Selective Catalytic Reduction of NOx by CO over Doubly Promoted MeMo/Nb2O5 Catalysts (Me = Pt, Ni, or Co)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structure by XRD and Raman Measurements
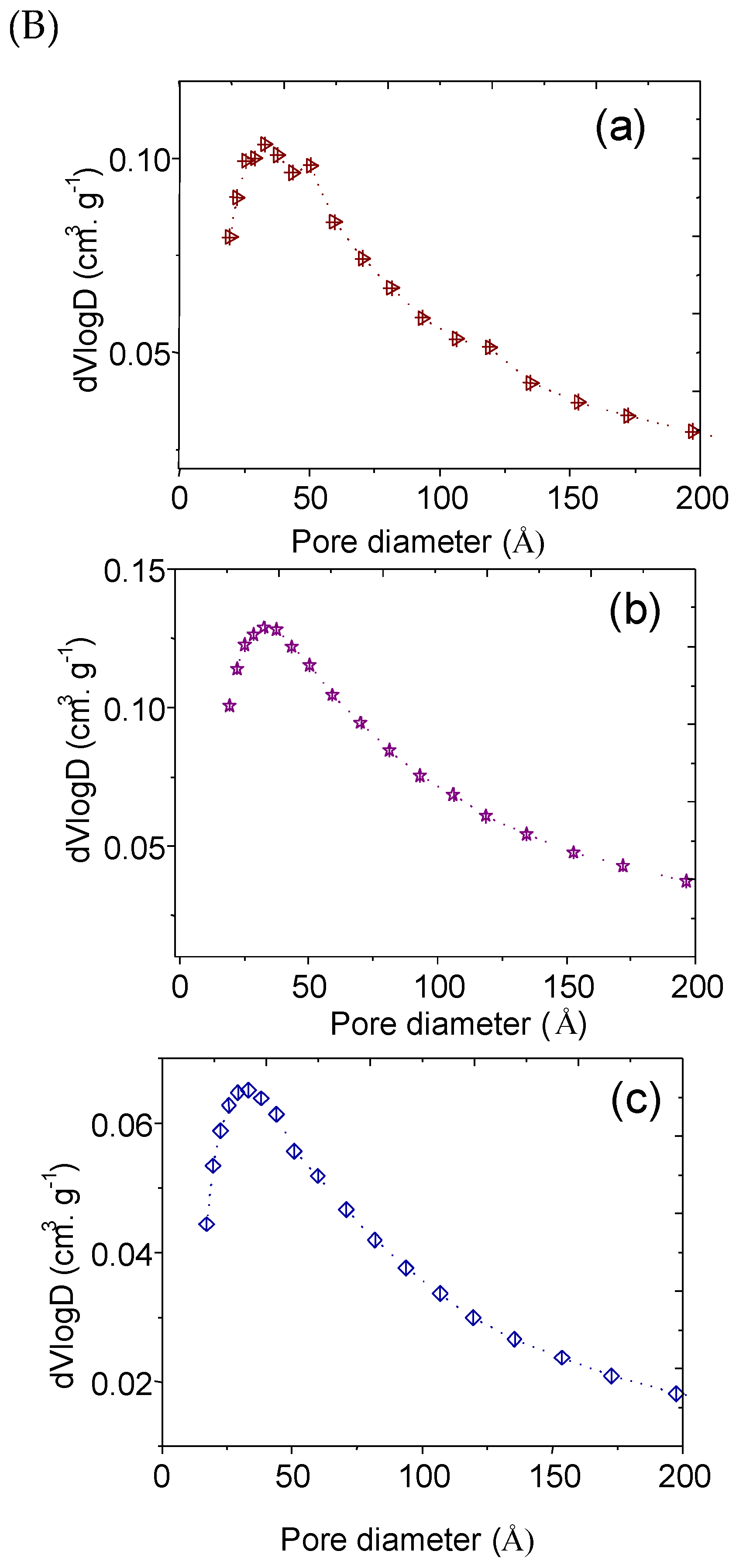
2.2. Texture and Morphology
2.3. Surface Acidity and Oxidation States
2.4. Catalytic Results
2.4.1. Temperature Effects
2.4.2. Stability of the Catalysts against Poisons
3. Materials and Methods
3.1. Nb2O5 Supported Transition Metal Catalysts Preparation
3.2. Characterizations
3.3. Catalytic Activity Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhou, H.; Ge, M.; Zhao, H.; Wu, S.; Li, M.; Su, Y. Selective Catalytic Reduction of Nitric Oxide with Propylene over Fe/Beta Catalysts Under Lean-Burn Conditions. Catalysts 2019, 9, 205. [Google Scholar] [CrossRef] [Green Version]
- He, D.; Wang, Z.; Deng, D.; Deng, S.; He, H.; Liu, L. Synthesis of Cu-SSZ-13 catalyst by using different silica sources for NO-SCR by NH3. Mol. Catal. 2020, 484, 110738. [Google Scholar] [CrossRef]
- Oton, L.F.; Oliveira, A.C.; De Araujo, J.C.; Araujo, R.S.; De Sousa, F.F.; Saraiva, G.D.; Lang, R.; Otubo, L.; Duarte, G.C.D.S.; Campos, A. Selective catalytic reduction of NOx by CO (CO-SCR) over metal-supported nanoparticles dispersed on porous alumina. Adv. Powder Technol. 2020, 31, 464–476. [Google Scholar] [CrossRef]
- Chavez-Rivas, F.; Rodriguez-Iznaga, I.; Berlier, G.; Ferro, D.T.; Concepción-Rosabal, B.; Petranovskii, V. Fe Speciation in Iron Modified Natural Zeolites as Sustainable Environmental Catalysts. Catalysts 2019, 9, 866. [Google Scholar] [CrossRef] [Green Version]
- Nascimento, J.P.S.; Oliveira, A.C.; Araujo, J.C.; De Sousa, F.F.; Saraiva, G.D.; Rodríguez-Aguado, E.; Rodríguez-Castellón, E. Combined promoting effect of molybdenum on the bimetallic Al2O3-La2O3 catalysts for NOx reduction by CO. Fuel 2020, 275, 117872. [Google Scholar] [CrossRef]
- Święs, A.; Kowalczyk, A.; Rutkowska, M.; Diaz, U.; Palomares, A.E.; Chmielarz, L. Ferrierite and Its Delaminated and Silica-Intercalated Forms Modified with Copper as Effective Catalysts for NH3-SCR Process. Catalysts 2020, 10, 734. [Google Scholar] [CrossRef]
- Ouyang, F.; Zhu, R.-S.; Sato, K.; Haneda, M.; Hamada, H. Promotion of surface SOx on the selective catalytic reduction of NO by hydrocarbons over Ag/Al2O3. Appl. Surf. Sci. 2006, 252, 6390–6393. [Google Scholar] [CrossRef]
- Resitoglu, I.A.; Altinisik, K.; Keskin, A.; Ocakoglu, K. The effects of Fe2O3 based DOC and SCR catalyst on the exhaust emissions of diesel engines. Fuel 2020, 262, 116501. [Google Scholar] [CrossRef]
- Li, L.; Wang, Y.; Zhang, L.; Yu, Y.; He, H. Low-Temperature Selective Catalytic Reduction of NOx on MnO2 Octahedral Molecular Sieves (OMS-2) Doped with Co. Catalysts 2020, 10, 396. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Li, D.; Li, K.; Farrauto, R. Enhanced propane and carbon monoxide oxidation activity by structural interactions of CeO2 with MnOx/Nb2O5-x catalysts. Appl. Catal. B Environ. 2020, 267, 118363. [Google Scholar] [CrossRef]
- Gao, E.; Pan, H.; Wang, L.; Shi, Y.; Chen, J. Identification of Main Active Sites and the Role of NO2 on NOx Reduction with CH4 over In/BEA Catalyst: A Computational Study. Catalysts 2020, 10, 572. [Google Scholar] [CrossRef]
- Kim, H.-S.; Kasipandi, S.; Kim, J.; Kang, S.-H.; Kim, J.-H.; Ryu, J.-H.; Bae, J.W. Kim Current Catalyst Technology of Selective Catalytic Reduction (SCR) for NOx Removal in South Korea. Catalysts 2020, 10, 52. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhao, L.; Duan, J.; Bi, S. Insights into deNOx processing over Ce-modified Cu-BTC catalysts for the CO-SCR reaction at low temperature by In Situ DRIFTS. Sep. Purif. Technol. 2020, 234, 116081. [Google Scholar] [CrossRef]
- Pinto, M.B.; Soares, A.L., Jr.; Quintão, M.C.; Duarte, H.A.; De Abreu, H.A. Unveiling the Structural and Electronic Properties of the B-Nb2O5 Surfaces and Their Interaction with H2O and H2O2. J. Phys. Chem. C 2018, 122, 6618–6628. [Google Scholar] [CrossRef]
- Kantcheva, M.; Cayirtepe, I.; Naydenov, A.; Ivanov, G. FT-IR spectroscopic investigation of the effect of SO2 on the SCR of NOx with propene over ZrO2–Nb2O5 catalyst. Catal. Today 2011, 176, 437–440. [Google Scholar] [CrossRef] [Green Version]
- Chary, K.V.R.; Praveen, C.; Reddy, K.K.R.; Bhaskar, T.; Rajiah, T. Characterization and catalytic properties of MoO3–V2O5/Nb2O5 catalysts. Catal. Commun. 2002, 3, 7–13. [Google Scholar] [CrossRef]
- Liu, Y.; Tursun, M.; Yu, H.; Wang, X. Surface property and activity of Pt/Nb2O5-ZrO2 for selective catalytic reduction of NO by H2. Mol. Catal. 2019, 464, 22–28. [Google Scholar] [CrossRef]
- Chary, K.V.R.; Kumar, C.P.; Murali, A.; Tripathi, A. Clearfield, studies on catalytic functionality of V2O5/Nb2O5 catalysts. J. Mol. Catal. A Chem. 2004, 216, 139–146. [Google Scholar] [CrossRef]
- Cayirtepe, I.; Naydenov, A.; Ivanov, G.; Kantcheva, M. Characterization of Niobium-zirconium Mixed Oxide as a Novel Catalyst for Selective Catalytic Reduction of NOx. Catal. Lett. 2009, 132, 438–449. [Google Scholar] [CrossRef]
- Xu, H.; Lin, Q.; Wang, Y.; Lan, L.; Liu, S.; Lin, C.; Wang, Q.; Wang, J.; Chen, Y. Promotional effect of niobium substitution on the low-temperature activity of a WO3/CeZrOx monolithic catalyst for the selective catalytic reduction of NOx with NH3. RSC Adv. 2017, 7, 47570. [Google Scholar] [CrossRef] [Green Version]
- Ye, D.; Ren, X.; Qu, R.; Liu, S.; Zheng, C.; Gao, X. Designing SO2-resistant cerium-based catalyst by modifying with Fe2O3 for the selective catalytic reduction of NO with NH3. Mol. Catal. 2019, 462, 10–18. [Google Scholar] [CrossRef]
- Zhang, S.; Tang, Y.; Nguyen, L.; Zhao, Y.-F.; Wu, Z.; Goh, T.W.; Liu, J.J.; Li, Y.; Zhu, T.; Huang, W.; et al. Catalysis on Singly Dispersed Rh Atoms Anchored on an Inert Support. ACS Catal. 2017, 8, 110–121. [Google Scholar] [CrossRef]
- Usha, N.; Sivakumar, R.; Sanjeeviraja, C.; Kuroki, Y. Effect of substrate temperature on the properties of Nb2O5: MoO3 (90:10) thin films prepared by rf magnetron sputtering technique. J. Alloys Compd. 2015, 649, 112–121. [Google Scholar] [CrossRef]
- Xin, Y.; Dong, L.; Guo, Y.; Liu, X.; Hu, Y.; Wang, Y. Correlation of the catalytic performance with Nb2O5 surface properties in the hydrodeoxygenation of lignin model compound. J. Catal. 2019, 375, 202–212. [Google Scholar] [CrossRef]
- Li, S.; Xu, Q.; Uchaker, E.; Cao, X.; Cao, G. Comparison of amorphous, pseudohexagonal and orthorhombic Nb2O5 for high-rate lithium ion insertion. CrystEngComm 2016, 18, 2532–2540. [Google Scholar] [CrossRef] [Green Version]
- Jehng, J.M.; Wachs, I.E. Structural chemistry and Raman spectra of niobium oxides. Chem. Mater. 1991, 3, 100–107. [Google Scholar] [CrossRef]
- Sobczak, I.; Ziolek, M.; Nowacka, M. Preparation and characterisation of Pt containing NbMCM-41 mesoporous molecular sieves addressed to catalytic NO reduction by hydrocarbons. Microporous Mesoporous Mater. 2005, 78, 103–116. [Google Scholar] [CrossRef]
- El-Eskandarany, M.S.; Banyan, M.; Al-Ajmi, F. Synergistic Effect of New ZrNi5/Nb2O5 Catalytic Agent on Storage Behavior of Nanocrystalline MgH2 Powders. Catalysts 2019, 9, 306. [Google Scholar] [CrossRef] [Green Version]
- Agudo, A.L.; Gil Llambias, F.; Reyes, P.; Fierro, J.G. Dispersion of molybdena and hydrodesulphurization activity of Mo/γ-Al2O3, AND Co (or Ni) Mo/γ-Al2O3 catalysts. Appl. Catal. 1981, 1, 59–70. [Google Scholar] [CrossRef]
- Yamashita, K.; Hirano, M.; Okumura, K.; Niwa, M. Activity and acidity of Nb2O5-MoO3 and Nb2O5-WO3 in the Friedel-Crafts alkylation. Catal. Today 2006, 118, 385–391. [Google Scholar] [CrossRef]
- Francisco, M.S.P.; Landers, R.; Gushikem, Y. Local order structure and surface acidity properties of a Nb2O5/SiO2 mixed oxide prepared by the sol–gel processing method. J. Solid State Chem. 2004, 177, 2432–2439. [Google Scholar] [CrossRef]
- Damyanova, S.; Dimitrov, L.; Petrov, L.; Grangé, P. Effect of niobium on the surface properties of Nb2O5-SiO2-supported Mo catalysts. Appl. Surf. Sci. 2003, 214, 68–74. [Google Scholar] [CrossRef]
- Arakawa, S.; Matsuura, Y.; Okamoto, M. Allophane–Pt nanocomposite: Synthesis and MO simulation. Appl. Clay Sci. 2014, 95, 191–196. [Google Scholar] [CrossRef]
- Oton, L.F.; Coelho, D.C.; Oliveira, A.C.; De Araujo, J.C.; Lang, R.; Rodríguez-Castellón, E.; Rodríguez-Aguado, E.; Lucredio, A.F.; Assaf, E.M.; Reyna-Alvarado, J.; et al. Structural transformation of vanadate nanotubes into vanadate oxides nanostructures during the dry reforming of methane. Mol. Catal. 2020, 480, 110641. [Google Scholar] [CrossRef]
- Araújo, J.C.; Oton, L.F.; Oliveira, A.C.; Lang, R.; Otubo, L.; Bueno, J.M. On the role of size controlled Pt particles in nanostructured Pt-containing Al2O3 catalysts for partial oxidation of methane. Int. J. Hydrogen Energy 2019, 44, 27329–27342. [Google Scholar] [CrossRef]
- Odenbrand, I. High Temperature and High Concentration SCR of NO with NH3 for the Oxyfuel Combustion Process: Fitting of Kinetics to Data from a Laboratory Reactor Experiment. Top. Catal. 2017, 60, 1317–1332. [Google Scholar] [CrossRef]
- Zhang, S.; Nguyen, L.; Liang, J.-X.; Shan, J.; Liu, J.; Frenkel, A.I.; Patlolla, A.; Huang, W.; Li, J.; Tao, F. Catalysis on singly dispersed bimetallic sites. Nat. Commun. 2015, 6, 7938. [Google Scholar] [CrossRef] [Green Version]
- Lu, Y.; Wang, J.; Yu, L.; Kovarik, L.; Zhang, X.; Hoffman, A.S.; Gallo, A.; Bare, S.R.; Sokaras, D.; Kroll, T.; et al. Identification of the active complex for CO oxidation over single-atom Ir-on-MgAl2O4 catalysts. Nat. Catal. 2018, 2, 149–156. [Google Scholar] [CrossRef]
- Hirunsit, P.; Toyao, T.; Siddiki, S.M.A.H.; Shimizu, K.-I.; Ehara, M. Origin of Nb2O5 Lewis Acid Catalysis for Activation of Carboxylic Acids in the Presence of a Hard Base. ChemPhysChem 2018, 19, 2848–2857. [Google Scholar] [CrossRef] [Green Version]
- Rasband, P.; Hecker, W. No reduction activity and FTIR characterization of rhodium on niobia-modified SiO2. Catal. Today 1990, 8, 99–111. [Google Scholar] [CrossRef]
- De Jesus, J.C.; Carrazza, J.; Pereira, P.; Zaera, F. Hydroxylation of NiO films: The effect of water and ion bombardment during the oxidation of nickel foils with O2 under vacuum. Surf. Sci. 1998, 397, 34–47. [Google Scholar] [CrossRef]
- Schulze, M.; Reissner, R.; Lorenz, M.; Radke, U.; Schnurnberger, W. Photoelectron study of electrochemically oxidized nickel and water adsorption on defined NiO surface layers. Electrochim. Acta 1999, 44, 3969–3976. [Google Scholar] [CrossRef]
- Zhuang, S. Catalytic conversion of CO, NO and SO2 on supported sulfide catalysts II. Catalytic reduction of NO and SO2 by CO. Appl. Catal. B Environ. 2001, 31, 133–143. [Google Scholar] [CrossRef]
- Pérez-Martínez, D.; Giraldo, S.A.; Centeno, A. Effects of the H2S partial pressure on the performance of bimetallic noble-metal molybdenum catalysts in simultaneous hydrogenation and hydrodesulfurization reactions. Appl. Catal. A Gen. 2006, 315, 35–43. [Google Scholar] [CrossRef]
- Zhu, J.; Gao, F.; Dong, L.; Yu, W.; Qi, L.; Wang, Z.; Dong, L.; Chen, Y. Studies on surface structure of MxOy/MoO3/CeO2 system (M = Ni, Cu, Fe) and its influence on SCR of NO by NH3. Appl. Catal. B Environm. 2010, 95, 144–152. [Google Scholar] [CrossRef]
- Zhuang, S.-X.; Magara, H.; Yamazaki, M.; Takahashi, Y.; Yamada, M. Catalytic conversion of CO, NO and SO2 on the supported sulfide catalyst: I. Catalytic reduction of SO2 by CO. Appl. Catal. B Environ. 2000, 24, 89–96. [Google Scholar] [CrossRef]
- Travert, A.; Dujardin, C.; Mauge, F.; Veilly, E.; Cristol, S.; Paul, J.-F.; Payen, E. CO Adsorption on CoMo and NiMo Sulfide Catalysts: A Combined IR and DFT Study. J. Phys. Chem. B 2006, 110, 1261–1270. [Google Scholar] [CrossRef] [PubMed]
- Xia, M.; Yue, R.; Chen, P.; Wang, M.; Jiao, T.; Zhang, L.; Zhao, Y.; Gao, F.; Wei, Z.; Li, L. Density functional theory investigation of the adsorption behaviors of SO2 and NO2 on a Pt(111) surface. Colloids Surf. A Physicochem. Eng. Asp. 2019, 568, 266–270. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, W.; Xie, W.; Qi, Z.; Wu, G.; Lv, M.; Sun, S.; Bao, J. Effects of cobalt promoter and reduction temperature on the surface species and syngas adsorption of K–Co–Mo/C catalyst for mixed alcohols synthesis. J. Mol. Catal. A Chem. 2014, 395, 269–275. [Google Scholar] [CrossRef]
- Gunasooriya, G.T.K.K.; Van Bavel, A.P.; Kuipers, H.P.; Saeys, M. CO adsorption on cobalt: Prediction of stable surface phases. Surf. Sci. 2015, 642, L6–L10. [Google Scholar] [CrossRef]
- De Sousa, H.S.A.; Da Silva, A.N.; Castro, A.J.; Campos, A.; Filho, J.M.; Oliveira, A.C. Mesoporous catalysts for dry reforming of methane: Correlation between structure and deactivation behaviour of Ni-containing catalysts. Int. J. Hydrogen Energy 2012, 37, 12281–12291. [Google Scholar] [CrossRef]











| Catalysts | a Total Acidity (µmol NH3·g−1) | Density of the Acid Sites (µmol NH3·m−2) | Textural Properties | ||
|---|---|---|---|---|---|
| BET Surface Area (m2·g−1) | Pore Volume (cm3·g−1) | b Pore Diameter (Å) | |||
| Nb2O5 | 275 | 3.2 | 87 | 0.16 | 46 |
| CoMo/Nb2O5 | 88.5 | 1.8 | 50 | 0.10 | 38 |
| NiMo/Nb2O5 | 358 | 8.3 | 43 | 0.09 | 35 |
| PtMo/Nb2O5 | 438 | 17.5 | 25 | 0.06 | 21 |
| Sample | C 1s | O 1s | Nb 3d5/2 | Mo 3d5/2 | Co 2p3/2 | Pt 4f7/2 |
|---|---|---|---|---|---|---|
| Nb2O5 | 284.8 (80) 286.2 (12) 288.6 (8) | 530.0 (90) 531.6 (10) | 207.0 | - | - | - |
| CoMo/Nb2O5 | 284.8 (76) 286.7 (11) 289.0 (13) | 530.2 (85) 531.6 (15) | 207.2 | 232.4 | 780.5 | - |
| PtMo/Nb2O5 | 284.8 (83) 286.2 (13) 288.9 (4) | 530.4 (90) 531.8 (10) | 207.3 | 231.7 (29) 232.9 (71) | - | 71.6 (16) 73.0 (70) 74.6 (14) |
| PtMo/ Nb2O5 Spent | 284.8 (78) 286.4 (18) 288.7 (4) | 530.4 (89) 532.0 (11) | 207.3 | 231.7 (19) 232.9 (81) | - | 71.4 (80) 72.7 (20) |
| Sample | C | O | Nb | Mo | Co | Pt | Mo/Nb | Pt/Nb | Co/Nb |
|---|---|---|---|---|---|---|---|---|---|
| Nb2O5 | - | 56.14 | 21.62 | - | - | - | - | - | - |
| CoMo/Nb2O5 | - | 53.98 | 16.67 | 2.75 | 5.38 | - | 0.165 | - | 0.035 |
| PtMo/Nb2O5 | - | 54.38 | 17.82 | 4.53 | - | 0.78 | 0.254 | 0.042 | - |
| PtMo/Nb2O5 Spent | 20.27 | 57.03 | 17.74 | 4.53 | - | 0.32 | 0.255 | 0.018 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nascimento, J.P.S.; Oton, L.F.; Oliveira, A.C.; Rodríguez-Aguado, E.; Rodríguez-Castellón, E.; Araujo, R.S.; Souza, M.S.; Lang, R. Selective Catalytic Reduction of NOx by CO over Doubly Promoted MeMo/Nb2O5 Catalysts (Me = Pt, Ni, or Co). Catalysts 2020, 10, 1048. https://doi.org/10.3390/catal10091048
Nascimento JPS, Oton LF, Oliveira AC, Rodríguez-Aguado E, Rodríguez-Castellón E, Araujo RS, Souza MS, Lang R. Selective Catalytic Reduction of NOx by CO over Doubly Promoted MeMo/Nb2O5 Catalysts (Me = Pt, Ni, or Co). Catalysts. 2020; 10(9):1048. https://doi.org/10.3390/catal10091048
Chicago/Turabian StyleNascimento, João Pedro S., Lais F. Oton, Alcineia C. Oliveira, Elena Rodríguez-Aguado, Enrique Rodríguez-Castellón, Rinaldo S. Araujo, Monique S. Souza, and Rossano Lang. 2020. "Selective Catalytic Reduction of NOx by CO over Doubly Promoted MeMo/Nb2O5 Catalysts (Me = Pt, Ni, or Co)" Catalysts 10, no. 9: 1048. https://doi.org/10.3390/catal10091048






