N-Formylation of Carbon Dioxide and Amines with EDTA as a Recyclable Catalyst under Ambient Conditions
Abstract
:1. Introduction
2. Results and Discussions
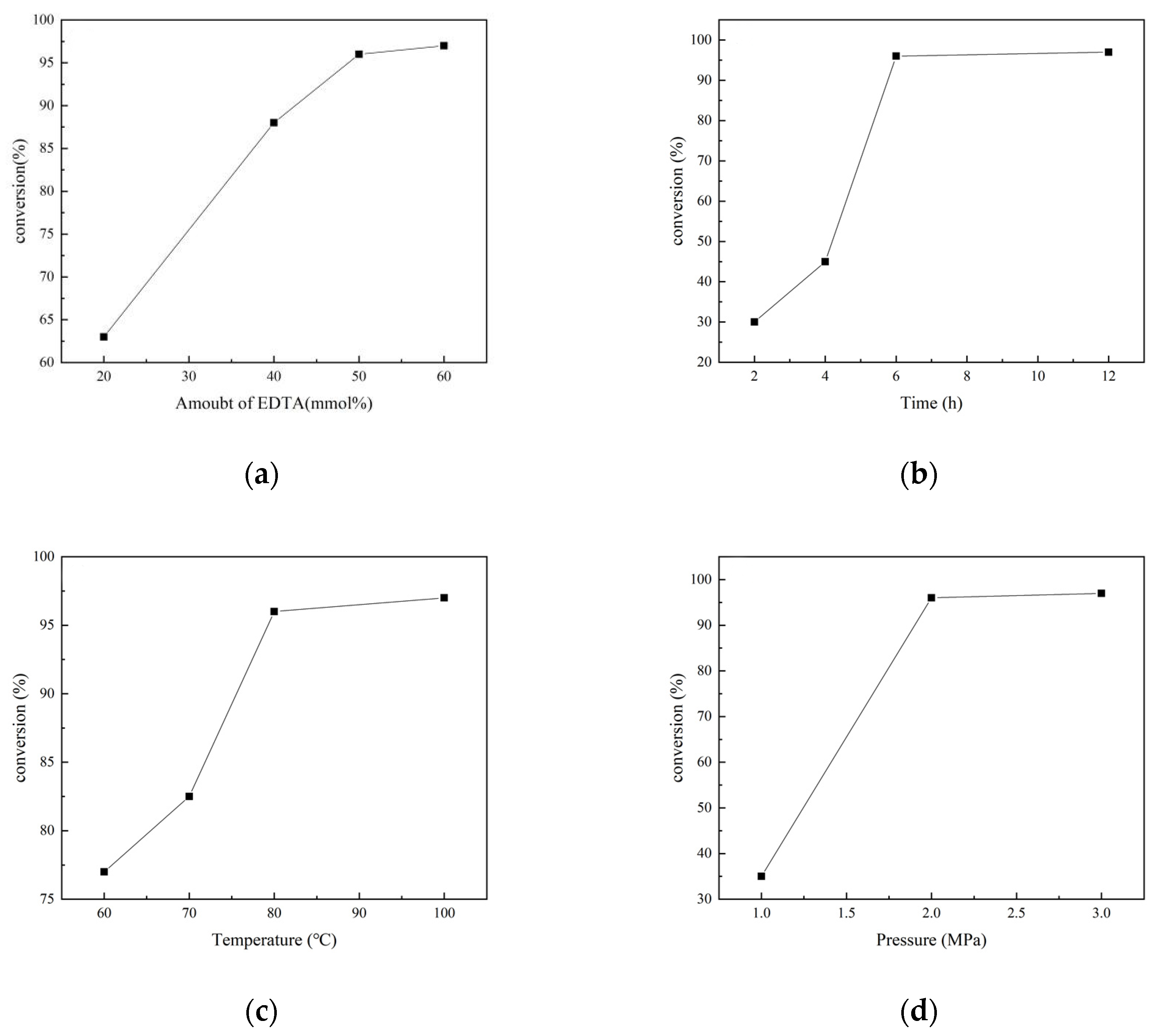
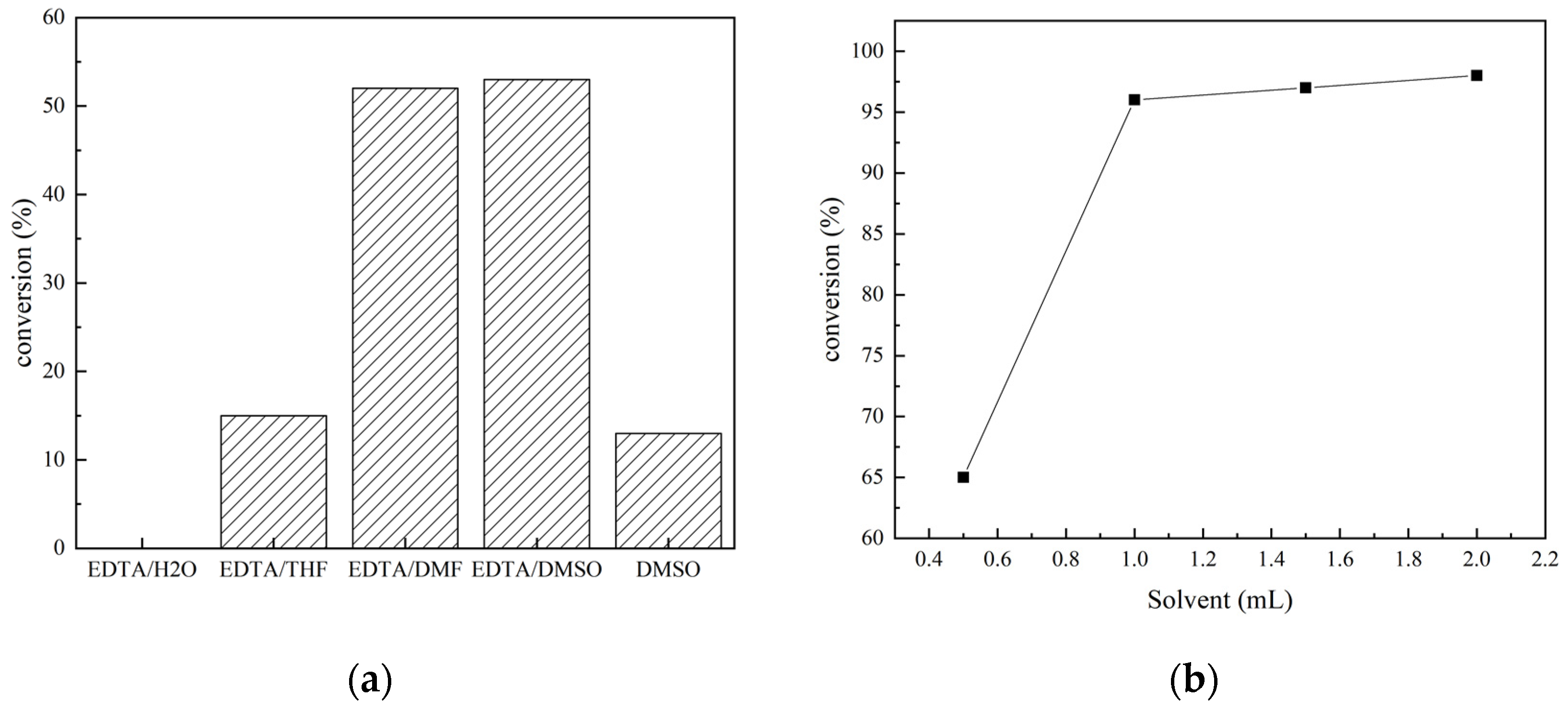
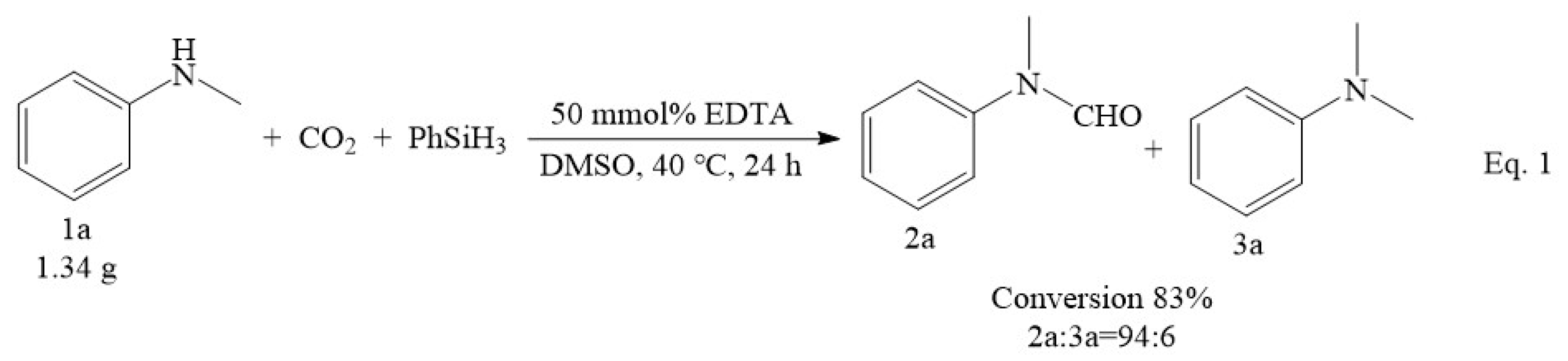
2.1. Screening of Optimal Reaction Conditions for N-Formylation Reaction of CO2 with Amines
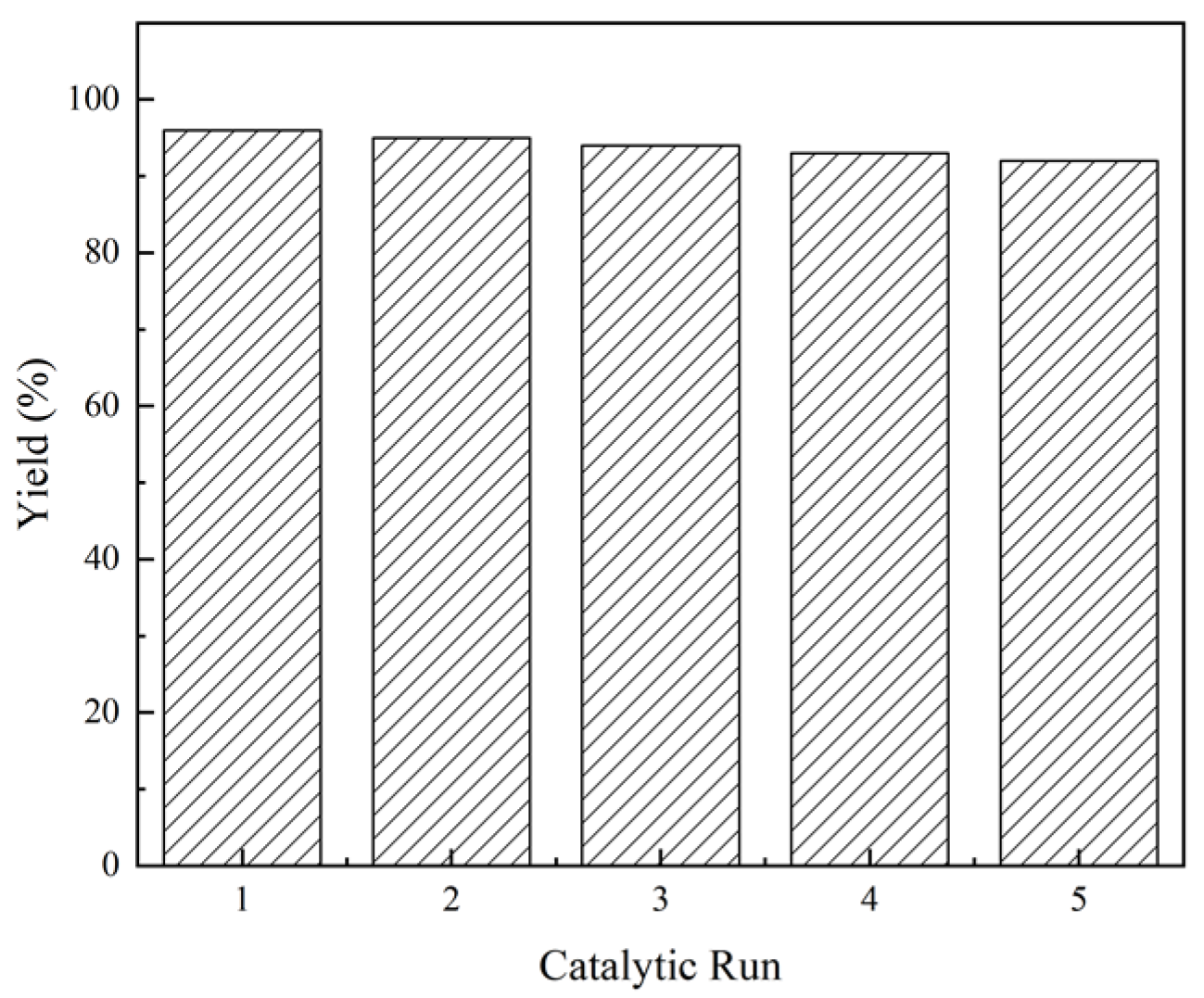
2.2. Reusability of the Catalyst
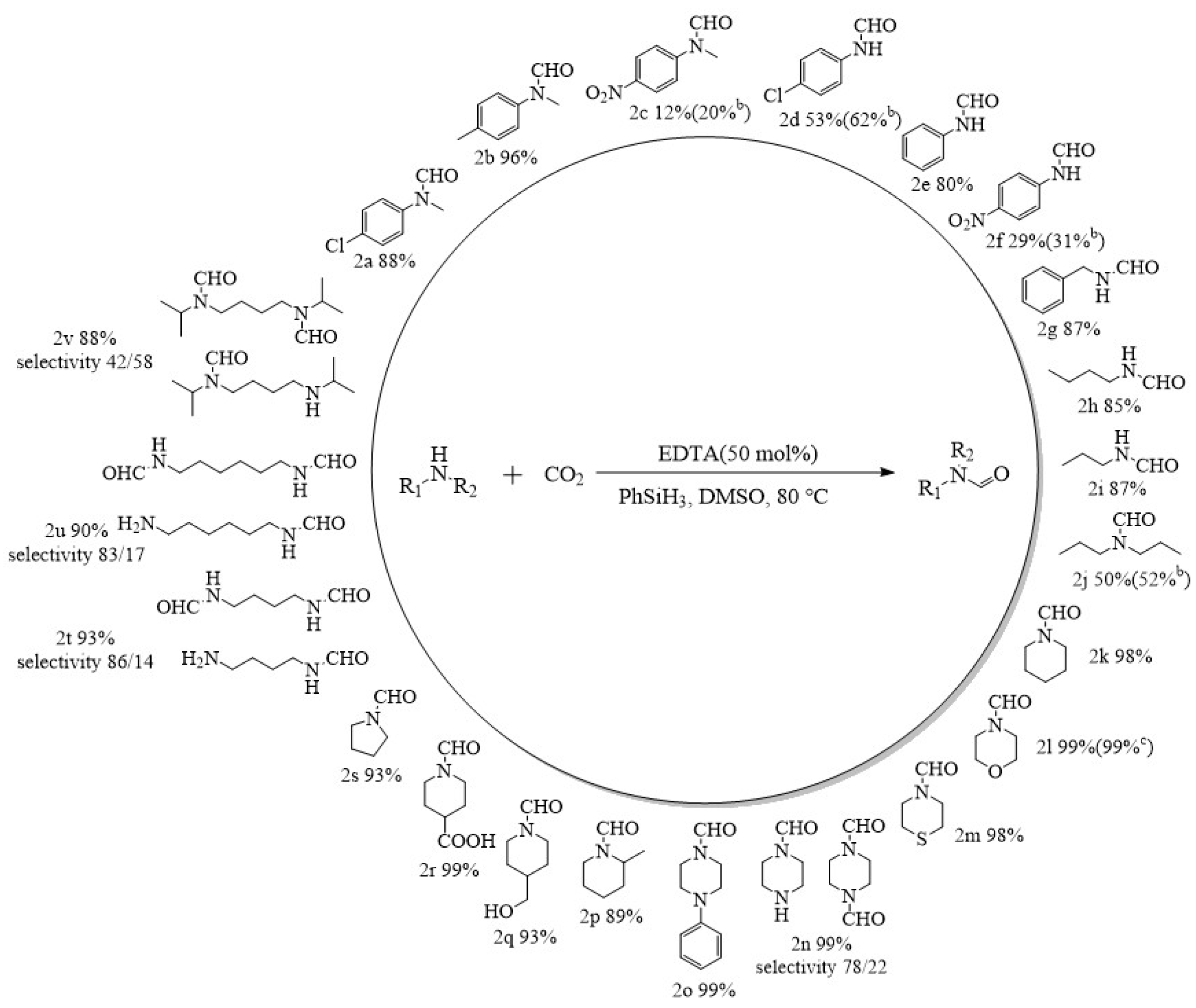
2.3. Application Scope Study: N-Formylation of CO2 with Other Amines Catalyzed by EDTA under High Pressure
2.4. EDTA-Catalyzed Formylation of Different Cyclic Amines and Carbon Dioxide at Room Temperature and Atmospheric Pressure
2.5. Scale-Up Experiment
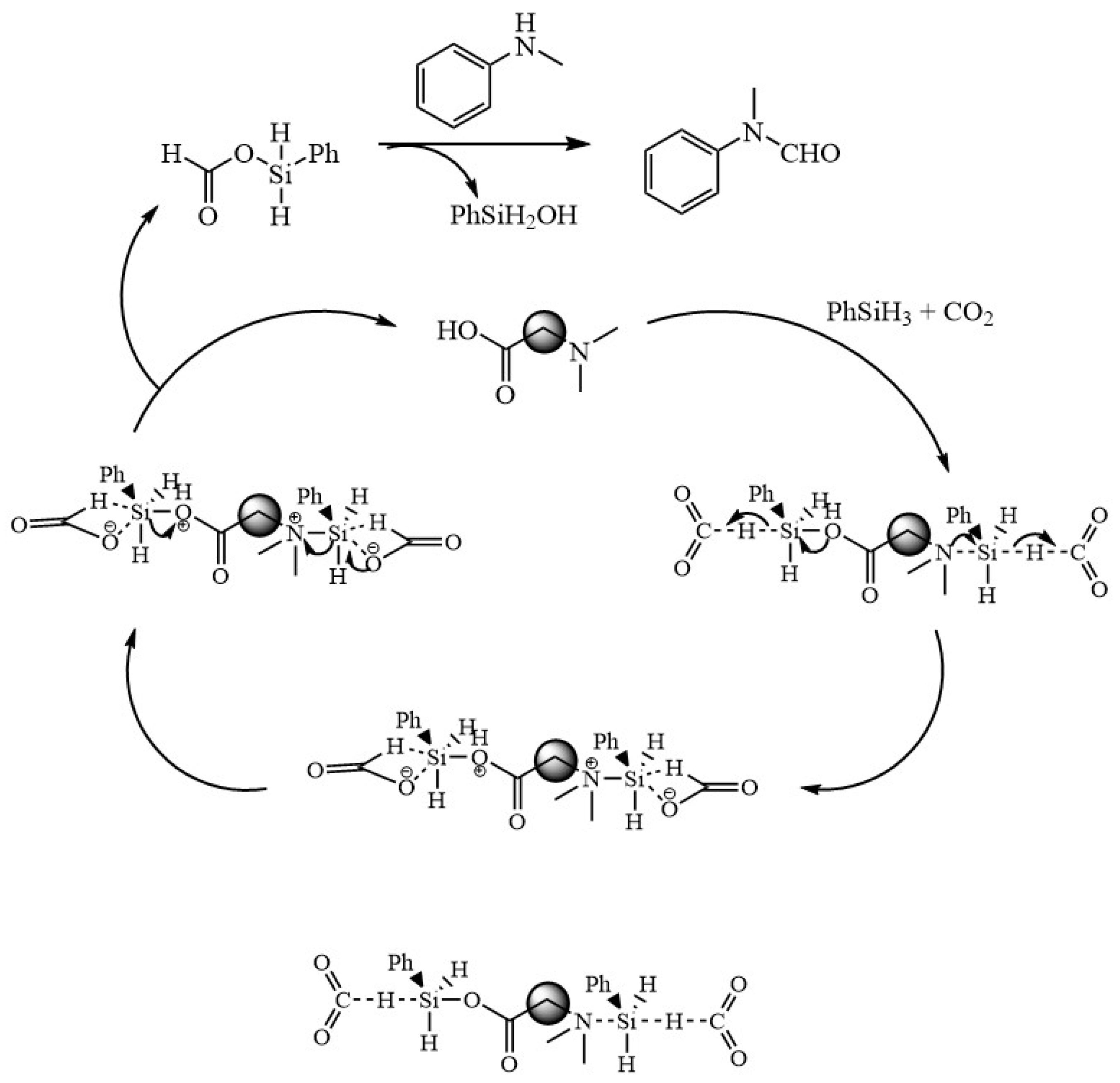
2.6. Possible Mechanism for the Coupling of Carbon Dioxide and Amines
3. Materials and Methods
3.1. Materials and Instruments
3.2. General Procedure of EDTA-Catalyzed N-Formylation of Carbon Dioxide with Amines
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Nema, P.; Nema, S.; Roy, P.J. An overview of global climate changing in current scenario and mitigation action. Renew. Sustain. Energy Rev. 2012, 16, 2329–2336. [Google Scholar] [CrossRef]
- Saptal, V.B.; Bhanage, B.M. N-Heterocyclic olefins as robust organocatalyst for the chemical conversion of carbon dioxide to value-added chemicals. ChemSusChem 2016, 9, 1980–1985. [Google Scholar] [CrossRef] [PubMed]
- Simsa, R.E.H.; Rognerb, H.; Gregoryc, K. Carbon emission and mitigation cost comparisons between fossil fuel, nuclear and renewable energy resources for electricity generation. Energy Policy 2003, 31, 1315–1326. [Google Scholar] [CrossRef]
- Zahasky, C.; Krevor, S. Global geologic carbon storage requirements of climate change mitigation scenarios. Energy Environ. Sci. 2020, 13, 1561–1567. [Google Scholar] [CrossRef]
- Chen, J.M.; Qi, L.; Zhang, L.L.; Li, L.J.; Hou, C.Y.; Li, W.; Wang, L.J. Copper/DTBP-promoted oxyselenation of propargylic amines with diselenides and CO2: Synthesis of selenyl 2-oxazolidinones. J. Org. Chem. 2020, 85, 10924–10933. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Jayakumar, S.; Chen, J.; Tao, L.; Li, H.; Yang, Q.H.; Li, C. Synthesis of bifunctional porphyrin polymers for catalytic conversion of dilute CO2 to cyclic carbonates. ACS Appl. Mater. 2021, 13, 29522–29531. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.P.; Tu, X.W.; Chen, Y.T.; Han, W.H.; Chen, L.C.; Sun, C.; Zhu, S.X.; Song, Y.J.; Zheng, H. Photothermal catalysis without solvent for fixing CO2 to cyclic carbonate. Mol. Catal. 2023, 538, 112971–112979. [Google Scholar] [CrossRef]
- Wen, Q.; Yuan, X.X.; Zhou, Q.Q.; Yang, H.J.; Jiang, Q.Q.; Hu, J.C.; Guo, C.Y. Solvent-free coupling reaction of carbon dioxide and epoxides catalyzed by quaternary ammonium functionalized schiff base metal complexes under mild conditions. Materials 2023, 16, 1646. [Google Scholar] [CrossRef]
- Ran, C.K.; Niu, Y.N.; Song, L.; Wei, M.K.; Cao, Y.F.; Luo, S.P.; Yu, Y.M.; Liao, L.L.; Yu, D.G. Visible-light photoredox-catalyzed carboxylation of activated C(sp3)-O bonds with CO2. ACS Catal. 2022, 12, 18–24. [Google Scholar] [CrossRef]
- Xiao, H.Z.; Yu, B.; Yan, S.S.; Zhang, W.; Li, X.X.; Bao, Y.; Luo, S.P.; Ye, J.H.; Yu, D.G. Photocatalytic 1,3-dicarboxylation of unactivated alkenes with CO2. Chin. J. Catal. 2023, 50, 222–228. [Google Scholar] [CrossRef]
- Gerack, C.J.; White, L.M. Formylation of amines. Molecules 2014, 19, 7689–7713. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.H.; Zhang, J.; Chen, H.J.; Zhang, Z.X.; Zhang, C.F.; Li, M.R.; Wang, F. Ru/ceria-catalyzed direct formylation of amines and CO to produce formamides. Green Chem. 2017, 19, 88–92. [Google Scholar] [CrossRef]
- Beyazay, T.; Martin, W.F.; Tuysuz, H. Direct synthesis of formamide from CO2 and H2O with nickel–iron nitride heterostructures under mild hydrothermal conditions. J. Am. Chem. Soc. 2023, 145, 19768–19779. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.F.; Li, X.Y.; Qiao, C. Transition-metal-free catalysis for the reductive functionalization of CO2 with amines. Synlett 2018, 29, 548–555. [Google Scholar]
- Zou, Q.Z.; Yi, Y.; Zhao, T.X.; Liu, F.; Kang, C.; Hu, X.B. Catalyst-free hierarchical reduction of CO2 with BH3N(C2H5)3 for selective N-methylation and N-formylation of amines. J. CO2 Util. 2021, 50, 101590–101596. [Google Scholar] [CrossRef]
- Li, W.D.; Zhu, D.Y.; Li, G.; Chen, J.; Xia, J.B. Iron-catalyzed selective N-methylation and N-formylation of amines with CO2. Adv. Synth. Catal. 2019, 361, 5098–5104. [Google Scholar] [CrossRef]
- Akhtar, N.; Chauhan, M.; Gupta, P.; Antil, N.; Manna, K. A supported pyridylimine-cobalt catalyst for N-formylation of amines using CO2. Dalton Trans. 2023, 52, 15384–15393. [Google Scholar] [CrossRef]
- Yoo, D.K.; Jhung, S.H. N-formylation of amines with CO2 by using Zr-based metal-organic frameworks: Contribution of defect sites of MOFs to N-formylation. Appl. Catal. A-Gen. 2023, 659, 119170–119177. [Google Scholar] [CrossRef]
- Li, R.P.; Zhao, Y.F.; Wang, H.; Li, D.Y.; Wu, Y.Y.; Zhang, H.Y.; Tang, M.H.; Liu, Z.M. Ionic liquid-promoted formylation of N(sp2)-heteroarenes with CO2/H2 over Pd/C. ACS Sustain. Chem. Eng. 2021, 9, 2507–2514. [Google Scholar] [CrossRef]
- Lai, W.; Jiang, Y.; Liao, H.; Wei, X.; Xu, Z.; Ding, J.; An, N.; Dai, S.; Hou, Z. Multivacant polyoxometalate-stabilizing palladium nanoparticles catalyze the N-formylation of amines with CO2 and H2. New J. Chem. 2024, 48, 11014–11024. [Google Scholar] [CrossRef]
- Wang, G.; Jiang, M.; Ji, G.; Sun, Z.; Ma, L.; Li, C.; Du, H.; Yan, L.; Ding, Y. Highly efficient heterogeneous Pd@POPs catalyst for the N-formylation of amine and CO2. Catalysts 2021, 11, 220. [Google Scholar] [CrossRef]
- Zhang, L.; Han, Z.B.; Zhao, X.Y.; Wang, Z.; Ding, K.L. Highly efficient ruthenium-catalyzed N-formylation of amines with H2 and CO2. Angew. Chem. Int. Ed. 2015, 127, 6186–6189. [Google Scholar] [CrossRef] [PubMed]
- Park, K.; Gunasekar, G.H.; Prakash, N.; Jung, K.; Yoon, S. A highly efficient heterogenized iridium complex for the catalytic hydrogenation of carbon dioxide to formate. ChemSusChem 2015, 8, 3410–3413. [Google Scholar] [CrossRef] [PubMed]
- Toyao, T.; Siddiki, S.M.A.H.; Morita, Y.; Kamachi, T.; Touchy, A.S.; Onodera, W.; Kon, K.; Furukawa, S.; Ariga, H.; Asakura, K.; et al. Rhenium-loaded TiO2: A highly versatile and chemoselective catalyst for the hydrogenation of carboxylic acid derivatives and the N-methylation of amines using H2 and CO2. Chem. Eur. J. 2017, 23, 14848–14859. [Google Scholar] [CrossRef]
- Mitsudome, T.; Urayama, T.; Fujita, S.; Maeno, Z.; Mizugaki, T.; Jitsukawa, K.; Kaneda, K. A titanium dioxide supported gold nanoparticle catalyst for the selective N-formylation of functionalized amines with carbon dioxide and hydrogen. ChemCatChem 2017, 9, 3632–3636. [Google Scholar] [CrossRef]
- Bai, P.; Zhao, Y.C.; Li, Y.D. Efficient photocatalytic CO2 N-formylation of amines over Pd/Bi-ZnOx without extra reductant. Catal. Today. 2024, 430, 114551–114561. [Google Scholar] [CrossRef]
- He, Y.; Bai, P.; Yuan, S.B.; Chen, J.F.; Zhao, Y.C.; Li, Y.D. Photocatalytic N-formylation of amines with CO2 over Pt-Bi bimetallic decorated CeO2−x. Mol. Catal. 2024, 559, 114086–114093. [Google Scholar] [CrossRef]
- Zhang, S.Q.; Mei, Q.Q.; Liu, H.Y.; Liu, H.Z.; Zhang, Z.P.; Han, B.X. Copper-catalyzed N-formylation of amines with CO2 under ambient conditions. RSC Adv. 2016, 6, 32370–32373. [Google Scholar] [CrossRef]
- Fang, C.; Lu, C.L.; Liu, M.H.; Zhu, Y.L.; Fu, Y.; Lin, B.L. Selective formylation and methylation of amines using carbon dioxide and hydrosilane catalyzed by alkali-metal carbonates. ACS Catal. 2016, 6, 7876–7881. [Google Scholar] [CrossRef]
- Luo, R.C.; Lin, X.W.; Chen, Y.J.; Zhang, W.Y.; Zhou, X.T.; Ji, H.B. Cooperative catalytic activation of Si-H bonds: CO2-based synthesis of formamides from amines and hydrosilanes under mild conditions. ChemSusChem 2017, 10, 1224–1232. [Google Scholar] [CrossRef]
- Gomes, C.D.N.; Jacquet, O.; Villiers, C.; Thury, P.; Ephritikhine, M.; Cantat, T. A diagonal approach to chemical Recycling of carbon dioxide: Organocatalytic transformation for the reductive functionalization of CO2. Angew. Chem. Int. Ed. 2012, 51, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Jacquet, O.; Gomes, C.D.N.; Ephritikhine, M.; Cantat, T. Recycling of Carbon and Silicon Wastes: Room Temperature Formylation of N–H Bonds Using Carbon Dioxide and Polymethylhydrosiloxane. J. Am. Chem. Soc. 2012, 134, 2934–2937. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.D.; Zhao, Y.F.; Yu, B.; Yang, Z.Z.; Zhang, H.Y.; Han, B.X.; Gao, X.; Liu, Z.M. Imidazolium-based ionic liquids catalyzed formylation of amines using carbon dioxide and phenylsilane at room temperature. ACS Catal. 2015, 5, 4989–4993. [Google Scholar] [CrossRef]
- Chong, C.C.; Kinjo, R. Hydrophosphination of CO2 and subsequent formate transfer in the 1,3,2-diazaphospholene-catalyzed N-formylation of amines. Angew. Chem. Int. Ed. 2015, 127, 12116–12120. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Yu, D.K.; Xue, Z.M.; Zhang, B.; Mu, T.C. Dihydrolevoglucosenone as a bio-based catalytic solvent for efficient reductive-transformation of CO2 with amines into formamides and benzothiazoles. Chem. Eng. J. 2022, 431, 133397–133405. [Google Scholar] [CrossRef]
- Lu, H.; Xing, Q.; Yue, C.T.; Lei, Z.Q.; Li, F.W. Solvent-promoted catalyst-free N-formylation of amines using carbon dioxide under ambient conditions. Chem. Commun. 2016, 52, 6545–6548. [Google Scholar]
- Niu, H.Y.; Lu, L.J.; Shi, R.Y.; Chiang, C.W.; Lei, A. Catalyst-free N-methylation of amines using CO2. Chem. Commun. 2017, 53, 1148–1151. [Google Scholar] [CrossRef]
- Chen, X.C.; Guo, L.; Shi, G.H.; Zhao, K.C.; Lu, Y.; Liu, Y. Can CO2 be a catalyst? Yes, CO2-catalyzed N-formylation of aliphatic amines with DMF. Mol. Catal. 2022, 528, 112431–112439. [Google Scholar] [CrossRef]
- Chakrabortty, P.; Kumar, S.; Chowdhury, A.; Khan, A.; Bhaumik, A.; Islam, S.M. Ag-nanocatalysts based on porous organic polymers in chemical fixation of CO2 for the N-methylation and N-formylation of amines. ChemCatChem 2024, 16, e202301539. [Google Scholar] [CrossRef]
- Liu, X.F.; Li, X.Y.; Qiao, C.; Fu, H.C.; He, L.N. Betaine Catalysis for Hierarchical Reduction of CO2 with amines and hydrosilane to form formamides, aminals, and methylamines. Angew. Chem. Int. Ed. 2017, 56, 7533–7537. [Google Scholar] [CrossRef]
- Hota, P.K.; Sau, S.C.; Mandal, S.K. Metal-free catalytic formylation of amides using CO2 under ambient conditions. ACS Catal. 2018, 8, 11999–12003. [Google Scholar] [CrossRef]
- Li, X.Y.; Zheng, S.S.; Liu, X.F.; Yang, Z.W.; Tan, T.Y.; Yu, A.; He, L.N. Waste recycling: Ionic liquid-catalyzed 4-electron peduction of CO2 with pmines and polymethylhydrosiloxane combining experimental and theoretical study. ACS Sustain. Chem. Eng. 2018, 6, 8130–8135. [Google Scholar] [CrossRef]
- Wen, Q.; Yuan, X.X.; Zhou, Q.Q.; Yang, H.J.; Jiang, Q.Q.; Hu, J.C.; Guo, C.Y. Solvent- and co-catalyst-free cycloaddition of carbon dioxide and epoxides catalyzed by recyclable bifunctional niobium complexes. Materials 2023, 16, 3531. [Google Scholar] [CrossRef] [PubMed]
- Wen, Q.; Yuan, X.; Zhou, Q.; Yang, H.J.; Jiang, Q.; Hu, J.; Guo, C.Y. Effcient N-formylation of carbon dioxide and amines with alkanolamine as eco-friendly catalyst under mild conditions. J. CO2 Util. 2023, 69, 102398–102408. [Google Scholar] [CrossRef]
- Lin, W.W.; Cheng, H.Y.; Wu, Q.F.; Zhang, C.; Arai, M.; Zhao, F.Y. Selective N-methylation of N-methylaniline with CO2 and H2 over TiO2-supported PdZn catalyst. ACS Catal. 2020, 10, 3285–3296. [Google Scholar] [CrossRef]
- Hulla, M.; Laurenczy, G.; Dyson, P.J. Mechanistic study of the N-formylation of amines with carbon dioxide and hydrosilanes. ACS Catal. 2018, 8, 10619–10630. [Google Scholar] [CrossRef]
- Liu, X.F.; Qiao, C.; Li, X.Y. DMF-promoted reductive functionalization of CO2 with secondary amines and phenylsilane to methylamines. Pure Appl. Chem. 2018, 90, 1099–1107. [Google Scholar] [CrossRef]
- Wang, J.X.; Li, S.H.; Wang, Y.P. Unsupported nanoporous palladium catalyst for N-formylation of amines using CO2 as a sustainable C1 source. Asian J. Org. Chem. 2022, 11, e202200064. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Wang, H.L.; Yuan, H.K.; Shi, F. Hydroxyl group-regulated active nano-Pd/C catalyst generation via in situ Reduction of Pd(NH3)xCly/C for N-formylation of amines with CO2/H2. ACS Sustain. Chem. Eng. 2017, 5, 5758–5765. [Google Scholar] [CrossRef]
- Gopakumar, A.; Lombardo, L.; Fei, Z.F.; Shyshkanov, S. A polymeric ionic liquid catalyst for the N-formylation and N-methylation of amines using CO2/PhSiH3. J. CO2 Util. 2020, 41, 101240–101245. [Google Scholar] [CrossRef]


| Entry | Silane | Amount (mmol) | Yield a,b |
|---|---|---|---|
| 1 | PhSiH3 | 5 | 96% |
| 2 | (EtO)3SiH | 5 | 0 |
| 3 | Ph2SiH2 | 5 | 0 |
| 4 | (CH3)2PhSiH | 5 | 0 |
| 5 | DMAB | 5 | 0 |
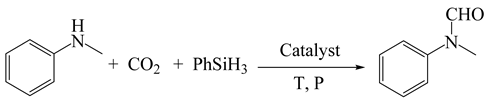
| Entry | Catalyst | Conversion a,b |
|---|---|---|
| 1 | 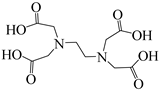 | 96% |
| 2 | 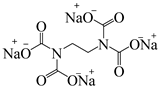 | 57% |
| 3 | 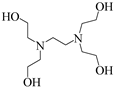 | 27% |
| 4 |  | 56% |
| 5 | 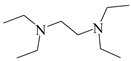 | 48% |
| 6 | 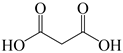 | 59% |
| 7 | 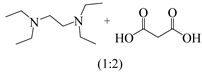 | 67% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, Q.; Chen, Y.; Yuan, X.; Yang, H.-J.; Jiang, Q.; Hu, J.; Guo, C.-Y. N-Formylation of Carbon Dioxide and Amines with EDTA as a Recyclable Catalyst under Ambient Conditions. Catalysts 2024, 14, 492. https://doi.org/10.3390/catal14080492
Zhou Q, Chen Y, Yuan X, Yang H-J, Jiang Q, Hu J, Guo C-Y. N-Formylation of Carbon Dioxide and Amines with EDTA as a Recyclable Catalyst under Ambient Conditions. Catalysts. 2024; 14(8):492. https://doi.org/10.3390/catal14080492
Chicago/Turabian StyleZhou, Qiqi, Yu Chen, Xuexin Yuan, Hai-Jian Yang, Qingqing Jiang, Juncheng Hu, and Cun-Yue Guo. 2024. "N-Formylation of Carbon Dioxide and Amines with EDTA as a Recyclable Catalyst under Ambient Conditions" Catalysts 14, no. 8: 492. https://doi.org/10.3390/catal14080492
APA StyleZhou, Q., Chen, Y., Yuan, X., Yang, H.-J., Jiang, Q., Hu, J., & Guo, C.-Y. (2024). N-Formylation of Carbon Dioxide and Amines with EDTA as a Recyclable Catalyst under Ambient Conditions. Catalysts, 14(8), 492. https://doi.org/10.3390/catal14080492







