Ethylene and propylene are raw materials used in the production of plastic materials, synthetic polymers, and a varied range of chemicals [
1,
2]. Demand for ethylene production is rapidly increasing. In China, ethylene production was 10 Mt in 2007 and 15 Mt in 2011. Propylene is the second largest raw material [
3]. A conventional method for production of olefins is steam cracking. Steam cracking has been the most common method of producing light olefins for 90 years. This method has disadvantages, such as high energy consumption and emissions [
1]. Some other methods are catalytic cracking, fluid catalytic cracking (FCC), methanol-to-olefins (MTO) technology, and coal-to-methanol-to-olefin (CTO) [
4]. Oxidative dehydrogenation of alkanes includes partial oxidation, oxidative dehydrogenation, and ammoxidation (suggested as an alternative) [
5]. Disadvantages of the steam cracking method are low energy efficiency and low selectivity of the desired product. Among the proposed methods, the oxidative dehydrogenation reaction is exothermic, with side products being easily separable. The oxidative dehydrogenation method has not been used in industry [
6]. The thermodynamic limitation, the rapid formation of coke, and the high energy consumption are the common problems of conventional processes. Oxidative dehydrogenation of C
2H
6 with CO
2 as oxidant can be attractive from an industrial and environmental perspective. From an environmental point of view, the use of carbon dioxide as an oxidant reduces the effects of greenhouse gases. The use of a suitable catalyst can overcome the thermodynamic and kinetic barriers of carbon dioxide activation. So far, the appropriate catalysts are H-ZSM-5 supported Cr-catalyst, Cr/SBA-15, Cr
2O
3/ZrO
2, SiO
2, Al
2O
3, and TiO
2 supported and unsupported Cr catalysts, Ni–Nb–O and Ca doped ThO
2 [
7]. HZSM-5 is a microporous, crystalline aluminosilicate zeolite with a regular structure [
8]. Zeolites have been used as catalysts in many chemical processes, especially hydrocarbon conversion [
9]. Also, properties such as high uniformity, controllable acidic properties, and strong electrostatic field within its cages have attracted much attention [
10]. Early theories regarding zeolite showed that the electrostatic field of zeolites leads to their better activity, because catalytic activity increases with the electrostatic field within their cages [
11]. By replacing organic cations, which induce electrostatic field gradients within the cages, the activity could be developed [
12].
To obtain a high reaction rate, many alternatives have been investigated by researchers, such as plasma technology, applying an external electric field and sono-processing. Applying an external direct current (DC) electric field consumes lower energy compared to plasma [
13]. Deren and Mania investigated the role of an external DC electric field in oxidation of carbon monoxide to carbon dioxide on a NiO catalyst and presented a relation between the catalytic performance and electric property of the catalyst surface. Activation energy in the external electric field declined almost about 20 kJ/mole, and energy bands were curved, leading to an increase in the catalytic activity [
14]. Andres et al. expressed that the electric field acts as a catalyst in chemical reactions. Ab initio SCF calculations were performed on the fluoride exchange reaction. The results showed that strong electric fields decreased the reaction’s activation energy [
15]. Sekine et al. studied degradation of ethanol over Pt/CeO
2 catalyst in electric field at 423 K. Thermal analyses indicated that about 90–95% of input power was used in the endothermic reaction [
16]. Sekine et al. carried out ethanol steam reformation, dissociation of ethanol, water gas shift, and methane steam reformation in an external DC electric field where the common reaction could barely be activated with conventional methods. In the electric field, feed conversion was drastically increased, and the activation energy was reduced. Feed conversion was drastically enhanced with the DC electric field, and activation energies for these reactions were decreased when utilizing an electric field, and the reaction temperature decreased by about 150 K, as well [
17]. Sekine et al. investigated methane steam reforming via metal catalysts supported on CeO
2 and Ce
xZr
1−xO
2 at 423 K. The conversion of CH
4 increased the external electric field, especially for Ce
xZr
1−xO
2 due to the lattice oxygen of Ce
xZr
1−xO
2 [
18]. Tanaka et al. studied methane oxidative coupling over SrLa
2O
3 catalysts at 423 K in the DC external electric field. Electrical conductivity had a key role in the reaction. The highest yield for C
2 (49% selectivity) was achieved with 2.7 W electrical power [
19]. Oshima et al. studied the role of input electric current, loading of Pt, and the support properties on the catalytic methane steam reforming in an external electric field at 423 K over CeO
2, Ce
xZr
1−xO
2 and a mixture of CeO
2 and ZrO
2, Al
2O
3 or SiO
2. The activity increased with input electrical current. Pt/Ce
xZr
1−xO
2 solid had the highest catalytic activity (methane conversion = 40.6% at 535.1 K) [
20]. Oshima et al. studied methane oxidative coupling with carbon dioxide in the presence of an electric field through La-ZrO
2. 5 mol % La–ZrO
2 had the highest catalytic performance, which is attributed to the synergy between La, structure of ZrO
2, and the external electric field [
21]. Oshima et al. studied catalytic reverse water gas shift reaction in the electric field at 423 K. Pt/La-ZrO
2 resulted in the highest yield (40%) [
22]. Catalytic water gas shifts for hydrogen production was reported by Sekine et al. at 423–873 K under external electric field over Pt/La–ZrO
2. Activation energy decreased drastically and the redox mechanism in the lattice oxygen of the surface had a crucial effect in water-gas shift reaction (WGS) reaction in the DC electric field [
23]. Methane dry reforming was studied by Yabe et al. over different transition metal-supported ZrO
2 under electric field. 1 wt. % Ni/10 mol % La-ZrO
2 had high activity at 423 K. The electric power consumption was related to the catalytic activity and reaction rate [
24]. Yabe et al. investigated methane oxidative coupling by carbon dioxide as oxidant in the electric field at 423 K over different perovskite oxide catalysts. La
0.7Ca
0.3AlO
3−δ had high activity (7.4% C
2 yield at 348 K). The input electrical current was related to the conversion of methane [
25]. To determine the mechanism for the steam reforming reaction of CH
4 in the electric field, operando- diffuse reflectance infrared Fourier transform spectroscopy was employed to study CH
4 dissociative adsorption. The results showed that CH
4 was dissociated to CO, CO
2 when CD
4 and D
2O were provided instead of CH
4 and H
2O. The proton collision activated methane at low temperatures in the presence of the DC electric field [
26]. Ogo et al. reported methane oxidative coupling and ethane oxidative dehydrogenation using different polyoxometalate-supported CeO
2 at 423 K under external electric field. Tetrabutylammonium salt-supported CeO
2 catalysts had high catalytic activity for these two reactions at 3 mA and 423 K. FTIR and XRD analyses showed that the structure of Tetrabutylammonium salt-supported CeO
2 was transferred to Ce
2(WO
4)
3/CeO
2 after applying an electric field, and it was a proper active site for these two reactions [
27]. The oxidative cracking of LPG was investigated in the external DC electric field. CrHZSM-5 had more catalytic activity compared to HZSM-5 and NaZSM-5. According to Faradaic numbers, the reaction progressed electrocatalytically. The best catalytic activity was achieved at 625 °C and 12 mA (olefin yield = 42.58% and LPG conversion = 92.12%) [
28].
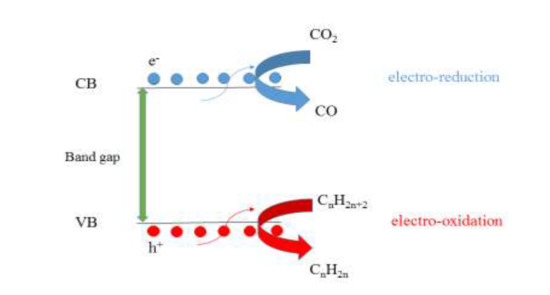
In this research, a novel catalytic reaction system was investigated for oxidative dehydrogenation of Liquefied Petroleum Gas (LPG) with CO2 in a DC external electric field over transition metals supported on HZSM-5. Due to the synergy effect of the electric field and electrostatic field gradient in the support catalyst, there is the possibility to improve the catalytic performance. This study is the first report of ethylene and propylene production with the synergetic effect between transition metals impregnated on HZSM-5 and an external electric field. We studied the role of a DC electric field on the HZSM-5 impregnated with transition metal oxides to obtain high catalytic activity. The beneficial use of an external electric field depends on the electronic properties of the catalyst as a semiconductor. These properties include the electrical conductivity, dielectric constant, Fermi surface and band gap of the catalyst. In an effort to explain the relation between catalytic activity and semiconductor property, the modified HZSM-5 with transition metal oxide was characterized by XRD, FTIR, SEM, NH3-TPD, H2-TPR, BET, TGA, UV-visible, Fermi measurement, Hall and Impedance measurements.