41, 1657–1669. [Google Scholar] [CrossRef]
- Figueiredo, J.L.; Pereira, M.F.R.; Freitas, M.M.A.; Orfao, J.J.M. Modification of the surface chemistry of activated carbons. Carbon 1999, 37, 1379–1389. [Google Scholar] [CrossRef]
- Lippens, B.C.; De Boer, J.H.; De Boer, J.H. Studies on pore systems in catalysts: V. The T method. J. Catal. 1965, 4, 319–323. [Google Scholar] [CrossRef]
- Bagreev, A.; Bandosz, T.J. A role of sodium hydroxide in the process of hydrogen sulfide adsorption/oxidation on caustic c-impregnated activated carbons. Ind. Eng. Chem. Res. 2002, 41, 672–679. [Google Scholar] [CrossRef]
- Turk, A.; Sakalis, E.; Lessuck, J.; Karamitsos, H.; Rago, O. Ammonia injection enhances capacity of activated carbon for hydrogen sulfide and methyl mercaptan. Environ. Sci. Technol. 1989, 23, 1242–1245. [Google Scholar] [CrossRef]
- Adib, F.; Bagreev, A.; Bandosz, T.J. Adsorption/oxidation of hydrogen sulfide on nitrogen-containing activated carbons. Langmuir 2000, 16, 1980–1986. [Google Scholar] [CrossRef]
- Tian, S.; Mo, H.; Zhang, R.; Ning, P.; Zhou, T. Enhanced removal of hydrogen sulfide from a gas stream by 3-aminopropyltriethoxysilane-surface-functionalized activated carbon. Adsorption 2009, 15, 477–488. [Google Scholar] [CrossRef]
- Adib, F.; Bagreev, A.; Bandosz, T.J. Effect of surface characteristics of wood-based activated carbons on adsorption of hydrogen sulfide. J. Colloid Interface Sci. 1999, 214, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Bagreev, A.; Adib, F.; Bandosz, T.J. Initial heats of H2S adsorption on activated carbons: Effect of surface features. J. Colloid Interface Sci. 1999, 219, 327–332. [Google Scholar] [CrossRef]
- Bandosz, T.J. Effect of pore structure and surface chemistry of virgin activated carbons on removal of hydrogen sulfide. Carbon 1999, 37, 483–491. [Google Scholar] [CrossRef]
- Bagreev, A.; Rahman, H.; Bandosz, T.J. Study of H2S adsorption and water regeneration of spent coconut-based activated carbon. Environ. Sci. Technol. 2000, 34, 4587–4592. [Google Scholar] [CrossRef]
- Bagreev, A.; Rahman, H.; Bandosz, T.J. Thermal regeneration of a spent activated carbon previously used as hydrogen sulfide adsorbent. Carbon 2001, 39, 1319–1326. [Google Scholar] [CrossRef]
- Boudou, J.P.; Chehimi, M.; Broniek, E.; Siemieniewska, T.; Bimer, J. Adsorption of H2S or SO2 on an activated carbon cloth modified by ammonia treatment. Carbon 2003, 41, 1999–2007. [Google Scholar] [CrossRef]
- Farooq, M.; Bell, A.H.; Almustapha, M.; Andresen, J.M. Bio-methane from anaerobic digestion using activated carbon adsorption. Anaerobe 2017, 46, 33–40. [Google Scholar] [CrossRef]
- Skouteris, G.; Saroj, D.; Melidis, P.; Hai, F.I.; Ouki, S. The effect of activated carbon addition on membrane bioreactor processes for wastewater treatment and reclamation—A critical review. Bioresour. Technol. 2015, 185, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Chen, C.; Chu, S. Effect of moisture on H2S adsorption by copper impregnated activated carbon. J. Hazard Mater. 2006, 136, 866–873. [Google Scholar] [CrossRef]
- Papurello, D.; Tomasi, L.; Silvestri, S.; Santarelli, M. Evaluation of the Wheeler-Jonas parameters for biogas trace compounds removal with activated carbons. Fuel Process. Technol. 2016, 152, 93–101. [Google Scholar] [CrossRef]
- Sitthikhankaew, R.; Chadwik, D.; Assabumrungrat, S.; Laosiripojana, N. Effects of humidity, O2, and CO2 on H2S adsorption onto upgraded and KOH impregnated activated carbons. Fuel Process. Technol. 2014, 124, 249–257. [Google Scholar] [CrossRef]
- Feng, W.; Kwon, S.; Borguet, E.; Vidic, R. Adsorption of hydrogen sulfide onto activated carbon fibers: Effect of pore structure and surface chemistry. Environ. Sci. Technol. 2005, 39, 9744–9749. [Google Scholar] [CrossRef] [PubMed]
- Primavera, A.; Trovarelli, A.; Andreussi, P.; Dolcetti, G. The effect of water in the low-temperature catalytic oxidation of hydrogen sulphide to sulfur over activated carbon. Appl. Catal. A Gen. 1998, 173, 185–192. [Google Scholar] [CrossRef]
- Bagreev, A.; Bandosz, T.J. H2S adsorption/oxidation on unmodified activated carbons: Importance of prehumidification. Carbon 2001, 39, 2303–2311. [Google Scholar] [CrossRef]
- Le-Minh, N.; Sivret, E.C.; Shammay, A.; Stuetz, R.M. Factors affecting the adsorption of gaseous environmental odors by activated carbon: A critical review. Crit. Rev. Environ. Sci. Technol. 2018, 48, 341–375. [Google Scholar] [CrossRef]
- Papurello, D.; Gandiglio, M.; Lanzini, A. Experimental analysis and model validation on the performance of impregnated activated carbons for the removal of hydrogen sulfide (H2S) from sewage biogas. Processes 2019, 7, 548. [Google Scholar] [CrossRef]
- Ciahotny, K.; Kyselova, V. Hydrogen sulfide removal from biogas using carbon impregnated with oxidants. Energy Fuels 2019, 33, 5316–5321. [Google Scholar] [CrossRef]
- Barelli, L.; Bidini, G.; De Arespacochaga, N.; Perez, L.; Sisani, E. Biogas use in high temperature fuel cells: Enhancement of KOH-KI activated carbon performance toward H2S removal. Int. J. Hydrog. Energy 2017, 42, 10341–10353. [Google Scholar] [CrossRef]
- McIntyre, N.S.; Mount, G.R.; Lipson, T.C.; Humphrey, R.; Harrison, B.; Liang, S.; Pagotto, J. Surface and microanalytical studies of whetlerite charcoals: 1. Effects of aging. Carbon 1991, 29, 1071–1079. [Google Scholar] [CrossRef]
- Awe, O.W.; Minh, D.P.; Lyczkob, N.; Nzihoub, A.; Zhao, Y. Laboratory-scale investigation of the removal of hydrogen sulfide from biogas and air using industrial waste-based sorbents. J. Environ. Chem. Eng. 2017, 5, 1809–1820. [Google Scholar] [CrossRef]
- Skerman, A.G.; Heubeck, S.; Batstone, D.J.; Tait, S. Low-cost filter media for removal of hydrogen sulphide from piggery biogas. Process Saf. Environ. 2017, 105, 117–126. [Google Scholar] [CrossRef]
- Rambabu, N.; Azargohar, R.; Dalai, A.K.; Adjaye, J. Evaluation and comparison of enrichment efficiency of physical/chemical activations and functionalized activated carbons derived from fluid petroleum coke for environmental applications. Fuel Process. Technol. 2013, 106, 501–510. [Google Scholar] [CrossRef]
- Mochizuki, T.; Kubota, M.; Matsuda, H.; D’Elia Camacho, L.F. Adsorption behaviors of ammonia and hydrogen sulfide on activated carbon prepared from petroleum coke by KOH chemical activation. Fuel Process. Technol. 2016, 144, 164–169. [Google Scholar] [CrossRef]
- Asaoka, S.; Okamura, H.; Morisawa, R.; Murakami, H.; Fukushi, K.; Okajima, T.; Katayama, M.; Inada, Y.; Yogi, C.; Ohta, T. Removal of hydrogen sulfide using carbonated steel slag. Chem. Eng. J. 2013, 228, 843–849. [Google Scholar] [CrossRef]
- Kim, K.H.; Asaoka, S.; Yamamoto, T.; Hayakawa, S.; Takeda, K.; Katayama, M.; Onoue, T. Mechanisms of hydrogen sulfide removal with steel making slag. Environ. Sci. Technol. 2012, 46, 10169–10174. [Google Scholar] [CrossRef]
- Asaoka, S.; Okamura, H.; Kim, K.; Hatanaka, Y.; Nakamoto, K.; Hino, K.; Oikawa, T.; Hayakawa, S.; Okuda, T. Optimum reaction ratio of coal fly ash to blast furnace cement for effective removal of hydrogen sulfide. Chemosphere 2017, 168, 384–389. [Google Scholar] [CrossRef]
- Asaoka, S.; Okamura, H.; Akita, Y.; Nakano, K.; Nakamoto, K.; Hino, K.; Saito, T.; Hayakawa, S.; Katayama, M.; Inada, Y. Regeneration of manganese oxide as adsorption sites for hydrogen sulfide on granulated coal ash. Chem. Eng. J. 2014, 254, 531–537. [Google Scholar] [CrossRef]
- Bazan-Wozniak, A.; Nowicki, P.; Pietrzak, R. The influence of activation procedure on the physicochemical and sorption properties of activated carbons prepared from pistachio nutshells for removal of NO2/H2S gases and dyes. J. Clean. Prod. 2017, 152, 211–222. [Google Scholar] [CrossRef]
- Zulkefli, N.N.; Masdar, M.S.; Wan Isahak, W.N.R.; Md Jahim, J.; Md Rejab, S.A.; Lye, C.C. Removal of hydrogen sulfide from a biogas mimic by using impregnated activated carbon adsorbent. PLoS ONE 2019, 14, e0211713. [Google Scholar] [CrossRef] [PubMed]
- Nam, H.; Wang, S.; Jeong, H.R. TMA and H2S gas removals using metal loaded on rice husk activated carbon for indoor air purification. Fuel 2018, 213, 186–194. [Google Scholar] [CrossRef]
- Leboda, R.; Skubiszewska-Zieba, J.; Tomaszewski, W.; Gun’ko, V.M. Structural and adsorptive properties of activated carbons prepared by carbonization and activation of resins. J. Colloid Interface Sci. 2003, 263, 533–541. [Google Scholar] [CrossRef]
- Kazmierczak-Razna, J.; Gralak-Podemska, B.; Nowicki, P.; Pietrzak, R. The use of microwave radiation for obtaining activated carbons from sawdust and their potential application in removal of NO2 and H2S. Chem. Eng. J. 2015, 269, 352–358. [Google Scholar] [CrossRef]
- De Oliveira, L.H.; Meneguin, J.G.; Pereira, M.V.; Do Nascimento, J.F.; Arroyo, P.A. Adsorption of hydrogen sulfide; carbon dioxide; methane; and their mixtures on activated carbon. Chem. Eng. Commun. 2019, 206, 1533–1553. [Google Scholar] [CrossRef]
- Montes, D.; Tocuyo, E.; Gonzaĺez, E.; Rodriǵuez, D.; Solano, R.; Atencio, R.; Ramos, M.A.; Moronta, A. Reactive H2S chemisorption on mesoporous silica molecular sieve-supported CuO or ZnO. Microporous Mesoporous Mater. 2013, 168, 111–120. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Tzounis, L.; Sebastian, V.; Baker, M.A.; Hinder, S.J.; Polychronopoulou, K.; Goula, M.A. Investigating the correlation between deactivation and the carbon deposited on the surface of Ni/Al2O3 and Ni/La2O3-Al2O3 catalysts during the biogas reforming reaction. Appl. Surf. Sci. 2019, 474, 42–56. [Google Scholar] [CrossRef]
- Gangwal, S.K.; Harkins, S.M.; Stogner, J.M.; Woods, M.C.; Rogers, T.N. Bench Scale Testing of Novel High-Temperature Desulfurization Sorbents; Final Report No. DOE/MC/23126–2662; NTIS/DE89000935; Research Triangle Institute: Research Triangle Park, NC, USA, 1988. [Google Scholar]
- Garces, H.F.; Galindo, H.M.; Garces, L.J.; Hunt, J.; Morey, A.; Suib, S.L. Low temperature H2S dry-desulfurization with zinc oxide. Microporous Mesoporous Mater. 2010, 127, 190–197. [Google Scholar] [CrossRef]
- Westmoreland, P.R.; Harrison, D.P. Evaluation of candidate solids for high-temperature desulfurization of low-Btu gases. Environ. Sci. Technol. 1976, 10, 659–661. [Google Scholar] [CrossRef]
- Portela, R.; Marcos, F.R.; Leret, P.; Fernández, J.F.; Bañares, M.A.; Ávila, P. Nanostructured ZnO/sepiolite monolithic sorbents for H2S removal. J. Mater. Chem. A 2015, 3, 1306–1316. [Google Scholar] [CrossRef]
- Samokhvalov, A.; Tatarchuk, B.J. Characterization of active sites, determination of mechanisms of H2S, COS and CS2 sorption and regeneration of ZnO low-temperature sorbents: Past current and perspectives. Phys. Chem. Chem. Phys. 2011, 13, 3197–3209. [Google Scholar] [CrossRef] [PubMed]
- Jalan, V. High-temperature desulfurization of coal gases by regenerative sorption. In Proceedings of the 1981 International Gas Research Conference, Los Angeles, CA, USA, 28 September–1 October 1981; pp. 291–303. [Google Scholar]
- Davidson, J.M.; Lawrie, C.H.; Sohail, K. Kinetics of the absorption of hydrogen sulfide by high purity and doped high surface area zinc oxide. Ind. Eng. Chem. Res. 1995, 34, 2981–2989. [Google Scholar] [CrossRef]
- Sasaoka, E.; Hirano, S.; Kasasoka, S.; Sakata, Y. Characterization of reaction between zinc oxide and hydrogen sulfide. Energy Fuels 1994, 8, 1100–1105. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Maiti, A. Adsorption and decomposition of H2S on MgO(100), NiMgO(100), and ZnO(0001) surfaces: A first-principles density functional study. J. Phys. Chem. B 2000, 104, 3630–3638. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Chaturvedi, S.; Kuhn, M.; Hrbek, J. Reaction of H2S and S2 with metal/oxide surfaces: Band-gap size and chemical reactivity. J. Phys. Chem. B 1998, 102, 5511–5519. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Jirsak, T.; Chaturvedi, S. Reaction of H2S with MgO (100) and Cu/MgO (100) surfaces: Band-gap size and chemical reactivity. J. Chem. Phys. 1999, 111, 8077–8087. [Google Scholar] [CrossRef]
- Carnes, C.L.; Klabunde, K.J. Unique chemical reactivities of nanocrystalline metal oxides toward hydrogen sulfide. Chem. Mater. 2002, 14, 1806–1811. [Google Scholar] [CrossRef]
- Rosso, I.; Galletti, C.; Bizzi, M.; Saracco, G.; Specchia, V. Zinc oxide sorbents for the removal of hydrogen sulfide from syngas. Ind. Eng. Chem. Res. 2003, 42, 1688–1697. [Google Scholar] [CrossRef]
- Neveux, L.; Chiche, D.; Perez-Pellitero, J.; Favergeon, L.; Gay, A.S.; Pijolat, M. New insight into the ZnO sulfidation reaction: Mechanism and kinetics modeling of the ZnS outward growth. Phys. Chem. Chem. Phys. 2013, 15, 1532–1545. [Google Scholar] [CrossRef]
- Bezverkhyy, I.; Skrzypski, J.; Safonova, O.; Bellat, J.-P. Sulfidation mechanism of pure and Cu-doped ZnO nanoparticles at moderate temperature: TEM and in situ XRD studies. J. Phys. Chem. C 2012, 116, 14423–14430. [Google Scholar] [CrossRef]
- Patrick, V.; Gavalas, G.R.; Flytzani-Stephanophoulos, M.; Jothimurugesan, K. High-temperature sulfidation-regeneration of CuO-Al2O3 sorbents. Ind. Eng. Chem. Res. 1989, 28, 931–940. [Google Scholar] [CrossRef]
- Tamhankar, S.S.; Bagajewicz, M.; Sharma, P.K.; Flytzani-Stephanopoulous, M.; Gavalas, G.R. Mixed oxide sorbents for high temperature removal hydrogen sulfide. Ind. Eng. Chem. Process Des. Dev. 1986, 25, 429–437. [Google Scholar] [CrossRef]
- Li, Z.; Flytzani-Stephanopoulous, M. Cu-Cr-O and Cu-Ce-O regenerable oxide sorbents for hot gas desulphurization. Ind. Eng. Chem. Res. 1997, 36, 187–196. [Google Scholar] [CrossRef]
- Pola-Albores, F.; Zambrano–Solís, K.; Ríos–Valdovinos, E.; Conde-Díaz, J.; Vilchis–Bravo, H.; Reyes–Nava, J.A.; Pantoja-Enríquez, J.; Moreira–Acosta, J. ZnO and Cu-based adsorbents for biogas desulfurization at room temperature. J. Mater. Sci. Mater. Electron. 2018, 9, 15597–15603. [Google Scholar] [CrossRef]
- Xue, M.; Chitrakar, R.; Sakane, K.; Ooi, K. Screening of adsorbents for removal of H2S at room temperature. Green Chem. 2003, 5, 529–534. [Google Scholar] [CrossRef]
- Skrzypski, J.; Bezverkhyy, I.; Heintz, O.; Bellat, J.P. Low temperature H2S removal with metal-doped nanostructure ZnO sorbents: Study of the origin of enhanced reactivity in Cu-containing materials. Ind. Eng. Chem. Res. 2011, 50, 5714–5722. [Google Scholar] [CrossRef]
- Elyassi, B.; Al Wahedi, Y.; Rajabbeigi, N.; Kumar, P.; Jeong, J.S.; Zhang, X.; Kumar, P.; Balasubramanian, V.V.; Katsiotis, M.S.; Mkhoyan, K.A.; et al. A high-performance adsorbent for hydrogen sulfide removal. Microporous Mesoporous Mater. 2014, 190, 152–155. [Google Scholar] [CrossRef]
- Yang, H.; Tatarchuk, B. Novel-doped zinc oxide sorbents for low temperature regenerable desulfurization applications. AIChE J. 2010, 56, 2898–2904. [Google Scholar] [CrossRef]
- Cimino, S.; Lisi, L.; De Falco, G.; Montagnaro, F.; Balsamo, M.; Erto, A. Highlighting the effect of the support during H2S adsorption at low temperature over composite Zn-Cu sorbents. Fuel 2018, 221, 374–379. [Google Scholar] [CrossRef]
- Dhage, P.; Samokhvalov, A.; Repala, D.; Duin, C.E.; Bowman, M.; Tatarchuk, B.J. Copper promoted ZnO/SiO2 regenerable sorbents for the room temperature removal of H2S from reformate gas streams. Ind. Eng. Chem. Res. 2010, 49, 8388–8396. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Fan, H.; Wang, H.; Hu, Y.; Wang, Z. Highly porous copper oxide sorbent for H2S capture at ambient temperature. Fuel 2017, 209, 329–338. [Google Scholar] [CrossRef]
- Jiang, D.; Su, L.; Ma, L.; Yao, N.; Xu, X.; Tang, H.; Li, X. Cu-Zn-Al mixed metal oxides derived from hydroxycarbonate precursors for H2S removal at low temperature. Appl. Surf. Sci. 2010, 256, 3216–3223. [Google Scholar] [CrossRef]
- Baird, T.; Denny, P.J.; Hoyle, R.; McMonagle, F.; Stirling, D.; Tweedy, J. Modified zinc oxide absorbents for low-temperature gas desulfurisation. J. Chem. Soc. Faraday Trans. 1992, 88, 3375–3382. [Google Scholar] [CrossRef]
- Baird, T.; Campbell, K.C.; Holliman, P.J.; Hoyle, R.; Stirling, D.; Williams, P.B. Mixed Co-Zn-al oxides as absorbents for low-temperature gas desulfurisation. J. Chem. Soc. Faraday Trans. 1995, 91, 3219–3230. [Google Scholar] [CrossRef]
- Shah, M.S.; Tsapatsis, M.; Siepmann, J.I. Hydrogen sulfide capture: From absorption in polar liquids to oxide, zeolite, and metal-organic framework adsorbents and membranes. Chem. Rev. 2017, 117, 9755–9803. [Google Scholar] [CrossRef]
- Polychronopoulou, K.; Fierro, J.L.G.; Efstathiou, A.M. Novel Zn-Ti-based mixed metal oxides for low-temperature adsorption of H2S from industrial gas streams. Appl. Catal. B Environ. 2005, 57, 125–137. [Google Scholar] [CrossRef]
- Liu, D.; Chen, S.; Fei, X.; Huang, C.; Zhang, Y. Regenerable CuO-based adsorbents for low temperature desulfurization application. Ind. Eng. Chem. Res. 2015, 54, 3556–3562. [Google Scholar] [CrossRef]
- Jung, S.Y.; Lee, S.J.; Lee, T.J.; Ryu, C.K.; Kim, J.C. H2S removal and regeneration properties of Zn-Al-based sorbents promoted with various promoters. Catal. Today 2006, 111, 217–222. [Google Scholar] [CrossRef]
- Thanakunpaisit, N.; Jantarachat, N.; Onthong, U. Removal of hydrogen sulfide from biogas using laterite materials as an adsorbent. Energy Procedia 2017, 138, 1134–1139. [Google Scholar] [CrossRef]
- Wang, Z.; Flytzani-Stephanopoulos, M. Cerium oxide-based sorbents for regenerative hot reformate gas desulfurization. Energy Fuels 2005, 19, 2089–2097. [Google Scholar] [CrossRef]
- Kobayashi, M.; Shirai, H.; Nunokawa, M. High-temperature sulfidation behavior of reduced zinc ferrite in simulated coal gas revealed by in situ X–ray diffraction analysis and mössbauer spectroscopy. Energy Fuels 2002, 16, 601–607. [Google Scholar] [CrossRef]
- Zhang, R.; Huang, J.; Zhao, J.; Sun, Z.; Wang, Y. Sol-Gel auto-combustion synthesis of zinc ferrite for moderate temperature desulfurization. Energy Fuels 2007, 21, 2682–2687. [Google Scholar] [CrossRef]
- Pahalagedara, L.R.; Poyraz, A.S.; Song, W.; Kuo, C.H.; Pahalagedara, M.N.; Meng, Y.T.; Suib, S.L. Low temperature desulfurization of H2S: High sorption capacities by mesoporous cobalt oxide via increased H2S diffusion. Chem. Mater. 2014, 26, 6613–6621. [Google Scholar] [CrossRef]
- Van Nisselrooy, P.F.M.T.; Lagas, J.A. Superclaus reduces SO2 emission by the use of a new selective oxidation catalyst. Catal. Today 1993, 16, 263–271. [Google Scholar] [CrossRef]
- DesMarias, T.L.; Costa, M. Mechanisms of chromium-induced toxicity. Curr. Opin. Toxicol. 2019, 14, 1–7. [Google Scholar] [CrossRef]
- Barba, D.; Palma, V.; Ciambelli, P. Screening of catalysts for H2S abatement from biogas to feed molten carbonate fuel cells. Int. J. Hydrog. Energy 2013, 38, 328–335. [Google Scholar] [CrossRef]
- Palma, V.; Barba, D. Vanadium-ceria catalysts for H2S abatement from biogas to feed to MCFC. Int. J. Hydrog. Energy 2017, 42, 1891–1898. [Google Scholar] [CrossRef]
- Fan, H.L.; Sun, T.; Zhao, Y.P.; Shangguan, J.; Lin, J.Y. Three dimensionally ordered macroporous iron oxide for removal of H2S at medium temperatures. Environ. Sci. Technol. 2013, 47, 4859–4865. [Google Scholar] [CrossRef]
- Huang, G.; He, E.; Wang, Z.; Fan, H.; Shangguan, J.; Croiset, E.; Chen, Z. Synthesis and characterization of γ-Fe2O3 for H2S removal at low temperature. Ind. Eng. Chem. Res. 2015, 54, 8469–8478. [Google Scholar] [CrossRef]
- Wang, L.J.; Fan, H.L.; Shangguan, J.; Croiset, E.; Chen, Z.; Wang, H.; Mi, J. Design of a sorbent to enhance reactive adsorption of hydrogen sulfide. Appl. Mater. Interfaces 2014, 6, 21167–21177. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yang, C.; Zhao, Y.R.; Fan, H.L.; De Wang, Z.; Shangguan, J.; Mi, J. Synthesis of porous cobalt oxide and its performance for H2S removal at room temperature. Ind. Eng. Chem. Res. 2017, 56, 12621–12629. [Google Scholar] [CrossRef]
- Wang, X.; Sun, T.; Yang, J.; Zhao, L.; Jia, J. Low-temperature H2S removal from gas streams with SBA-15 supported ZnO nanoparticles. Chem. Eng. J. 2008, 142, 48–55. [Google Scholar] [CrossRef]
- Wang, X.; Jia, J.; Zhao, L.; Sun, T. Mesoporous SBA-15 supported iron oxide: A potent catalyst for hydrogen sulfide removal. Water Air Soil Pollut. 2008, 193, 247–257. [Google Scholar] [CrossRef]
- Geng, Q.; Wang, L.J.; Yang, C.; Zhang, H.Y.; Zhao, Y.R.; Fan, H.L.; Huo, C. Room-temperature hydrogen sulfide removal with zinc oxide nanoparticle/molecular sieve prepared by melt infiltration. Fuel Process. Technol. 2019, 185, 26–37. [Google Scholar] [CrossRef]
- Zeng, H.Z.; Qiu, K.Q.; Du, Y.Y.; Li, W.Z. A new way to synthesize ZnS nanoparticles. Chin. Chem. Lett. 2007, 18, 483–486. [Google Scholar] [CrossRef]
- Hussain, M.; Abbas, N.; Fino, D.; Russo, N. Novel mesoporous silica supported ZnO adsorbents for the desulphurization of biogas at low temperatures. Chem. Eng. J. 2012, 188, 222–232. [Google Scholar] [CrossRef]
- Li, L.; Sun, T.H.; Shu, C.H.; Zhang, H.B. Low temperature H2S removal with 3-D structural mesoporous molecular sieves supported ZnO from gas stream. J. Hazard. Mater. 2016, 311, 142–150. [Google Scholar] [CrossRef]
- Hong, Y.S.; Sin, K.R.; Pak, J.S.; Kim, C.J.; Liu, B.S. Kinetic analysis of H2S removal over mesoporous Cu-Mn mixed oxide/SBA-15 and La-Mn mixed oxide/KIT-6 sorbents during hot coal gas desulfurization using the deactivation kinetics model. Energy Fuels 2017, 31, 9874–9880. [Google Scholar] [CrossRef]
- Daneshyar, A.; Ghaedi, M.; Sabzehmeidani, M.M.; Daneshyar, A. H2S adsorption onto Cu-Zn-Ni nanoparticles loaded activated carbon and Ni-Co nanoparticles loaded γ-Al2O3: Optimization and adsorption isotherms. J. Colloid Interface Sci. 2017, 490, 553–561. [Google Scholar] [CrossRef]
- de Falco, G.; Montagnaro, F.; Balsamo, M.; Erto, A.; Deorsola, F.A.; Lisi, L.; Cimino, S. Synergic effect of Zn and Cu oxides dispersed on activated carbon during reactive adsorption of H2S at room temperature. Microporous Mesoporous Mater. 2018, 257, 135–146. [Google Scholar] [CrossRef]
- Balsamo, M.; Cimino, S.; De Falco, G.; Erto, A.; Lisi, L. ZnO-CuO supported on activated carbon for H2S removal at room temperature. Chem. Eng. J. 2016, 304, 399–407. [Google Scholar] [CrossRef]
- Tian, H.; Wu, J.; Zhang, W.; Yang, S.; Li, F.; Qi, Y.; Zhou, R.; Qi, X.; Zhao, L.; Wang, X. High performance of Fe nanoparticles/carbon aerogel sorbents for H2S Removal. Chem. Eng. J. 2017, 313, 1051–1060. [Google Scholar] [CrossRef]
- Wu, M.; Su, Z.; Fan, H.; Mi, J. New way of removing hydrogen sulfide at a high temperature: Microwave desulfurization using an iron-based sorbent supported on active coke. Energy Fuels 2017, 31, 4263–4272. [Google Scholar] [CrossRef]
- Gupta, R.P.; Gangwal, S.K. Enhanced Durability of Desulfurization Sorbents for Fluidized-Bed Applications; Topical ReportUnder U.S. Department of Energy Contract No. DOE/MC/250063271; NTIS/DE9300247; Research Triangle Institute: Research Triangle Park, NC, USA, 1992. [Google Scholar]
- UOPLLC. Handling of Zeolite Molecular Sieve Adsorbents in Process Units. UOP5085. Available online: https://www.uop.com/?document=uop-molsiv-handling-procedures-brochure&download=1 (accessed on 14 April 2009).
- Lee, S.; Lee, T.; Kim, D. Adsorption of hydrogen sulfide from gas streams using the amorphous composite of α-FeOOH and activated carbon powder. Ind. Eng. Chem. Res. 2017, 56, 3116–3122. [Google Scholar] [CrossRef]
- Jie, M.; Yongyan, Z.; Yongsheng, Z.; Ting, G.; Huiling, F. Semi-coke-supported mixed metal oxides for hydrogen sulfide removal at high temperatures. Environ. Eng. Sci. 2012, 29, 611–616. [Google Scholar] [CrossRef]
- Ko, T.H. Removal of hydrogen sulfur from coal-derived gas by iron oxides in various oxisols. Environ. Eng. Sci. 2008, 25, 969–974. [Google Scholar] [CrossRef]
- Song, H.S.; Park, M.G.; Kwon, S.J.; Yi, K.B.; Croiset, E.; Chen, Z.; Nam, S.C. Hydrogen sulfide adsorption on nano-sized zinc oxide/reduced graphite oxide composite at ambient condition. Appl. Surf. Sci. 2013, 276, 646–652. [Google Scholar] [CrossRef]
- Seredych, M.; Mabayoje, O.; Bandosz, T.J. Visible-light-enhanced interactions of hydrogen sulfide with composites of zinc (oxy) hydroxide with graphite oxide and grapheme. Langmuir 2011, 28, 1337–1346. [Google Scholar] [CrossRef]
- Mabayoje, O.; Seredych, M.; Bandosz, T.J. Enhanced adsorption of hydrogen sulfide on mixed zinc/cobalt hydroxides: Effect of morphology and an increased number of surface hydroxyl groups. J. Colloid Interface Sci. 2013, 405, 218–225. [Google Scholar] [CrossRef]
- Lee, S.; Kim, D. Enhanced adsorptive removal of hydrogen sulfide from gas stream with zinc iron hydroxide at room temperature. Chem. Eng. J. 2019, 363, 43–48. [Google Scholar] [CrossRef]
- Arcibar-Orozco, J.A.; Wallace, R.; Mitchell, J.K.; Bandosz, T.J. Role of surface chemistry and morphology in the reactive adsorption of H2S on iron (HYDR) oxide/graphite oxide composites. Langmuir 2015, 31, 2730–2742. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.C.; Song, C.S.; Andresen, J.M.; Miller, B.G.; Scaroni, A.W. Novel polyethylenimine-modified mesoporous molecular sieve of MCM-41 type as high-capacity adsorbent for CO2 capture. Energy Fuels 2002, 16, 1463–1469. [Google Scholar] [CrossRef]
- Wang, X.X.; Schwartz, V.; Clark, J.C.; Ma, X.L.; Overbury, S.H.; Xu, X.C.; Song, C.S. Infrared study of CO2 sorption over “molecular basket” sorbent consisting of polyethylenimine-modified mesoporous molecular sieve. J. Phys. Chem. C 2009, 113, 7260–7268. [Google Scholar] [CrossRef]
- Xu, X.C.; Song, C.S.; Andresen, J.M.; Miller, B.G.; Scaroni, A.W. Preparation and characterization of novel CO2 “molecular basket” adsorbents based on polymer-modified mesoporous molecular sieve MCM-41. Microporous Mesoporous Mater. 2003, 62, 29–45. [Google Scholar] [CrossRef]
- Xu, X.C.; Song, C.S.; Miller, B.G.; Scaroni, A.W. Influence of moisture on CO2 separation from gas mixture by a nanoporous adsorbent based on polyethylenimine-modified molecular sieve MCM-41. Ind. Eng. Chem. Res. 2005, 44, 8113–8119. [Google Scholar] [CrossRef]
- Xu, X.C.; Song, C.S.; Miller, B.G.; Scaroni, A.W. Adsorption separation of carbon dioxide from flue gas of natural gas-fired boiler by a novel nanoporous “molecular basket” adsorbent. Fuel Process. Technol. 2005, 86, 1457–1472. [Google Scholar] [CrossRef]
- Ma, X.L.; Wang, X.X.; Song, C.S. “Molecular basket” sorbents for separation of CO2 and H2S from various gas streams. J. Am. Chem. Soc. 2009, 131, 5777–5783. [Google Scholar] [CrossRef]
- Tsuda, T.; Fujiwara, T. Polyethyleneimine and macrocyclic polyamine silica gels acting as carbon dioxide absorbents. J. Chem. Soc. Chem. Commun. 1992, 1659–1661. [Google Scholar] [CrossRef]
- Tsuda, T.; Fujiwara, T.; Taketani, Y.; Saegusa, T. Amino silica gels acting as a carbon dioxide absorbent. Chem. Lett. 1992, 21, 2161–2164. [Google Scholar] [CrossRef]
- Franchi, R.S.; Harlick, P.J.E.; Sayari, A. Applications of pore–expanded mesoporous silica. 2. Development of a high–capacity, water-tolerant adsorbent for CO2. Ind. Eng. Chem. Res. 2005, 44, 8007–8013. [Google Scholar] [CrossRef]
- Harlick, P.J.E.; Sayari, A. Applications of pore-expanded mesoporous silica. 5. Triamine grafted material with exceptional CO2 dynamic and equilibrium adsorption performance. Ind. Eng. Chem. Res. 2007, 46, 446–458. [Google Scholar] [CrossRef]
- Harlick, P.J.E.; Sayari, A. Applications of pore-expanded mesoporous silicas. 3. Triamine silane grafting for enhanced CO2 adsorption. Ind. Eng. Chem. Res. 2006, 45, 3248–3255. [Google Scholar] [CrossRef]
- Harlick, P.J.E.; Sayari, A. Amine grafted; pore-expanded MCM-41 for acid gas removal: Effect of grafting temperature; water; and amine type on performance. Stud. Surf. Sci. Catal. 2005, 158, 987–994. [Google Scholar]
- Serna-Guerrero, R.; Dana, E.; Sayari, A. New insights into the interactions of CO2 with amine-functionalized silica. Ind. Eng. Chem. Res. 2008, 47, 9406–9412. [Google Scholar] [CrossRef]
- Belmabkhout, Y.; De Weireld, G.; Sayari, A. Amine-bearing mesoporous silica for CO2 and H2S removal from natural gas and biogas. Langmuir 2009, 25, 13275–13278. [Google Scholar] [CrossRef]
- Belmabkhout, Y.; Sayari, A. Effect of pore expansion and amine functionalization of mesoporous silica on CO2 adsorption over a wide range of conditions. Adsorption 2009, 15, 318–328. [Google Scholar] [CrossRef]
- Belmabkhout, Y.; Serna-Guerrero, R.; Sayari, A. Adsorption of CO2-containing gas mixtures over amine-bearing pore-expanded MCM-41 silica: Application for gas purification. Ind. Eng. Chem. Res. 2010, 49, 359–365. [Google Scholar] [CrossRef]
- Wang, X.; Ma, X.; Xu, X.; Sun, L.; Song, C. Mesoporous-molecular-sieve-supported polymer sorbents for removing H2S from hydrogen gas streams. Top. Catal. 2008, 49, 108–117. [Google Scholar] [CrossRef]
- Yoosuk, B.; Wongsanga, T.; Prasassarakich, P. CO2 and H2S binary sorption on polyamine modified fumed silica. Fuel 2016, 168, 47–53. [Google Scholar] [CrossRef]
- Quan, W.; Wang, X.; Song, C. Selective removal of H2S from biogas using solid amine-based “molecular basket” sorbent. Energy Fuels 2017, 31, 9517–9528. [Google Scholar] [CrossRef]
- Deng, Y.; Vellingiri, K.; Kim, K.-H.; Boukhvalov, D.W.; Philip, L. Activation strategies of metal-organic frameworks for the sorption of reduced sulfur compounds. Chem. Eng. J. 2015, 350, 747–756. [Google Scholar] [CrossRef]
- Hamon, L.; Serre, C.; Devic, T.; Loiseau, T.; Millange, F.; Férey, G.; Weireld, G.D. Comparative study of hydrogen sulfide adsorption in the MIL-53(Al, Cr, Fe), MIL-47(V), MIL-100(Cr), and MIL-101(Cr) metal-organic frameworks at room temperature. J. Am. Chem. Soc. 2009, 131, 8775–8777. [Google Scholar] [CrossRef] [PubMed]
- Allan, P.K.; Wheatley, P.S.; Aldous, D.; Mohideen, M.I.; Tang, C.; Hriljac, J.A.; Megson, I.L.; Chapman, K.W.; De Weireld, G.; Vaesen, S.; et al. Metal–organic frameworks for the storage and delivery of biologically active hydrogen sulfide. Dalton Trans. 2012, 41, 4060–4066. [Google Scholar] [CrossRef]
- Chavan, S.; Bonino, F.; Valenzano, L.; Civalleri, B.; Lamberti, C.; Acerbi, N.; Cavka, J.H.; Leistner, M.; Bordiga, S. Fundamental aspects of H2S adsorption on CPO-27-Ni. J. Phys. Chem. C 2013, 117, 15615–15622. [Google Scholar] [CrossRef]
- Yang, Q.; Vaesen, S.; Vishnuvarthan, M.; Ragon, F.; Serre, C.; Vimont, A.; Daturi, M.; De Weireld, G.; Maurin, G. Probing the adsorption performance of the hybrid porous MIL-68(Al): A synergic combination of experimental and modelling tools. J. Mater. Chem. 2012, 22, 10210–10220. [Google Scholar] [CrossRef]




| Property | Value |
|---|---|
| Molar mass | 34.08 g mol−1 |
| Melting point | −82 °C |
| Boiling point | −60.25 °C |
| Density | 1.363 g dm−3 |
| Dipole moment | 0.97 D |
| Critical temperature | 100.25 °C |
| Critical pressure | 89.7 bar |
| Acidity | 7 pKa |
| Solubility in water (H2O) | 4 g dm−3 (at 20 °C) |
| Lower explosive limit (LEL) | 4% |
| Upper explosive limit (UEL) | 44% |
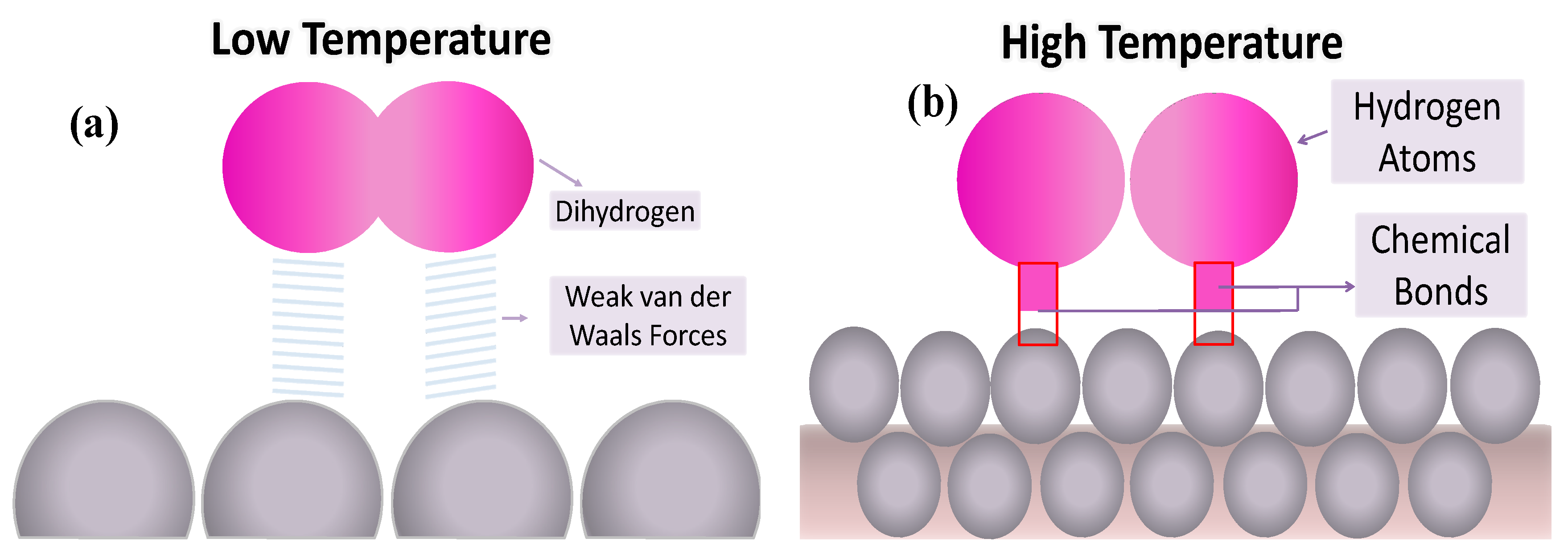
| Physisorption | Chemisorption |
|---|---|
| Electrostatic interactions and Van der Waals forces are involved | Covalent bonds are formed between the surface and the adsorbed molecules |
| Fast | Slow |
| Occurs in at low temperature | Occurs at high temperature |
| Is reversible | Is irreversible |
| Multilayers are formed | Monolayers are formed |
| Activation energy is not required | Activation energy is required |
| Mixture recommended by Norm ASTM D6646–03 (containing approximately 20% O2, 15 g/m3 H2O, and 7.5 g/m3 H2S, balanced with nitrogen). | Sample label | Sample weight (g) | Total feed volume (m3) | H2S concentration (g/m3) | Adsorption capacity for S (%) |
| I | 49.55 | 1.380 | 7.88 | 22.78 | |
| Cu I | 54.40 | 0.606 | 7.93 | 9.88 | |
| Mn | 65.50 | 0.158 | 7.89 | 4.64 | |
| Cu II | 50.40 | 0.424 | 7.89 | 6.45 | |
| K | 48.35 | 0.686 | 6.76 | 9.57 | |
| Mg | 55.68 | 0.289 | 6.76 | 3.47 | |
| Mixture without oxygen | I | 50.18 | 0.016 | 7.44 | 0.33 |
| Cu I | 54.59 | 0.031 | 6.87 | 0.26 | |
| Mn | 67.33 | 0.056 | 6.87 | 0.13 | |
| Cu II | 50.93 | 0.077 | 6.87 | 2.27 | |
| K | 50.33 | 0.062 | 6.87 | 0.38 | |
| Mg | 56.99 | 0.027 | 7.44 | 0.17 | |
| Mixture without water vapor | I | 48.83 | 1.004 | 6.09 | 11.90 |
| Cu I | 53.86 | 0.421 | 5.63 | 9.77 | |
| Mn | 68.75 | 0.248 | 5.63 | 1.87 | |
| Cu II | 50.00 | 0.420 | 5.56 | 7.48 | |
| K | 51.54 | 0.871 | 5.16 | 11.21 | |
| Mg | 55.56 | 0.230 | 5.16 | 1.85 | |
| Mixture without oxygen and water vapor | I | 50.04 | 0.027 | 6.36 | 0.47 |
| Cu I | 53.31 | 0.043 | 6.36 | 0.56 | |
| Mn | 66.83 | 0.042 | 6.36 | 0.23 | |
| Cu II | 50.47 | 0.075 | 6.36 | 2.87 | |
| K | 0.079 | 6.36 | 0.97 | 51.62 | |
| Mg | 0.028 | 6.36 | 0.31 | 57.12 |
| Adsorbent | SSA (m2/g) | mg S/g Sorbent | T (°C) | Gas Composition |
|---|---|---|---|---|
| CuO | 16.4 | 17.7 | 40 | 0.1% H2S in CO2 |
| Cu3Al | 72.3 | 86.0 | ||
| Co0.44Cu3Al | 87.4 | 73.4 | ||
| Ni0.44Cu3Al | 86.0 | 55.7 | ||
| Ce0.44Cu3Al | 82.1 | 91.1 | ||
| 207 | 70 | |||
| 212 | 100 | |||
| 220 | 130 | |||
| CuO | 16.4 | 25.3 | 40 | 0.1% S in N2 |
| Cu3Al | 72.3 | 106.0 | ||
| Fe0.44Cu3Al | 96.7 | 125.0 |
| Sorbent | Inlet H2S (mg/m3) | Sulfur capacity mg S/g | Utilization ration of ZnO (%) | Source |
|---|---|---|---|---|
| Commercial ZnO | 500 | 37.7 | 16.00 | [141] |
| ZnO (15%)/MCM–41 | 300 | 5.9 | 10.01 | [142] |
| ZnO (15%)/SBA–15–S | 300 | 18.5 | 31.37 | [142] |
| ZnO(15%)/SBA–15–F | 300 | 15.6 | 26.46 | [142] |
| Z30/MCM–48 | 1200 | 14.8 | 12.60 | [143] |
| M–Z20/MCM–41 | 500 | 54.9 | 69.80 | [140] |
| M–Z20/SBA–15 | 500 | 41.0 | 52.20 | [140] |
| M–Z20/MCM–48 | 500 | 53.2 | 45.10 | [140] |
| T (°K) | Adsorbent | Kc | r2 | ΔG°ads (kj·mol−1) | ΔH°ads (kj·mol−1) |
|---|---|---|---|---|---|
| 288 | Cu–Zn–Ni/AC | 6.74 | 0.970 | −4.54 | −38.07 |
| 298 | 3.58 | −3.14 | |||
| 308 | 2.22 | −2.04 | |||
| 318 | 1.66 | −1.40 | |||
| 288 | Ni–Co/γ–Al2O3 | 6.08 | 0.975 | −4.32 | −37.98 |
| 298 | 3.23 | −2.90 | |||
| 308 | 2.09 | −1.88 | |||
| 318 | 1.53 | −1.13 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgiadis, A.G.; Charisiou, N.D.; Goula, M.A. Removal of Hydrogen Sulfide From Various Industrial Gases: A Review of The Most Promising Adsorbing Materials. Catalysts 2020, 10, 521. https://doi.org/10.3390/catal10050521
Georgiadis AG, Charisiou ND, Goula MA. Removal of Hydrogen Sulfide From Various Industrial Gases: A Review of The Most Promising Adsorbing Materials. Catalysts. 2020; 10(5):521. https://doi.org/10.3390/catal10050521
Chicago/Turabian StyleGeorgiadis, Amvrosios G., Nikolaos D. Charisiou, and Maria A. Goula. 2020. "Removal of Hydrogen Sulfide From Various Industrial Gases: A Review of The Most Promising Adsorbing Materials" Catalysts 10, no. 5: 521. https://doi.org/10.3390/catal10050521
APA StyleGeorgiadis, A. G., Charisiou, N. D., & Goula, M. A. (2020). Removal of Hydrogen Sulfide From Various Industrial Gases: A Review of The Most Promising Adsorbing Materials. Catalysts, 10(5), 521. https://doi.org/10.3390/catal10050521







