The Effect of Si on CO2 Methanation over Ni-xSi/ZrO2 Catalysts at Low Temperature
Abstract
:1. Introduction
2. Results and Discussion
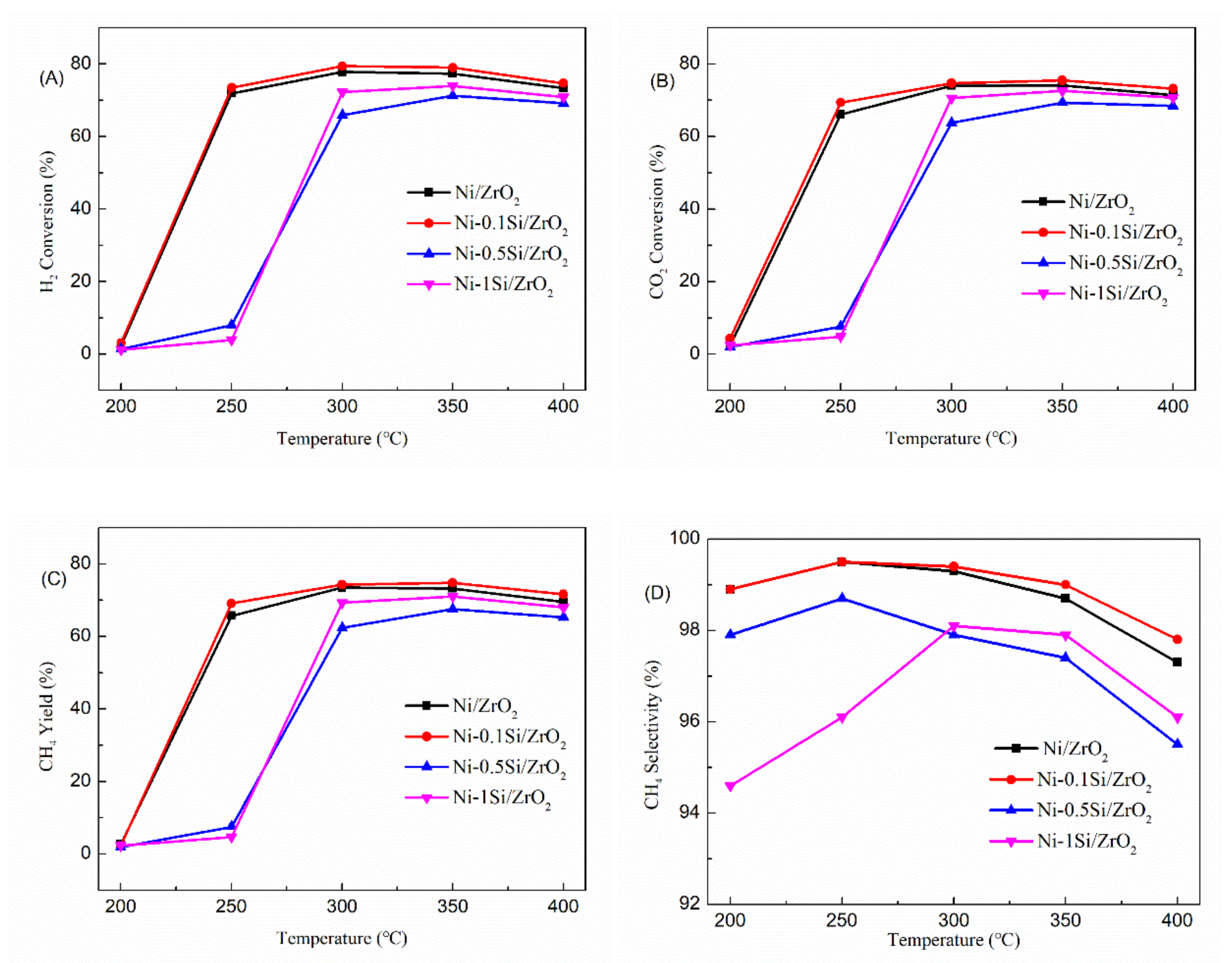
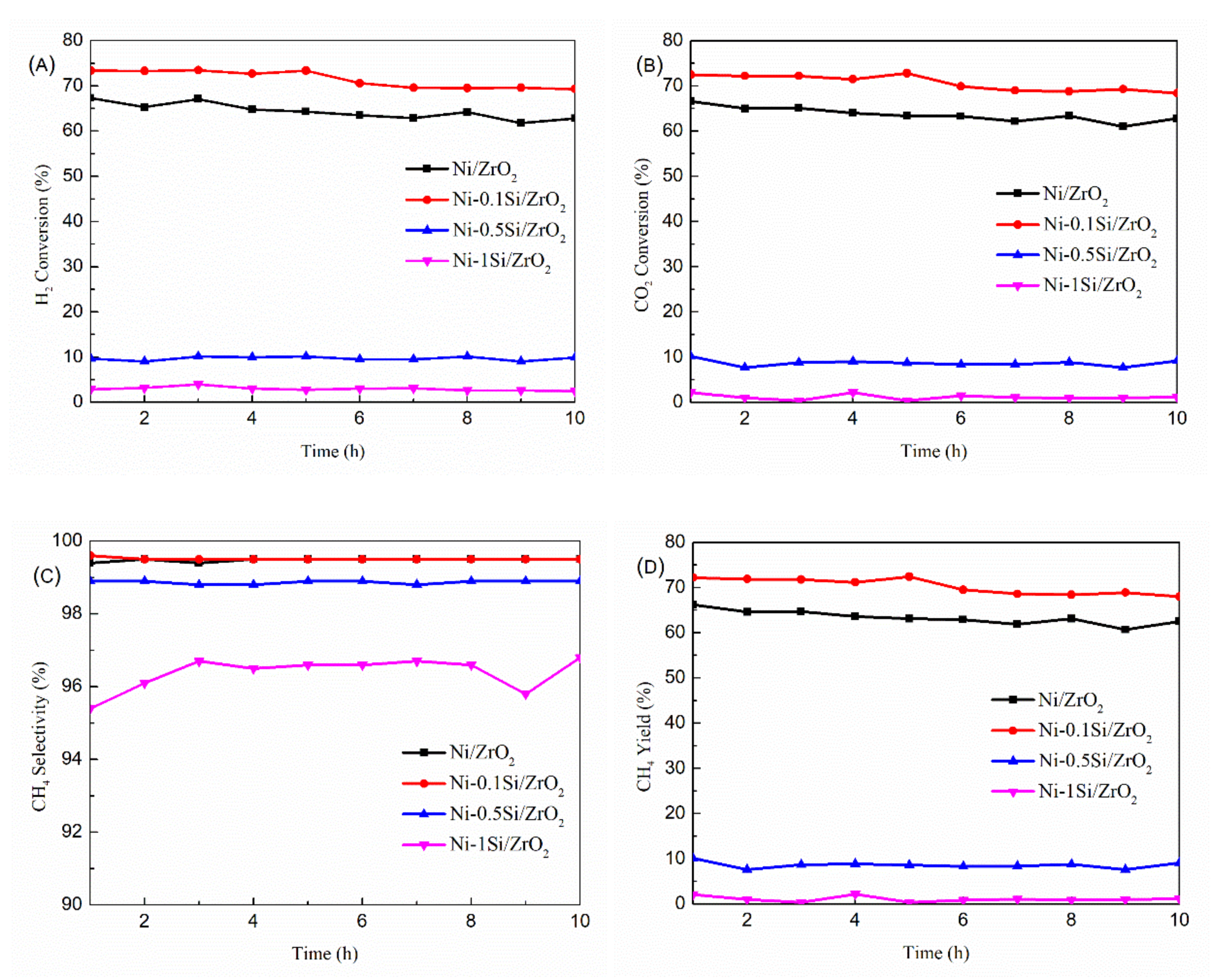
2.1. The Catalytic Performance of Ni-xSi/ZrO2 Catalysts
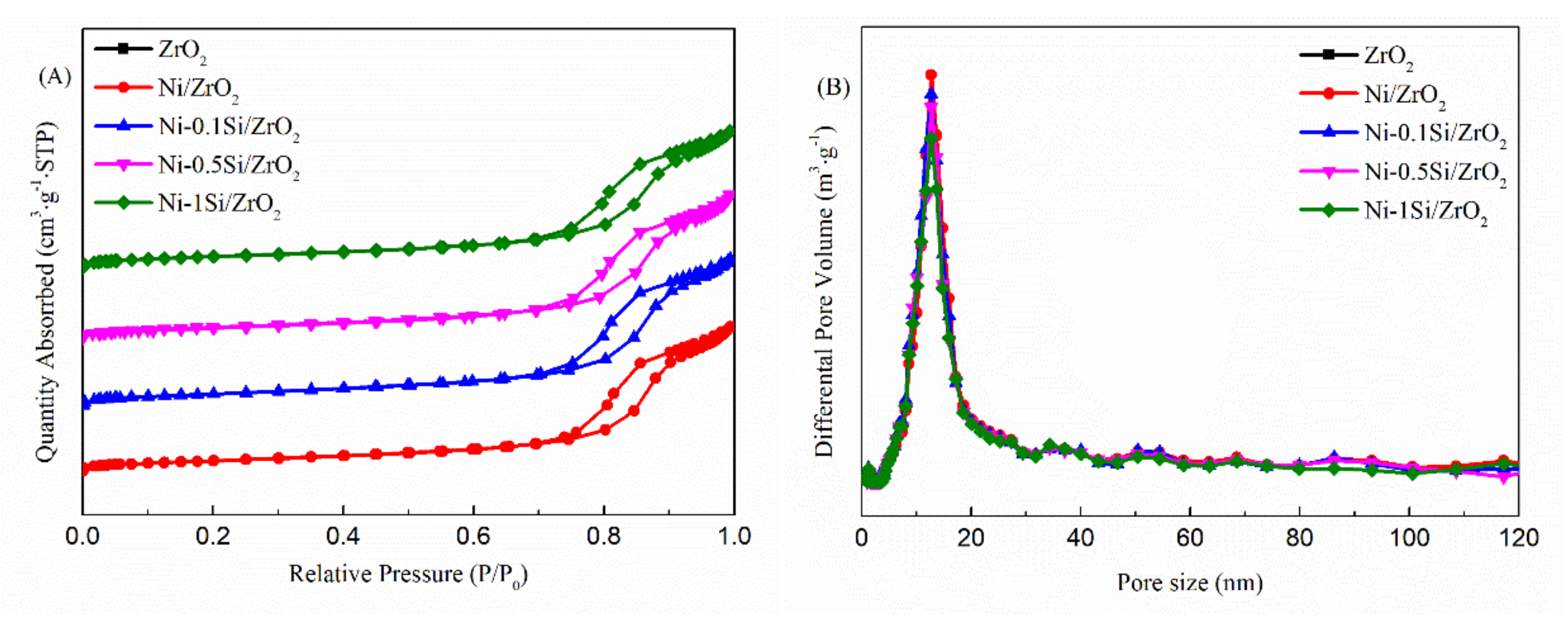
2.2. The Textural Properties of Ni-xSi/ZrO2 Catalysts
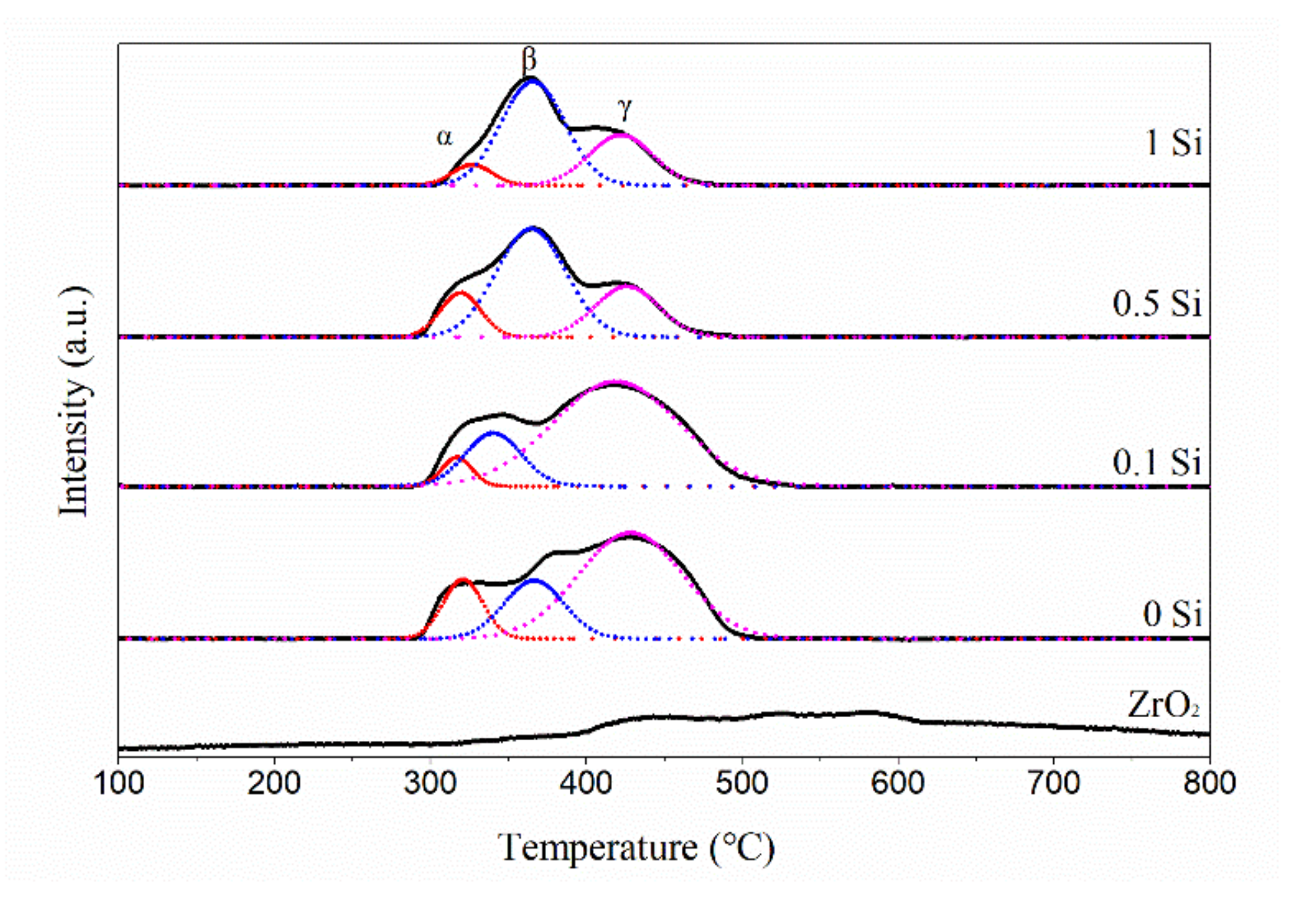
2.3. The Reducibility of Ni-xSi/ZrO2 Catalysts
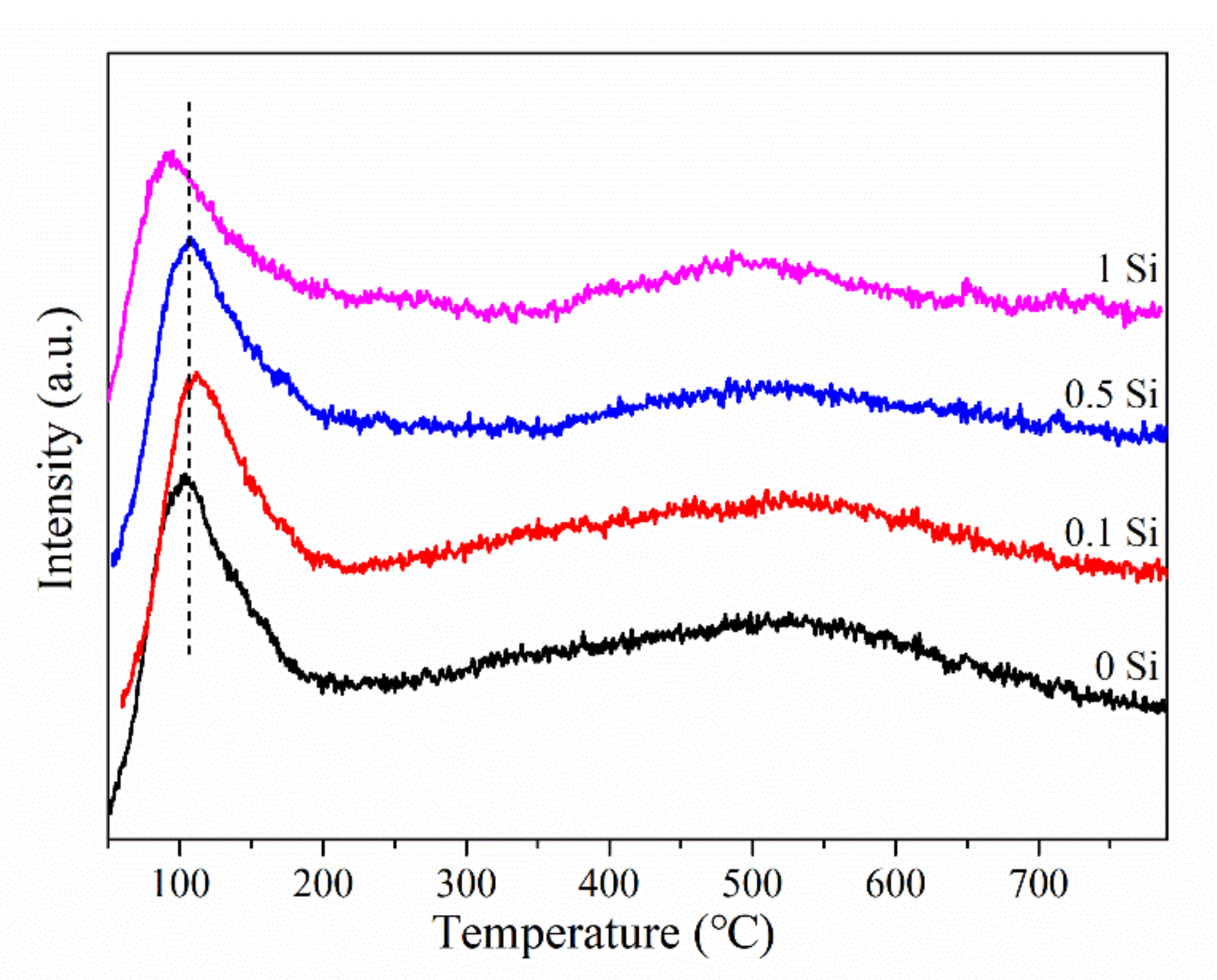
2.4. The H2-TPD and H2-Chemisorption of Ni-xSi/ZrO2 Catalysts
2.5. The Results of CO2-TPD on Ni-xSi/ZrO2 Catalysts
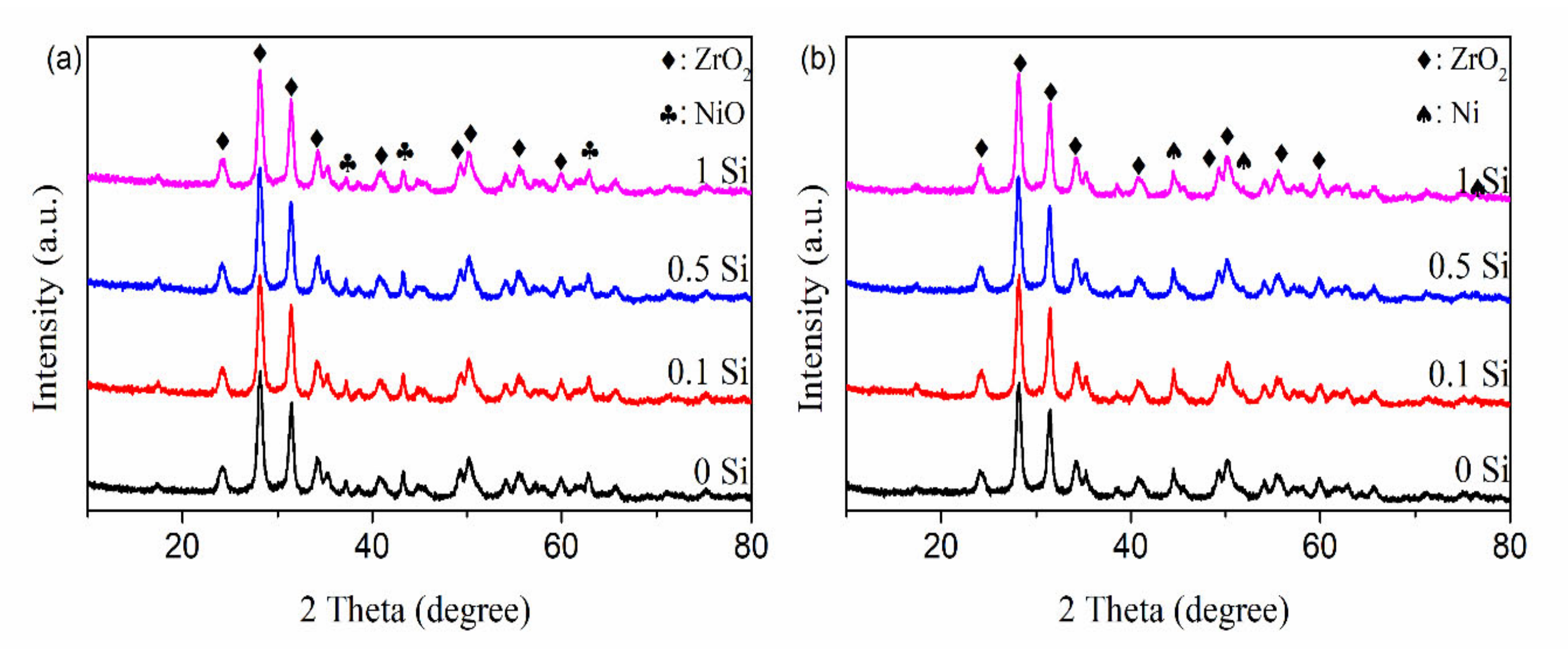
2.6. Crystallite Structure of Ni-xSi/ZrO2 Catalysts
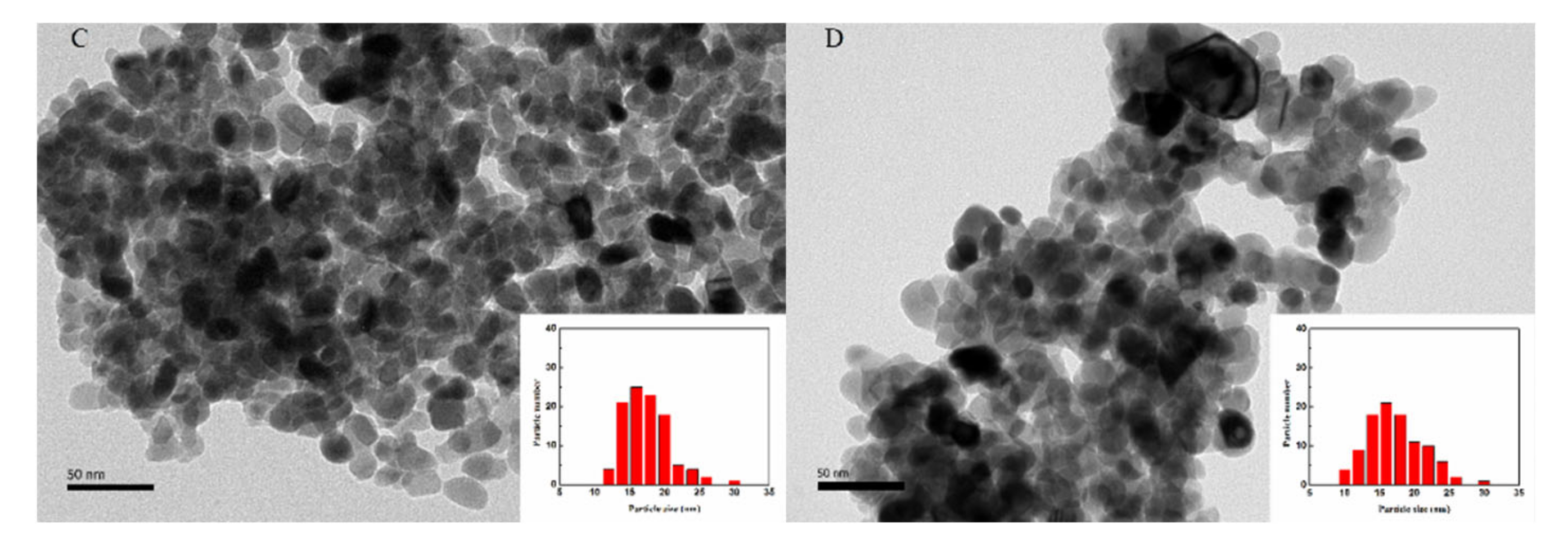
2.7. The TEM Images of Ni-xSi/ZrO2 Catalysts
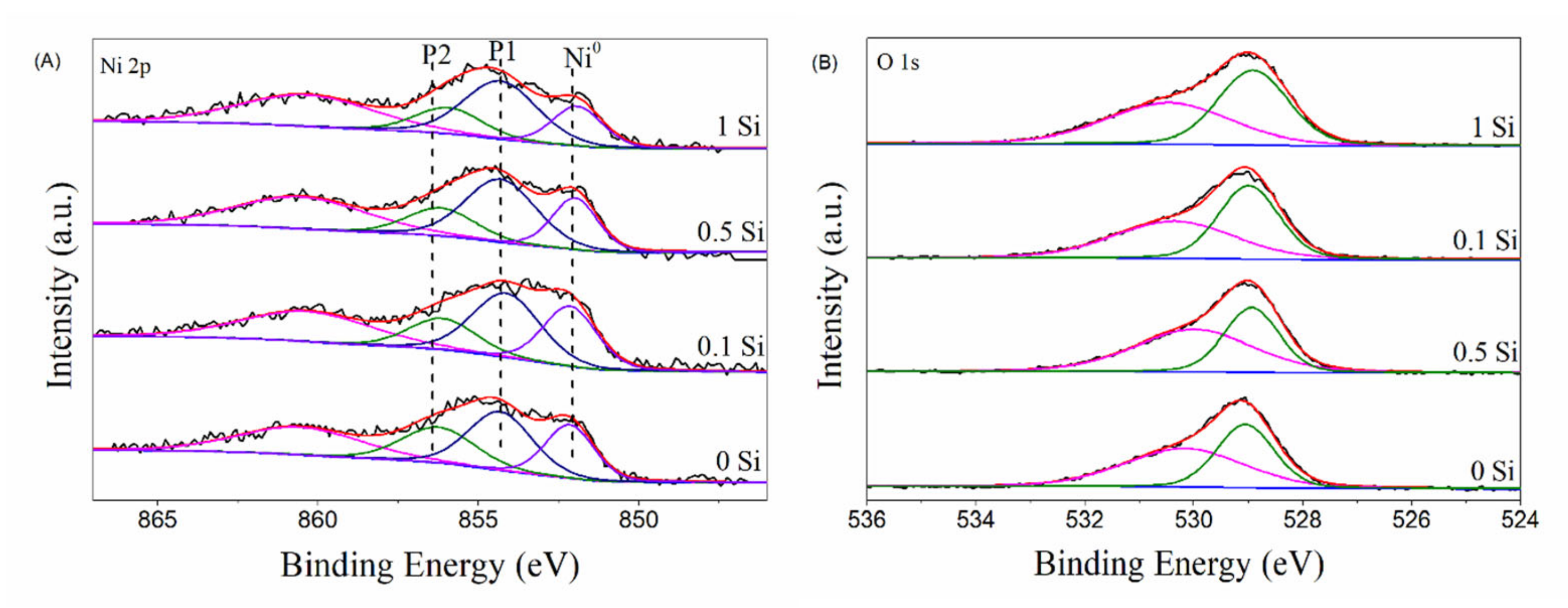
2.8. Chemical State of the Elements on Ni-xSi/ZrO2 Catalysts
3. Materials and Methods
3.1. Catalysts Preparation
3.2. Catalytic Activity Test
3.3. Catalysts Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iglesias, I.; Quindimil, A.; Mariño, F.; De-La-Torre, U.; González-Velasco, J.R. Zr promotion effect in CO2 methanation over ceria supported nickel catalysts. Int. J. Hydrogen Energy 2019, 44, 1710–1719. [Google Scholar] [CrossRef]
- Liu, Q.; Bian, B.; Fan, J.; Yang, J. Cobalt doped Ni based ordered mesoporous catalysts for CO2 methanation with enhanced catalytic performance. Int. J. Hydrogen Energy 2018, 43, 4893–4901. [Google Scholar] [CrossRef]
- Stangeland, K.; Kalai, D.; Li, H.; Yu, Z. CO2 Methanation: The Effect of Catalysts and Reaction Conditions. Energy Procedia 2017, 105, 2022–2027. [Google Scholar] [CrossRef]
- Anwar, M.N.; Fayyaz, A.; Sohail, N.F.; Khokhar, M.F.; Baqar, M.; Yasar, A.; Rasool, K.; Nazir, A.; Raja, M.U.F.; Rehan, M.; et al. CO2 utilization: Turning greenhouse gas into fuels and valuable products. J. Environ. Manag. 2020, 260, 110059–110072. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Nie, X.; Jiang, X.; Zhang, A.; Ding, F.; Liu, M.; Liu, Z.; Guo, X.; Song, C. ZrO2 support imparts superior activity and stability of Co catalysts for CO2 methanation. Appl. Catal. B Environ. 2018, 220, 397–408. [Google Scholar] [CrossRef]
- Do, J.Y.; Park, N.-K.; Seo, M.W.; Lee, D.; Ryu, H.-J.; Kang, M. Effective thermocatalytic carbon dioxide methanation on Ca-inserted NiTiO3 perovskite. Fuel 2020, 271, 117624–117641. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, S.; Zhao, G.; Yang, H.; Yuan, M.; An, X.; Zhou, H.; Qiao, Y.; Tian, Y. CO2 methanation over ordered mesoporous NiRu-doped CaO-Al2O3 nanocomposites with enhanced catalytic performance. Int. J. Hydrogen Energy 2018, 43, 239–250. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, L.; Wang, S.; Mao, D.; Hu, C. Low-temperature catalytic CO2 dry reforming of methane on Ni-based catalysts: A review. Fuel Process. Technol. 2018, 169, 199–206. [Google Scholar] [CrossRef]
- Li, S.; Guo, S.; Gong, D.; Kang, N.; Fang, K.-G.; Liu, Y. Nano composite composed of MoOx-La2O3-Ni on SiO2 for storing hydrogen into CH4 via CO2 methanation. Int. J. Hydrogen Energy 2019, 44, 1597–1609. [Google Scholar] [CrossRef]
- Alarcón, A.; Guilera, J.; Díaz, J.A.; Andreu, T. Optimization of nickel and ceria catalyst content for synthetic natural gas production through CO2 methanation. Fuel Process. Technol. 2019, 193, 114–122. [Google Scholar] [CrossRef]
- Lin, J.; Ma, C.; Wang, Q.; Xu, Y.; Ma, G.; Wang, J.; Wang, H.; Dong, C.; Zhang, C.; Ding, M. Enhanced low-temperature performance of CO2 methanation over mesoporous Ni/Al2O3-ZrO2 catalysts. Appl. Catal. B Environ. 2019, 243, 262–272. [Google Scholar] [CrossRef]
- Yan, X.; Sun, W.; Fan, L.; Duchesne, P.N.; Wang, W.; Kübel, C.; Wang, D.; Kumar, S.G.H.; Li, Y.F.; Tavasoli, A.; et al. Nickel@Siloxene catalytic nanosheets for high-performance CO2 methanation. Nat. Commun. 2019, 10, 2608–2618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bukhari, S.N.; Chong, C.C.; Teh, L.P.; Vo, D.-V.N.; Ainirazali, N.; Triwahyono, S.; Jalil, A.A.; Setiabudi, H.D. Promising hydrothermal technique for efficient CO2 methanation over Ni/SBA-15. Int. J. Hydrogen Energy 2019, 44, 20792–20804. [Google Scholar] [CrossRef]
- Xu, L.; Lian, X.; Chen, M.; Cui, Y.; Wang, F.; Li, W.; Huang, B. CO2 methanation over Co-Ni bimetal-doped ordered mesoporous Al2O3 catalysts with enhanced low-temperature activities. Int. J. Hydrogen Energy 2018, 43, 17172–17184. [Google Scholar] [CrossRef]
- Vita, A.; Italiano, C.; Pino, L.; Laganà, M.; Ferraro, M.; Antonucci, V. High-temperature CO2 methanation over structured Ni/GDC catalysts: Performance and scale-up for Power-to-Gas application. Fuel Process. Technol. 2020, 202, 106365–106375. [Google Scholar] [CrossRef]
- Atzori, L.; Cutrufello, M.G.; Meloni, D.; Monaci, R.; Cannas, C.; Gazzoli, D.; Sini, M.F.; Deiana, P.; Rombi, E. CO2 methanation on hard-templated NiO-CeO2 mixed oxides. Int. J. Hydrogen Energy 2017, 42, 20689–20702. [Google Scholar] [CrossRef]
- Jiang, Y.; Huang, T.; Dong, L.; Su, T.; Li, B.; Luo, X.; Xie, X.; Qin, Z.; Xu, C.; Ji, H. Mn Modified Ni/Bsentonite for CO2 Methanation. Catalysts 2018, 8, 646. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.; Lin, Q.; Su, X.; Duan, H.; Geng, H.; Huang, Y. CO2 methanation over TiO2–Al2O3 binary oxides supported Ru catalysts. Chin. J. Chem. Eng. 2016, 24, 140–145. [Google Scholar] [CrossRef]
- Younas, M.; Sethupathi, S.; Kong, L.L.; Mohamed, A.R. CO2 methanation over Ni and Rh based catalysts: Process optimization at moderate temperature. Int. J. Hydrogen Energy 2018, 42, 3289–3302. [Google Scholar] [CrossRef]
- Martin, N.M.; Velin, P.; Skoglundh, M.; Bauer, M.; Carlsson, P.-A. Catalytic hydrogenation of CO2 to methane over supported Pd, Rh and Ni catalysts. Catal. Sci. Technol. 2017, 7, 1086–1094. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Bian, L.; Zhu, Z.; Li, Z. La-promoted Ni/Mg-Al catalysts with highly enhanced low-temperature CO2 methanation performance. Int. J. Hydrogen Energy 2018, 43, 2197–2206. [Google Scholar] [CrossRef]
- Yu, Y.; Chan, Y.M.; Bian, Z.; Song, F.; Wang, J.; Zhong, Q.; Kawi, S. Enhanced performance and selectivity of CO2 methanation over g-C3N4 assisted synthesis of Ni CeO2 catalyst: Kinetics and DRIFTS studies. Int. J. Hydrogen Energy 2018, 43, 15191–15204. [Google Scholar] [CrossRef]
- Zhou, G.; Liu, H.; Cui, K.; Jia, A.; Hu, G.; Jiao, Z.; Liu, Y.; Zhang, X. Role of surface Ni and Ce species of Ni/CeO2 catalyst in CO2 methanation. Appl. Surf. Sci. 2016, 383, 248–252. [Google Scholar] [CrossRef]
- Lin, J.; Ma, C.; Luo, J.; Kong, X.; Xu, Y.; Ma, G.; Wang, J.; Zhang, C.; Li, Z.; Ding, M. Preparation of Ni based mesoporous Al2O3 catalyst with enhanced CO2 methanation performance. RSC Adv. 2019, 9, 8684–8694. [Google Scholar] [CrossRef] [Green Version]
- Garbarino, G.; Wang, C.; Cavattoni, T.; Finocchio, E.; Riani, P.; Flytzani-Stephanopoulos, M.; Busca, G. A study of Ni/La-Al2O3 catalysts: A competitive system for CO2 methanation. Appl. Catal. B Environ. 2019, 248, 286–297. [Google Scholar] [CrossRef]
- Gnanakumar, E.S.; Chandran, N.; Kozhevnikov, I.V.; Grau-Atienza, A.; Ramos Fernández, E.V.; Sepulveda-Escribano, A.; Shiju, N.R. Highly efficient nickel-niobia composite catalysts for hydrogenation of CO2 to methane. Chem. Eng. Sci. 2019, 194, 2–9. [Google Scholar] [CrossRef]
- Czuma, N.; Zarębska, K.; Motak, M.; Gálvez, M.E.; Da Costa, P. Ni/zeolite X derived from fly ash as catalysts for CO2 methanation. Fuel 2020, 267, 117139–117147. [Google Scholar] [CrossRef]
- Ye, R.-P.; Li, Q.; Gong, W.; Wang, T.; Razink, J.J.; Lin, L.; Qin, Y.-Y.; Zhou, Z.; Adidharma, H.; Tang, J.; et al. High-performance of nanostructured Ni/CeO2 catalyst on CO2 methanation. Appl. Catal. B Environ. 2020, 268, 118474–118484. [Google Scholar] [CrossRef]
- Loder, A.; Siebenhofer, M.; Lux, S. The reaction kinetics of CO2 methanation on a bifunctional Ni/MgO catalyst. J. Ind. Eng. Chem. 2020, 85, 196–207. [Google Scholar] [CrossRef]
- Guilera, J.; Del Valle, J.; Alarcón, A.; Díaz, J.A.; Andreu, T. Metal-oxide promoted Ni/Al2O3 as CO2 methanation micro-size catalysts. J. CO2 Util. 2019, 30, 11–17. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, J.; Liu, F.; Wu, D. Reaction mechanism of CO2 methanation over Rh/TiO2 catalyst. Fuel 2020, 276, 118093–118103. [Google Scholar] [CrossRef]
- Zhou, R.; Rui, N.; Fan, Z.; Liu, C.-J. Effect of the structure of Ni/TiO2 catalyst on CO2 methanation. Int. J. Hydrogen Energy 2016, 41, 22017–22025. [Google Scholar] [CrossRef]
- Moghaddam, S.V.; Rezaei, M.; Meshkani, F.; Daroughegi, R. Synthesis of nanocrystalline mesoporous Ni/Al2O3-SiO2 catalysts for CO2 methanation reaction. Int. J. Hydrogen Energy 2018, 43, 19038–19046. [Google Scholar] [CrossRef]
- Zeng, L.; Wang, Y.; Li, Z.; Song, Y.; Zhang, J.; Wang, J.; He, X.; Wang, C.; Lin, W. Highly Dispersed Ni Catalyst on Metal–Organic Framework-Derived Porous Hydrous Zirconia for CO2 Methanation. ACS Appl. Mater. Interfaces 2020, 12, 17436–17442. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Tong, Y.; Huang, J.; Zhu, J.; Fang, X.; Xu, J.; Wang, X. Insights into CO2 methanation mechanism on cubic ZrO2 supported Ni catalyst via a combination of experiments and DFT calculations. Fuel 2021, 283, 118867–118876. [Google Scholar] [CrossRef]
- Martínez, J.; Hernández, E.; Alfaro, S.; López Medina, R.; Valverde Aguilar, G.; Albiter, E.; Valenzuela, M. High Selectivity and Stability of Nickel Catalysts for CO2 Methanation: Support Effects. Catalysts 2018, 9, 24. [Google Scholar] [CrossRef] [Green Version]
- Hu, L.; Urakawa, A. Continuous CO2 capture and reduction in one process: CO2 methanation over unpromoted and promoted Ni/ZrO2. J. CO2 Util. 2018, 25, 323–329. [Google Scholar] [CrossRef] [Green Version]
- Jia, X.; Zhang, X.; Rui, N.; Hu, X.; Liu, C.-J. Structural effect of Ni/ZrO2 catalyst on CO2 methanation with enhanced activity. Appl. Catal. B Environ. 2019, 244, 159–169. [Google Scholar] [CrossRef]
- Yan, Y.; Dai, Y.; Yang, Y.; Lapkin, A.A. Improved stability of Y2O3 supported Ni catalysts for CO2 methanation by precursor-determined metal-support interaction. Appl. Catal. B Environ. 2018, 237, 504–512. [Google Scholar] [CrossRef]
- Shang, X.; Deng, D.; Wang, X.; Xuan, W.; Zou, X.; Ding, W.; Lu, X. Enhanced low-temperature activity for CO2 methanation over Ru doped the Ni/CexZr(1−x)O2 catalysts prepared by one-pot hydrolysis method. Int. J. Hydrogen Energy 2018, 43, 7179–7189. [Google Scholar] [CrossRef]
- Ashok, J.; Ang, M.L.; Kawi, S. Enhanced activity of CO2 methanation over Ni/CeO2-ZrO2 catalysts: Influence of preparation methods. Catal. Today 2017, 281, 304–311. [Google Scholar] [CrossRef]
- Pan, Q.; Peng, J.; Sun, T.; Gao, D.; Wang, S.; Wang, S. CO2 methanation on Ni/Ce0.5Zr0.5O2 catalysts for the production of synthetic natural gas. Fuel Process. Technol. 2014, 123, 166–171. [Google Scholar] [CrossRef]
- Mihet, M.; Lazar, M.D. Methanation of CO2 on Ni/γ-Al2O3: Influence of Pt, Pd or Rh promotion. Catal. Today 2018, 306, 294–299. [Google Scholar] [CrossRef]
- Xu, L.; Yang, H.; Chen, M.; Wang, F.; Nie, D.; Qi, L.; Lian, X.; Chen, H.; Wu, M. CO2 methanation over Ca doped ordered mesoporous Ni-Al composite oxide catalysts: The promoting effect of basic modifier. J. CO2 Util. 2017, 21, 200–210. [Google Scholar] [CrossRef]
- Baysal, Z.; Kureti, S. CO2 methanation on Mg-promoted Fe catalysts. Appl. Catal. B Environ. 2020, 262, 118300–118310. [Google Scholar] [CrossRef]
- Xu, L.; Wang, F.; Chen, M.; Nie, D.; Lian, X.; Lu, Z.; Chen, H.; Zhang, K.; Ge, P. CO2 methanation over rare earth doped Ni based mesoporous catalysts with intensified low-temperature activity. Int. J. Hydrogen Energy 2017, 42, 15523–15539. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, L.; Wang, Y.; Wang, S.; Zhao, Q.; Mao, D.; Hu, C. Low-Temperature Catalytic CO2 Dry Reforming of Methane on Ni-Si/ZrO2 Catalyst. ACS Catal. 2018, 8, 6495–6506. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Y.; Huang, T.; Dong, L.; Qin, Z.; Ji, H. Ni/bentonite catalysts prepared by solution combustion method for CO2 methanation. Chin. J. Chem. Eng. 2018, 26, 2361–2367. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, L.; Liu, Y.; Wang, S. CO2 methanation on the catalyst of Ni/MCM-41 promoted with CeO2. Sci. Total Environ. 2018, 625, 686–695. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhu, L.; Wang, X.; Yin, S.; Leng, F.; Zhang, F.; Lin, H.; Wang, S. Catalytic methanation of syngas over Ni-based catalysts with different supports. Chin. J. Chem. Eng. 2017, 25, 602–608. [Google Scholar] [CrossRef]
- Ray, K.; Deo, G. A potential descriptor for the CO2 hydrogenation to CH4 over Al2O3 supported Ni and Ni-based alloy catalysts. Appl. Catal. B Environ. 2017, 218, 525–537. [Google Scholar] [CrossRef]
- Kim, H.-M.; Jang, W.-J.; Yoo, S.-Y.; Shim, J.-O.; Jeon, K.-W.; Na, H.-S.; Lee, Y.-L.; Jeon, B.-H.; Bae, J.W.; Roh, H.-S. Low temperature steam reforming of methane using metal oxide promoted Ni-Ce0.8Zr0.2O2 catalysts in a compact reformer. Int. J. Hydrogen Energy 2018, 43, 262–270. [Google Scholar] [CrossRef]
- Roh, H.-S.; Jun, K.-W.; Dong, W.-S.; Chang, J.-S.; Park, S.-E.; Joe, Y.U.-I. Highly active and stable Ni/Ce–ZrO2 catalyst for H2 production from methane. J. Mol. Catal. A Chem. 2002, 181, 137–142. [Google Scholar] [CrossRef]
- Le, T.A.; Kim, M.S.; Lee, S.H.; Kim, T.W.; Park, E.D. CO and CO2 methanation over supported Ni catalysts. Catal. Today 2017, 293–294, 89–96. [Google Scholar]
- Wang, Y.; Li, L.; Wang, Y.; Costa, P.D.; Hu, C. Highly Carbon-Resistant Y Doped NiO–ZrOm Catalysts for Dry Reforming of Methane. Catalysts 2019, 9, 1055. [Google Scholar] [CrossRef] [Green Version]
- Rui, N.; Zhang, X.; Zhang, F.; Liu, Z.; Cao, X.; Xie, Z.; Zou, R.; Senanayake, S.D.; Yang, Y.; Rodriguez, J.A.; et al. Highly active Ni/CeO2 catalyst for CO2 methanation: Preparation and characterization. Appl. Catal. B Environ. 2021, 282, 119581–119592. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, Q.; Wang, Y.; Hu, C.; Da Costa, P. One-Step Synthesis of Highly Active and Stable Ni–ZrOx for Dry Reforming of Methane. Ind. Eng. Chem. Res. 2020, 59, 11441–11452. [Google Scholar] [CrossRef]


| Catalyst | SBET a | VBJH b | Dp c | Ni (%) d | Si (%) d |
|---|---|---|---|---|---|
| (m2·g−1) | (m3·g−1) | (nm) | |||
| Ni/ZrO2 | 42.5 | 0.15 | 13.5 | 6.74 | - |
| Ni-0.1Si/ZrO2 | 46.0 | 0.16 | 12.8 | 6.66 | 0.11 |
| Ni-0.5Si/ZrO2 | 44.6 | 0.15 | 13.2 | 6.51 | 0.34 |
| Ni-1Si/ZrO2 | 44.4 | 0.15 | 13.2 | 5.79 | 0.58 |
| Catalyst | α | β | γ | |||
|---|---|---|---|---|---|---|
| Position (°C) | Content (%) | Position (°C) | Content (%) | Position (°C) | Content (%) | |
| Ni/ZrO2 | 321 | 13.9 | 366 | 20.5 | 428 | 65.6 |
| Ni-0.1Si/ZrO2 | 317 | 5.0 | 340 | 17.5 | 419 | 77.5 |
| Ni-0.5Si/ZrO2 | 319 | 14.2 | 365 | 60.1 | 426 | 25.7 |
| Ni-1Si/ZrO2 | 327 | 7.8 | 366 | 62.1 | 422 | 30.1 |
| Catalyst | Peak 1 | Peak 2 | Total H2 Uptake (µmol/g) a | Ni Dispersion (%) b | ||
|---|---|---|---|---|---|---|
| T (°C) | H2 Uptake (µmol/g) | T (°C) | H2 Uptake (µmol/g) | |||
| Ni/ZrO2 | 102 | 4.64 | 512 | 5.02 | 9.66 | 0.65 |
| Ni-0.1Si/ZrO2 | 111 | 4.64 | 517 | 4.95 | 9.59 | 0.67 |
| Ni-0.5Si/ZrO2 | 103 | 4.28 | 505 | 2.59 | 6.87 | 0.34 |
| Ni-1Si/ZrO2 | 92 | 4.06 | 503 | 2.51 | 6.57 | 0.28 |
| Catalyst | Peak 1 | Peak 2 | Total Basicity (µmol/g) | ||
|---|---|---|---|---|---|
| Position (°C) | Content (%) | Position (°C) | Content (%) | ||
| Ni/ZrO2 | 372 | 44 | 577 | 56 | 123 |
| Ni-0.1Si/ZrO2 | 402 | 29 | 588 | 71 | 124 |
| Ni-0.5Si/ZrO2 | 361 | 49 | 595 | 51 | 127 |
| Ni-1Si/ZrO2 | 410 | 46 | 604 | 54 | 124 |
| Catalyst | Before Reduction | After Reduction |
|---|---|---|
| NiO | Ni Metal | |
| Ni/ZrO2 | 24 nm | 22 nm |
| Ni-0.1Si/ZrO2 | 26 nm | 23 nm |
| Ni-0.5Si/ZrO2 | 26 nm | 23 nm |
| Ni-1Si/ZrO2 | 25 nm | 21 nm |
| Catalyst | Ni0 (%) | Si (%) | Oβ/OT |
|---|---|---|---|
| Ni/ZrO2 | 1.88 | 0 | 0.56 |
| Ni-0.1Si/ZrO2 | 2.15 | 0.45 | 0.59 |
| Ni-0.5Si/ZrO2 | 1.78 | 1.91 | 0.51 |
| Ni-1Si/ZrO2 | 1.27 | 3.74 | 0.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Wang, Y.; Zhao, Q.; Hu, C. The Effect of Si on CO2 Methanation over Ni-xSi/ZrO2 Catalysts at Low Temperature. Catalysts 2021, 11, 67. https://doi.org/10.3390/catal11010067
Li L, Wang Y, Zhao Q, Hu C. The Effect of Si on CO2 Methanation over Ni-xSi/ZrO2 Catalysts at Low Temperature. Catalysts. 2021; 11(1):67. https://doi.org/10.3390/catal11010067
Chicago/Turabian StyleLi, Li, Ye Wang, Qing Zhao, and Changwei Hu. 2021. "The Effect of Si on CO2 Methanation over Ni-xSi/ZrO2 Catalysts at Low Temperature" Catalysts 11, no. 1: 67. https://doi.org/10.3390/catal11010067
APA StyleLi, L., Wang, Y., Zhao, Q., & Hu, C. (2021). The Effect of Si on CO2 Methanation over Ni-xSi/ZrO2 Catalysts at Low Temperature. Catalysts, 11(1), 67. https://doi.org/10.3390/catal11010067





