The Use of Lignin as a Microbial Carrier in the Co-Digestion of Cheese and Wafer Waste
Abstract
:1. Introduction
2. Materials and Methods
2.1. Feedstock and Inoculum
2.2. Experimental Procedure
2.2.1. Sample Preparation
2.2.2. Carrier Preparation
2.2.3. Anaerobic Digester Setup
2.2.4. Qualitative and Quantitative Analysis of Biogas
2.3. Analytical Techniques
2.3.1. Physicochemical Analysis
2.3.2. Microbial Analysis
2.3.3. Biochemical Analysis
2.3.4. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Properties of Substrates
3.2. Carrier Characteristics
3.3. Batch Experiments
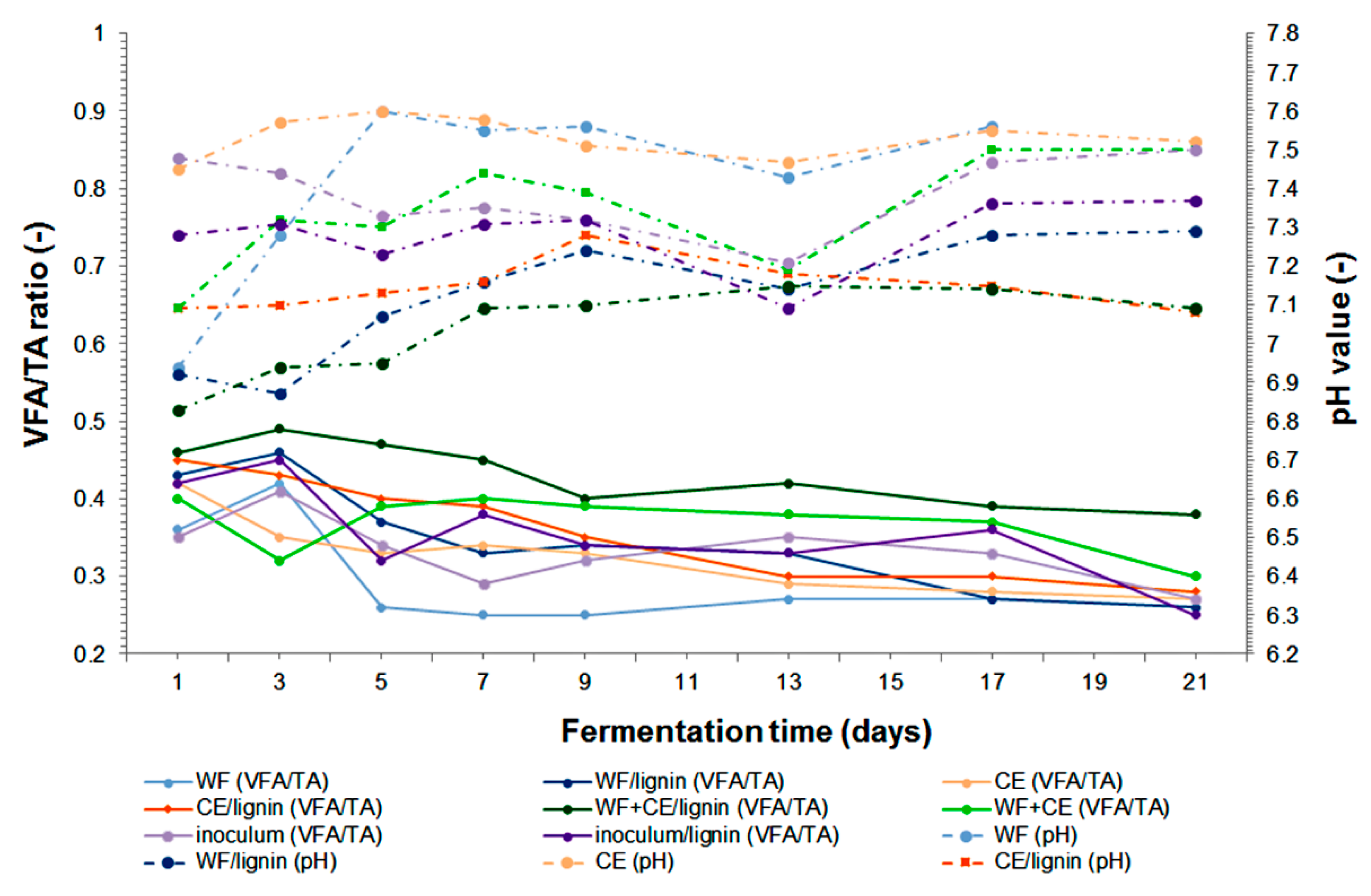
3.3.1. Digestion Process Monitoring
3.3.2. Bacterial Count and Enzymatic Activity
3.3.3. Process Performance
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fontana, A.; Campanaro, S.; Treu, L.; Kougias, P.G.; Cappa, F.; Morelli, L.; Angelidaki, I. Performance and genome-centric metagenomics of thermophilic single and two-stage anaerobic digesters treating cheese wastes. Water Res. 2018, 134, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.N.; Nelson, B.K. Invited review: Anaerobic fermentation of dairy food wastewater. J. Dairy Sci. 2012, 95, 6188–6203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pilarska, A.A.; Pilarski, K.; Witaszek, K.; Waliszewska, H.; Zborowska, M.; Waliszewska, B.; Kolasiński, M.; Szwarc-Rzepka, K. Treatment of dairy waste by anaerobic digestion with sewage sludge. Ecol. Chem. Eng. S 2016, 23, 99–115. [Google Scholar] [CrossRef] [Green Version]
- Prazeres, A.R.; Carvalho, F.; Rivas, J. Cheese whey management: A review. J. Environ. Manag. 2012, 110, 48–68. [Google Scholar] [CrossRef] [PubMed]
- Vlyssides, A.G.; Tsimas, E.S.; Barampouti, E.M.P.; Mai, S.T. Anaerobic digestion of cheese dairy wastewater following chemical oxidation. Biosyst. Eng. 2012, 113, 253–258. [Google Scholar] [CrossRef]
- Rico, C.; Muńoz, N.; Fernández, J.; Rico, J.L. High-load anaerobic co-digestion of cheese whey and liquid fraction of dairy manure in a one-stage UASB process: Limits in co-substrates ratio and organic loading rate. Chem. Eng. J. 2015, 262, 794–802. [Google Scholar] [CrossRef]
- Fernández, C.; Cuetos, M.J.; Martínez, E.J.; Gómez, X. Thermophilic anaerobic digestion of cheese whey: Coupling H2 and CH4 production. Biomass Bioenerg. 2015, 81, 55–62. [Google Scholar] [CrossRef]
- Seo, Y.H.; Yun, Y.M.; Lee, H.; Han, J.I. Pretreatment of cheese whey for hydrogen production using a simple hydrodynamic cavitation system under alkaline condition. Fuel 2015, 150, 202–207. [Google Scholar] [CrossRef]
- Basak, B.; Fatima, A.; Jeon, B.H.; Ganguly, A.; Chatterjee, P.K.; Dey, A. Process kinetic studies of biohydrogen production by co-fermentation of fruit-vegetable wastes and cottage cheese whey. Energy Sustain. Dev. 2018, 47, 39–52. [Google Scholar] [CrossRef]
- Maragkaki, A.E.; Vasileiadis, I.; Fountoulakis, M.; Kyriakou, A.; Lasaridi, K.; Manios, T. Improving biogas production from anaerobic co-digestion of sewage sludge with a thermal dried mixture of food waste, cheese whey and olive mill wastewater. Waste Manag. 2018, 71, 644–651. [Google Scholar] [CrossRef]
- Szaja, A.; Montusiewicz, A. Enhancing the co-digestion efficiency of sewage sludge and cheese whey using brewery spent grain as an additional substrate. Bioresour. Technol. 2019, 291, 121863. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Wickham, R.; Nghiem, L.D. Synergistic effect from anaerobic co-digestion of sewage sludge and organic wastes. Int. Biodeterior. Biodegrad. 2017, 116, 191–197. [Google Scholar] [CrossRef]
- Rusín, J.; Kašáková, K.; Chamrádová, K. Anaerobic digestion of waste wafer material from the confectionery production. Energy 2015, 85, 194–199. [Google Scholar] [CrossRef]
- Pilarska, A.A. Anaerobic co-digestion of waste wafers from the confectionery production with sewage sludge. Polish J. Environ. Stud. 2018, 27, 237–245. [Google Scholar] [CrossRef]
- Pilarska, A.A.; Pilarski, K.; Wolna-Maruwka, A.; Boniecki, P.; Zaborowicz, M. Use of confectionery waste in biogas production by the anaerobic digestion process. Molecules 2019, 24, 37. [Google Scholar] [CrossRef] [Green Version]
- Pagliano, G.; Ventorino, V.; Antonio Panico, A.; Romano, I.; Francesco Pirozzi, F.; Pepe, O. Anaerobic process for bioenergy recovery from dairy waste: Meta-analysis and enumeration of microbial community related to intermediates production. Front. Microbiol. 2019, 9. [Google Scholar] [CrossRef]
- Dzionek, A.; Wojcieszyńska, D.; Guzik, U. Natural carriers in bioremediation: A review. Electron. J. Biotechnol. 2016, 23, 28–36. [Google Scholar] [CrossRef] [Green Version]
- Guisan, J.M. Methods in Biotechnology: Immobilization of Enzymes and Cells, 2nd ed.; Humana Press: Totowa, NJ, USA, 2006. [Google Scholar]
- Purnomo, C.W.; Mellyanawaty, M.; Budhijanto, W. Simulation and experimental study on iron impregnated microbial immobilization in zeolite for production of biogas. Waste Biomass Valorization 2017, 8, 2413–2421. [Google Scholar] [CrossRef]
- Gellerstedt, G. Softwood kraft lignin: Raw material for the future. Ind. Crops Prod. 2015, 77, 845–854. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, D.; Xu, L.; Kong, F.; Wang, S.; Yang, G. Preparation and characterization of softwood kraft lignin copolymers as a paper strength additive. Polymers 2018, 10, 743. [Google Scholar] [CrossRef] [Green Version]
- Qin, Y.; Lin, X.; Lu, Y.; Wu, S.; Yang, D.; Qiu, X.; Fang, Y.; Wang, T. Preparation of a low reducing effect sulfonated alkali lignin and application as dye dispersant. Polymers 2018, 10, 982. [Google Scholar] [CrossRef] [Green Version]
- Zdarta, J.; Meyer, A.S.; Jesionowski, J.; Pinelo, M. A general overview of support materials for enzyme immobilization: Characteristics, properties, practical utility. Catalysts 2018, 8, 92. [Google Scholar] [CrossRef] [Green Version]
- Ciesielczyk, F.; Klapiszewski, F.; Szwarc-Rzepka, K.; Jesionowski, T. A novel method of combination of Kraft lignin with synthetic mineral support. Adv. Powder Technol. 2014, 25, 695–703. [Google Scholar] [CrossRef]
- Pilarska, A.A.; Wolna-Maruwka, A.; Pilarski, K. Kraft lignin grafted with polyvinylpyrrolidone as a novel microbial carrier in biogas production. Energies 2018, 11, 3246. [Google Scholar] [CrossRef] [Green Version]
- Pilarska, A.A.; Pilarski, K.; Wolna-Maruwka, A. Cell immobilization on lignin–polyvinylpyrrolidone material used for anaerobic digestion of waste wafers and sewage sludge. Environ. Eng. Sci. 2019, 36, 478–490. [Google Scholar] [CrossRef]
- Klapiszewski, Ł.; Bartczak, P.; Wysokowski, M.; Jankowska, M.; Kabat, K.; Jesionowski, T. Silica conjugated with kraft lignin and its use as a novel ‘‘green’’ sorbent for hazardous metal ions removal. Chem. Eng. J. 2015, 260, 684–693. [Google Scholar] [CrossRef]
- Li, Z.; Xiao, D.; Ge, Y.; Koehler, S. Surface-functionalized porous lignin for fast and efficient lead removal from aqueous solution. ACS Appl. Mater. Inter. 2015, 7, 15000–15009. [Google Scholar] [CrossRef]
- Singleton, P.; Keyser, H.; Sand, E. Development and evaluation of liquid inoculants. Inoculants and Nitrogen Fixation of Legumes in Vietnam; Herridge, D., Ed.; ACIAR Proceedings 109e; Australian Centre for International Agricultural Research: Canberra, Australia, 2002; pp. 52–66. [Google Scholar]
- Norm VDI 4630. Fermentation of Organic Materials Characterization of the Substrate, Sampling, Collection of Material Data, Fermentation Tests; German Engineers Club: Düsseldorf, Germany, 2006. [Google Scholar]
- Pilarska, A.A.; Pilarski, K.; Ryniecki, A.; Tomaszyk, K.; Dach, J.; Wolna-Maruwka, A. Utilization of vegetable dumplings waste from industrial production by anaerobic digestion. Int. Agrophys. 2017, 31, 93–102. [Google Scholar] [CrossRef]
- Montalvo, S.; Díaz, F.; Guerrero, L.; Sánchez, E.; Borja, R. Effect of particle size and doses of zeolite addition on anaerobic digestion processes of synthetic and piggery wastes. Process. Biochem. 2005, 40, 1475–1481. [Google Scholar] [CrossRef]
- Ivanova, G.; Rákhely, G.; Kovács, K.L. Hydrogen production from biopolymers by Caldicellulosiruptor saccharolyticus and stabilization of the system by immobilization. Int. J. Hydrog. Energy 2008, 33, 6953–6961. [Google Scholar] [CrossRef]
- Pilarski, K.; Pilarska, A.A.; Witaszek, K.; Dworecki, Z.; Żelaziński, T.; Ekielski, A.; Makowska, A.; Michniewicz, J. The impact of extrusion on the biogas and biomethane yield of plant substrates. J. Ecol. Eng. 2016, 17, 264–272. [Google Scholar] [CrossRef]
- DIN Guideline 38 414-S8. Characterisation of the Substrate, Sampling, Collection of Material Data, Fermentation Tests; German Institute for Standardization: Berlin, Germany, 1985. [Google Scholar]
- Wolna-Maruwka, A.; Piechota, T.; Niewiadomska, A.; Dach, J.; Szczech, M.; Jędryczka, M.; Pilarska, A.A. An assessment of adaptive and antagonistic properties of Trichoderma sp. strains in vegetable waste composts. Arch. Environ. Prot. 2017, 43, 2–81. [Google Scholar] [CrossRef] [Green Version]
- Amann, R.I.; Krumholz, L.; Stahl, D.A. Fluorescent-oligonucleotide probing of whole cells for determinative, phylogenetic, and environmental studies in microbiology. J. Bacteriol. 1990, 172, 762–770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amann, R.I.; Binder, B.J.; Olson, R.J.; Chisholm, S.W.; Devereux, R.; Stahl, D.A. Combination of 16S rRNAtargeted oligonucleotide probes with flow cytometry for analyzing mixed microbial populations. Appl. Environ. Microbiol. 1990, 56, 1919–1925. [Google Scholar] [PubMed]
- Stahl, D.A.; Amann, R. Development and application of nucleic acid probes. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E., Goodfellow, M., Eds.; Wiley: New York, NY, USA, 1991; pp. 205–248. [Google Scholar]
- Camiña, F.; Trasar-Cepeda, C.; Gil-Sotres, F.; Leirós, C. Measurement of dehydrogenase activity in acid soils rich in organic matter. Soil Biol. Biochem. 1998, 30, 1005–1011. [Google Scholar] [CrossRef]
- Wolna-Maruwka, A.; Niewiadomska, A.; Piechota, T.; Karwatka, K.; Pilarska, A.A. The handling of composted onion waste in the form of substrates for the proliferation of the Trichoderma sp. Ann. Set Environ. Prot. 2019, 21. In Press. [Google Scholar]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef]
- Pilarska, A.A.; Pilarski, K.; Waliszewska, B.; Zborowska, M.; Witaszek, K.; Waliszewska, H.; Kolasiński, M.; Szwarc-Rzepka, K. Evaluation of bio-methane yields for high-energy organic waste and sewage sludge: A pilot-scale study for a wastewater treatment plant. Environ. Eng. Manag. J. 2019, 18, 2019–2030. [Google Scholar]
- Li, X.; Li, M.; Pu, Y.; Ragauskas, A.J.; Klett, A.S.; Thies, M.; Zheng, Y. Inhibitory effects of lignin on enzymatic hydrolysis: The role of lignin chemistry and molecular weight. Renew. Energ. 2018, 123, 664–674. [Google Scholar] [CrossRef]
- Klapiszewski, L.; Rzemieniecki, T.; Krawczyk, M.; Malina, D.; Norman, M.; Zdarta, J.; Majchrzak, I.; Dobrowolska, A.; Czaczyk, K.; Jesionowski, T. Kraft lignin/silica–AgNPs as a functional material with antibacterial activity. Colloid. Surf. B 2015, 134, 220–228. [Google Scholar] [CrossRef]
- Zhao, Y.; Tagami, A.; Dobele, G.; Lindström, M.E.; Sevastyanova, O. The impact of lignin structural diversity on performance of cellulose nanofiber (CNF)-starch composite films. Polymers 2019, 11, 538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kosmulski, M. Zeta potentials in nonaqueous media: How to measure and control them. Colloids Surf. A Physicochem. Eng. Asp. 2009, 159, 277–281. [Google Scholar] [CrossRef]
- Latif, M.A.; Mehta, C.M.; Batstone, D.J. Influence of low pH on continuous anaerobic digestion of waste activated sludge. Water Res. 2017, 113, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Krakat, N.; Demirel, B.; Anjum, R.; Dietz, D. Methods of ammonia removal in anaerobic digestion: A review. Water Sci. Technol. 2017, 76, 1925–1938. [Google Scholar] [CrossRef] [PubMed]
- Suschka, J.; Grübel, K. Nitrogen in the process of waste activated sludge anaerobic digestion. Arch. Environ. Prot. 2014, 40, 123–136. [Google Scholar] [CrossRef] [Green Version]
- Snell, J.R. Anaerobic digestion: Nitrogen changes and losses during anaerobic digestion. Sew. Work. J. 1943, 15, 56–70. [Google Scholar]
- Sheng, K.; Chen, X.; Pan, J.; Klossa, R.; Wei, Y.; Ying, Y. Effect of ammonia and nitrate on biogas production from food waste via anaerobic digestion. Biosyst. Eng. 2013, 116, 205–212. [Google Scholar] [CrossRef]
- Yuan, H.; Zhu, N. Progress in inhibition mechanisms and process control of intermediates and by-products in sewage sludge anaerobic digestion. Renew. Sustain. Energy Rev. 2016, 58, 429–438. [Google Scholar] [CrossRef]
- Gong, W.; Ran, Z.; Ye, F.; Zhao, G. Lignin from bamboo shoot shells as an activator and novel immobilizing support for α-amylase. Food Chem. 2017, 228, 455–462. [Google Scholar] [CrossRef]
- Lalov, I.G.; Krysteva, M.A.; Phelouzat, J.L. Improvement of biogas production from vinasse via covalently immobilized methanogens. Bioresour. Technol. 2001, 79, 83–85. [Google Scholar] [CrossRef]
- Tan, X.; Wang, Z.; Lu, G.; He, W.; Wei, G.; Huang, F.; Xu, X.; Shen, W. Kinetics of soil dehydrogenase in response to exogenous Cd toxicity. J. Hazard. Material. 2017, 329, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Cheng, J.; Qiao, D.; Yue, L.; Li, Y.Y.; Zhou, J.; Cen, K. Investigating hydrothermal pretreatment of food waste for two-stage fermentative hydrogen and methane co-production. Bioresour. Technol. 2017, 241, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xiao, L.; Yang, Y.C.; Wang, Z.X.; Li, G.X. Lignin binding to pancreatic lipase and its influence on enzymatic activity. Food Chem. 2014, 149, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Kim, S.H.; Kim, J.H.; Yu, H.; Kim, H.J. Application of cellulose/lignin hydrogel beads as novel supports for immobilizing lipase. J. Mol. Catal. B Enzym. 2015, 119, 33–39. [Google Scholar] [CrossRef]










| Waste | pH | TS | VS | C/N Ratio | C | N | N–NH4 | P | Light Metal Ions | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| K | Na | Mg | Ca | |||||||||
| ̶ | (wt %) | (wt %TS) | ̶ | (wt %TS) | (wt %TS) | (wt %TS) | (mg kg−1) | (mg kg−1) | ||||
| CE | 4.67 | 24.89 | 95.75 | 3.48 | 52.19 | 15.01 | 0.44 | 1,452 | 351 | 433 | 84 | 849 |
| WF | 6.80 | 82.24 | 98.37 | 46.21 | 41.59 | 0.90 | 0.29 | 152 | 37 | 154 | 49 | 154 |
| Inoc. | 7.19 | 2.15 | 58.33 | 3.33 | 27.68 | 8.32 | 3.86 | 2560 | 3236 | 6300 | 33 | 52 |
| Batch | CE (g) | WF (g) | Lignin+PVP (g) | Inoculum (g) | pH | TS (%) | VS (%) |
|---|---|---|---|---|---|---|---|
| WF | ̶ | 14.0 | ̶ | 1186.0 | 7.24 | 4.24 | 70.15 |
| WF/lignin | ̶ | 14.0 | 23.5 | 1186.0 | 7.16 | 4.07 | 67.34 |
| CE | 30.7 | ̶ | ̶ | 1169.9 | 7.38 | 2.73 | 59.29 |
| CE/lignin | 30.7 | ̶ | 23.5 | 1170.3 | 7.04 | 2.76 | 60.47 |
| WF+CE | 4.1 | 7.8 | ̶ | 1189.0 | 7.02 | 2.74 | 58.72 |
| WF+CE/lignin | 4.1 | 7.8 | 23.5 | 1169.9 | 6.78 | 2.75 | 58.84 |
| inoculum | ̶ | ̶ | ̶ | 1200.0 | 7.22 | 3.17 | 69.82 |
| inoculum/lignin | ̶ | ̶ | 23.5 | 1200.0 | 7.13 | 4.85 | 71.01 |
| Parameters | WF | WF/Lignin | CE | CE/Lignin | WF+CE | WF+CE/Lignin | Inoculum | Inoculum/Lignin |
|---|---|---|---|---|---|---|---|---|
| Bacteria Number | ||||||||
| N–NH4+ | −0.19 | 0.21 | −0.13 | 0.81 * | 0.67 | 0.31 | 0.81 * | 0.80 * |
| TKN | −0.19 | 0.24 | −0.23 | 0.79 | 0.60 | 0.47 | 0.41 | −0.14 |
| pH | −0.46 | −0.69 | −0.44 | 0.98 * | −0.42 | 0.74 | −0.88 * | −0.92 * |
| DHA | −0.70 | −0.93 * | −0.73 | −0.03 | −0.33 | −0.28 | −0.85 * | −0.70 |
| DHA | ||||||||
| N–NH4+ | 0.75 | 0.03 | −0.03 | −0.56 | 0.08 | −0.23 | −0.49 | −0.57 |
| TKN | 0.79 | 0.01 | 0.44 | −0.56 | −0.04 | −0.26 | −0.46 | −0.08 |
| pH | 0.86 * | 0.67 | 0.24 | −0.22 | 0.79 | 0.13 | 0.54 | 0.86 * |
| Batch | Biogas | Methane | CH4 (%) | ||
|---|---|---|---|---|---|
| (m3 Mg−1 FM) | (m3 Mg−1 VS) | (m3 Mg−1 FM) | (m3 Mg−1 VS) | ||
| WF | 866.33 | 897.22 | 479.16 | 496.39 | 55.31 |
| WF/lignin | 966.28 | 1201.45 | 551.34 | 685.53 | 57.06 |
| CE | 210.04 | 881.26 | 132.61 | 556.42 | 63.14 |
| CE/lignin | 235.87 | 989.65 | 151.43 | 635.36 | 64.20 |
| WF+CE | 674.48 | 1102.00 | 349.83 | 571.57 | 51.87 |
| WF+CE/lignin | 769.58 | 1257.38 | 402.18 | 657.68 | 52.26 |
| inoculum | 1.51 | 120.07 | 0.94 | 74.90 | 62.38 |
| inoculum/lignin | 1.29 | 170.43 | 2.14 | 103.10 | 60.49 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pilarska, A.A.; Wolna-Maruwka, A.; Pilarski, K.; Janczak, D.; Przybył, K.; Gawrysiak-Witulska, M. The Use of Lignin as a Microbial Carrier in the Co-Digestion of Cheese and Wafer Waste. Polymers 2019, 11, 2073. https://doi.org/10.3390/polym11122073
Pilarska AA, Wolna-Maruwka A, Pilarski K, Janczak D, Przybył K, Gawrysiak-Witulska M. The Use of Lignin as a Microbial Carrier in the Co-Digestion of Cheese and Wafer Waste. Polymers. 2019; 11(12):2073. https://doi.org/10.3390/polym11122073
Chicago/Turabian StylePilarska, Agnieszka A., Agnieszka Wolna-Maruwka, Krzysztof Pilarski, Damian Janczak, Krzysztof Przybył, and Marzena Gawrysiak-Witulska. 2019. "The Use of Lignin as a Microbial Carrier in the Co-Digestion of Cheese and Wafer Waste" Polymers 11, no. 12: 2073. https://doi.org/10.3390/polym11122073






