Aqueous Solution Spray Drying Preparations of Binary Amorphous Solid Dispersions
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results
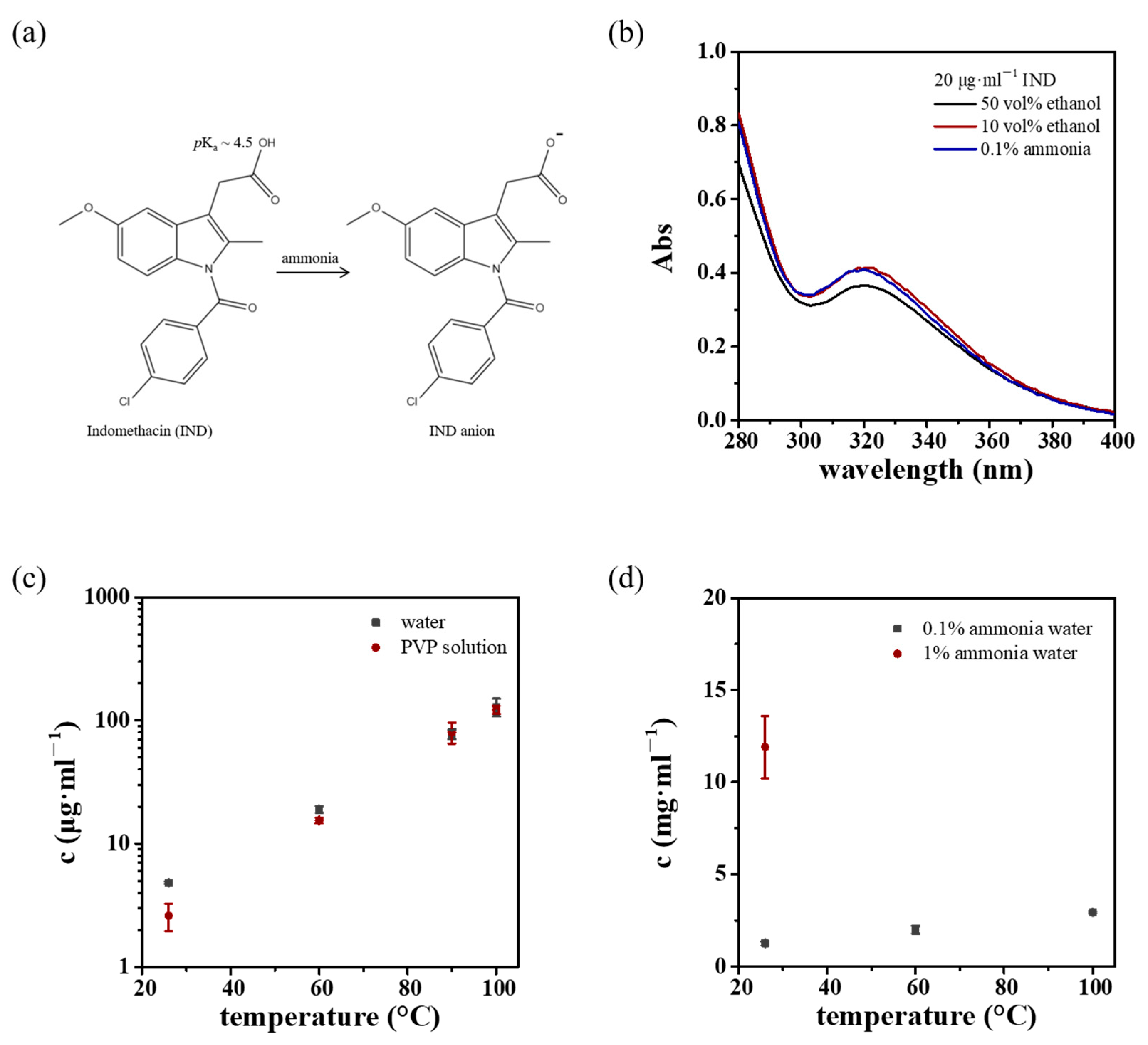
3.1. Aqueous Solution Spray Drying of IND
3.1.1. IND Solubility
3.1.2. High-Temperature Aqueous Solution Spray Drying
3.1.3. Ammonia-Assisted Aqueous Solution Spray Drying
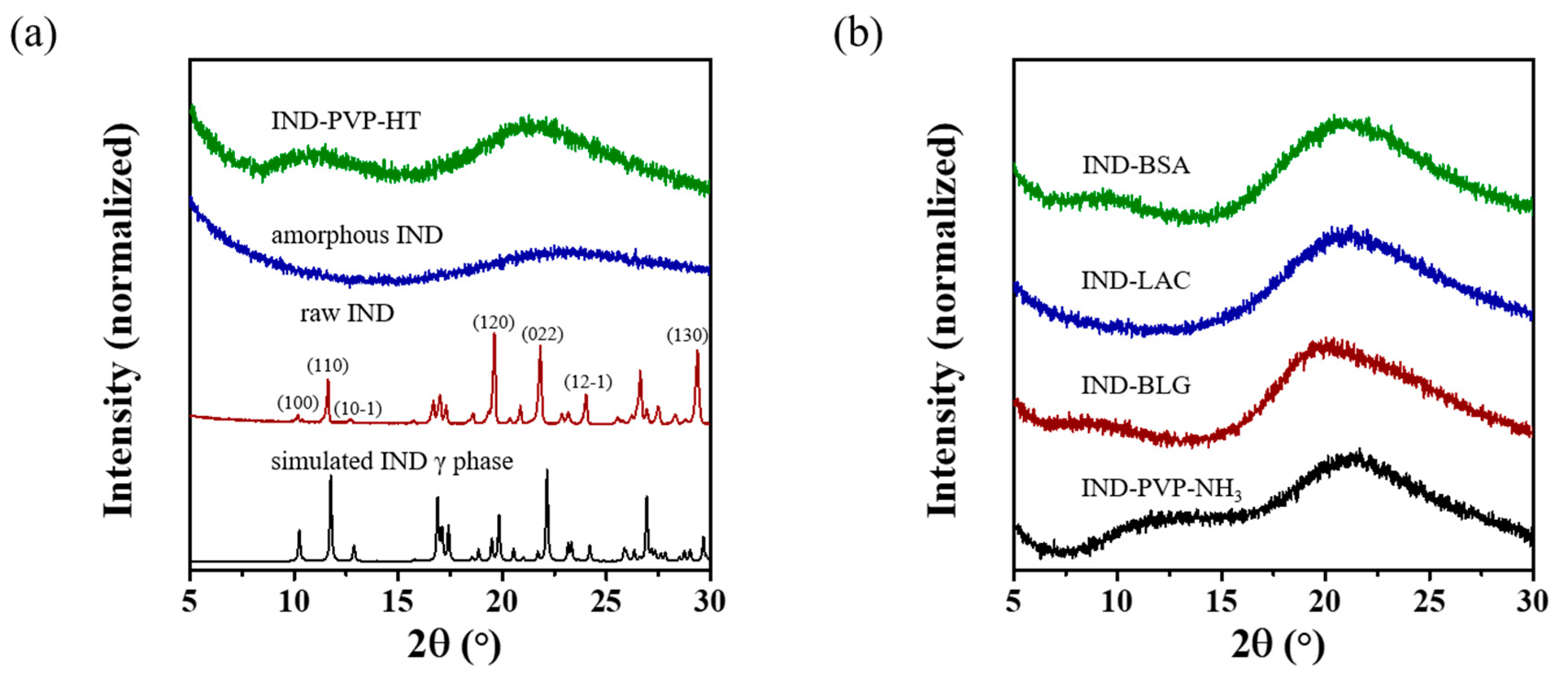
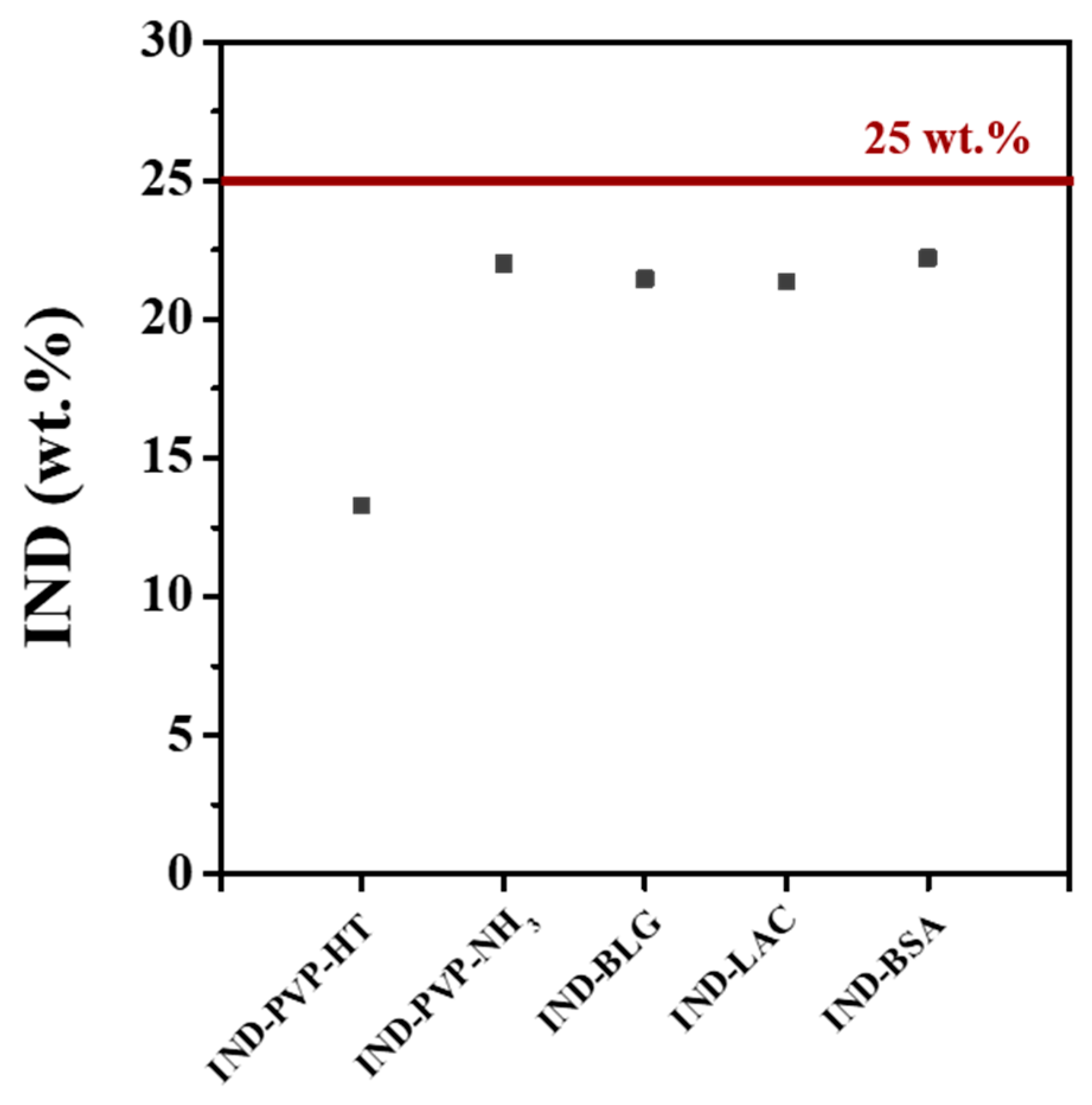
3.2. Drug Loadings and Morphology of IND ASDs
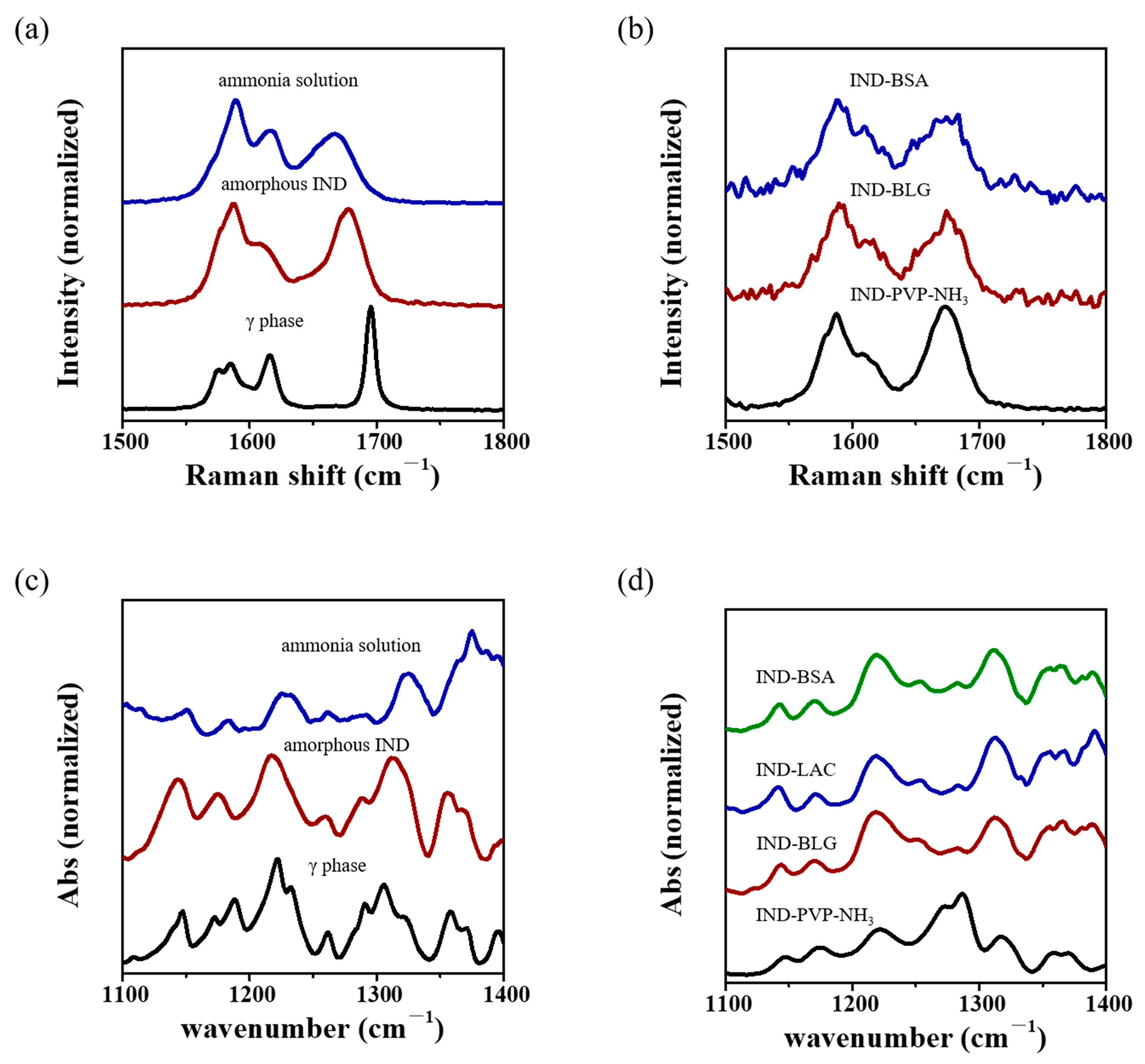
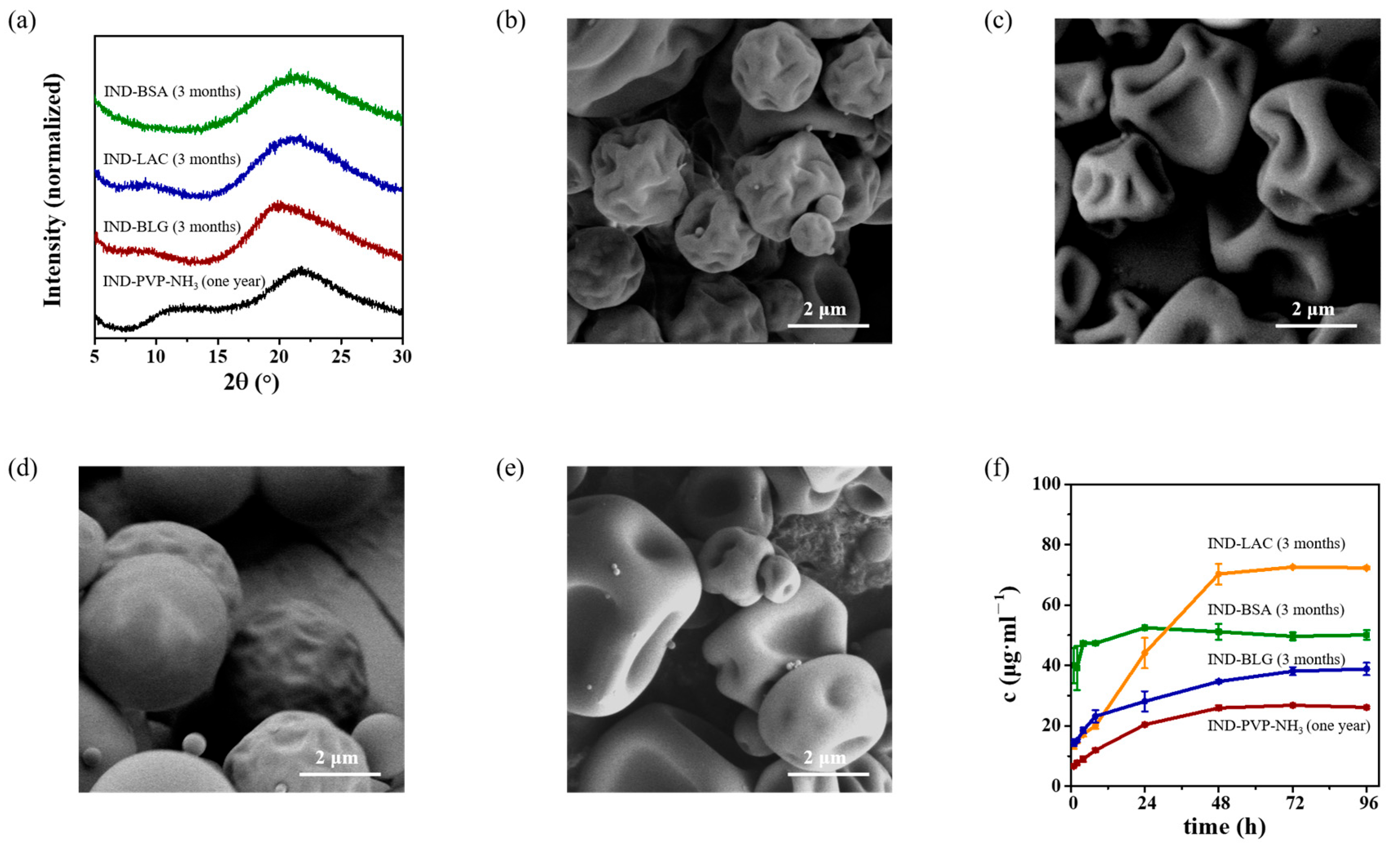
3.3. Spectroscopy Analysis of IND ASDs
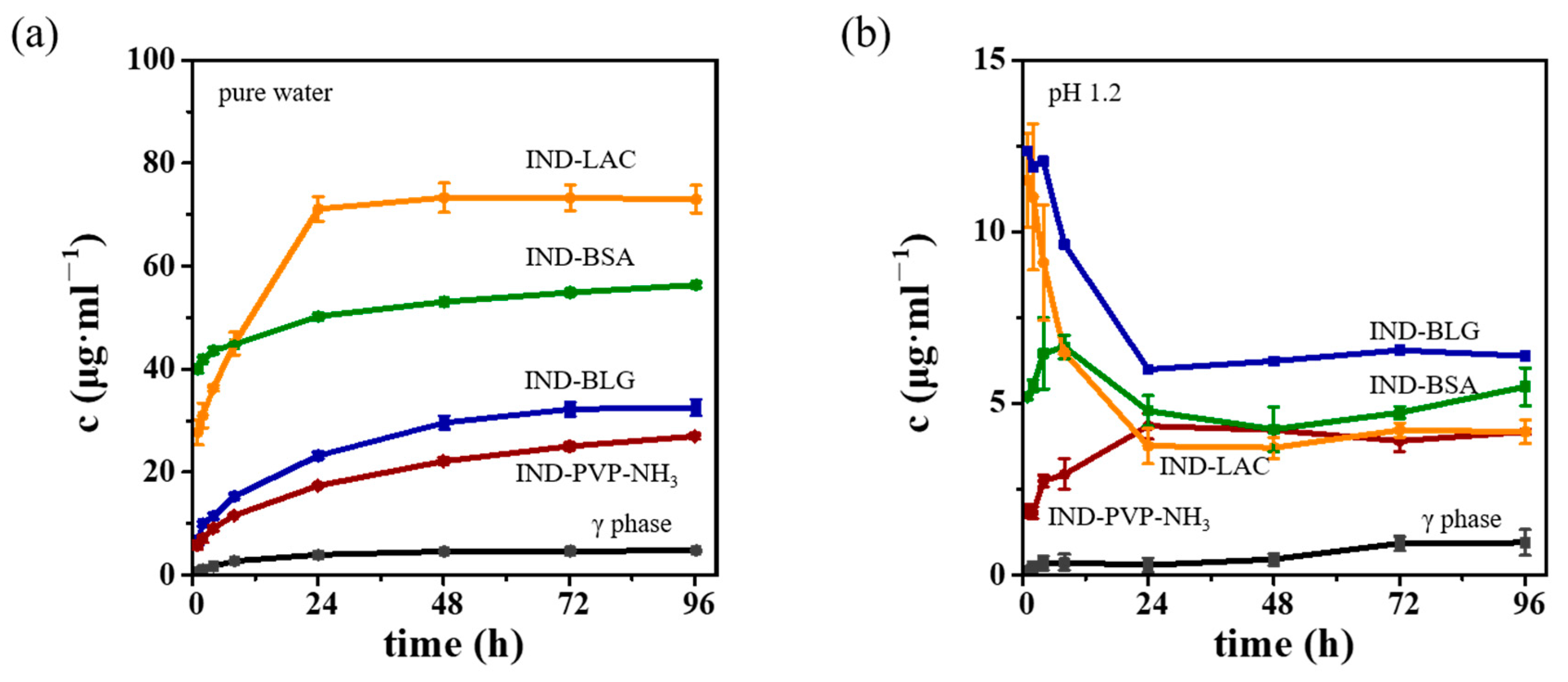
3.4. Dissolution Profiles of IND ASDs
3.5. Storage Stability of IND ASDs
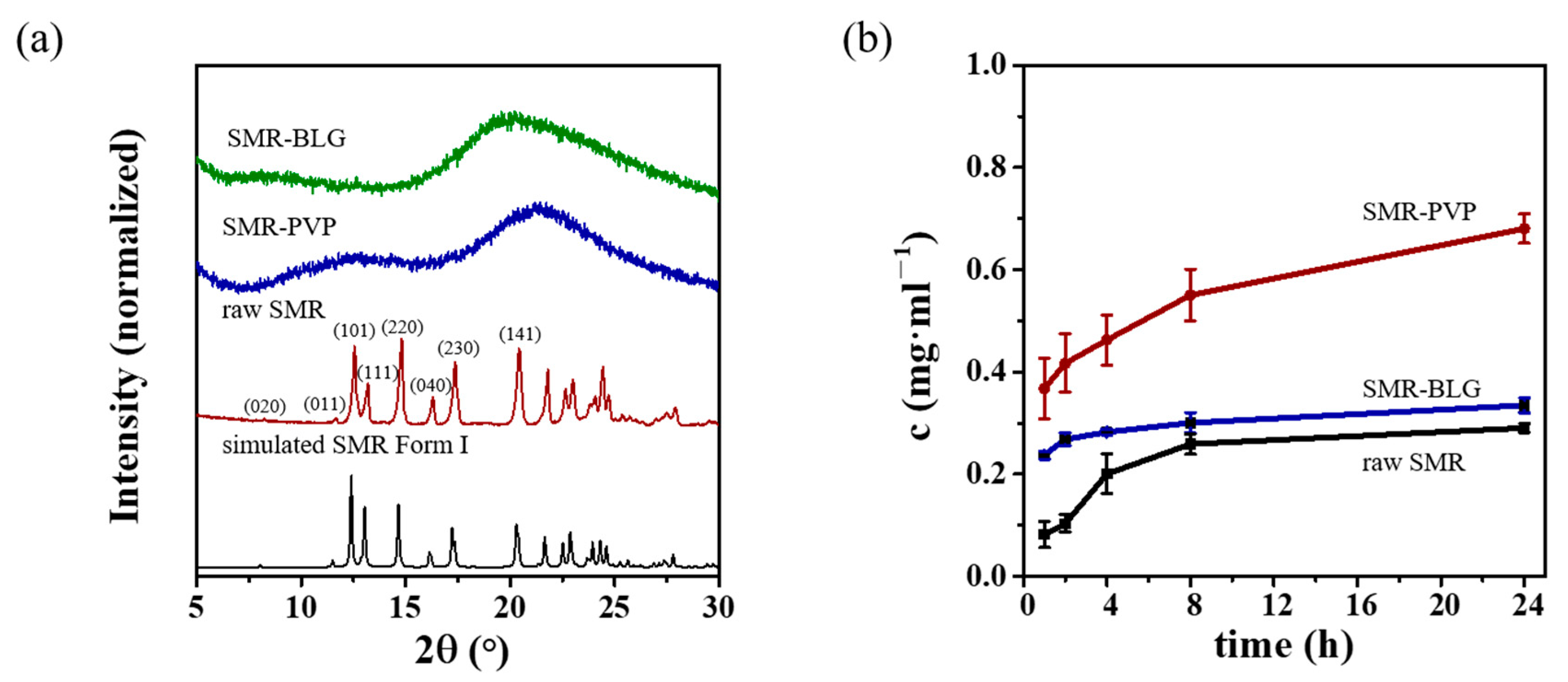
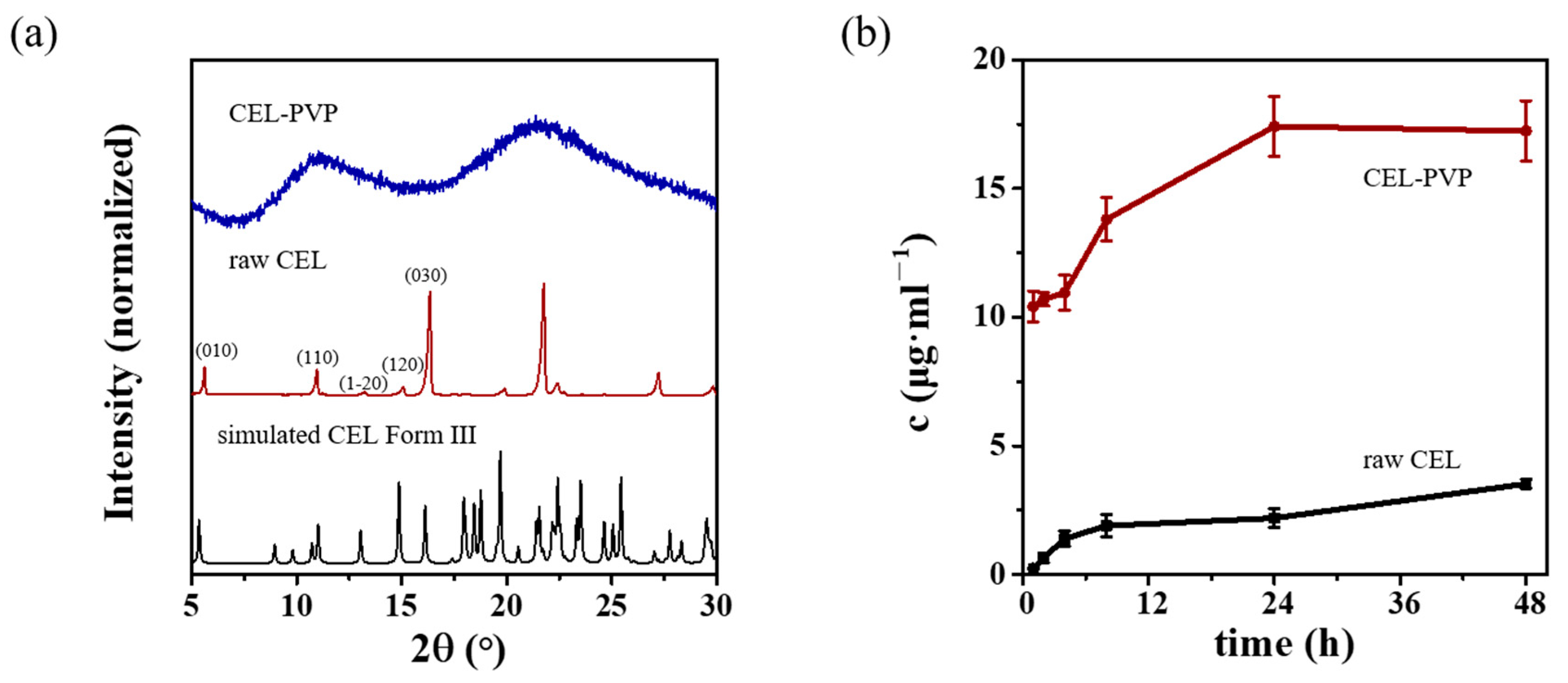
3.6. Method Verification by Additional APIs
4. Discussion
4.1. Feasibility of Aqueous Solution Spray Drying for ASDs Preparation
4.2. Disadvantages and Prospect of Aqueous Solution Spray Drying
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| IND | Indomethacin |
| SMR | Sulfamerazine |
| CEL | Celecoxib |
| PVP | Polyvinylpyrrolidone |
| BLG | β-Lactoglobulin |
| LAC | Lactalbumin Hydrolysate |
| BSA | Bovine Serum Albumin |
| ASDs | Amorphous Solid Dispersions |
| IND-polymer | ASDs of IND (25 %) and polymer |
| PXRD | Powder X-Ray Diffraction |
| SEM | Scanning Electron Microscope |
References
- Bhujbal, S.V.; Mitra, B.; Jain, U.; Gong, Y.; Agrawal, A.; Karki, S.; Taylor, L.S.; Kumar, S.; Zhou, Q. Pharmaceutical amorphous solid dispersion: A review of manufacturing strategies. Acta Pharm. Sin. B 2021, 11, 2505–2536. [Google Scholar] [CrossRef] [PubMed]
- Pisay, M.; Padya, S.; Mutalik, S.; Koteshwara, K.B. Stability challenges of amorphous solid dispersions of drugs: A critical review on mechanistic aspects. Crit. Rev. Ther. Drug Carrier Syst. 2024, 41, 45–94. [Google Scholar] [CrossRef] [PubMed]
- Moseson, D.E.; Tran, T.B.; Karunakaran, B.; Ambardekar, R.; Hiew, T.N. Trends in amorphous solid dispersion drug products approved by the U.S. Food and Drug Administration between 2012 and 2023. Int. J. Pharm. X 2024, 7, 100259. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yan, A.; Sun, T.; Wang, X.; Sun, W.; Pan, B. Self-nanomicellizing solid dispersion: A promising platform for oral drug delivery. Colloids Surf. B 2024, 241, 114057. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Van den Mooter, G. Spray drying formulation of amorphous solid dispersions. Adv. Drug Deliv. Rev. 2016, 100, 27–50. [Google Scholar] [CrossRef]
- Patil, H.; Vemula, S.K.; Narala, S.; Lakkala, P.; Munnangi, S.R.; Narala, N.; Jara, M.O.; Williams, R.O.; Terefe, H.; Repka, M.A. Hot-melt extrusion: From theory to application in pharmaceutical formulation where are we now? AAPS PharmSciTech 2024, 25, 37. [Google Scholar] [CrossRef]
- Ramachandran, R.P.; Nadimi, M.; Cenkowski, S.; Paliwal, J. Advancement and Innovations in Drying of Biopharmaceuticals, Nutraceuticals, and Functional Foods. Food Eng. Rev. 2024, 16, 540–566. [Google Scholar] [CrossRef]
- Dedroog, S.; Adriaensens, P.; Van den Mooter, G. Gaining Insight into the Role of the Solvent during Spray Drying of Amorphous Solid Dispersions by Studying Evaporation Kinetics. Mol. Pharm. 2022, 19, 1604–1618. [Google Scholar] [CrossRef]
- Li, N.; Cape, J.L.; Mankani, B.R.; Zemlyanov, D.Y.; Shepard, K.B.; Morgen, M.M.; Taylor, L.S. Water-induced phase separation of spray-dried amorphous solid dispersions. Mol. Pharm. 2020, 17, 4004–4017. [Google Scholar] [CrossRef]
- Shepard, K.B.; Dower, A.M.; Ekdahl, A.M.; Morgen, M.M.; Baumann, J.M.; Vodak, D.T. Solvent-assisted secondary drying of spray-dried polymers. Pharm. Res. 2020, 37, 156. [Google Scholar] [CrossRef]
- Luebbert, C.; Real, D.; Sadowski, G. Choosing Appropriate Solvents for ASD Preparation. Mol. Pharm. 2018, 15, 5397–5409. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, X.; Foderà, V.; Larsson, P.; Schaal, Z.; Bergström, C.A.S.; Löbmann, K.; Kabedev, A. Analysis of stabilization mechanisms in β-lactoglobulin-based amorphous solid dispersions by experimental and computational approaches. Eur. J. Pharm. Sci. 2024, 192, 106639. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, X.; Tozzetti, M.; Arnous, A.; Leng, D.; Foderà, V.; Löbmann, K. Investigating the influence of protein secondary structure on the dissolution behavior of β-lactoglobulin-based amorphous solid dispersions. Int. J. Pharm. 2024, 653, 123887. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, X.; Jasiukenaite, I.; Löbmann, K. β-Lactoglobulin-based amorphous solid dispersions: A graphical review on the state-of-the-art. Eur. J. Pharm. Biopharm. 2024, 202, 114396. [Google Scholar] [CrossRef]
- Ren, S.; Nian, F.; Chen, X.; Xue, R.; Chen, F. Routes of theophylline monohydrate dehydration process proposed by mid-frequency raman difference spectra. J. Pharm. Sci. 2023, 112, 2863–2868. [Google Scholar] [CrossRef]
- Mazák, K.; Noszál, B. Advances in microspeciation of drugs and biomolecules: Species-specific concentrations, acid-base properties and related parameters. J. Pharm. Biomed. Anal. 2016, 130, 390–403. [Google Scholar] [CrossRef]
- Avdeef, A.; Sugano, K. Salt Solubility and Disproportionation—Uses and Limitations of Equations for pHmax and the In-silico Prediction of pHmax. J. Pharm. Sci. 2022, 111, 225–246. [Google Scholar] [CrossRef]
- Yan, H.; Zhong, X.; Liu, Y. Improving the Solubility, Stability, and Bioavailability of Albendazole through Synthetic Salts. Molecules 2024, 29, 3571. [Google Scholar] [CrossRef]
- Di Martino, P.; Scoppa, M.; Joiris, E.; Palmieri, G.F.; Andres, C.; Pourcelot, Y.; Martelli, S. The spray drying of acetazolamide as method to modify crystal properties and to improve compression behaviour. Int. J. Pharm. 2001, 213, 209–221. [Google Scholar] [CrossRef]
- Lu, H. Fabrication and characterization of porous silver powder prepared by spray drying and calcining technology. Powder Technol. 2010, 203, 176–179. [Google Scholar] [CrossRef]
- Long, B.; Walker, G.M.; Ryan, K.M.; Padrela, L. Controlling polymorphism of carbamazepine nanoparticles in a continuous supercritical-CO2-assisted spray drying process. Cryst. Growth Des. 2019, 19, 3755–3767. [Google Scholar] [CrossRef]
- Nuchuchua, O.; Every, H.A.; Jiskoot, W. Critical processing parameters of carbon dioxide spray drying for the production of dried protein formulations: A study with myoglobin. Eur. J. Pharm. Biopharm. 2016, 103, 200–209. [Google Scholar] [CrossRef]
- Zhuo, X.; Ochner, J.; Leng, D.; Foderà, V.; Löbmann, K. Exploring the effect of protein secondary structure on the solid state and physical stability of protein-based amorphous solid dispersions. Eur. J. Pharm. Biopharm. 2024, 198, 114274. [Google Scholar] [CrossRef]
- Hou, A.; Li, L.; Huang, Y.; Singh, V.; Zhu, C.; Pan, X.; Quan, G.; Wu, C. Fragmented particles containing octreotide acetate prepared by spray drying technique for dry powder inhalation. Drug Delivery Transl. Res. 2018, 8, 693–701. [Google Scholar] [CrossRef]
- Zhang, X.; Yue, X.; Cui, Y.; Zhao, Z.; Huang, Y.; Cai, S.; Wang, G.; Wang, W.; Hugh, S.; Pan, X.; et al. A systematic safety evaluation of nanoporous mannitol material as a dry-powder inhalation carrier system. J. Pharm. Sci. 2020, 109, 1692–1702. [Google Scholar] [CrossRef]
- Anita, C.; Munira, M.; Mural, Q.; Shaily, L. Topical nanocarriers for management of Rheumatoid Arthritis: A review. Biomed. Pharmacother. 2021, 141, 111880. [Google Scholar] [CrossRef]
- Gogic, A.; Vesovic, M.; Nedeljkovic, N.; Nikolic, M.; Jurisevic, M.; Zdravkovic, N.; Zivanovic, A. Indomethacin Derivatives as Potential Anticancer Agents-Daybreak of New Epoch. ChemistrySelect 2024, 9, e202402050. [Google Scholar] [CrossRef]
- Leung, H.W.; Copley, R.C.B.; Lampronti, G.I.; Day, S.J.; Saunders, L.K.; Johnstone, D.N.; Midgley, P.A. From formulation to structure: 3D electron diffraction for the structure solution of a new indomethacin polymorph from an amorphous solid dispersion. IUCrJ 2024, 11, 744–748. [Google Scholar] [CrossRef]
- Keßler, L.; Mishra, R.; Hietala, S.; Lammens, M.; Peltonen, L.; Rades, T.; van Veen, B.; Juppo, A.; Laaksonen, T.; Strachan, C.; et al. Amorphous solid dispersions of amphiphilic polymer excipients and indomethacin prepared by hot melt extrusion. Eur. J. Pharm. Sci. 2025, 204, 106960. [Google Scholar] [CrossRef]
- Autzen Virtanen, A.; Myślińska, M.; Healy, A.M.; Power, E.; Madi, A.; Sivén, M. The challenge of downstream processing of spray dried amorphous solid dispersions into minitablets designed for the paediatric population—A sustainable product development approach. Eur. J. Pharm. Sci. 2024, 196, 106752. [Google Scholar] [CrossRef]
- Zhuo, X.; Sener, Z.; Kabedev, A.; Zhao, M.; Arnous, A.; Leng, D.; Foderà, V.; Löbmann, K. Mechanisms of drug solubility enhancement induced by β-lactoglobulin-based amorphous solid dispersions. Mol. Pharm. 2023, 20, 5206–5213. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Mooter, G.V.D. The influence of hydrogen bonding between different crystallization tendency drugs and PVPVA on the stability of amorphous solid dispersions. Int. J. Pharm. 2023, 646, 123440. [Google Scholar] [CrossRef] [PubMed]
- Hermeling, M.; Nueboldt, C.; Heumann, R.; Hoheisel, W.; Breitkreutz, J. Nano-Dry-Melting: A novel technology for manufacturing of pharmaceutical amorphous solid dispersions. Pharmaceutics 2022, 14, 2145. [Google Scholar] [CrossRef]
- Kabedev, A.; Zhuo, X.; Leng, D.; Fodera, V.; Zhao, M.; Larsson, P.; Bergstrom, C.A.S.; Lobmann, K. Stabilizing mechanisms of beta-lactoglobulin in amorphous solid dispersions of indomethacin. Mol. Pharm. 2022, 19, 3922–3933. [Google Scholar] [CrossRef]
- Dedroog, S.; Boel, E.; Kindts, C.; Appeltans, B.; Van den Mooter, G. The underestimated contribution of the solvent to the phase behavior of highly drug loaded amorphous solid dispersions. Int. J. Pharm. 2021, 609, 121201. [Google Scholar] [CrossRef]
- Holzapfel, K.; Rades, T.; Leopold, C.S. Co-amorphous systems consisting of indomethacin and the chiral co-former tryptophan: Solid-state properties and molecular mobilities. Int. J. Pharm. 2023, 636, 122840. [Google Scholar] [CrossRef]
- Rathod, V.; Gajera, B.; Pinninti, A.; Mohammed, I.A.; Dave, R.H. Strategizing spray drying process optimization for the manufacture of redispersible indomethacin nanoparticles using quality-by-design principles. AAPS PharmSciTech 2023, 24, 133. [Google Scholar] [CrossRef]
- Méndez Cañellas, F.; Geertman, R.; Tajber, L.; Padrela, L. Sorbitol-coated indomethacin and naproxen particles produced by supercritical CO2-assisted spray drying. J. Supercrit. Fluids 2023, 199, 105969. [Google Scholar] [CrossRef]
- Li, J.; Zordan, C.; Ponce, S.; Lu, X. Impact of swelling of spray dried dispersions in dissolution media on their dissolution: An investigation based on UV imaging. J. Pharm. Sci. 2022, 111, 1761–1769. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Saipul Bahri, N.S.N.; Rahmatika, A.M.; Cao, K.L.A.; Hirano, T.; Ogi, T. Rapid indomethacin release from porous pectin particles as a colon-targeted drug delivery system. ACS Appl. Bio Mater. 2023, 6, 2725–2737. [Google Scholar] [CrossRef]
- Larkin, P.J.; Wasylyk, J.; Raglione, M. Application of low- and mid-frequency Raman spectroscopy to characterize the amorphous-crystalline transformation of indomethacin. Appl. Spectrosc. 2015, 69, 1217–1228. [Google Scholar] [CrossRef] [PubMed]
- Shen, P.; Zhang, C.; Hu, E.; Pang, Z.; Gao, Y.; Qian, S.; Zhang, J.; Wei, Y.; Heng, W. Gelation switch of polyamorphic indomethacin depending on the thermal procedure. Eur. J. Pharm. Biopharm. 2022, 177, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Dunn, C.; Perrier, J.; Khadra, I.; Wilson, C.G.; Halbert, G.W. Topography of simulated intestinal equilibrium solubility. Mol. Pharm. 2019, 16, 1890–1905. [Google Scholar] [CrossRef] [PubMed]
- Shakeel, F.; Alanazi, F.K.; Alsarra, I.A.; Haq, N. Solubility prediction of indomethacin in PEG 400+water mixtures at various temperatures. J. Mol. Liq. 2013, 188, 28–32. [Google Scholar] [CrossRef]
- Alsalhi, M.S.; Chan, K.L.A. Amino acid hydrotropes to increase the solubility of indomethacin and carbamazepine in aqueous solution. Int. J. Pharm. 2022, 617, 121591. [Google Scholar] [CrossRef]
- Bolten, D.; Lietzow, R.; Türk, M. Solubility of ibuprofen, phytosterol, salicylic acid, and naproxen in aqueous solutions. Chem. Eng. Technol. 2013, 36, 426–434. [Google Scholar] [CrossRef]
- Xin, X.; Xu, Y.; Shi, H.; Liu, X. General method to stabilize mesophilic proteins in hyperthermal water. iScience 2021, 24, 102503. [Google Scholar] [CrossRef]
- Jiménez-Rodríguez, A.; Heredia-Olea, E.; Barba-Dávila, B.A.; Gutiérrez-Uribe, J.A.; Antunes-Ricardo, M. Polysaccharides from Agave salmiana bagasse improves the storage stability and the cellular uptake of indomethacin nanoemulsions. Food Bioprod. Process. 2021, 127, 114–127. [Google Scholar] [CrossRef]
- Li, Y.; Yu, J.; Tan, X.; Yu, L. Surface Mobility of Amorphous Indomethacin Containing Moisture and a Surfactant: A Concentration–Temperature Superposition Principle. Mol. Pharm. 2022, 19, 2962–2970. [Google Scholar] [CrossRef]
- Thayumanasundaram, S.; Venkatesan, T.R.; Ousset, A.; Van Hollebeke, K.; Aerts, L.; Wübbenhorst, M.; Van den Mooter, G. Complementarity of mDSC, DMA, and DRS Techniques in the Study of Tg and Sub-Tg Transitions in Amorphous Solids: PVPVA, Indomethacin, and Amorphous Solid Dispersions Based on Indomethacin/PVPVA. Mol. Pharm. 2022, 19, 2299–2315. [Google Scholar] [CrossRef]
- Bērziņš, K.; Fraser-Miller, S.J.; Rades, T.; Gordon, K.C. Low-frequency Raman spectroscopic study on compression-induced destabilization in melt-quenched amorphous celecoxib. Mol. Pharm. 2019, 16, 3678–3686. [Google Scholar] [CrossRef]
- Bērziņš, K.r.; Sutton, J.J.; Fraser-Miller, S.J.; Rades, T.; Korter, T.M.; Gordon, K.C. Solving the computational puzzle: Toward a pragmatic pathway for modeling low-energy vibrational modes of pharmaceutical crystals. Cryst. Growth Des. 2020, 20, 6947–6955. [Google Scholar] [CrossRef]
- Bērziņš, K.r.; Fraser-Miller, S.J.; Gordon, K.C. A new frontier for nondestructive spatial analysis of pharmaceutical solid dosage forms: Spatially offset low-frequency raman spectroscopy. Anal. Chem. 2021, 93, 3698–3705. [Google Scholar] [CrossRef]
- Bērziņš, K.r.; Fraser-Miller, S.J.; Gordon, K.C. Pseudo-3D subsurface imaging of pharmaceutical solid dosage forms using micro-spatially offset low-frequency Raman spectroscopy. Anal. Chem. 2021, 93, 8986–8993. [Google Scholar] [CrossRef]
- Jantschke, A.; Pinkas, I.; Hirsch, A.; Elad, N.; Schertel, A.; Addadi, L.; Weiner, S. Anhydrous β-guanine crystals in a marine dinoflagellate: Structure and suggested function. J. Struct. Biol. 2019, 207, 12–20. [Google Scholar] [CrossRef]
- Ali, H.R.H.; Alhalaweh, A.; Velaga, S.P. Vibrational spectroscopic investigation of polymorphs and cocrystals of indomethacin. Drug Dev. Ind. Pharm. 2013, 39, 625–634. [Google Scholar] [CrossRef]
- Badawi, H.M.; Förner, W. Analysis of the molecular structure and vibrational spectra of the indole based analgesic drug indomethacin. Spectrochim. Acta Part A 2014, 123, 447–454. [Google Scholar] [CrossRef]
- Aitipamula, S.; Chow, P.S.; Tan, R.B.H. The solvates of sulfamerazine: Structural, thermochemical, and desolvation studies. CrystEngComm 2012, 14, 691–699. [Google Scholar] [CrossRef]
- Hossain, G.M.G. Sulfamerazine tetrahydrofuran monosolvate. IUCrData 2016, 1, x160596. [Google Scholar] [CrossRef]
- Zhang, G.G.Z.; Gu, C.; Zell, M.T.; Burkhardt, R.T.; Munson, E.J.; Grant, D.J.W. Crystallization and Transitions of Sulfamerazine Polymorphs. J. Pharm. Sci. 2002, 91, 1089–1100. [Google Scholar] [CrossRef]
- Hyun, S.-M.; Lee, B.J.; Abuzar, S.M.; Lee, S.; Joo, Y.; Hong, S.-H.; Kang, H.; Kwon, K.-A.; Velaga, S.; Hwang, S.-J. Preparation, characterization, and evaluation of celecoxib eutectic mixtures with adipic acid/saccharin for improvement of wettability and dissolution rate. Int. J. Pharm. 2019, 554, 61–71. [Google Scholar] [CrossRef]
- Feng, P.; Yang, W.; Xu, D.; Ma, M.; Guo, Y.; Jing, Z. Characteristics, mechanisms and measurement methods of dissolution and deposition of inorganic salts in sub-/supercritical water. Water Res. 2022, 225, 119167. [Google Scholar] [CrossRef]

| Form | Solutions | Ref. |
|---|---|---|
| ASDs | A mixed solution of water, acetic acid, and ethanol with a v/v% ratio of 10%: 5%: 85%. Briefly, indomethacin (1.5 g) was dissolved in 425 mL ethanol, β-lactoglobulin (3.5 g) was dissolved in 50 mL water and 25 mL acetic acid. | [31] |
| Drug and poly (vinylpyrrolidone-co-vinyl acetate) 64 were dissolved in dichloromethane with a 10% w/v solid concentration. | [32] | |
| Nano-Dry-Melting process. An aqueous drug slurry in a wet-stirred media mill was nano-milled and spray-dried. | [33] | |
| A mixed solution of acetic acid (5%) and ethanol (95%). Indomethacin was dissolved in 190 mL ethanol, and β-lactoglobulin was dissolved in 10 mL acetic acid with a total of 1 g solid. | [34] | |
| Indomethacin (2% w/v) and poly(vinylpyrrolidone-co-vinyl acetate) 64 were dissolved in one of seven solvents (methanol, ethanol, 2-propanol, acetone, dichloromethane, acetonitrile, ethyl acetate). | [35] | |
| co-amorphous | Indomethacin and tryptophan were dissolved in a mixture of acetone and water (70:30 v/v) with a 1.0% or 0.5% w/v solid concentration. | [36] |
| crystals | Aqueous nanosuspension prepared using the wet media milling technique. | [37] |
| partially crystalline | Supercritical CO2-assisted spray drying. 400 mg indomethacin and 400 mg sorbitol were dissolved in 20 mL methanol. | [38] |
| not mentioned | 10 g of indomethacin and polymers were dissolved in 300 mL acetone (hydroxypropyl cellulose, hydroxypropyl methylcellulose acetate succinate) or methanol (polyvinylpyrrolidone). | [39] |
| loading | 0.1 wt.% indomethacin, 1.0 wt.% pectin and 0.5–12 wt.% CaCO3 aqueous dispersions. | [40] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, W.; Ke, J.; Liu, K.; Xue, R.; Chen, F. Aqueous Solution Spray Drying Preparations of Binary Amorphous Solid Dispersions. Crystals 2025, 15, 323. https://doi.org/10.3390/cryst15040323
Zheng W, Ke J, Liu K, Xue R, Chen F. Aqueous Solution Spray Drying Preparations of Binary Amorphous Solid Dispersions. Crystals. 2025; 15(4):323. https://doi.org/10.3390/cryst15040323
Chicago/Turabian StyleZheng, Wenling, Junni Ke, Kaerdun Liu, Rongrong Xue, and Fenghua Chen. 2025. "Aqueous Solution Spray Drying Preparations of Binary Amorphous Solid Dispersions" Crystals 15, no. 4: 323. https://doi.org/10.3390/cryst15040323
APA StyleZheng, W., Ke, J., Liu, K., Xue, R., & Chen, F. (2025). Aqueous Solution Spray Drying Preparations of Binary Amorphous Solid Dispersions. Crystals, 15(4), 323. https://doi.org/10.3390/cryst15040323







