Abstract
Intramolecular transfer of t-Bu3P-ligated Pd catalyst on a carbon–carbon triple bond (C≡C) and nitrogen–nitrogen double bond (N=N) was investigated and compared with the case of a carbon–carbon double bond (C=C), which is resistant to intramolecular transfer of the Pd catalyst. Suzuki–Miyaura coupling reaction of equimolar 4,4’-dibromotolan (1a) or 4,4’-dibromoazobenzene (1b) with 3-isobutoxyphenylboronic acid (2) was carried out in the presence of t-Bu3P-ligated Pd precatalyst 3 and KOH/18-crown-6 as a base at room temperature. In both cases, the diphenyl-substituted product was selectively obtained, indicating that the Pd catalyst walked from one benzene ring to the other through the C≡C or N=N bond after the first substitution with 2. Taking advantage of this finding, we conducted unstoichiometric Suzuki–Miyaura polycondensation of 1.3 equiv. of 1 and 1.0 equiv. of phenylenediboronic acid (ester) 6 in the presence of 3 and CsF/18-crown-6 as a base, obtaining high-molecular-weight conjugated polymer with a boronic acid (ester) moiety at both ends, contrary to the Flory principle.
1. Introduction
Suzuki–Miyaura coupling reaction is a powerful protocol for the synthesis of polyarylenes containing π-conjugated polymers [1]. We have found that t-Bu3PPd(Ar)Br [2,3] initiates chain-growth Suzuki–Miyaura coupling polymerization of haloarylboronic acid (ester) as an AB type monomer to afford well-defined polyfluorene [4], poly(p-phenylene) [5], and poly(hexylthiophene) [6], and other researchers have obtained well-defined poly(phenanthrene) [7] and poly(fluorene-alt-benzothiadiazole) [8]. The chain-growth polymerization progresses via intramolecular transfer of the Pd catalyst to the terminal C–X (X = halogen) bond after reductive elimination of polymer–Pd–ArX. Therefore, these types of chain-growth polymerizations, including Kumada–Tamao and other coupling polymerizations, are known as catalyst-transfer condensation polymerizations (CTCPs) [9,10].
When this t-Bu3PPd(0) catalyst, which has a propensity for intramolecular catalyst transfer on a π-electron face, was used for Suzuki–Miyaura coupling polymerization of dibromoarene and arenyldiboronic acid ester (AA + BB polycondensation), high-molecular-weight π-conjugated polymer with a boronate moiety at both ends was obtained, even though excess dibromoarene was used [11]. This unstochiometric polycondensation behavior is accounted for by successive substitution of the bromides in dibromoarene with arenyldiboronic acid ester or oligomers having boronate moieties at both ends through intramolecular transfer of the Pd catalyst on the π face of dibromoarene. However, Suzuki–Miyaura coupling reaction of 4,4’-dibromostilbene with phenylboronic acid in the presence of t-Bu3PPd(0) catalyst did not selectively afford diphenyl-substituted stilbene, implying that the Pd catalyst did not walk from one benzene ring to the other through the carbon–carbon double bond (C=C) in the stilbene after the first substitution of 4,4’-dibromostilbene with phenylboronic acid [12]. We found that this failure of intramolecular catalyst transfer is due to bimolecular intermolecular transfer of t-Bu3PPd(0) on the C=C of dibromostilbene to the C=C of another dibromostilbene. However, this intermolecular transfer of the catalyst could be suppressed by introduction of alkoxy groups at the ortho positions of the C=C.
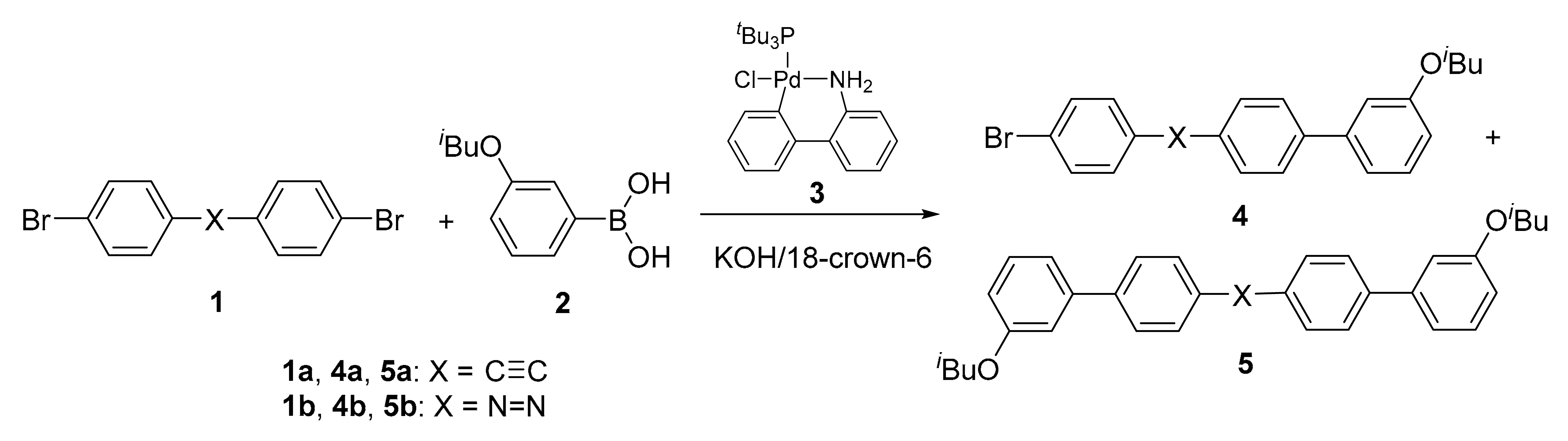
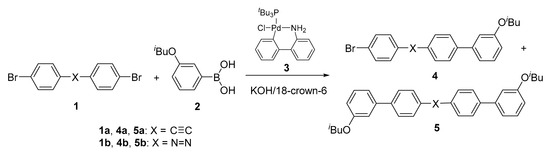
We were next interested in whether or not the Pd catalyst undergoes intramolecular catalyst transfer on other multiple bonds. In the present study, we investigated catalyst transfer on a carbon–carbon triple bond (C≡C) and nitrogen–nitrogen double bond (N=N) by means of Suzuki–Miyaura coupling reaction of 4,4’-dibromotolan (1a) or 4,4’-dibromoazobenzene (1b) with phenylboronic acid 2 in the presence of t-Bu3PPd(0) precatalyst 3 [13] (Scheme 1). If the catalyst undergoes intramolecular transfer on the multiple bond X in 1 and then inserts itself into the C–Br bond after the first substitution, the main product would be disubstituted 5. On the other hand, if the catalyst diffuses into the reaction mixture after the first substitution, the main product would be monosubstituted 4. Bielawski and coworkers have demonstrated intramolecular catalyst transfer of t-Bu3PPd(0) catalyst on C≡C in Stille CTCP of 4-(tributylstannylethynyl)-2,5-dialkoxyiodobenzene as an AB monomer [14]. However, this catalyst transfer on C≡C might be dependent on the steric effect of the tributylstannyl and/or alkoxy groups, as in the case of catalyst transfer on C=C in o-alkoxy-substituted stilbene. In the present work, we found that t-Bu3PPd(0) catalyst undergoes intramolecular catalyst transfer on C≡C and N=N even if the benzene rings adjacent to these multiple bonds are unsubstituted, in contrast to the case of catalyst transfer on C=C. To demonstrate effective catalyst transfer on C≡C and N=N, we further conducted unstoichiometric Suzuki–Miyaura polycondensation of excess 1 and 1.0 equiv. of phenylenediboronic acid (ester) in the presence of 3, obtaining π-conjugated polymer with a boronate moiety at both ends.
Scheme 1.
Suzuki–Miyaura coupling reaction of 1 with 2 in the presence of 3 to examine intramolecular catalyst transfer on the multiple bond X in 1.
2. Results and Discussion
2.1. Suzuki–Miyaura Coupling Reaction of 1 with 2
Intramolecular catalyst transfer on C≡C was first investigated by Suzuki–Miyaura coupling reaction of equimolar 1a with 2 in the presence of 5 mol % of t-Bu3PPd(0) precatalyst 3 and KOH/18-crown-6 (Table 1). In order to determine the product ratio of monosubstituted 4a to disubstituted 5a by means of gas chromatography (GC), authentic 4a and 5a were prepared by the reaction of equimolar 1a and 2 in the presence of (Ph3P)4Pd, which has no propensity for intramolecular catalyst transfer on a π face, and by the reaction of 1a and 2 equiv. of 2 in the presence of 3, respectively.

Table 1.
Suzuki–Miyaura coupling reaction of 1 with 2 in the presence of 3 1.
In the reaction of 1a with 2, disubstituted 5a was obtained with high selectivity (Entry 1). Furthermore, the product ratio of 5a was consistently more than 90% from the beginning of the reaction (Figure S1). This is important, because if 5a was selectively formed due to higher reactivity of 4a than 1a—i.e., not via the catalyst-transfer mechanism—then 4a should be accumulated at the beginning of the reaction, in which the concentration of 1a is much higher than that of 4a [15]. Therefore, these results indicate that intramolecular transfer of t-Bu3PPd(0) catalyst on C≡C took place, in contrast to the case of the reaction of 4,4’-dibromostilbene with 2 in the presence of t-Bu3PPd(0) catalyst, which afforded mainly monosubstituted stilbene [12]. We next examined the effects of additives to see whether intramolecular catalyst transfer on C≡C is disturbed by additives containing C=C or C≡C. If the affinity of an additive for t-Bu3PPd(0) is stronger than that of C≡C, the additive would trap the Pd catalyst during intramolecular catalyst transfer on 1a, resulting in a decrease of the ratio of disubstituted 5a. In the case of styrene as an additive, the ratio of 5a was drastically decreased to 26% (Entry 2), whereas stilbene resulted in decrease to 77% (Entry 3). It turned out that terminal C=C disturbed intramolecular catalyst transfer on C≡C more strongly than did internal C=C. As for additives containing C≡C, phenylacetylene also induced a decrease of the ratio of 5a (entry 4), whereas tolan and 1-phenyl-1-propyne did not affect the intramolecular transfer of the catalyst, and 5a was selectively formed (Entries 5 and 6). Consequently, compounds bearing internal C≡C, including substrate 1a, turned out not to disturb intramolecular catalyst transfer on C≡C.
We next investigated intramolecular catalyst transfer on N=N. Suzuki–Miyaura coupling reaction of 4,4’-dibromoazobenzene (1b) with 2 was similarly carried out. Products 4b and 5b were isolated by preparative HPLC, and we confirmed that aromatic proton signals of 1b, 4b, and 5b appeared separately in 1H NMR spectra (Figure S2). The same reaction was conducted again in the presence of 1,4-bis(hexyloxy)benzene as an internal standard, and conversion of 1b and the product ratio of 4b to 5b were determined by 1H NMR spectroscopy. The ratio of 4b to 5b was 11:89, indicating that intramolecular catalyst transfer also took place on N=N (Entry 7), although the ratio of 5b was slightly decreased as compared to the case of reaction of 1a with 2 (Entry 1).
The observation that t-Bu3PPd(0) catalyst underwent intramolecular catalyst transfer on C≡C and N=N, in contrast to the case of C=C, might be accounted for by lower stability of the η2 coordination complexes of these multiple bonds to t-Bu3PPd, compared with the η2 complex of C=C. As the η2 complex becomes less stable, the chance of bimolecular ligand exchange of η2 complex between multiple bonds would be decreased. The observation that intramolecular catalyst transfer on C≡C is disturbed by additives containing C=C implies that the η2 complex of C=C is more stable than that of C≡C. In addition, density functional theory (DFT) calculations were performed to estimate the stability of the η2 coordination complexes of these multiple bonds with t-Bu3PPd. The values of stabilization energy of complexation of stilbene, tolan, and azobenzene with t-Bu3PPd were −27.9, −25.6, and −20.8 kcal/mol, respectively (Figures S3–S5). These results support our interpretation, although calculations for model PdPH3 systems with ethylene and acetylene indicated that the Pd–C bonds are stronger in the acetylene complex than in the ethylene complex [16,17].
2.2. Unstoichiometric Suzuki–Miyaura Polycondensation
2.2.1. Polycondensation of Dibromotolan and Phenylenediboronic Acid (Ester)
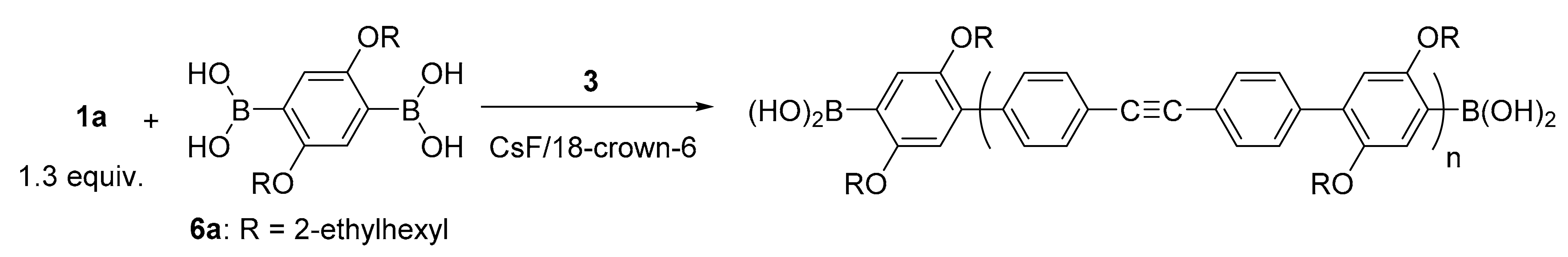
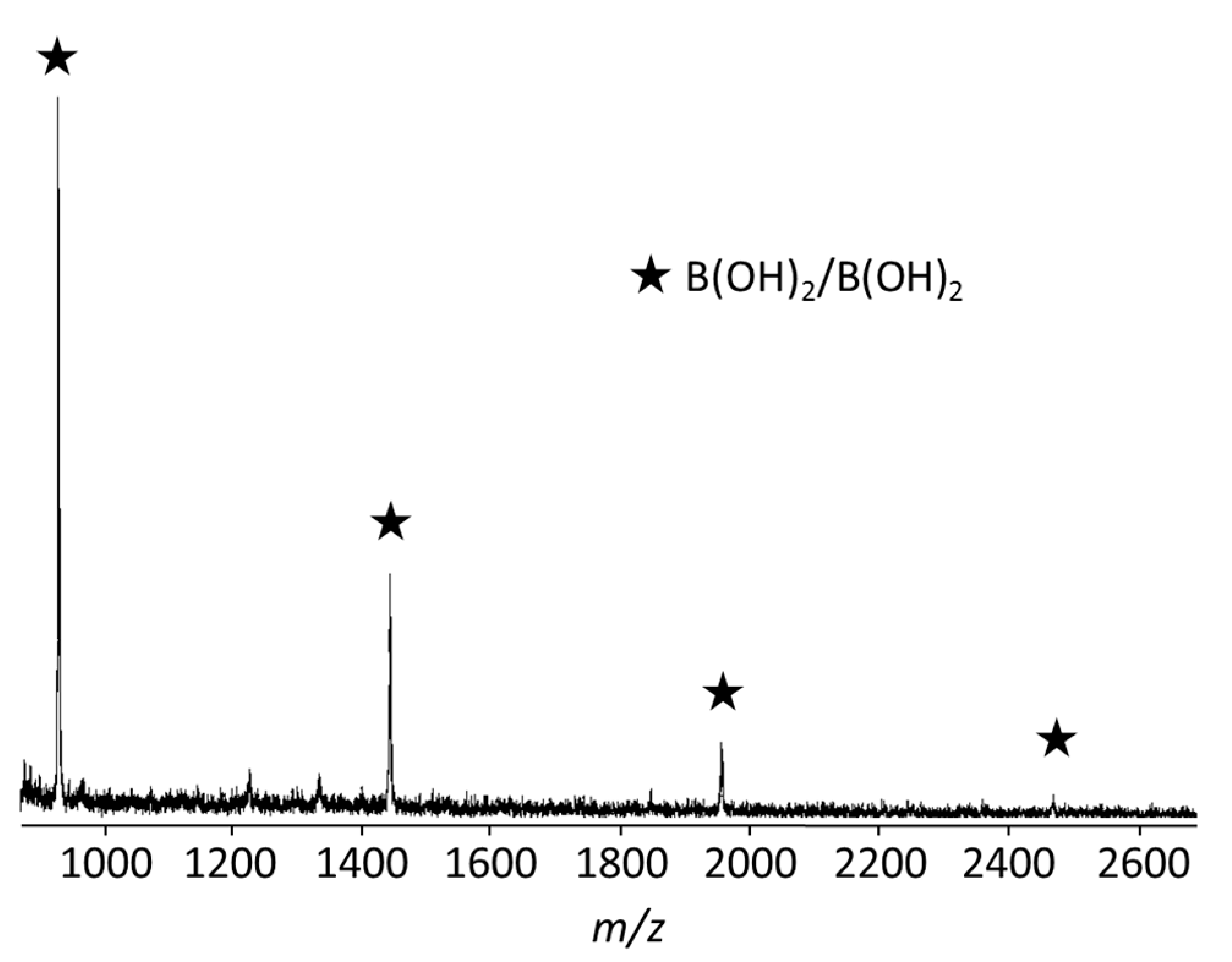
Since we found that t-Bu3PPd(0) catalyst undergoes intramolecular catalyst transfer on C≡C, we conducted unstoichiometric Suzuki–Miyaura polycondensation of 1.3 equiv. of 1a and 1.0 equiv. of phenylenediboronic acid 6a in the presence of Pd catalyst 3 and CsF/18-crown-6 as a base at room temperature to afford polymer with Mn of 5500 Da and Mw/Mn of 3.95 (Scheme 2). The matrix-assisted laser desorption ionization time-of-flight (MALDI-TOF) mass spectrum of the polymer showed one series of peaks due to polymer with a boronic acid moiety at both ends (Figure 1). These results indicated that the polymerization involved intramolecular catalyst transfer on C≡C-containing monomer 1a.
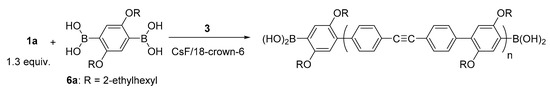
Scheme 2.
Unstoichiometric Suzuki–Miyaura polycondensation of 1a with 6a in the presence of 3.
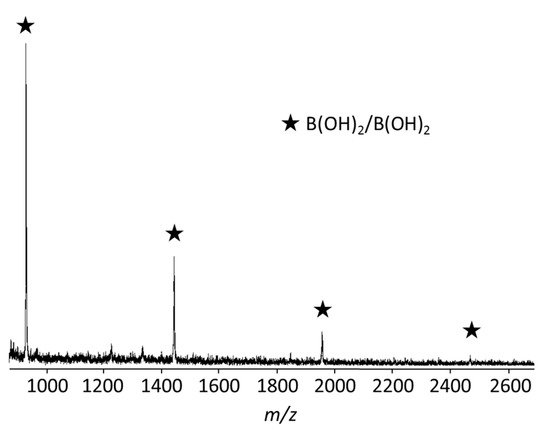
Figure 1.
MALDI-TOF MS spectrum of polymer obtained by the polymerization of 1.3 equiv. of 1a and 1.0 equiv. of 6a in the presence of 2.0 mol % of 3, CsF (5 equiv.), and 18-crown-6 (8 equiv.) in THF ([6a] = 8.33 × 10−3 M) and water (THF/water = 30/1, v/v) at rt for 15 h, followed by quenching with 1 M hydrochloric acid.
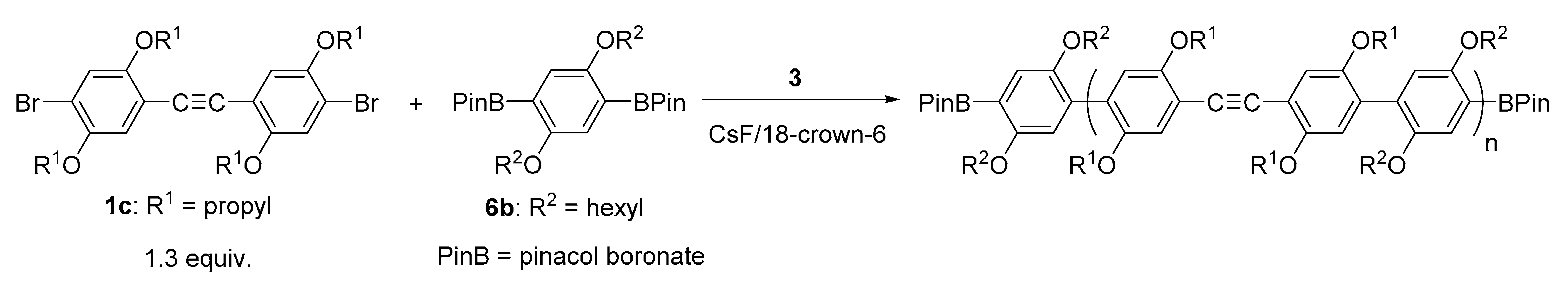
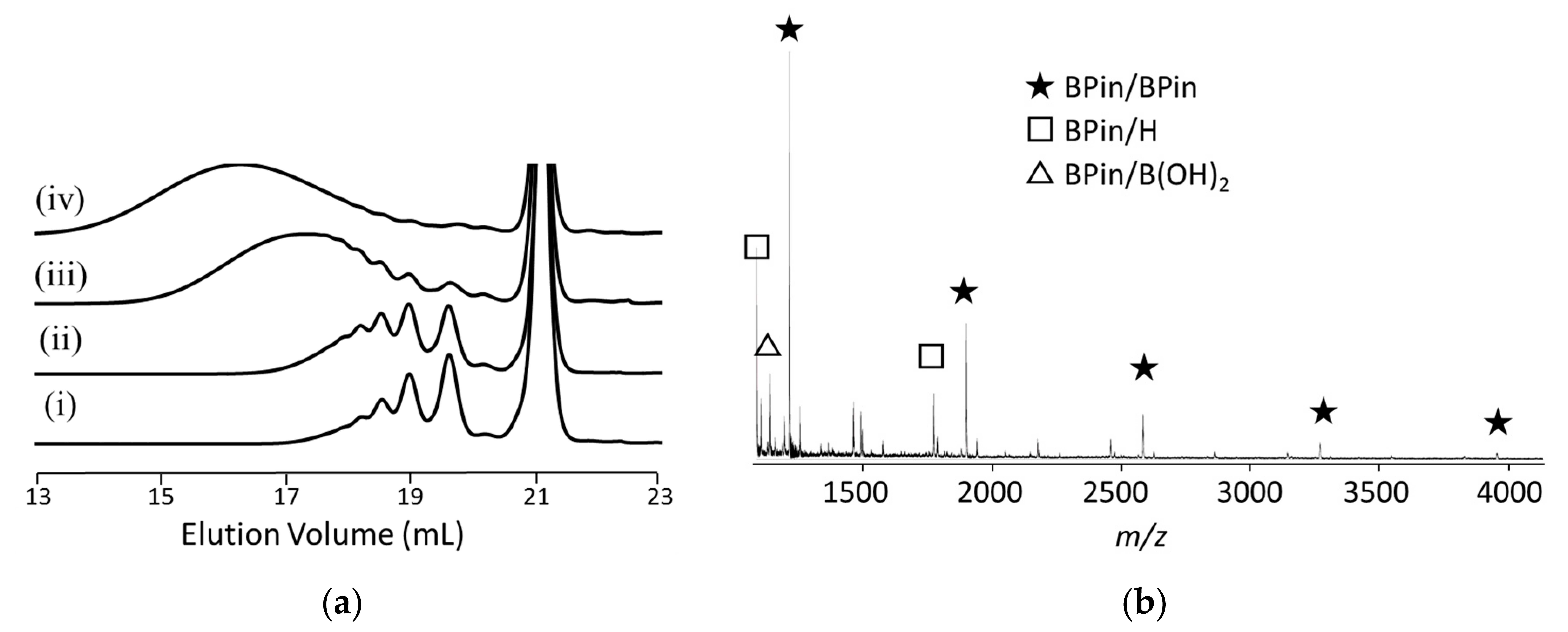
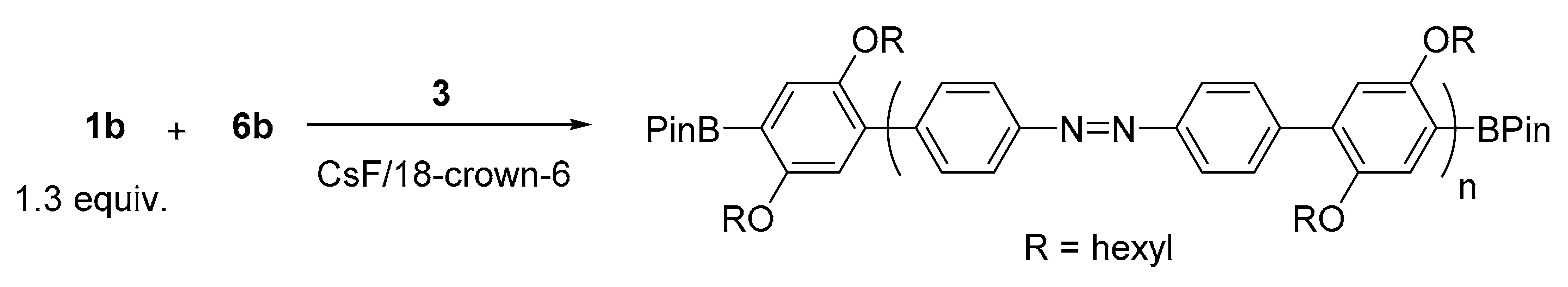
The polymerization of alkoxy-substituted dibromotolan 1c and alkoxy-substituted phenylene diboronic acid ester 6b was also conducted under similar conditions (Scheme 3). The molecular weight increased with reaction time and reached Mn = 10,500 Da (Mw/Mn = 2.77) (Figure 2a), even though 1.3 equiv. of 1c was used. The MALDI-TOF mass spectrum of polymer obtained at 24 h similarly showed major peaks due to polymer with a boronic acid ester moiety at both ends (BPin/BPin). Minor peaks in the lower-molecular-weight region are assignable to BPin/H and BPin/B(OH)2, which would be formed by hydrolysis of polymer–Pd–Br and the pinacol boronate moiety, respectively, by hydrochloric acid used to quench the polymerization. Formation of higher-molecular-weight polymer, in contrast to the polymerization of unsubstituted tolan 1a with 6a, is presumably accounted for by a higher propensity for intramolecular catalyst transfer on 1c than 1a, as in the case of Suzuki–Miyaura coupling reaction of dibromostilbene with phenylboronic acid: alkoxy groups at the ortho position of C=C of stilbene promoted intramolecular catalyst transfer, as mentioned in the Introduction.
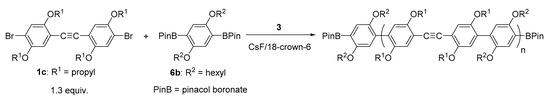
Scheme 3.
Unstoichiometric Suzuki–Miyaura polycondensation of 1c with 6b in the presence of 3.
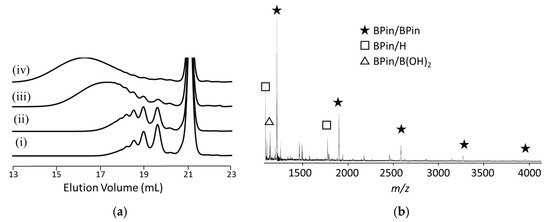
Figure 2.
Unstoichiometric Suzuki–Miyaura polycondensation of 1.3 equiv. of 1c with 1.0 equiv. of 6b in the presence of 2.0 mol % of 3, CsF (5.0 equiv.), and 18-crown-6 (8.0 equiv.) in THF ([6b] = 8.33 × 10−3 M) and water (THF/water = 30/1, v/v) at rt: (a) GPC profiles of polymer obtained at (i) 1 h (Mn = 2470 Da, Mw/Mn = 1.29), (ii) 5 h (Mn = 3000 Da, Mw/Mn = 1.39), (iii) 24 h (Mn = 6640 Da, Mw/Mn = 2.03), (iv) 48 h (Mn = 10,500 Da, Mw/Mn = 2.77; (b) MALDI-TOF mass spectra of polymer obtained at 24 h (Mn = 6640 Da, Mw/Mn = 2.03).
2.2.2. Polycondensation of Dibromoazobenzene and Phenylenediboronic Acid Ester
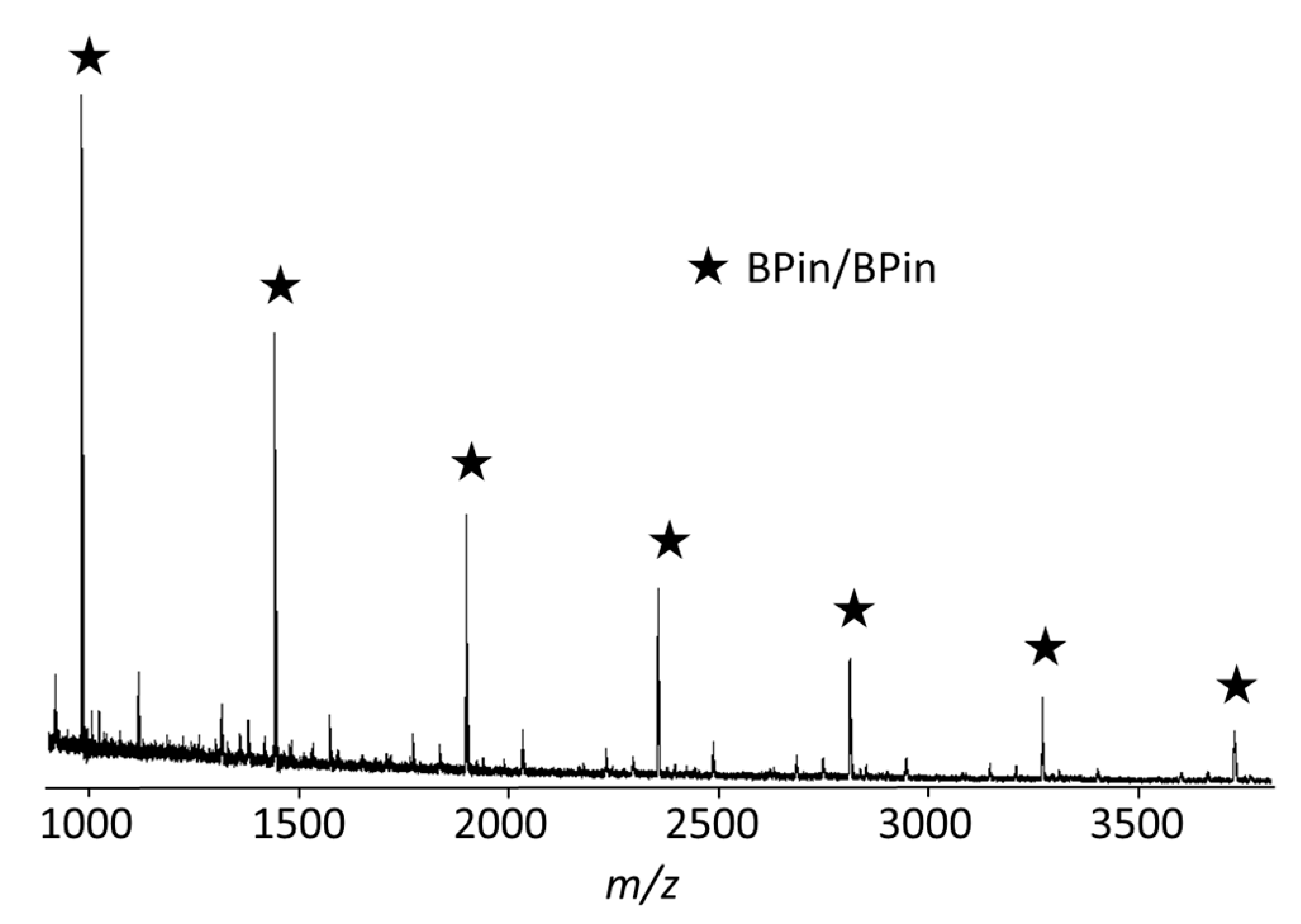
We applied intramolecular catalyst transfer on N=N, as mentioned in Section 2.1, for unstoichiometric polycondensation of 1.3 equiv. of dibromoazobenzene 1b and 1.0 equiv. of phenylenediboronic acid ester 6b in the presence of Pd catalyst 3 (Scheme 4). The polymerization proceeded homogeneously, but polymer was precipitated at 42 h. Accordingly, the THF-soluble part of the polymer obtained at 42 h was analyzed. The GPC elution curve of the obtained polymer showed Mn = 5500 Da and Mw/Mn = 1.44, and the polymer ends were confirmed to be BPin/BPin by MALDI-TOF mass spectrometry (Figure 3). Therefore, it turned out that polycondensation involving intramolecular catalyst transfer on 1b proceeded, although high-molecular-weight polymer was not obtained due to the low solubility of the polymer in THF.
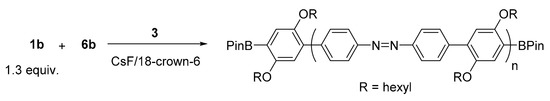
Scheme 4.
Unstoichiometric Suzuki–Miyaura polycondensation of 1b with 6b in the presence of 3.
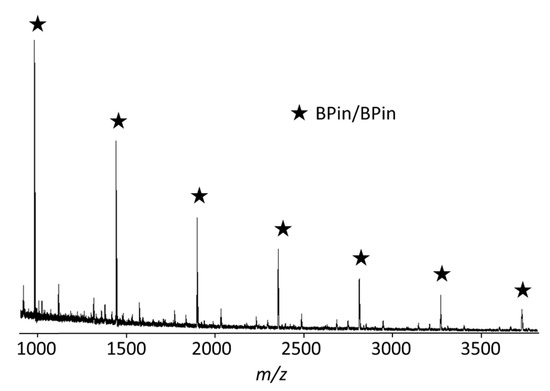
Figure 3.
MALDI-TOF MS spectrum of polymer obtained by the polymerization of 1.3 equiv. of 1b and 1.0 equiv. of 6b in the presence of 2.0 mol % of 3, CsF (5 equiv.), and 18-crown-6 (8 equiv.) in THF ([6b] = 8.33 × 10−3 M) and water (THF/water = 30/1, v/v) at rt for 42 h, followed by quenching with 1 M hydrochloric acid.
In order to obtain more soluble polymer, polymerization of 1b with 6a, bearing a 2-ethylhexyloxy group, was carried out. The polymerization proceeded homogeneously, and the molecular weight was increased to 8300. However, the molecular weight distribution was exceptionally broad (Mw/Mn = 12.4); the reason for this is not clear at present. Probably due to this broad molecular weight distribution, the MALDI-TOF mass spectrum did not show clear peaks in the region of molecular weight higher than 2000 Da.
3. Materials and Methods
3.1. Materials
All starting materials were purchased from commercial suppliers (TCI, Aldrich, Wako, and Kanto, Tokyo, Japan) and used without further purification. Dry tetrahydrofuran (THF, stabilizer-free, Kanto, Tokyo, Japan) and distilled water (Wako, Tokyo, Japan) were used as received. Dibromoazobenzene 1b [18] and phenylenediboronic acid 6a [11] were prepared according to the literature.
3.2. Measurements
1H and 13C NMR spectra were obtained on JEOL ECA-500 (JEOL, Tokyo, Japan and ECA-600 spectrometers (JEOL, Tokyo, Japan). The internal standard for 1H NMR spectra in CDCl3 was tetramethylsilane (0.00 ppm) and the internal standard for 13C NMR spectra in CDCl3 was the midpoint of CDCl3 (77.0 ppm). IR spectra were recorded on a JASCO FT/IR-410 (JASCO, Tokyo, Japan). All melting points were measured with a Yanagimoto hot stage melting point apparatus (Yanaco, Tokyo, Japan) without correction. GC was performed on a Shimadzu GC-14B gas chromatograph (Shimazu, Kyoto, Japan) equipped with a Shimadzu fused silica capillary column CBP1-W12–100 (12 m length, 0.53 mm i.d.) and a flame ionization detector (FID). Isolation of 4b and 5b was carried out on a Japan Analytical Industry LC908-C60 recycling preparative HPLC (eluent, CHCl3) (JAI, Tokyo, Japan) with two JAIGEL columns (1H-40 and 2H-40). Column chromatography was performed on silica gel (Kieselgel 60, 230–400 mesh, Merck, Darmstadt, Germany) with a specified solvent. The Mn and Mw/Mn values of polymer were measured on a Tosoh HLC-8020 gel permeation chromatography (GPC) unit (eluent, THF; calibration, polystyrene standards) (Tosoh, Yamaguchi, Japan) with two TSK-gel columns (2 × Multipore HXL-M). MALDI-TOF mass spectra were recorded on a Shimadzu/Kratos AXIMA-CFR plus (Shimadzu, Kyoto, Japan) in the reflectron ion mode (laser λ = 337 nm). DCTB (trans-2-[3-(4-tert-butylphenyl)-2-methyl-2-propenylidene]malononitrile) was used as the matrix for the MALDI-TOF mass measurements.
3.3. Synthesis of Monosubstituted Tolan 4a
Transfer of reagents and withdrawal of small aliquots of the reaction mixture for analysis were carried out via a syringe from a three-way stopcock under a stream of nitrogen. A two-necked round-bottomed flask was equipped with a three-way stopcock and a dimroth condenser. Dibromotolan 1a (134.9 mg, 0.40 mmol), phenylboronic acid 2 (81.4 mg, 0.42 mmol), (PPh3)4Pd (23.3 mg, 0.020 mmol), K3PO4 (370.9 mg, 1.75 mmol), and 18-crown-6 (846 mg, 3.26 mmol) were placed in the flask, and the flask was flushed with argon. Dry THF (10.0 mL) and distilled water (0.60 mL) were added to the flask via a syringe, and the reaction mixture was refluxed for 3 h. The reaction was quenched with 1 M hydrochloric acid, and the mixture was extracted with CHCl3. The organic layer was washed with water and dried over anhydrous MgSO4. The solvent was removed under reduced pressure, and the residue was purified by means of column chromatography (SiO2, hexane) to afford 4a as a pale yellow viscous liquid (111.3 mg, 69%); mp 96–99 °C.
1H NMR (500 MHz, CDCl3) δ 7.58 (s, 4H), 7.50 (d, J = 8.6 Hz, 2H), 7.40 (d, J = 8.6 Hz, 2H), 7.35 (t, J = 8.0 Hz, 1H), 7.18 (d, J = 6.9 Hz, 1H), 7.13 (s, 1H), 6.90 (dd, J = 5.7 Hz and 2.3 Hz, 1H), 3.78 (d, J = 6.9 Hz, 2H), 2.14-2.10 (m, 1H), 1.05 (d, J = 6.3 Hz, 6H); 13C NMR (126 MHz, CDCl3) δ 159.7, 141.8, 140.9, 132.0, 129.8, 127.3, 122.3, 119.3, 113.4, 90.0, 75.6, 30.9, 28.3, 19.3; IR (KBr) 3460, 2908, 1606, 1471, 1350, 1206, 1105, 965, 776, 690, 513 cm−1.
3.4. Synthesis of Disubstituted Tolan 5a
Transfer of reagents and withdrawal of small aliquots of the reaction mixture for analysis were carried out via a syringe from a three-way stopcock under a stream of nitrogen. Dibromotolan 1a (134.3 mg, 0.40 mmol), 2 (157.7 mg, 0.81 mmol), 3 (10.2 mg, 0.020 mmol), KOH (105.4 mg, 1.88 mmol), and 18-crown-6 (861.0 mg, 3.26 mmol) were placed in a flask equipped with a three-way stop cock, and the atmosphere in the flask was replaced with argon. Dry THF (10.0 mL) and distilled water (0.60 mL) were added to the flask via a syringe, and the mixture was degassed under reduced pressure and filled with argon. The reaction mixture was stirred at room temperature for 2 h. The reaction was quenched with hydrochloric acid, and the mixture was extracted with CHCl3. The organic layer was washed with water and dried over anhydrous MgSO4. The solvent was removed under reduced pressure, and the residue was purified by means of column chromatography (SiO2, hexane) to afford 5a as a dark yellow viscous solid (184.1 mg, 96%); mp 130–134 °C.
1H NMR (500 MHz, CDCl3) δ 7.60 (s, 8H), 7.35 (t, J = 7.7 Hz, 2H), 7.18 (d, J = 7.7 Hz, 2H), 7.15 (s, 2H), 6.90 (dd, J = 6.0 and 2.0 Hz, 2H), 3.79 (d, J = 6.6 Hz, 4H), 2.13-2.10 (m, 2H), 1.04 (d, J = 6.9 Hz, 12H); 13C NMR (126 MHz, CDCl3) δ 167.7, 152.2, 147.2, 144.7, 130.8, 124.5, 115.5, 114.2, 83.9, 69.8, 29.7, 24.8; IR (KBr) 3461, 2907, 1606, 1522, 1295, 1206, 1050, 1013, 873, 830, 776, 690, 538 cm−1.
3.5. Synthesis of Mono- and Disubstituted Azobenzene 4b and 5b
Transfer of reagents and withdrawal of small aliquots of the reaction mixture for analysis were carried out via a syringe from a three-way stopcock under a stream of nitrogen. Dibromoazobenzene 1b (50.9 mg, 0.15 mmol), 2 (30.6 mg, 0.16 mmol), 3 (4.2 mg, 0.016 mmol), KOH (37.8 mg, 0.68 mmol), and 18-crown-6 (318.4 mg, 1.2 mmol) were placed in a flask equipped with a three-way stop cock, and the atmosphere in the flask was replaced with argon. Dry THF (6.0 mL) and distilled water (0.36 mL) were added to the flask via a syringe, and the mixture was degassed under reduced pressure and filled with argon. The reaction mixture was stirred at room temperature for 24 h. The reaction was quenched with hydrochloric acid, and the mixture was extracted with CHCl3. The organic layer was washed with water and dried over anhydrous MgSO4. The solvent was removed under reduced pressure, and the residue was purified by means of HPLC to afford 4b as a pale yellow viscous solid (10.4 mg, 17%) and 5b as a pale yellow viscous solid (22.3 mg, 31%).
4b: mp 89–95 °C; 1H NMR (500 MHz, CDCl3) δ 7.98 (d, J = 8.6 Hz, 2H), 7.82 (d, J = 8.9 Hz, 2H), 7.74 (d, J = 8.6 Hz, 2H), 7.66 (d, J = 8.6 Hz, 2H), 7.37 (t, J = 8.0 Hz, 1H), 7.24 (d, J = 7.7 Hz, 1H), 7.20 (s, 1H), 6.94 (dd, J = 7.7 and 1.7 Hz, 1H), 3.80 (d, J = 6.6 Hz, 1H), 2.16–2.10 (m, 1H), 1.06 (d, J = 6.6 Hz, 6H), 13C NMR (126 MHz, CDCl3) δ 159.7, 151.9, 143.6, 141.6, 129.8, 127.8, 123.3, 119.4, 113.8, 113.6, 74.5, 28.3, 19.3, IR (KBr) 3460, 2911, 1599, 1469, 1397, 1217, 1108, 965, 840, 610, 552 cm−1.
5b: mp 195–198 °C; 1H NMR (500 MHz, CDCl3) δ 8.00 (d, J = 8.6 Hz, 4H), 7.75 (d, J = 8.3 Hz, 4H), 7.37 (t, J = 8.0 Hz, 2H), 7.24 (d, J = 8.3 Hz, 2H), 7.20 (s, 2H), 6.93 (dd, J = 6.3 and 2.0 Hz, 2H), 3.81 (d, J = 6.3 Hz, 4H), 2.17–2.11 (m, 2H), 1.06 (d, J = 6.6 Hz, 12H), 13C NMR (126 MHz, CDCl3) δ 159.7, 151.6, 132.3, 129.9, 128.3, 127.8, 124.3, 119.4, 113.9, 113.6, 77.3, 28.3, 19.3, IR (KBr) 3448, 2924, 2362, 1719, 1571, 1473, 1281, 1065, 1006, 835, 657, 577 cm−1.
3.6. Suzuki–Miyaura Coupling Reaction of 1a with 2
Transfer of reagents and withdrawal of small aliquots of the reaction mixture for analysis were carried out via a syringe from a three-way stopcock under a stream of nitrogen. Dibromotolan 1a (0.100 mmol), 2 (0.105 mmol), 3 (0.005 mmol), KOH (0.5 mmol), 18-crown-6 (0.80 mmol), and 1,4-bis(hexyloxy)benzene (0.040 mmol) as an internal standard substance were placed in the flask, and the atmosphere in the flask was replaced with argon. Dry THF (4.0 mL), distilled water (0.40 mL), and additive (none or 0.100 mmol) were added to the flask via a syringe, and the mixture was degassed under reduced pressure and filled with argon. The reaction mixture was stirred at room temperature for 2 h, and the reaction was quenched with 1 M hydrochloric acid. The mixture was extracted with CHCl3, and the organic layer was subjected to GC analysis for estimation of conversion of 1a and the product ratio of 4a to 5a.
3.7. Suzuki–Miyaura Coupling Reaction of 1b with 2
Transfer of reagents and withdrawal of small aliquots of the reaction mixture for analysis were carried out via a syringe from a three-way stopcock under a stream of nitrogen. Dibromoazobenzene 1b (50.9 mg, 0.15 mmol), 2 (30.6 mg, 0.16 mmol), 3 (4.2 mg, 0.008 mmol), KOH (37.8 mg, 0.68 mmol), 18-crown-6 (318.4 mg, 1.2 mmol), and 1,4-bis(hexyloxy)benzene (27.2 mg, 0.016 mmol) as an internal standard substance were placed in the flask, and the atmosphere in the flask was replaced with argon. Dry THF (6.0 mL) and distilled water (0.36 mL) were added to the flask via a syringe, and the mixture was degassed under reduced pressure and filled with argon. The reaction mixture was stirred at room temperature for 24 h, and the reaction was quenched with 1 M hydrochloric acid. The mixture was extracted with CHCl3 and dried over MgSO4. The solvent was removed under reduced pressure. The whole mixture was dissolved in CDCl3 and then subjected to 1H NMR analysis to determine conversion of 1b and the product ratio of 4b to 5b.
3.8. Polycondensation of 1c and 6b
Transfer of reagents and withdrawal of small aliquots of the reaction mixture for analysis was carried out via a syringe from a three-way stopcock under a stream of nitrogen. Dibromo monomer 1c (36.89 mg, 0.065 mmol), phenylenediboronic acid pinacol ester 6b (26.91 mg, 0.051 mmol), CsF (34.1 mg, 0.22 mmol), 18-crown-6 (119.1 mg, 0.45 mmol), and 3 (1.4 mg, 0.027 mmol) were placed in the flask, and the atmosphere in the flask was replaced with argon. Dry THF (6.0 mL) and distilled water (0.2 mL) were added to the flask via a syringe, and the mixture was degassed under reduced pressure and filled with argon. The reaction mixture was stirred at room temperature for two days, and the reaction was quenched with 1 M hydrochloric acid. The mixture was extracted with CHCl3, and the organic layer was washed with water, then dried over anhydrous MgSO4. Concentration under reduced pressure gave residue, which was purified by preparative HPLC to give polymer (32.7 mg, 94%).
4. Conclusions
We have demonstrated that t-Bu3PPd(0) catalyst undergoes intramolecular transfer on C≡C and N=N, in contrast to the case of C=C, by means of Suzuki–Miyaura coupling reaction of equimolar dibromotolan 1a or dibromoazobenzene 1b with phenylboronic acid 2 in the presence of t-Bu3P-ligated Pd precatalyst 3 and by unstoichiometric polycondensation of 1 and phenyleneboronic acid (ester) 6 with 3. In the former reaction, successive disubstitution of 1 with 2 selectively proceeded, and the latter polycondensation yielded high-molecular-weight conjugated polymer with a boronic acid (ester) moiety at both ends, even if excess dibromo monomer 1 was used. We believe these findings provide useful information about the requirements for Pd catalyst transfer on π-conjugated faces, as well as affording entry into a new class of π-conjugated polymer architectures via functionalization of chain-end boronate moieties.
Supplementary Materials
The following details are available online at www.mdpi.com/2073-4344/7/7/195/s1, Synthesis of mono- and disubtituted tolan 4a and 5a, Synthesis of mono- and disubstituted azobenzene 4b and 5b, The product ratio of 5a to 4a and 5a as a function of conversion of 1a, Estimation of product ratio of 4b to 5b by 1H NMR spectra, and DFT calculations for complexation of diphenylacetylene, stilbene, and azobenene with PdP(t-Bu)3.
Acknowledgments
This study was supported by a Grant in Aid (No. 15H03819) for Scientific Research from the Japan Society for the Promotion of Science (JSPS) and by the MEXT-Supported Program for the Strategic Research Foundation at Private Universities (No. S1311032), 2013-2018.
Author Contributions
H.S. and T.Y. conceived and designed the experiments; T.K. performed the experiments; K.M. and T.Y. analyzed the data; Y.O. contributed reagents/materials/analysis tools; T.Y. wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sakamoto, J.; Rehahn, M.; Wegner, G.; Schlüter, A.D. Suzuki polycondensation: Polyarylenes à la carte. Macromol. Rapid Commun. 2009, 30, 653–687. [Google Scholar] [CrossRef] [PubMed]
- Stambuli, J.P.; Bühl, M.; Hartwig, J.F. Synthesis, characterization, and reactivity of monomeric, arylpalladium halide complexes with a hindered phosphine as the only dative ligand. J. Am. Chem. Soc. 2002, 124, 9346–9347. [Google Scholar] [CrossRef] [PubMed]
- Stambuli, J.P.; Incarvito, C.D.; Buhl, M.; Hartwig, J.F. Synthesis, structure, theoretical studies, and ligand exchange reactions of monomeric, T-shaped arylpalladium(II) halide complexes with an additional, weak agostic interaction. J. Am. Chem. Soc. 2004, 126, 1184–1194. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, A.; Suzuki, H.; Kubota, Y.; Ohuchi, K.; Higashimura, H.; Yokozawa, T. Chain-growth polymerization for the synthesis of polyfluorene via Suzuki–Miyaura coupling reaction from an externally added initiator unit. J. Am. Chem. Soc. 2007, 129, 7236–7237. [Google Scholar] [CrossRef] [PubMed]
- Yokozawa, T.; Kohno, H.; Ohta, Y.; Yokoyama, A. Catalyst-transfer Suzuki–Miyaura coupling polymerization for precision synthesis of poly(p-phenylene). Macromolecules 2010, 43, 7095–7100. [Google Scholar] [CrossRef]
- Yokozawa, T.; Suzuki, R.; Nojima, M.; Ohta, Y.; Yokoyama, A. Precision synthesis of poly(3-hexylthiophene) from catalyst-transfer Suzuki–Miyaura coupling polymerization. Macromol. Rapid Commun. 2011, 32, 801–806. [Google Scholar] [CrossRef] [PubMed]
- Verswyvel, M.; Hoebers, C.; De Winter, J.; Gerbaux, P.; Koeckelberghs, G. Study of the controlled chain-growth polymerization of poly(3,6-phenanthrene). J. Polym. Sci. Part A: Polym. Chem. 2013, 51, 5067–5074. [Google Scholar] [CrossRef]
- Elmalem, E.; Kiriy, A.; Huck, W.T.S. Chain-growth Suzuki polymerization of n-type fluorene copolymers. Macromolecules 2011, 44, 9057–9061. [Google Scholar] [CrossRef]
- Yokozawa, T.; Ohta, Y. Transformation of step-growth polymerization into living chain-growth polymerization. Chem. Rev. 2016, 116, 1950–1968. [Google Scholar] [CrossRef] [PubMed]
- Yokozawa, T.; Ohta, Y. Chapter 1 controlled synthesis of conjugated polymers in catalyst-transfer condensation polymerization: Monomers and catalysts. In Semiconducting Polymers: Controlled Synthesis and Microstructure; The Royal Society of Chemistry: Cambridge, UK, 2017; pp. 1–37. [Google Scholar]
- Nojima, M.; Kosaka, K.; Kato, M.; Ohta, Y.; Yokozawa, T. Alternating intramolecular and intermolecular catalyst-transfer Suzuki–Miyaura condensation polymerization: Synthesis of boronate-terminated π-conjugated polymers using excess dibromo monomers. Macromol. Rapid Commun. 2016, 37, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Nojima, M.; Ohta, Y.; Yokozawa, T. Structural requirements for palladium catalyst transfer on a carbon–carbon double bond. J. Am. Chem. Soc. 2015, 137, 5682–5685. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-H.; Xing, C.-H.; Tsemo, G.B.; Hu, Q.-S. t-Bu3P-coordinated 2-phenylaniline-based palladacycle complex as a precatalyst for the Suzuki cross-coupling polymerization of aryl dibromides with aryldiboronic acids. ACS Macro Lett. 2013, 2, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Ono, R.J.; Bielawski, C.W. Controlled catalyst transfer polycondensation and surface-initiated polymerization of p-phenyleneethynylene-based monomer. J. Am. Chem. Soc. 2013, 135, 4984–4987. [Google Scholar] [CrossRef] [PubMed]
- Bryan, Z.J.; Hall, A.O.; Zhao, C.T.; Chen, J.; McNeil, A.J. Limitations of using small molecules to identify catalyst-transfer polycondensation reactions. ACS Macro Lett. 2016, 5, 69–72. [Google Scholar] [CrossRef]
- Zenkina, O.V.; Gidron, O.; Shimon, L.J.W.; Iron, M.A.; van der Boom, M.E. Mechanistic aspects of aryl–halide oxidative addition, coordination chemistry, and ring-walking by palladium. Chem. Eur. J. 2015, 21, 16113–16125. [Google Scholar] [CrossRef] [PubMed]
- Zenkina, O.; Altman, M.; Leitus, G.; Shimon, L.J.W.; Cohen, R.; van der Boom, M.E. From azobenzene coordination to aryl-halide bond activation by platinum. Organometallics 2007, 26, 4528–4534. [Google Scholar] [CrossRef]
- Ma, H.; Li, W.; Wang, J.; Xiao, G.; Gong, Y.; Qi, C.; Feng, Y.; Li, X.; Bao, Z.; Cao, W.; et al. Organocatalytic oxidative dehydrogenation of aromatic amines for the preparation of azobenzenes under mild conditions. Tetrahedron 2012, 68, 8358–8366. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).