1. Introduction
With the continuously growing demand for global technology chips, whether this is because of the development of science and technology or due to the issue of energy, they have received considerable attention. Through a careful study of the root cause for this, we found that the energy demand has also increased alongside the continuous improvement of semiconductor process capabilities (the feature size has continued to shrink). For example, the chip application of 5G communication systems reveals that they still use a multilayer board as the main circuit board design because of the need for a large amount of data transmission. The materials used need to possess suitable electromagnetic properties, high resistance to environmental degradation and oxidation, and appropriate hardness [
1,
2,
3,
4,
5]. Dielectric materials are currently a good choice for producing high-frequency components since they have characteristics such as a higher dielectric constant (ε
r), which can reduce the dimensions of the components; high-quality factors (Q
f), which can improve the energy of stored electromagnetic waves; and a temperature coefficient of the resonance frequency (τ
f) approaching zero, which can improve the thermal stability of the components. For example, when the base version of a filter has the above three characteristics, this will result in effective downsizing, lower dielectric loss, steeper filtering, and high stability unaffected by the external ambient temperature [
6,
7,
8,
9,
10,
11].
Ceramic systems based on MgTiO
3 have always received considerable attention and have been used in a wide range of applications, for example, as passive components in communication systems such as dielectric resonators (DRs), filters, and antennas. Therefore, it is quite important to enhance the dielectric performance of MgTiO
3 (ε
r~17, a Q
f~160,000 GHz, τ
f~−51 ppm/°C) [
12,
13,
14,
15,
16]. It has an ilmenite-type structure and can be represented by the general formula ABO
3. We previously tried to partially replace the position of A with a divalent element; the results of that experiment showed that (Mg
0.95A
2+0.05)TiO
3 (A = Co, Ni, and Zn) has better dielectric performances than MgTiO
3. For example, when A = cobalt and Mg
2+ are replaced with Co
2+ of 0.95 moles, the Mg
0.95Co
0.05TiO
3 (hereafter MCT) ceramic maintains the ilmenite-type structure and possesses a relatively good quality factor and dielectric performances with an ε
r of 16.8, a Q
f of 230,000 GHz, and a τ
f of −54 ppm/°C [
17]. However, because the MCT ceramic has negative temperature characteristics and the sintering temperature is too high, it cannot be widely used in the production of microwave components.
In our previous studies, to enhance the thermal stability of MCT ceramics, we tried to add perovskite ceramics with high positive temperature characteristics, such as CaTiO3, Ca0.6La0.8/3TiO3, and Ca0.61Nd0.78/3TiO3. When Ca0.6La0.8/3TiO3 (CLT) was added to Mg0.95Co0.05TiO3 to form the ceramic system of (1–x)Mg0.95Co0.05TiO3-xCa0.6La0.8/3TiO3 (MCT-CLT), it could effectively compensate for the negative temperature characteristics of the MCT. When the ratio of MCT to CLT was 9:1, a ceramic composition of 0.9Mg0.95Co0.05TiO3-0.1Ca0.6La0.8/3TiO3 (9MCT-CLT) was obtained. This composition has high practicality. 9MCT–CLT ceramics possess an εr of 21.8, a Qf value of 131,000 GHz, and a τf value of −15.5 ppm/°C. However, the sintering temperature of this composition is still as high as 1350 °C/4 h. Therefore, it is quite important to lower this composition’s sintering temperature to reduce manufacturing costs and enhance its practical value.
In general, the main considerations are the sintering temperature and thermal calculation to investigate the factors of phase forming. These two factors usually involve the cost of the manufacturing process. Therefore, a considerable amount of research has been invested in reducing the sintering temperature, and most of the methods discussed are as follows [
18,
19,
20,
21]:
From the relevant literature, the sol–gel process is quite complicated, and the second method, i.e., lowering the particle size of the starting powders, will increase the cost of the fabrication. Therefore, considering both the cost and time of fabrication, we chose to conduct experiments using liquid-phase sintering with the addition of other low-melting-point materials.
On the other hand, a single-sintering aid method has been employed to modify the MCT-CLT dielectric characteristics. In a 9MCT-CLT system with doped V
2O
5 [
22], the crystal structures of Mg
0.95Co
0.05TiO
3 and Ca
0.6La
0.8/3TiO
3 were found to be an ilmenite-type structure and a pseudo-cubic perovskite crystal. A significant change was not observed in the XRD patterns of the 9MCT-CLT ceramics with 0.25 wt% V
2O
5 sintered at 1200 °C–1300 °C. The sintering temperature of 9MCT–CLT ceramics with doped V
2O
5 can be decreased by about 100 °C. In a 9MCT-CLT system with doped B
2O
3 [
23], the peaks of the X-ray diffraction (XRD) pattern indicated the presence of Mg
0.95Co
0.05TiO
3 (JCPDS #06-0494) as the main crystalline phase, in association with Ca
0.6La
0.8/3TiO
3 (JCPDS #22-0153) and Mg
0.95Co
0.05Ti
2O
5 as minor phases. B
2O
3 additions could effectively lower the sintering temperature and improve the radio-frequency dielectric properties of the 9MCT-CLT ceramics.
In this study, we investigated the microstructure and radio-frequency dielectric characteristics of 9MCT-CLT ceramics to enhance their phase-forming capacity using multiple sintering aids, ZnO-B2O3 (ZB). X-ray diffraction (XRD), scanning electron microscopy (SEM), and energy-dispersive X-ray spectroscopy (EDS) analyses were also employed to study the microstructure grain boundary and compositions of the ceramic system.
2. Experimental Procedure
Mg0.95Co0.05TiO3 and Ca0.6La0.8/3TiO3 compositions were produced with mixed oxide solid-state reactions using high-purity chemical powders: magnesium oxide (MgO), cobalt oxide (CoO), calcium carbonate (CaCO3), lanthanum oxide (La2O3), and titanium dioxide (TiO2). The magnesium oxide needed to be dried at 600 °C for 2 h due to its hygroscopicity. The compositions were mixed using stoichiometry: Mg0.95Co0.05TiO3 and Ca0.6La0.8/3TiO3. They were then subjected to ball grinding in distilled water with a ball-grinding medium for 24 h. All mixed compositions were parched in the kiln and pre-phased (calcine) at 1100 °C for 4 h in a high-temperature furnace. Then, the pre-phased reagents were formulated again according to the chemical molar ratio of 0.9Mg0.95Co0.05TiO3-0.1Ca0.6La0.8/3TiO3 with different contents of sintering additives (ZnO-B2O3) and ball-ground into a fine powder for 24 h. Then, polyvinyl alcohol (PVA 500; Showa, Tokyo, Japan) as a binder was added to the calcined powder, and after complete granulation uniformity was achieved, it was screened with a 100-mesh screen and pressed with a pressure of 200 MPa to form cylinders with a height of 0.5 cm and a diameter of 1.1 cm. The sintering temperatures (STs) of the cylinders were set to 1100 °C–1300 °C for 4 h in the air. The rate of temperature rises and falls for the sintering equipment was set to 10 degrees per minute to ensure consistency for all specimens.
The crystallization-phase observations of the pre-phased powder and the mixed compositions were carried out via a Siemens D5000 X-Ray Powder diffractometer (XRD, Munich, Germany) with Cu-Kα radiation (at 40 kV and 40 mA). The XRD analysis of the sample was performed at a scan speed of 2 degrees/minute and a step size of 0.06 degrees. The range of the 2 theta was set to 20–60 degrees. The XRD data analysis and the lattice constants calculation were performed using the DIFFRAC (version 2.7) software. Scanning electron microscopy (SEM; Philips XL-40FEG, Eindhoven, The Netherlands) was employed to observe the structure generations of the specimen surfaces, and an energy-dispersive X-ray spectrometer (EDS) was utilized to show the composition of the mixed phases. The apparent densities of the mixed compositions were measured using the Archimedes method. The ε
r and Q
f at radio frequencies were measured using the Hakki–Coleman dielectric resonator method [
24,
25].
The test set was mainly composed of a vector network analyzer (HP8757D) and HP8350B sweep oscillator connections. The thermal coefficients (τ
f) of the mixed compositions were assessed by keeping the DR inside a temperature-controlled oven with a temperature range of 20 °C to 80 °C. The following formula was utilized to obtain τ
f (ppm/°C): by tracking the shift in the resonance peak frequency of the TE
011 mode as the temperature slowly changed from 20 °C to 80 °C, the τ
f could be calculated from the following equation:
where
f1 and
f2 represent the resonant frequencies at T
1 = 20 °C and T
2 = 80 °C, respectively.
3. Results and Discussion
Figure 1 presents the XRD analysis obtained after adding 1 wt% multiple sintering aids (ZnO-B
2O
3) to 9MCT-CLT ceramics at different sintering temperatures (1100–1200 °C) for 4 h. The 9MCT-CLT ceramics can be regarded as multi-phase systems, in which the MCT ceramic (JCPDS #06-0494) with an ilmenite structure as the main phase coexists with the CLT ceramic (JCPDS #22-0153) with a pseudo-cubic perovskite structure as the minor phase. From the figure, in addition to the above multi-phase compositions, very few numbers of second phases (MgTi
2O
5) appeared. Regarding the cause of the second phase, the particle size of the initial material was generally uneven, which increased the probability of crystallization secondary nucleation; the sintering temperature was too high; the grain boundary movement rate increased; and the local uneven liquid phase was stored. According to the relevant literature [
26,
27], when MgO reacts with a mole ratio of 1:1 TiO
2, MgTi
2O
5 usually forms an intermediate phase in the material system and can hardly be eliminated. As shown in
Table 1, the MCT phase ratio in the ceramics increased due to the increased grain boundary motion with the increasing sintering temperature. However, the ratio of the MgTi
2O
5 phase decreased, which might have been due to the influence of the liquid-phase sintering, resulting in the insufficient temperature of its phase formation.
Figure 2 presents the results of the XRD analysis obtained after adding multiple additives (ZnO-B
2O
3) to the 9MCT-CLT ceramics; the different additives did not influence the phase formation of the 9MCT-CLT ceramics. Only a few second phases remained in the system with the different amounts of multiple additives (ZnO-B
2O
3). As mentioned above, the existence of the inferior MgTi
2O
5 phase may have impacted the dielectric performances of the 9MCT-CLT ceramics [
28]. It also can be seen from the references that the sintering temperature of the MgTi
2O
5 phase was close to 1450 °C, which means that the higher the sintering temperature of the 9MCT-CLT ceramics, the greater the influence of the MgTi
2O
5 phase. In addition, due to the increase in the multiple additive content, the sintering temperature decreased, so the MgTi
2O
5 phase ratio decreased with the increase in ZB content. The lattice constants obtained after adding multiple additives (ZnO-B
2O
3) and the contents of the 9MCT-CLT ceramics are presented in
Table 2. All samples can be regarded as multi-phase systems, and according to the JCPDS card, MgTiO
3 has a hexagonal structure with the following lattice constants: a = b = 5.054 (Å), c = 13.898 (Å) (JCPDS #06-0494). The results showed that when cobalt (Co
2+) of 0.05 mole replaced the magnesium (Mg
2+) in MgTiO
3, it caused some slight changes in the lattice constant of MgTiO
3. This is mainly because the ion radius of cobalt (0.82 Å) is comparatively larger than that of magnesium, so when cobalt is added to MgTiO
3, it causes local lattice distortion. When CLT was blended with MCT, no obvious influence on the lattice constants of MCT could be found. In addition, the mixed phases forming in the 9MCT-CLT ceramic system might have represented the structural differences, and the average ionic radii of Ca
2+ (1.06 Å) and La
3+ (1.22 Å) were larger than those of Mg
2+ (0.78 Å) and Co
2+ (0.82 Å). This confirms the existence of a multi-phase ceramic system for MCT-CLT, with MCT as the primary crystalline phase and CLT as the minor phase. These results are in agreement with the XRD analysis, and the different additives and additions did not influence the lattice constants of the 9MCT-CLT ceramics.
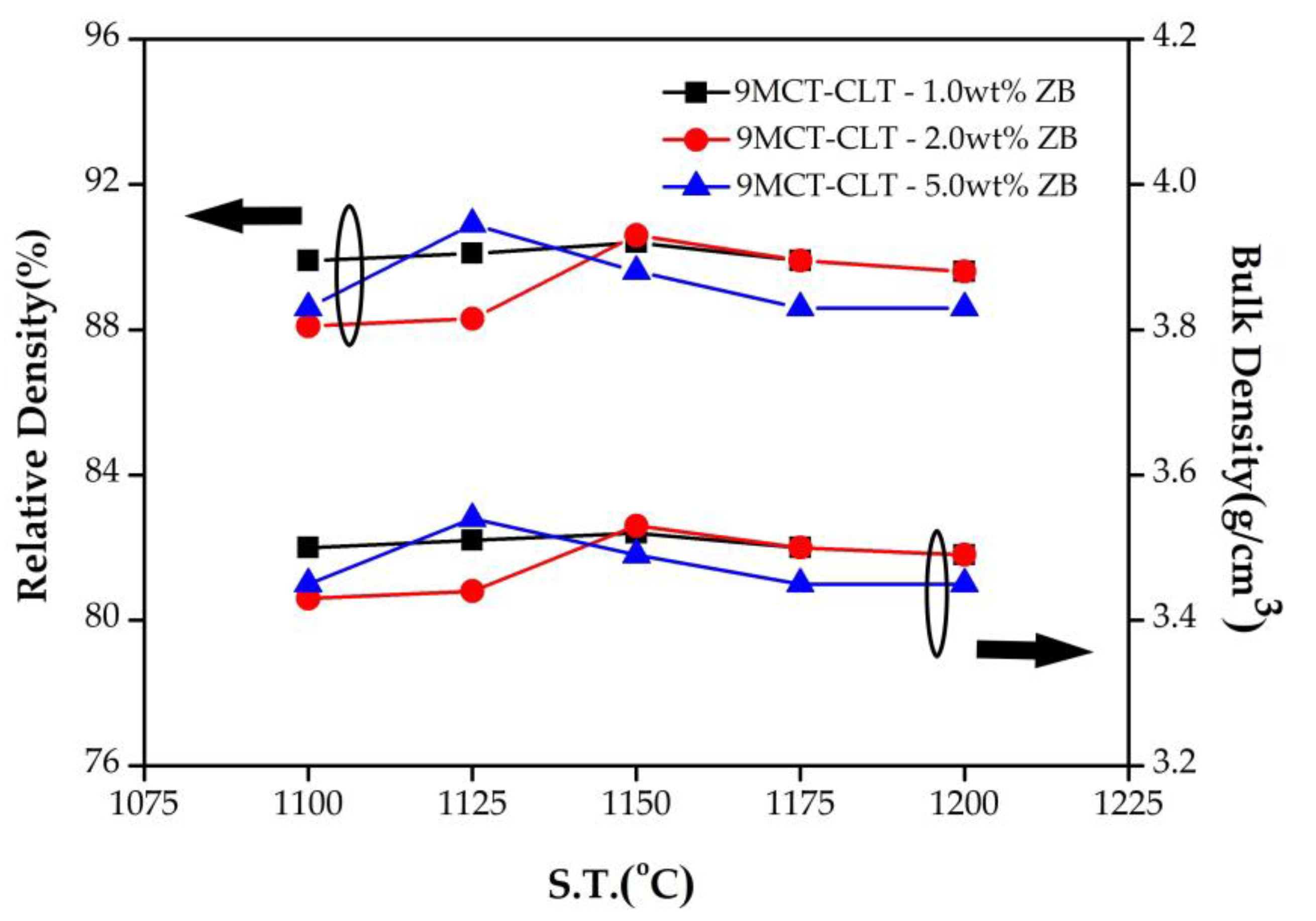
Figure 3 presents the results for the relative density and bulk density of 9MCT-CLT ceramics with multiple additives in various amounts that were sintered at 1100–1200 °C/4 h. As can be seen in the figure, there will be a period of density increase when the low temperature of the multiple additives reaches its optimal temperature point. The reason for the increase in density is that the thermal drive energy is relatively low at low temperatures, and it is not sufficient to connect and expand the neck between the grains so that the grains grow and eliminate the porosity. When the sintering temperature rises, the energy rises, meaning that the grain boundary moves better and it is easy for the small grains to dissolve so that the grain growth is more uniform and the pores decrease with the increase in the sintering temperature, and densification occurs. In addition, when multiple additives are added to the 9MCT-CLT ceramics to form liquid-phase sintering, this is more conducive to small-grain dissolution, an increase in the wetting boundary movement rate, and grain rearrangement to achieve densification. This can be compared with the SEM in
Figure 4 for the 9MCT-CLT ceramics with multiple sintering aids (ZnO-B
2O
3) that were sintered at 1100 °C/4 h, where the grains were smaller, more pores appeared, and the density was lower; when the sintering temperature rose to 1175 °C, the grain size was consistent, there were fewer pores, and the density was higher. Furthermore, when multiple sintering aids (ZnO-B
2O
3) were used, the sintering temperature could be effectively reduced to 1175 °C, and the optimum density could be obtained by using 3.8 g/cm
3 with 0.5 wt% ZB. It can be concluded that liquid-phase sintering occurs when using different additives, and when the additive exceeds 0.5 wt%, it will cause a slight decrease in the density. Generally, in cases of liquid-phase sintering, using appropriate amounts of sintering aids can effectively improve the phase-forming capacity and reduce the sintering temperature. However, when the sintering aid content is excessive, the grains grow excessively, and with a larger particle size, the rearrangement will be subject to greater resistance and there will be a small number of pores in the boundary, meaning it is not easy to discharge and other factors will affect the change in the density and radio-frequency dielectric performances. Furthermore, grains of 9MCT-CLT ceramics can be roughly divided into three shapes, as shown in
Figure 4b. The EDS results of each grain are summarized in
Table 3. The different grains were identified as follows: spot A is (Mg
0.95Co
0.05)TiO
3; spot B is Ca
0.6La
0.8/3TiO
3; spot C is (Mg
0.95Co
0.05)Ti
2O
5. The EDS results are consistent with the XRD analysis, verifying that a 9MCT-CLT ceramic is a three-phase coexistence system.
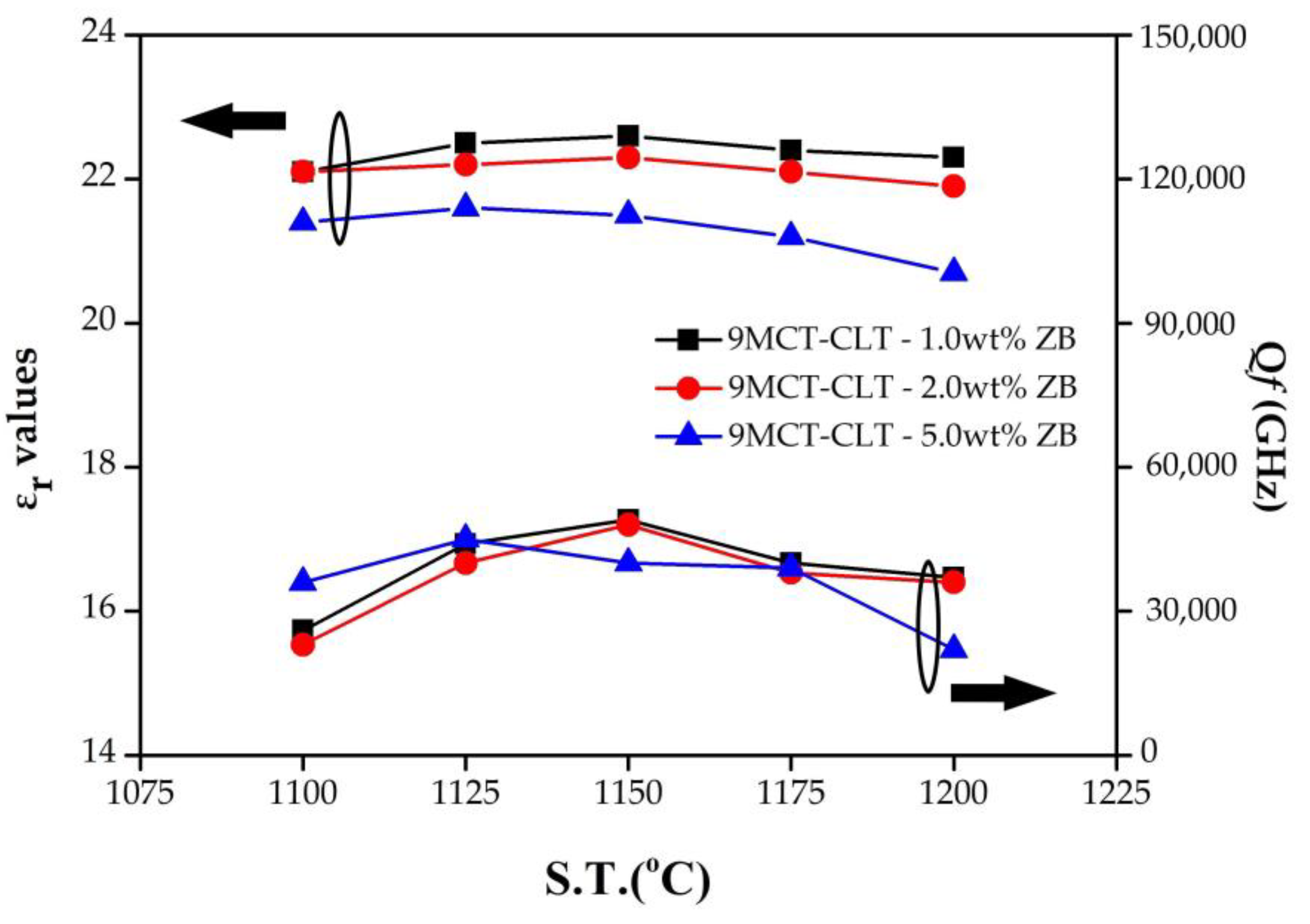
Figure 5 presents the results of the dielectric constant (ε
r) and quality factor (Q
f) of 9MCT-CLT ceramics with multiple additives in varying amounts that were sintered at 1100–1200 °C/4 h. As can be seen in
Figure 5, the ε
r values were positively correlated with the sintering temperatures, because the temperature rise caused the grain to continue to grow, the pore size to shrink (porosity decrease), and the structure of the 9MCT-CLT ceramics to reach densification. This implies that the density is dependent on the dielectric constant. In addition, it can also be seen from the SEM that when the temperature rises, the grains will be closer and the pores will gradually decrease, thereby increasing the ε
r. When adding 0.5 wt% ZB, the resulting ε
r was 22.5 at 1175 °C.
According to the literature, the factors that impact microwave dielectric loss are the sum of intrinsic loss and extrinsic loss. The intrinsic dielectric losses are the losses in the perfect crystals which depend on the crystal structure and can be described by the interaction of the phonon system with the ac electric field. Extrinsic losses are associated with imperfections in the crystal lattice such as the second phase, oxygen vacancy, grain size, and porosity [
29,
30,
31,
32]. In general, the relationship between the microstructure of the material and the sintering temperature affects the ability of phase formation and characteristics. Liquid-phase sintering using glass additives is one of the most effective and least expensive methods of reducing sintering temperature. At lower sintering temperatures, the Q
f product is lower because of the smaller grains (phase not formed). When the sintering temperature rises to 1175 °C, the grain size is consistent (phase formed), the pores on the microstructure are reduced, and the liquid-phase sintering of the sintering aids is affected, so that the microstructure achieves the most densification, and the optimal Q
f product can be obtained. The Q
f of 9MCT-CLT ceramics with 0.5 wt% ZB at 1175 °C/4 h is 57,000 (GHz). When the sintering temperature continues to rise, the Q
f will have a downward trend, which may be due to factors such as excessive grain growth, the additive content, or the second phase. In addition, 9MCT-CLT ceramics with different additives possess a lower Q
f compared to the pure type. This can also be explained because of the pores, the grain boundary phase, the liquid-phase sintering, and the second phase of MgTi
2O
5.
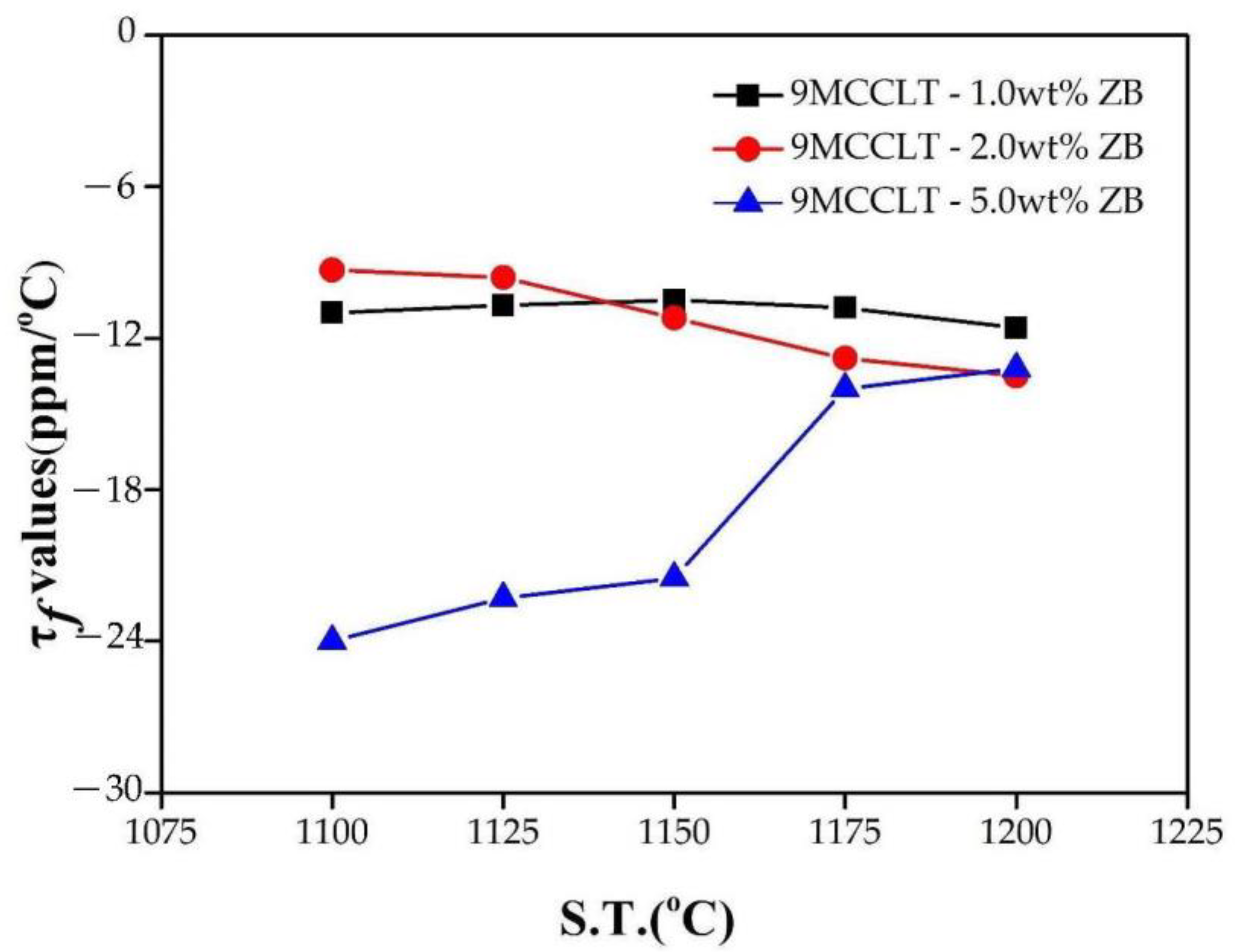
Figure 6 presents the temperature coefficients of the resonant frequency τ
f for 9MCT-CLT ceramics with different additives in varying amounts that were sintered at 1100~1200 °C/4 h. In general, the τ
f depends on the composition and the second phase of the material. The dielectric performances of individual phases are shown in
Table 4. As can be seen in
Table 5, the τ
f did not change significantly with the addition of different amounts of additives. The τ
f of 9MCT-CLT ceramics with 0.5 wt% ZB at 1175 °C/4 h was −13.6 ppm/°C. The sintering temperature of B
2O
3- and V
2O
5-doped 9MCT-CLT ceramics can only be reduced about by 125 °C. However, despite the decrease of 200 degrees in sintering temperature, the dielectric properties were sacrificed, especially with a reduction of more than half in the Q
f product. Even so, the dielectric characteristics of 9MCT-CLT ceramics with 0.5 wt% ZB are sufficient to be used in general component substrates and can be improved both in terms of size miniaturization and signal transmission.