Copper(II) Complexes of Arylhydrazone of 1H-Indene-1,3(2H)-dione as Catalysts for the Oxidation of Cyclohexane in Ionic Liquids
Abstract
1. Introduction
2. Results and Discussion
2.1. Conventional Medium
2.2. Unconventional Medium (Ionic Liquid)
3. Experimental Section
3.1. Materials and Equipment
3.2. Synthesis of the Catalysts
3.3. Oxidation of Cyclohexane and Products Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Labinger, J.A. Platinum-Catalyzed C-H Functionalization. Chem. Rev. 2017, 117, 8483–8496. [Google Scholar] [CrossRef]
- Pombeiro, A.J.L. Toward Functionalization of Alkanes Under Environmentally Benign Conditions. In Advances in Organometallic Chemistry and Catalysis: The Silver/Gold Jubilee International Conference on Organometallic Chemistry Celebratory Book; Pombeiro, A.J.L., Ed.; Wiley: Hoboken, NJ, USA, 2014; pp. 15–23. ISBN 978-1-11-851014-8. [Google Scholar]
- Crabtree, R.H. Alkane C-H activation and functionalization with homogeneous transition metal catalysts: A century of progress-a new millennium in prospect. J. Chem. Soc. Dalton Trans. 2001, 17, 2437–2450. [Google Scholar] [CrossRef]
- Crabtree, R.H.A.E. Shilov’s influence on early work in organometallic CH activation and functionalization. J. Org. Chem. 2015, 793, 41–46. [Google Scholar] [CrossRef]
- Shul’pin, G.B. New Trends in Oxidative Functionalization of Carbon-Hydrogen Bonds: A Review. Catalysts 2016, 6, 50. [Google Scholar] [CrossRef]
- Nesterov, D.S.; Nesterova, O.V.; Pombeiro, A.J.L. Homo- and heterometallic polynuclear transition metal catalysts for alkane C-H bonds oxidative functionalization: Recent advances. Coord Chem. Rev. 2018, 355, 199–222. [Google Scholar] [CrossRef]
- Banerjee, A.; Sarkar, S.; Patel, B.K. C-H functionalisation of cycloalkanes. Org. Biomol. Chem. 2017, 15, 505–530. [Google Scholar] [CrossRef]
- Schuchardt, U.; Carvalho, W.A.; Spinace, E.V. Why is it interesting to study cyclohexane oxidation. Synlett 1993, 10, 713–718. [Google Scholar] [CrossRef]
- Schuchardt, U.; Cardoso, D.; Sercheli, R.; Pereira, R.; de Cruz, R.S.; Guerreiro, M.C.; Mandelli, D.; Spinace, E.V.; Fires, E.L. Cyclohexane oxidation continues to be a challenge. Appl. Catal. A 2001, 211, 1–17. [Google Scholar] [CrossRef]
- Shilov, A.E.; Shul’pin, G.B. (Eds.) Activation and Catalytic Reactions of Saturated Hydrocarbons in the Presence of Metal Complexes; Springer Science & Business Media: New York, NY, USA, 2006; Volume 21. [Google Scholar]
- Wu, W.; Jiang, H. Palladium-Catalyzed Oxidation of Unsaturated Hydrocarbons Using Molecular Oxygen. Acc. Chem. Res. 2012, 45, 1736–1748. [Google Scholar] [CrossRef]
- Allen, S.E.; Walvoord, R.R.; Padilla-Salinas, R.; Kozlowski, M.C. Aerobic Copper-Catalyzed Organic Reactions. Chem. Rev. 2013, 113, 6234–6458. [Google Scholar] [CrossRef]
- Shul’pin, G.B.; Loginov, D.A.; Shul’pina, L.S.; Ikonnikov, N.S.; Idrisov, V.O.; Vinogradov, M.M.; Osipov, S.N.; Nelyubina, Y.V.; Tyubaeva, P.M. Stereoselective Alkane Oxidation with meta-Chloroperoxybenzoic Acid (MCPBA) Catalyzed by Organometallic Cobalt Complexes. Molecules 2016, 21, 1593. [Google Scholar] [CrossRef]
- Saisaha, P.; Dong, J.J.; Meinds, T.G.; de Boer, J.W.; Hage, R.; Mecozzi, F.; Kasper, J.B.; Browne, W.R. Mechanism of Alkene, Alkane, and Alcohol Oxidation with H2O2 by an in Situ Prepared Mn-II/Pyridine-2-carboxylic Acid Catalyst. ACS Catal. 2016, 6, 3486–3495. [Google Scholar] [CrossRef]
- Dragancea, D.; Talmaci, N.; Shova, S.; Novitchi, G.; Darvasiová, D.; Rapta, P.; Breza, M.; Galanski, M.; Kožıšek, J.; Martins, N.M.R.; et al. Vanadium(V) Complexes with Substituted 1,5-bis(2-hydroxybenzaldehyde)carbohydrazones and Their Use As Catalyst Precursors in Oxidation of Cyclohexane. Inorg. Chem. 2016, 55, 9187–9203. [Google Scholar] [CrossRef]
- Silva, T.F.S.; Martins, L.M.D.R.S.; Guedes da Silva, M.F.C.; Kuznetsov, M.L.; Fernandes, A.R.; Silva, A.; Pan, C.-J.; Lee, J.-F.; Hwang, B.J.; Pombeiro, A.J.L. Cobalt Complexes with Pyrazole Ligands as Catalyst Precursors for the Peroxidative Oxidation of Cyclohexane: X-ray Absorption Spectroscopy Studies and Biological Applications. Chem. Asian J. 2014, 9, 1132–1143. [Google Scholar] [CrossRef]
- Martins, L.M.D.R.S.; Pombeiro, A.J.L. Tris(pyrazol-1-yl)methane metal complexes for catalytic mild oxidative functionalizations of alkanes, alkenes and ketones. Coord. Chem. Rev. 2014, 265, 74–88. [Google Scholar] [CrossRef]
- Hazra, S.; Mukherjee, S.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. A cyclic tetranuclear cuboid type copper(II) complex doubly supported by cyclohexane-1,4-dicarboxylate: Molecular and supramolecular structure and cyclohexane oxidation activity. RSC Adv. 2014, 4, 48449–48457. [Google Scholar] [CrossRef]
- Timokhin, I.; Pettinari, C.; Marchetti, F.; Pettinari, R.; Condello, F.; Galli, S.; Alegria, E.C.B.A.; Martins, L.M.D.R.S.; Pombeiro, A.J.L. Novel Coordination Polymers with (Pyrazolato)-Based Tectons: Catalytic Activity in the Peroxidative Oxidation of Alcohols and Cyclohexane. Cryst. Growth Des. 2015, 15, 2303–2317. [Google Scholar] [CrossRef]
- Ribeiro, A.P.C.; Martins, L.M.D.R.S.; Pombeiro, A.J.L. N2O-Free single-pot conversion of cyclohexane to adipic acid catalysed by an iron(II) scorpionate complex. Green Chem. 2017, 19, 1499–1501. [Google Scholar] [CrossRef]
- Nesterova, O.V.; Nesterov, D.S.; Krogul-Sobczak, A.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. Synthesis, crystal structures and catalytic activity of Cu(II) and Mn(III) Schiff base complexes: Influence of additives on the oxidation catalysis of cyclohexane and 1-phenylethanol. J. Mol. Catal. A Chem. 2017, 426, 506–515. [Google Scholar] [CrossRef]
- Wasserscheid, P. Transition Metal Catalysis in Ionic Liquids. In Handbook of Green Chemistry; Dupont, J., Kollar, L., Eds.; Wiley-VCH Verlag GmbH & Co.: Weinheim, Germany, 2010; Volume 6, pp. 65–91. ISBN 978-3-52-762869-8. [Google Scholar]
- Pârvulescu, V.I.; Hardacre, C. Catalysis in ionic liquids. Chem. Rev. 2007, 107, 2615–2665. [Google Scholar] [CrossRef]
- Vekariya, R.L. A review of ionic liquids: Applications towards catalytic organic transformations. J. Mol. Liq. 2017, 227, 44–60. [Google Scholar] [CrossRef]
- Dai, C.; Zhang, J.; Huang, C.; Lei, Z. Ionic Liquids in Selective Oxidation: Catalysts and Solvents. Chem. Rev. 2017, 117, 6929–6983. [Google Scholar] [CrossRef]
- Li, Z.; Xia, C.-G. Oxidation of hydrocarbons with iodobenzene diacetate catalyzed by manganese(III) porphyrins in a room temperature ionic liquid. J. Mol. Catal. A Chem. 2004, 214, 95–101. [Google Scholar] [CrossRef]
- Li, Z.; Xia, C.-G.; Xu, C.-Z. Oxidation of alkanes catalyzed by manganese(III) porphyrin in an ionic liquid at room temperature. Tetrahedron Lett. 2003, 44, 9229–9232. [Google Scholar] [CrossRef]
- Ribeiro, A.P.C.; Martins, L.M.D.R.S.; Kuznetsov, M.L.; Pombeiro, A.J.L. Tuning Cyclohexane Oxidation: Combination of Microwave Irradiation and Ionic Liquid with the C-Scorpionate [FeCl2(Tpm)] Catalyst. Organometallics 2017, 36, 192–198. [Google Scholar] [CrossRef]
- Martins, L.M.D.R.S.; Martins, A.; Alegria, E.C.B.A.; Carvalho, A.P.; Pombeiro, A.J.L. Efficient cyclohexane oxidation with hydrogen peroxide catalysed by a C-scorpionate iron(II) complex immobilized on desilicated MOR zeolite. Appl. Catal. A-Gen. 2013, 464, 43–50. [Google Scholar] [CrossRef]
- Silva, T.F.S.; Guedes da Silva, M.F.C.; Mishra, G.S.; Martins, L.M.D.R.S.; Pombeiro, A.J.L. Synthesis and structural characterization of iron complexes with 2,2,2-tris(1-pyrazolyl)ethanol ligands: Application in the peroxidative oxidation of cyclohexane under mild conditions. J. Org. Chem. 2011, 696, 1310–1318. [Google Scholar] [CrossRef]
- Silva, T.F.S.; Alegria, E.C.B.A.; Martins, L.M.D.R.S.; Pombeiro, A.J.L. Half-sandwich scorpionate vanadium, iron and copper complexes: Synthesis and application in the catalytic peroxidative oxidation of cyclohexane under mild conditions. Adv. Synth. Catal. 2008, 350, 706–716. [Google Scholar] [CrossRef]
- Silva, T.F.S.; Luzyanin, K.V.; Kirillova, M.V.; Guedes da Silva, M.F.C.; Martins, L.M.D.R.S.; Pombeiro, A.J.L. Novel Scorpionate and Pyrazole Dioxovanadium Complexes, Catalysts for Carboxylation and Peroxidative Oxidation of Alkanes. Adv. Synth. Catal. 2010, 352, 171–187. [Google Scholar] [CrossRef]
- Martins, L.M.D.R.S.; Pombeiro, A.J.L. Water-Soluble C-Scorpionate Complexes–Catalytic and Biological Applications. Eur. J. Inorg. Chem. 2016, 2236–2252. [Google Scholar] [CrossRef]
- Sutradhar, M.; Martins, L.M.D.R.S.; Guedes da Silva, M.F.G.; Pombeiro, A.J.L. Vanadium complexes: Recent progress in oxidation catalysis. Coord. Chem. Rev. 2015, 301, 200–239. [Google Scholar] [CrossRef]
- Martins, L.M.D.R.S. C-Homoscorpionate Oxidation Catalysts-Electrochemical and Catalytic Activity. Catalysts 2017, 7, 12. [Google Scholar] [CrossRef]
- Paul, A.; Ribeiro, A.P.C.; Karmakar, A.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. A Cu(II) MOF with a flexible bifunctionalised terpyridine as an efficient catalyst for the single-pot hydrocarboxylation of cyclohexane to carboxylic acid in water/ionic liquid médium. Dalton Trans. 2016, 45, 12779–12789. [Google Scholar] [CrossRef]
- Hazra, S.; Ribeiro, A.P.C.; Guedes da Silva, M.F.C.; Nieto de Castro, C.A.; Pombeiro, A.J.L. Syntheses and crystal structures of benzene-sulfonate and -carboxylate copper polymers and their application in the oxidation of cyclohexane in ionic liquid under mild conditions. Dalton Trans. 2016, 45, 13957–13968. [Google Scholar] [CrossRef]
- Ribeiro, A.P.C.; Martins, L.M.D.R.S.; Hazra, S.; Pombeiro, A.J.L. Catalytic oxidation of cyclohexane with hydrogen peroxide and a tetracopper(II) complex in an ionic liquid. Comp. Rend. Chim. 2015, 18, 758–765. [Google Scholar] [CrossRef]
- Jlassi, R.; Ribeiro, A.P.C.; Guedes da Silva, M.F.C.; Mahmudov, K.T.; Kopylovich, M.N.; Anisimova, T.B.; Naïli, H.; Tiago, G.A.O.; Pombeiro, A.J.L. Polynuclear Copper(II) Complexes as Catalysts for the Peroxidative Oxidation of Cyclohexane in a Room-Temperature Ionic Liquid. Eur. J. Inorg. Chem. 2014, 2014, 4541–4550. [Google Scholar] [CrossRef]
- Mazzi, A.; Paul, S.; Cavani, F.; Wojcieszak, R. Cyclohexane Oxidation to Adipic Acid Under Green Conditions: A Scalable and Sustainable Process. ChemCatChem 2018, 10, 3680–3682. [Google Scholar] [CrossRef]
- Gryca, I.; Czerwinska, K.; Machura, B.; Chrobok, A.; Shul’pina, L.S.; Kuznetsov, M.L.; Nesterov, D.S.; Kozlov, Y.N.; Pombeiro, A.J.L.; Varyan, I.A.; et al. High Catalytic Activity of Vanadium Complexes in Alkane Oxidations with Hydrogen Peroxide: An Effect of 8-Hydroxyquinoline Derivatives as Noninnocent Ligands. Inorg. Chem. 2018, 57, 1824–1839. [Google Scholar] [CrossRef]
- Ribeiro, A.P.C.; Alegria, E.C.B.A.; Kopylovich, M.N.; Ferraria, A.M.; do Rego, A.M.B.; Pombeiro, A.J.L. Comparison of microwave and mechanochemical energy inputs in catalytic oxidation of cyclohexane. Dalton Trans. 2018, 47, 8193–8198. [Google Scholar] [CrossRef]
- Alegria, E.C.B.A.; Fontolan, E.; Ribeiro, A.P.C.; Kopylovich, M.N.; Domingos, C.; Ferraria, A.M.; Bertani, R.; do Rego, A.M.B.; Pombeiro, A.J.L. Simple solvent-free preparation of dispersed composites and their application as catalysts in oxidation and hydrocarboxylation of cyclohexane. Mater. Today Chem. 2017, 5, 52–62. [Google Scholar] [CrossRef]
- Aboelfetoh, E.F.; Pietschnig, R. Preparation and Catalytic Performance of Al2O3, TiO2 and SiO2 Supported Vanadium Based-Catalysts for C-H Activation. Catal. Lett. 2009, 127, 83–94. [Google Scholar] [CrossRef]
- Aboelfetoh, E.F.; Fechtelkord, M.; Pietschnig, R. Structure and catalytic properties of MgO-supported vanadium oxide in the selective oxidation of cyclohexane. J. Mol. Catal. A Chem. 2010, 318, 51–59. [Google Scholar] [CrossRef]
- Aboelfetoh, E.F.; Pietschnig, R. Preparation, Characterization and Catalytic Activity of MgO/SiO2 Supported Vanadium Oxide Based Catalysts. Catal. Lett. 2014, 144, 97–103. [Google Scholar] [CrossRef]
- Mahmudov, K.T.; Kopylovich, M.N.; Pombeiro, A.J.L. Coordination chemistry of arylhydrazones of methylene active compounds. Coord. Chem. Rev. 2013, 257, 1244–1281. [Google Scholar] [CrossRef]
- Mahmudov, K.T.; Kopylovich, M.N.; Sabbatini, A.; Drew, M.G.B.; Martins, L.M.D.R.S.; Pettinari, C.; Pombeiro, A.J.L. Cooperative Metal Ligand Assisted E/Z lsomerization and Cyano Activation at Cu-II and Co-II Complexes of Arylhydrazones of Active Methylene Nitriles. Inorg. Chem. 2014, 53, 9946–9958. [Google Scholar] [CrossRef]
- Mahmudov, K.T.; Pombeiro, A.J.L. Resonance-Assisted Hydrogen Bonding as a Driving Force in Synthesis and a Synthon in the Design of Materials. Chem. Eur. J. 2016, 22, 16356–16398. [Google Scholar] [CrossRef]
- Mahmudov, K.T.; Kopylovich, M.N.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L. Non-covalent interactions in the synthesis of coordination compounds: Recent advances. Coord. Chem. Rev. 2017, 345, 54–72. [Google Scholar] [CrossRef]
- Tiago, G.A.O.; Ribeiro, A.P.C.; Mahmudov, K.T.; Guedes da Silva, M.F.C.; Branco, L.C.; Pombeiro, A.J.L. Mononuclear copper(II) complexes of an arylhydrazone of 1H-indene-1,3(2H)-dione as catalysts for the oxidation of 1-phenylethanol in ionic liquid medium. RSC Adv. 2016, 6, 83412–83420. [Google Scholar] [CrossRef]
- Kopylovich, M.N.; Mac Leod, T.C.O.; Haukka, M.; Amanullayeva, G.I.; Mahmudov, K.T.; Pombeiro, A.J.L. Aquasoluble iron(III)-arylhydrazone-beta-diketone complexes: Structure and catalytic activity for the peroxidative oxidation of C-5-C-8 cycloalkanes. J. Inorg. Biochem. 2012, 115, 72–77. [Google Scholar] [CrossRef]
- Jlassi, R.; Ribeiro, A.P.C.; Alegria, E.C.B.A.; Naïli, H.; Tiago, G.A.O.; Rüffer, T.; Lang, H.; Zubkov, F.I.; Pombeiro, A.J.L.; Rekik, W. Copper(II) complexes with an arylhydrazone of methyl 2-cyanoacetate as effective catalysts in the microwave-assisted oxidation of cyclohexane. Inorg. Chim. Acta 2018, 471, 658–663. [Google Scholar] [CrossRef]
- Kustov, L.M.; Kucherov, A.V.; Finashina, E.D. Oxidative dehydrogenation of C-2-C-4 alkanes into alkenes: Conventional catalytic systems and microwave catalysis. Russ. J. Phys. Chem. A 2013, 87, 345–351. [Google Scholar] [CrossRef]
- Kopylovich, M.N.; Nunes, A.C.C.; Mahmudov, K.T.; Haukka, M.; Mac Leod, T.C.O.; Martins, L.M.D.R.S.; Kuznetsov, M.L.; Pombeiro, A.J.L. Complexes of copper(II) with 3-(ortho-substituted phenylhydrazo)pentane-2,4-diones: Syntheses, properties and catalytic activity for cyclohexane oxidation. Dalton Trans. 2011, 40, 2822–2836. [Google Scholar] [CrossRef]
- Tariq, M.; Carvalho, P.J.; Coutinho, J.A.P.; Marrucho, I.M.; Canongia Lopes, J.N.C.; Rebelo, L.P.N. Viscosity of (C-2-C-14) 1-alkyl-3-methylimidazolium bis(trifluoromethylsulfonyl)amide ionic liquids in an extended temperature range. Fluid Phase Equilib. 2011, 301, 22–32. [Google Scholar] [CrossRef]
- Weissermel, K.; Arpe, H.-J. Industrial Organic Chemistry, 2nd ed.; VCH Press: Weinheim, Germany, 1993; ISBN 978-3-52-761919-1. [Google Scholar]
- Di Nicola, C.; Garau, F.; Karabach, Y.Y.; Martins, L.M.D.R.S.; Monari, M.; Pandolfo, L.; Pettinari, C.; Pombeiro, A.J.L. Trinuclear Triangular Copper(II) Clusters. Synthesis, Electrochemical Studies and Catalytic Peroxidative Oxidation of Cycloalkanes. Eur. J. Inorg. Chem. 2009, 666–676. [Google Scholar] [CrossRef]
- Galassi, R.; Simon, O.C.; Burini, A.; Tosi, G.; Conti, C.; Graiff, C.; Martins, N.M.R.; Guedes da Silva, M.F.C.; Pombeiro, A.J.L.; Martins, L.M.D.R.S. Copper(I) and copper(II) metallacycles as catalysts for microwave assisted selective oxidation of cyclohexane. Polyhedron 2017, 134, 143–152. [Google Scholar] [CrossRef]
- Shul’pin, G.B.; Attanasio, D.; Suber, L. Oxidations by a H2O2-VO3−- pyrazine-2-carboxylic acid reagent. Russ. Chem. Bull. 1993, 42, 55–59. [Google Scholar] [CrossRef]
- Nizova, G.V.; Süss-Fink, G.; Shul’pin, G.B. Oxidations by the reagent O2–H2O2–vanadium complex–pyrazine-2-carboxylic acid: Efficient oxygenation of methane and other lower alkanes in acetonitrile. Tetrahedron 1997, 53, 3603–3614. [Google Scholar] [CrossRef]
- Guerreiro, M.C.; Schuchardt, U.; Shul’pin, G.B. Oxidations with the “O2-H2O2-VO3−- pyrazine-2-carboxylic acid” reagent. Russ. Chem. Bull. 1997, 46, 749–754. [Google Scholar] [CrossRef]


| Entry | Oxidant | Catalyst Amount (mol L−1) | Time (min) | Yield % (after PPh3) b | Total TON d | Total TOF e (h−1) | [Alc]/[Keto] f | ||
|---|---|---|---|---|---|---|---|---|---|
| Cyclo-Hexanone | Cyclo-Hexanol | Total c | |||||||
| 1 | H2O2 | 1 × 10−4 | 30 | 0.8 | 4.2 | 5.0 | 230 | 460 | 5.3 |
| 2 | 60 | 3.8 | 5.0 | 8.9 | 409 | 409 | 1.3 | ||
| 3 | 120 | 4.4 | 5.2 | 9.6 | 442 | 221 | 1.2 | ||
| 4 | 180 | 4.7 | 8.1 | 12.8 | 589 | 196 | 1.7 | ||
| 5 | H2O2 | 1 × 10−5 | 30 | 2.3 | 3.2 | 5.4 | 248 × 10 | 497 × 10 | 1.4 |
| 6 | 60 | 2.2 | 4.5 | 6.7 | 308 × 10 | 308 × 10 | 2.0 | ||
| 7 | 120 | 4.4 | 4.7 | 9.1 | 419 × 10 | 209 × 10 | 1.1 | ||
| 8 | 180 | 3.7 | 4.7 | 8.4 | 386 × 10 | 129 × 10 | 1.3 | ||
| 9 | TBHP | 1 × 10−4 | 30 | 4.7 | 1.7 | 6.4 | 294 | 588 | 0.4 |
| 10 | 60 | 8.4 | 2.2 | 10.6 | 488 | 488 | 0.3 | ||
| 11 | 120 | 13.0 | 2.3 | 15.3 | 704 | 352 | 0.2 | ||
| 12 | 180 | 21.7 | 2.8 | 24.6 | 113 × 10 | 377 | 0.1 | ||
| 13 | TBHP | 1 × 10−5 | 30 | 13.5 | 0.0 | 13.5 | 621 × 10 | 124 × 102 | 0.0 |
| 14 | 60 | 20.2 | 0.0 | 20.2 | 929 × 10 | 929 × 10 | 0.0 | ||
| 15 | 120 | 29.6 | 0.0 | 29.6 | 136 × 102 | 681 × 10 | 0.0 | ||
| 16 | 180 | 17.3 | 0.0 | 17.3 | 796 × 10 | 265 × 10 | 0.0 | ||
| 17 g | H2O2 | 1 × 10−5 | 120 | trace | 0.0 | 0.0 | - | - | - |
| 18 g | TBHP | 1 × 10−5 | 120 | trace | 0.0 | 0.0 | - | - | - |
| 19 h | TBHP | 4 × 10−4 | 120 | 1.6 | 3.8 | 5.4 | 49 | 24 | 2.4 |
| Entry | Oxidant | Catalyst Amount (mol L−1) | Time (min) | Yield (%) (after PPh3) b | Total TON d | Total TOF e (h−1) | [Alc]/[Keto] f | ||
|---|---|---|---|---|---|---|---|---|---|
| Cyclo-Hexanone | Cyclo-Hexanol | Total c | |||||||
| 1 | H2O2 | 1 × 10−4 | 30 | 1.1 | 0.3 | 1.4 | 64 | 128 | 0.3 |
| 2 | 60 | 1.0 | 0.5 | 1.5 | 69 | 69 | 0.5 | ||
| 3 | 120 | 1.4 | 0.9 | 2.3 | 106 | 53 | 0.6 | ||
| 4 | 180 | 2.0 | 0.8 | 2.8 | 129 | 43 | 0.4 | ||
| 5 | H2O2 | 1 × 10−5 | 30 | 1.2 | 0.3 | 1.5 | 690 | 138 × 10 | 0.3 |
| 6 | 60 | 3.5 | 1.7 | 5.2 | 239 × 10 | 239 × 10 | 0.5 | ||
| 7 | 120 | 3.9 | 2.2 | 6.1 | 281 × 10 | 140 × 10 | 0.6 | ||
| 8 | 180 | 5.4 | 3.5 | 8.9 | 409 × 10 | 137 × 10 | 0.6 | ||
| 9 | TBHP | 1 × 10−4 | 30 | 17.4 | 0.0 | 17.4 | 800 | 160 × 10 | 0.0 |
| 10 | 60 | 18.4 | 0.0 | 18.4 | 846 | 846 | 0.0 | ||
| 11 | 120 | 20.0 | 0.0 | 20.0 | 920 | 460 | 0.0 | ||
| 12 | 180 | 24.5 | 0.0 | 24.5 | 113 × 10 | 376 | 0.0 | ||
| 13 | TBHP | 1 × 10−5 | 30 | 7.7 | 0.0 | 7.7 | 354 × 10 | 708 × 10 | 0.0 |
| 14 | 60 | 15.2 | 0.0 | 15.2 | 699 × 10 | 699 × 10 | 0.0 | ||
| 15 | 120 | 15.4 | 0.0 | 15.4 | 708 × 10 | 354 × 10 | 0.0 | ||
| 16 | 180 | 11.1 | 0.0 | 11.1 | 511 × 10 | 170 × 10 | 0.0 | ||
| 17 g | H2O2 | 1 × 10−5 | 120 | trace | 0.0 | 0.0 | - | - | - |
| 18 g | TBHP | 1 × 10−5 | 120 | trace | 0.0 | 0.0 | - | - | - |
| Entry | Oxidant | Time (min) | Yield (%) (after PPh3) b | Total TON d | Total TOF e (h−1) | [Alc]/[Keto] f | ||
|---|---|---|---|---|---|---|---|---|
| Cyclohexanone | Cyclohexanol | Total c | ||||||
| [bmim][NTf2] | ||||||||
| 1 | H2O2 | 30 | 3.6 | 5.2 | 8.8 | 405 × 10 | 810 × 10 | 1.4 |
| 2 | 60 | 4.4 | 5.9 | 10.3 | 474 × 10 | 474 × 10 | 1.3 | |
| 3 | 120 | 3.5 | 8.5 | 12.0 | 552 × 10 | 276 × 10 | 2.4 | |
| 4 | 180 | 6.1 | 7.8 | 13.9 | 639 × 10 | 213 × 10 | 1.3 | |
| [hmim][NTf2] | ||||||||
| 5 | H2O2 | 30 | 0.0 | 0.0 | 0.0 | 0 | 0 | n.d. |
| 6 | 60 | 0.0 | 0.0 | 0.0 | 0 | 0 | n.d. | |
| 7 | 120 | 0.11 | 0.10 | 0.21 | 97 | 49 | 0.9 | |
| 8 | 180 | 0.18 | 0.16 | 0.34 | 156 | 52 | 0.9 | |
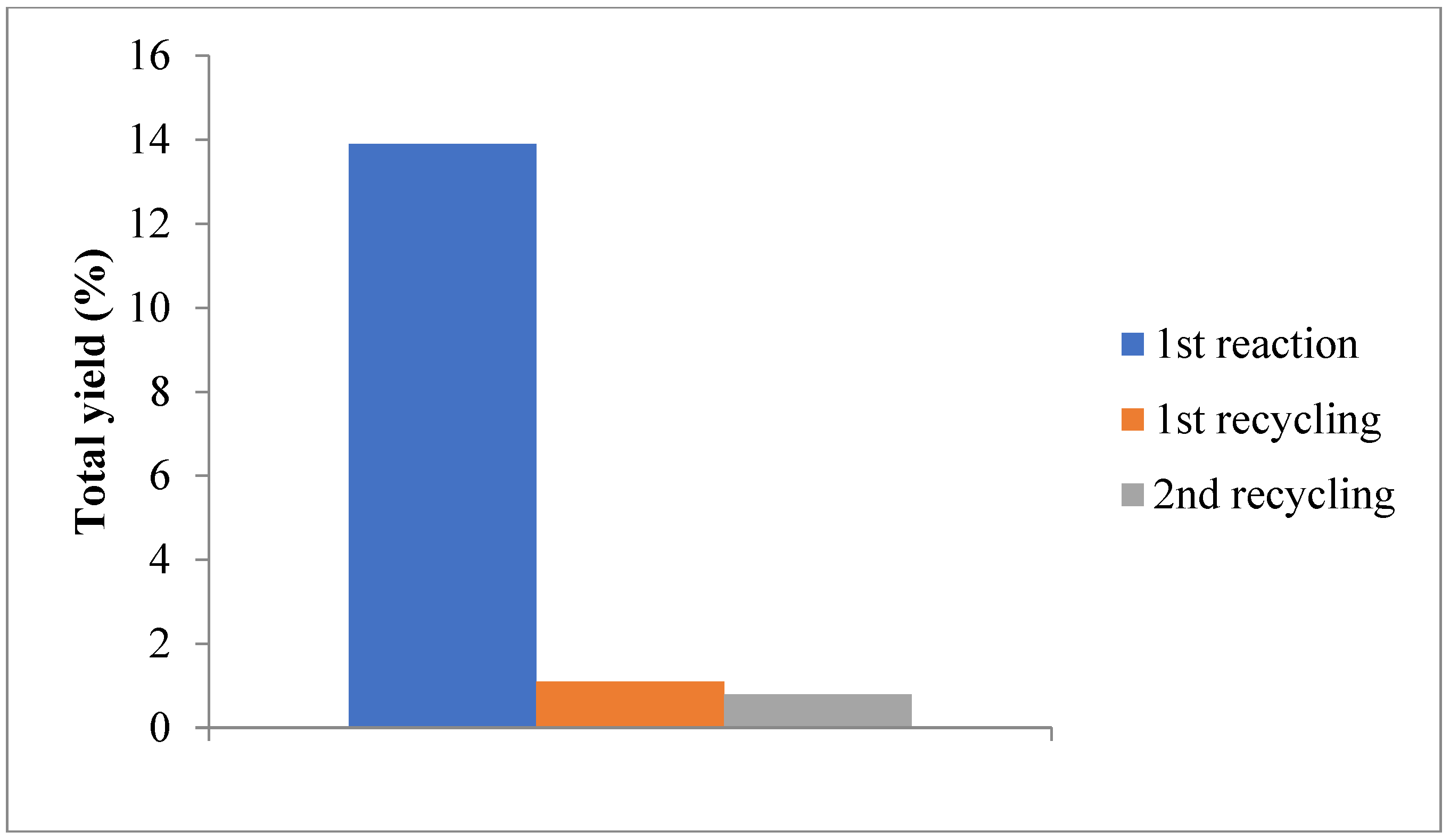
| Entry | Yield (%) (after PPh3) b | Activity (%) | Total TON d | Total TOF (h−1) e | [Alc]/[Keto] f | ||
|---|---|---|---|---|---|---|---|
| Cyclohexanone | Cyclohexanol | Total c | |||||
| 1 | 6.1 | 7.8 | 13.9 | 100 | 639 × 10 | 213 × 10 | 1.3 |
| 2 | 0.3 | 0.8 | 1.1 | 8 | 638 | 213 | 2.7 |
| 3 | 0.5 | 0.3 | 0.8 | 6 | 464 | 155 | 0.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tiago, G.A.O.; Ribeiro, A.P.C.; C. Guedes da Silva, M.F.; Mahmudov, K.T.; Branco, L.C.; Pombeiro, A.J.L. Copper(II) Complexes of Arylhydrazone of 1H-Indene-1,3(2H)-dione as Catalysts for the Oxidation of Cyclohexane in Ionic Liquids. Catalysts 2018, 8, 636. https://doi.org/10.3390/catal8120636
Tiago GAO, Ribeiro APC, C. Guedes da Silva MF, Mahmudov KT, Branco LC, Pombeiro AJL. Copper(II) Complexes of Arylhydrazone of 1H-Indene-1,3(2H)-dione as Catalysts for the Oxidation of Cyclohexane in Ionic Liquids. Catalysts. 2018; 8(12):636. https://doi.org/10.3390/catal8120636
Chicago/Turabian StyleTiago, Gonçalo A. O., Ana P. C. Ribeiro, M. Fátima C. Guedes da Silva, Kamran T. Mahmudov, Luís C. Branco, and Armando J. L. Pombeiro. 2018. "Copper(II) Complexes of Arylhydrazone of 1H-Indene-1,3(2H)-dione as Catalysts for the Oxidation of Cyclohexane in Ionic Liquids" Catalysts 8, no. 12: 636. https://doi.org/10.3390/catal8120636
APA StyleTiago, G. A. O., Ribeiro, A. P. C., C. Guedes da Silva, M. F., Mahmudov, K. T., Branco, L. C., & Pombeiro, A. J. L. (2018). Copper(II) Complexes of Arylhydrazone of 1H-Indene-1,3(2H)-dione as Catalysts for the Oxidation of Cyclohexane in Ionic Liquids. Catalysts, 8(12), 636. https://doi.org/10.3390/catal8120636