Molecular Mechanisms of Antitumor Activity of PAMAM Dendrimer Conjugates with Anticancer Drugs and a Monoclonal Antibody
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Conjugates
2.3. Cell Culture
2.4. Measurement of Reactive Oxygen Species (ROS)
2.5. Assessment of Mitochondrial Membrane Potential (ΔΨm)
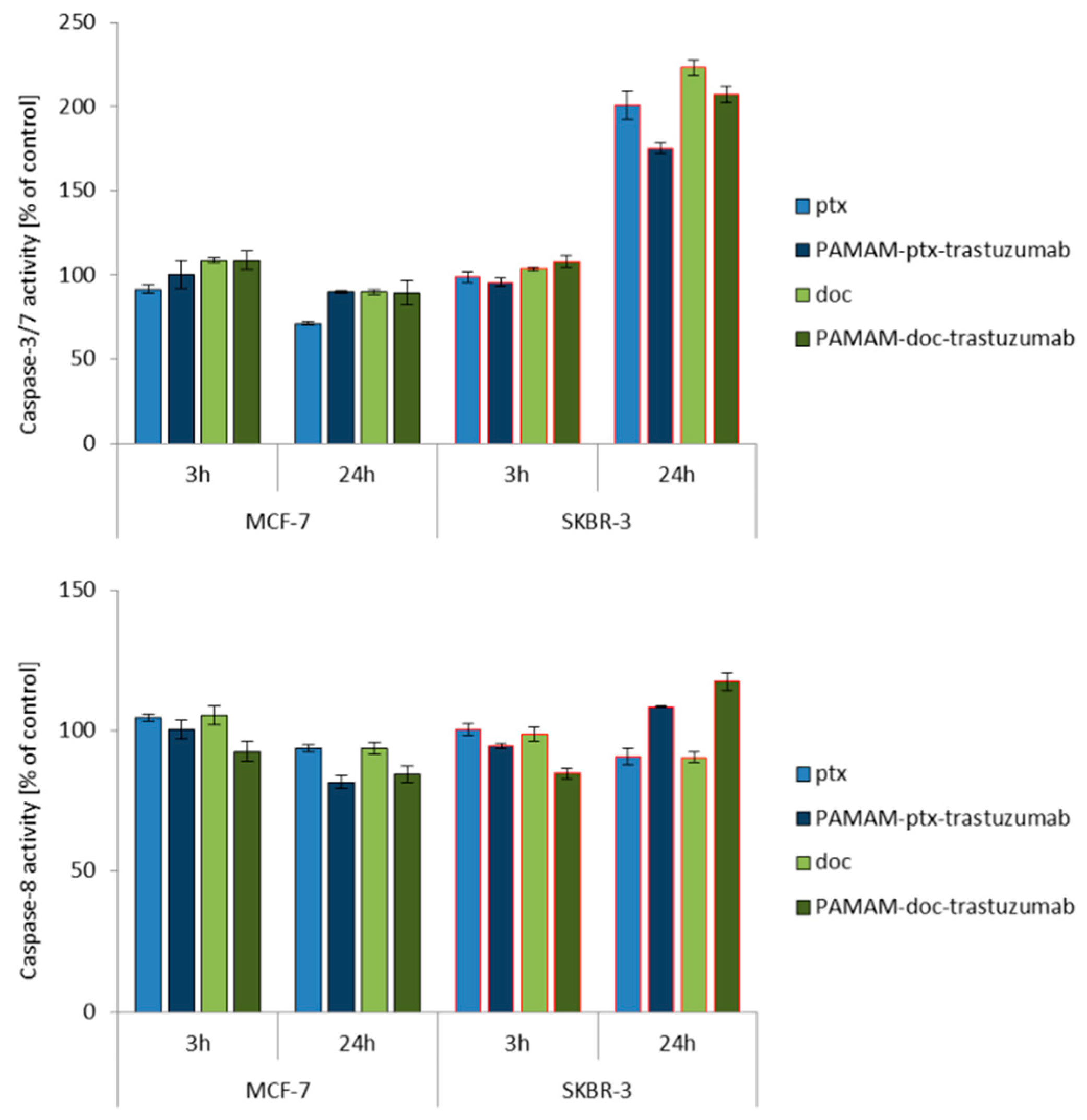
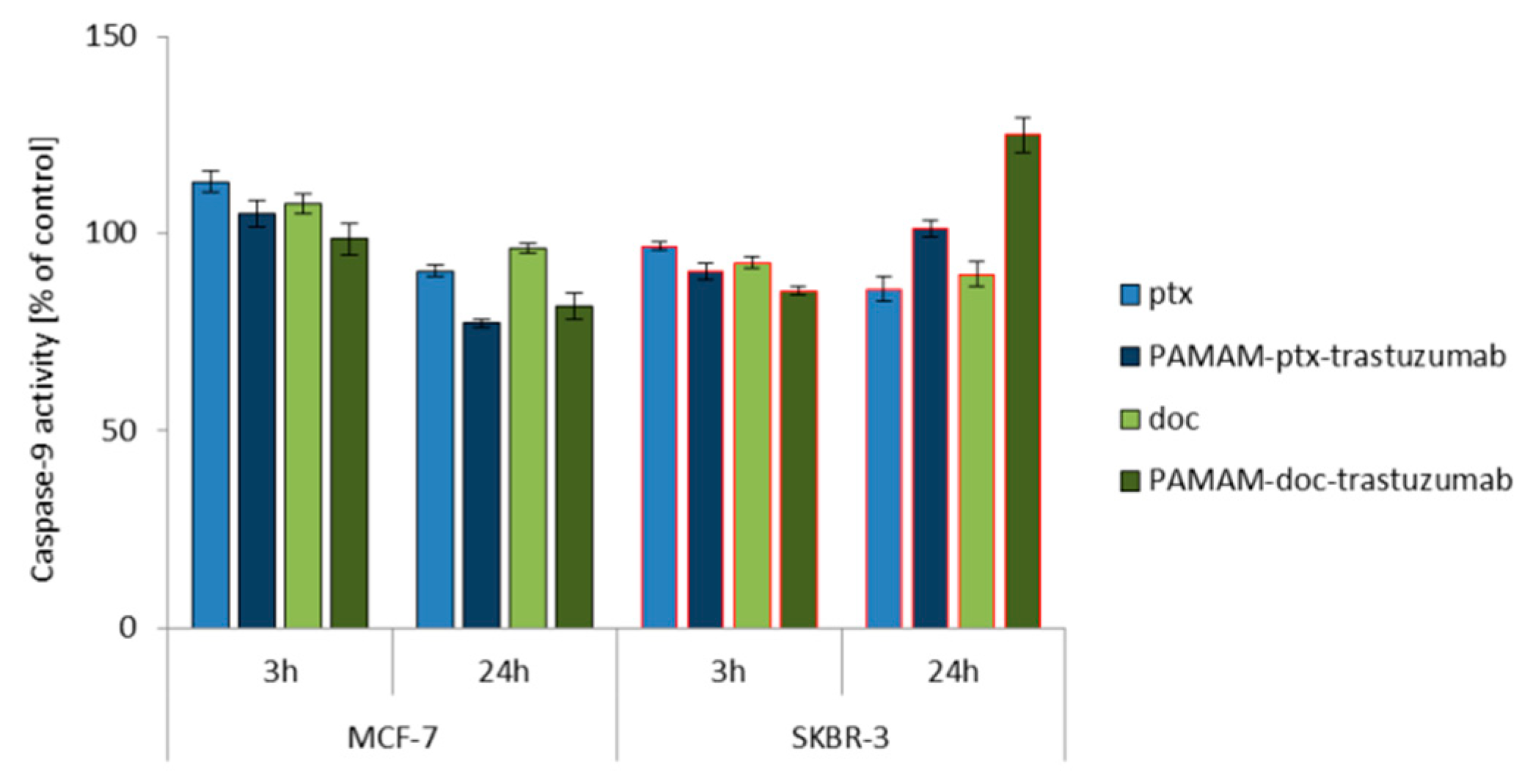
2.6. Measurement of Caspases Activity
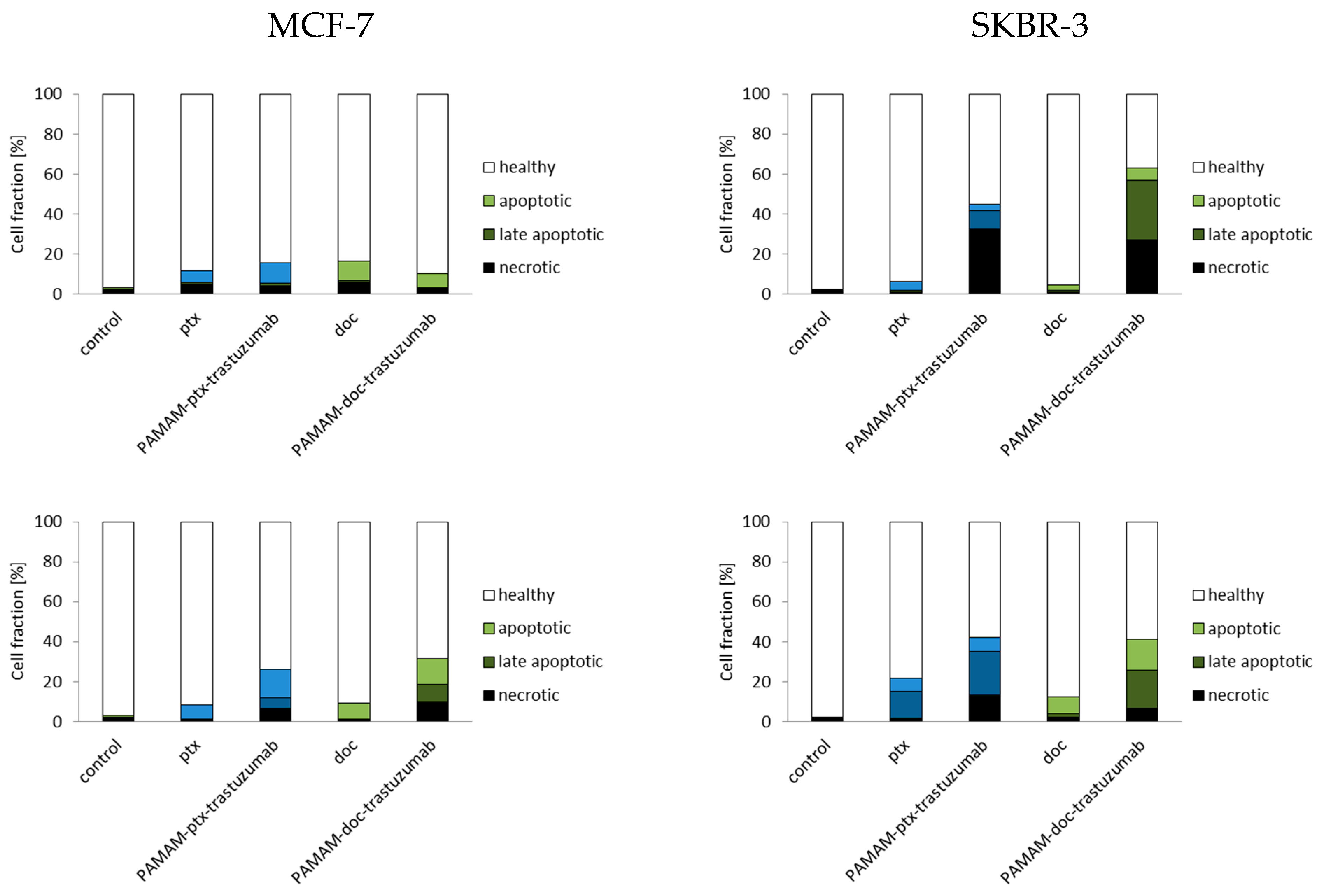
2.7. Detection of Apoptotic and Necrotic Cells
2.8. Confocal Microscopy
2.9. Cell Cycle Studies
2.10. Statistical Analysis
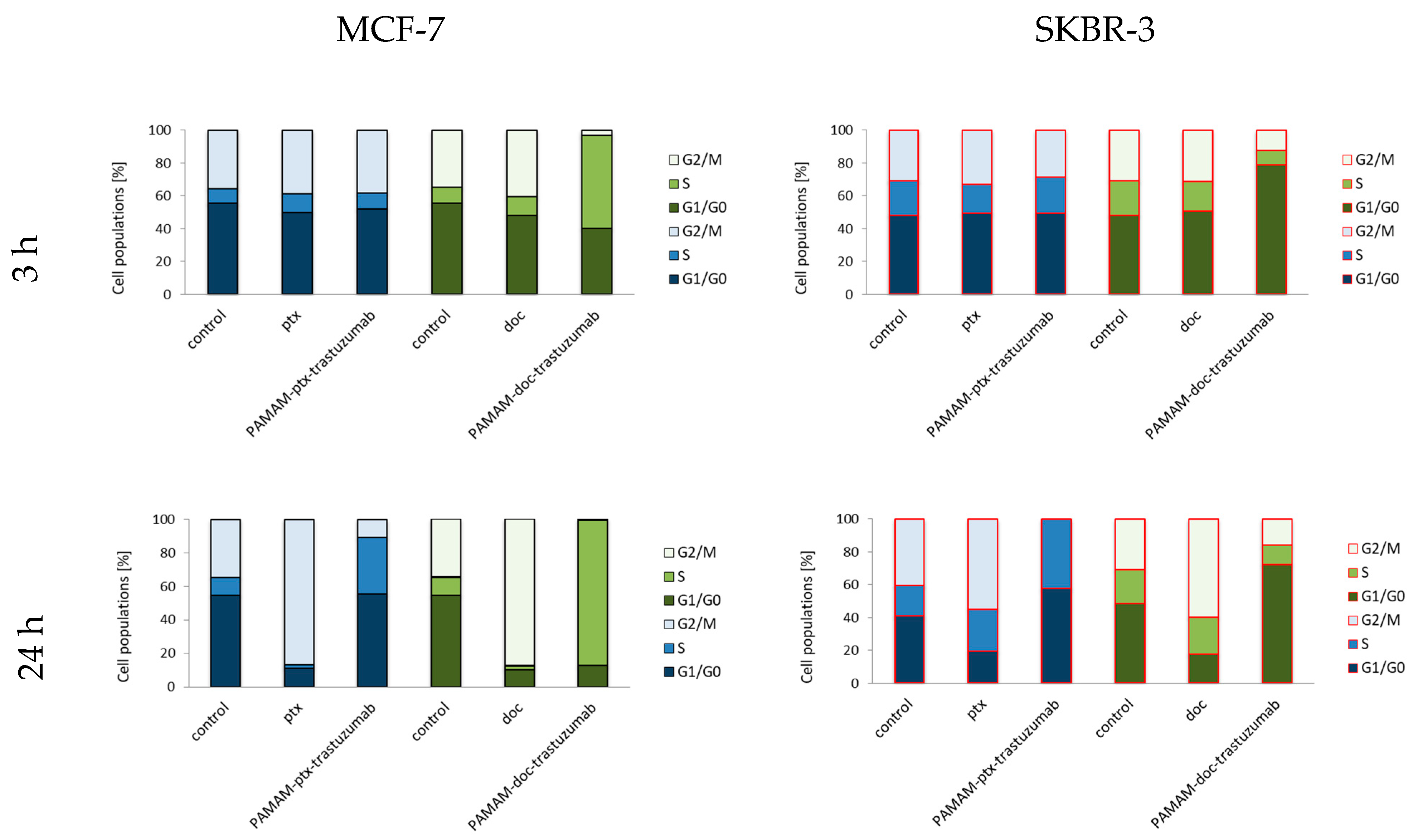
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wani, M.C.; Taylor, H.L.; Wall, M.E.; Coggon, P.; McPhail, A.T. Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J. Am. Chem. Soc. 1971, 93, 2325–2327. [Google Scholar] [CrossRef] [PubMed]
- Bissery, M.C.; Nohynek, G.; Sanderink, G.J.; Lavelle, F. Docetaxel (Taxotere): A review of preclinical and clinical experience. Part I: Preclinical experience. Anti-Cancer Drugs 1995, 6, 339–355, 363–368. [Google Scholar] [PubMed]
- Schiff, P.B.; Horwitz, S.B. Taxol stabilizes microtubules in mouse fibroblast cells. Proc. Natl. Acad. Sci. USA 1980, 77, 1561–1565. [Google Scholar] [CrossRef] [PubMed]
- Rowinsky, E.K.; Cazenave, L.A.; Donehower, R.C. Taxol: A Novel Investigational Antimicrotubule Agent. J. Natl. Cancer Inst. 1990, 82, 1247–1259. [Google Scholar] [PubMed]
- Dumontet, C.; Sikic, B.I. Mechanisms of Action of and Resistance to Antitubulin Agents: Microtubule Dynamics, Drug Transport, and Cell Death. J. Clin. Oncol. 1999, 17, 1061. [Google Scholar] [CrossRef] [PubMed]
- De Leeuw, R.B.-B.L.; Schiewer, M.J.; Ciment, S.J.; Den, R.B.; Dicker, A.P.; Kelly, W.K.; Trabulsi, E.J.; Lallas, C.D.; Gomella, L.G.; Knudsen, K.E. Novel actions of next-generation taxanes benefit advanced stages of prostate cancer. Clin. Cancer Res. 2015, 21, 795–807. [Google Scholar] [CrossRef] [PubMed]
- Jordan, M.A.; Wilson, L. Microtubules as a target for anticancer drugs. Nat. Rev. Cancer 2004, 4, 253–265. [Google Scholar] [CrossRef]
- Mozzetti, S.; Ferlini, C.; Concolino, P.; Filippetti, F.; Raspaglio, G.; Prislei, S.; Gallo, D.; Martinelli, E.; Ranelletti, F.O.; Ferrandina, G.; et al. Class III β-tubulin overexpression is a prominent mechanism of paclitaxel resistance in ovarian cancer patients. Clin. Cancer Res. 2005, 11, 298–305. [Google Scholar]
- Ten Tije, A.J.; Verweij, J.; Loos, W.J.; Sparreboom, A. Pharmacological effects of formulation vehicles: Implications for cancer chemotherapy. Clin. Pharmacokinet. 2003, 42, 665–685. [Google Scholar] [CrossRef]
- Gupta, S. Novel Antitumoral Use of Cabazitaxel in Metastatic Prostate Cancer. U.S. Patent WO2011051894A1, 5 May 2011. [Google Scholar]
- Desai, N.; Trieu, V.; Yao, Z.; Louie, S.; Ci, A.; Yang, C.; Tao, T.; De, B.; Beals, D.; Dykes, P.; et al. Soon-Shiong Increased antitumor activity, intratumor paclitaxel concentrations, and endothelial cell transport of cremophor-free, albumin-bound paclitaxel, ABI-007, compared with cremophor-based paclitaxel. Clin. Cancer Res. 2006, 12, 1317–1324. [Google Scholar]
- Desai, N.P.; Soon-Shiong, P. Nanoparticles of Paclitaxel and Albumin in Combination with Bevacizumab against Cancer. U.S. Patent US20100112077A1, 6 May 2010. [Google Scholar]
- Swain, S.M.; Kim, S.B.; Cortés, J.; Ro, J.; Semiglazov, V.; Campone, M.; Ciruelos, E.; Ferrero, J.M.; Schneeweiss, A.; Knott, A.; et al. Pertuzumab, trastuzumab, and docetaxel for HER2-positive metastatic breast cancer (CLEOPATRA study): Overall survival results from a randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2013, 14, 461–471. [Google Scholar] [CrossRef]
- Marcinkowska, M.; Stanczyk, M.; Janaszewska, A.; Sobierajska, E.; Chworos, A.; Klajnert-Maculewicz, B. Multicomponent conjugates of anticancer drugs and monoclonal antibody with PAMAM dendrimers to increase efficacy of HER-2 positive breast cancer therapy. Pharm. Res. 2019. [Google Scholar] [CrossRef]
- Schaefer, N.G.; Pestalozzi, B.C.; Knuth, A.; Renner, C. Potential use of humanized antibodies in the treatment of breast cancer. Expert Rev. Anticancer Ther. 2006, 6, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Hynes, N.E.; Stern, D.F. The biology of erbB-2/neu/HER-2 and its role in cancer. Biochim. Biophys. Acta 1994, 1198, 165–184. [Google Scholar] [PubMed]
- Ross, J.S.; Fletcher, J.A. The HER-2/neu Oncogene in Breast Cancer: Prognostic Factor, Predictive Factor, and Target for Therapy. Stem Cells 1998, 3, 413–428. [Google Scholar] [CrossRef]
- Pegram, M.D.; Konecny, G.E.; O’Callaghan, C.; Beryt, M.; Pietras, R.; Slamon, D.J. Rational combinations of trastuzumab with chemotherapeutic drugs used in the treatment of breast cancer. J. Natl. Cancer Inst. 2004, 96, 739–749. [Google Scholar] [CrossRef]
- Nahta, R.; Esteva, F.J. Herceptin: Mechanisms of action and resistance. Cancer Lett. 2006, 232, 123–138. [Google Scholar] [CrossRef]
- Kono, K.; Takahashi, A.; Ichihara, F.; Sugai, H.; Fujii, H.; Matsumoto, Y. Impaired antibody-dependent cellular cytotoxicity mediated by herceptin in patients with gastric cancer. Cancer Res. 2002, 62, 5813–5817. [Google Scholar]
- Bartosz, G. Use of spectroscopic probes for detection of reactive oxygen species. Clin. Chim. Acta 2006, 368, 53–76. [Google Scholar] [CrossRef]
- Salvioli, S.; Ardizzoni, A.; Franceschi, C.; Cossarizza, A. JC-1, but not DiOC6(3) or rhodamine 123, is a reliable fluorescent probe to assess delta psi changes in intact cells: Implications for studies on mitochondrial functionality during apoptosis. FEBS Lett. 1997, 411, 77–82. [Google Scholar] [CrossRef]
- Rieger, A.M.; Nelson, K.L.; Konowalchuk, J.D.; Barreda, D.R. Modified Annexin V/Propidium Iodide Apoptosis Assay For Accurate Assessment of Cell Death. J. Vis. Exp. 2011, 24, e2597. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.-L.; Kung, M.-L.; Chow, N.-H.; Su, S.-J. Genistein arrests hepatoma cells at G2/M phase: Involvement of ATM activation and upregulation of p21waf1/cip1 and Wee1. Biochem. Pharmacol. 2004, 67, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Marcinkowska, M.; Sobierajska, E.; Stanczyk, M.; Janaszewska, A.; Chworos, A.; Klajnert-Maculewicz, B. Conjugate of PAMAM Dendrimer, Doxorubicin and Monoclonal Antibody—Trastuzumab: The New Approach of a Well-Known Strategy. Polymers 2018, 10, 187. [Google Scholar] [CrossRef] [PubMed]
- Janaszewska, A.; Mączyńska, K.; Matuszko, G.; Appelhans, D.; Voit, B.; Klajnert, B.; Bryszewska, M. Cytotoxicity of PAMAM, PPI and maltose modified PPI dendrimers in Chinese hamster ovary (CHO) and human ovarian carcinoma (SKOV3) cells. New J. Chem. 2012, 36, 428–437. [Google Scholar] [CrossRef]
- Pathania, D.; Millard, M.; Neamati, N. Opportunities in discovery and delivery of anticancer drugs targeting mitochondria and cancer cell metabolism. Adv. Drug Deliv. Rev. 2009, 61, 1250–1275. [Google Scholar] [CrossRef] [PubMed]
- Spierings, D.; Berkovitch, F.; Nicolet, Y.; Wan, J.T.; Jarrett, J.T.; Drennan, C.L. Connected to Death: The (Unexpurgated) Mitochondrial Pathway of Apoptosis. Science 2005, 310, 66–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaufmann, S.H.; Earnshaw, W.C. Induction of Apoptosis by Cancer Chemotherapy. Exp. Cell Res. 2000, 256, 42–49. [Google Scholar] [CrossRef]
- Mukherjee, S.P.; Lyng, F.M.; Garcia, A.; Davoren, M.; Byrne, H.J. Mechanistic studies of in vitro cytotoxicity of poly(amidoamine) dendrimers in mammalian cells. Toxicol. Appl. Pharmacol. 2010, 248, 259–268. [Google Scholar] [CrossRef] [Green Version]
- Mohan, N.; Shen, Y.; Endo, Y.; ElZarrad, M.K.; Wu, W.J. Trastuzumab, but Not Pertuzumab, Dysregulates HER2 Signaling to Mediate Inhibition of Autophagy and Increase in Reactive Oxygen Species Production in Human Cardiomyocytes. Mol. Cancer Ther. 2016, 15, 1321–1331. [Google Scholar] [CrossRef] [Green Version]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial ROS-induced ROS release: An update and review. Biochim. Biophys. Acta BBA 2006, 1757, 509–517. [Google Scholar] [CrossRef] [Green Version]
- Lucken-Ardjomande, S.; Martinou, J.-C. Regulation of Bcl-2 proteins and of the permeability of the outer mitochondrial membrane. C. R. Boil. 2005, 328, 616–631. [Google Scholar] [CrossRef] [PubMed]
- Evtodienko, Y.V.; Teplova, V.V.; Sidash, S.S.; Ichas, F.; Mazat, J.-P. Microtubule-active drugs suppress the closure of the permeability transition pore in tumour mitochondria. FEBS Lett. 1996, 393, 86–88. [Google Scholar] [CrossRef] [Green Version]
- Kidd, J.; Pilkington, M.; Schell, M.; Fogarty, K.; Skepper, J.; Taylor, C.; Thorn, P. Paclitaxel affects cytosolic Ca2+ signals by opening the mitochondrial permeability transition pore. J. Biol. Chem. 2002, 277, 6504–6510. [Google Scholar] [CrossRef] [PubMed]
- Mironov, K.; Ivannikov, M.; Johansson, M. [Ca2+]i signaling between mitochondria and endoplasmic reticulum in neurons is regulated by microtubules. From mitochondrial permeability transition pore to Ca2+-induced Ca2+ release. J. Biol. Chem. 2005, 280, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Cristofani, R.; Marelli, M.M.; Cicardi, M.E.; Fontana, F.; Marzagalli, M.; Limonta, P.; Poletti, A.; Moretti, R.M. Dual role of autophagy on docetaxel-sensitivity in prostate cancer cells. Cell Death Dis. 2018, 9, 889. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.P.; Byrne, H.J. Polyamidoamine dendrimer nanoparticle cytotoxicity, oxidative stress, caspase activation and inflammatory response: Experimental observation and numerical simulation. Nanomed. Nanotechnol. Boil. Med. 2013, 9, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Cain, K.; Bratton, S.B.; Cohen, G.M. The Apaf-1 apoptosome: A large caspase-activating complex. Biochimie 2002, 84, 203–214. [Google Scholar] [CrossRef]
- Woo, M.; Hakem, R.; Soengas, M.S.; Duncan, G.S.; Shahinian, A.; Kägi, D.; Hakem, A.; McCurrach, M.; Khoo, W.; Kaufman, S.A.; et al. Essential contribution of caspase 3/CPP32 to apoptosis and its associated nuclear changes. Genes Dev. 1998, 12, 806–819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Debatin, K.-M. Activation of apoptosis pathways by anticancer treatment. Toxicol. Lett. 2000, 112, 41–48. [Google Scholar] [CrossRef]
- Slee, E.A.; Adrain, C.; Martin, S.J. Executioner caspase-3, -6, and -7 perform distinct, non-redundant roles during the demolition phase of apoptosis. J. Biol. Chem. 2001, 276, 7320–7326. [Google Scholar] [CrossRef]
- Kovár, J.; Ehrlichová, M.; Smejkalová, B.; Zanardi, I.; Ojima, I.; Gut, I. Comparison of Cell Death-inducing Effect of Novel Taxane SB-T-1216 and Paclitaxel in Breast Cancer Cells. Anticancer Res. 2009, 29, 2951–2960. [Google Scholar] [PubMed]
- Osaki, S.; Nakanishi, Y.; Takayama, K.; Pei, X.H.; Ueno, H.; Hara, N. Transfer of IkappaBalpha gene increase the sensitivity of paclitaxel mediated with caspase 3 activation in human lung cancer cell. J. Exp. Clin. Cancer Res. 2003, 22, 69–75. [Google Scholar] [PubMed]
- Ueno, N.T.; Bartholomeusz, C.; Herrmann, J.L.; Estrov, Z.; Shao, R.; Andreeff, M.; Price, J.; Paul, R.W.; Anklesaria, P.; Yu, D.; et al. E1A-mediated paclitaxel sensitization in HER-2/neu-overexpressing ovarian cancer SKOV3.ip1 through apoptosis involving the caspase-3 pathway. Clin. Cancer Res. 2000, 6, 250–259. [Google Scholar] [PubMed]
- Kim, J.Y.; Chung, J.-Y.; Lee, S.G.; Kim, Y.-J.; Park, J.-E.; Yoo, K.S.; Yoo, Y.H.; Park, Y.C.; Kim, B.G.; Kim, J.-M. Nuclear interaction of Smac/DIABLO with Survivin at G2/M arrest prompts docetaxel-induced apoptosis in DU145 prostate cancer cells. Biochem. Biophys. Res. Commun. 2006, 350, 949–954. [Google Scholar] [CrossRef] [PubMed]
- Vikhanskaya, F.; Vignati, S.; Beccaglia, P.; Ottoboni, C.; Russo, P.; D’Incalci, M.; Broggini, M. Inactivation of p53 in a Human Ovarian Cancer Cell Line Increases the Sensitivity to Paclitaxel by Inducing G2/M Arrest and Apoptosis. Exp. Cell Res. 1998, 241, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Huisman, C.; Ferreira, C.G.; Bröker, L.E.A.; Rodriguez, J.; Smit, E.F.; Postmus, P.E.; Kruyt, F.A.E.; Giaccone, G. Paclitaxel triggers cell death primarily via caspase-independent routes in the non-small cell lung cancer cell line NCI-H460. Clin. Cancer Res. 2002, 8, 596–606. [Google Scholar] [PubMed]
- Ofir, R.; Seidman, R.; Rabinski, T.; Krup, M.; Yavelsky, V.; Weinstein, Y.; Wolfson, M. Taxol-induced apoptosis in human SKOV3 ovarian and MCF7 breast carcinoma cells is caspase-3 and caspase-9 independent. Cell Death Differ. 2002, 9, 636–642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kulhari, H.; Pooja, D.; Shrivastava, S.; Kuncha, M.; Naidu, V.G.M.; Bansal, V.; Sistla, R.; Adams, D.J. Trastuzumab-grafted PAMAM dendrimers for the selective delivery of anticancer drugs to HER2-positive breast cancer. Sci. Rep. 2016, 6, 23179. [Google Scholar] [CrossRef]
- Conklin, K.A. Chemotherapy-Associated Oxidative Stress: Impact on Chemotherapeutic Effectiveness. Integr. Cancer Ther. 2004, 3, 294–300. [Google Scholar] [CrossRef]
- Huang, J.; Wang, S.; Lyu, H.; Cai, B.; Yang, X.; Wang, J.; Liu, B. The anti-erbB3 antibody MM-121/SAR256212 in combination with trastuzumab exerts potent antitumor activity against trastuzumab-resistant breast cancer cells. Mol. Cancer 2013, 12, 134. [Google Scholar] [CrossRef]
- Mohsin, S.K.; Weiss, H.L.; Gutierrez, M.C.; Chamness, G.C.; Schiff, R.; DiGiovanna, M.P.; Wang, C.-X.; Hilsenbeck, S.G.; Osborne, C.K.; Allred, D.C.; et al. Neoadjuvant Trastuzumab Induces Apoptosis in Primary Breast Cancers. J. Clin. Oncol. 2005, 23, 2460–2468. [Google Scholar] [CrossRef] [PubMed]
- Lane, H.A.; Motoyama, A.B.; Beuvink, I.; Hynes, N.E. Modulation of p27/Cdk2 complex formation through 4D5-mediated inhibition of HER2 receptor signaling. Ann. Oncol. 2001, 12, 21–22. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, T.; Outhoff, K. The in vitro influences of epidermal growth factor and heregulin-β1 on the efficacy of trastuzumab used in Her-2 positive breast adenocarcinoma. Cancer Cell Int. 2013, 13, 97. [Google Scholar] [CrossRef] [PubMed]
- Beer, T.M.; El-Geneidi, M.; Eilers, K.M. Docetaxel (Taxotere®) in the treatment of prostate cancer. Expert Rev. Anticancer Ther. 2003, 3, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Collins, D.M.; O’Donovan, N.; McGowan, P.M.; O’Sullivan, F.; Duffy, M.J.; Crown, J. Trastuzumab induces antibody-dependent cell-mediated cytotoxicity (ADCC) in HER-2-non-amplified breast cancer cell lines. Ann. Oncol. 2012, 23, 1788–1795. [Google Scholar] [CrossRef] [PubMed]
- Pegram, M.; Hsu, S.; Lewis, G.; Pietras, R.; Beryt, M.; Sliwkowski, M.; Coombs, D.; Baly, D.; Kabbinavar, F.; Slamon, D. Inhibitory effects of combinations of HER-2/neu antibody and chemotherapeutic agents used for treatment of human breast cancers. Oncogene 1999, 18, 2241–2251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baselga, J.; Norton, L.; Albanell, Y.; Kim, M.; Mendelsohn, J. Recombinant humanized anti-HER2 antibody (Herceptin™) enhances the antitumor activity of paclitaxel and doxorubicin against HER2/neu overexpressing human breast cancer xenografts. Cancer Res. 1998, 58, 2825–2831. [Google Scholar] [PubMed]


 ), giant cells and elongated cells with protrusions from the plasma membrane and cytoplasmic bridges between cells (marked by the yellow arrows
), giant cells and elongated cells with protrusions from the plasma membrane and cytoplasmic bridges between cells (marked by the yellow arrows  ). For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.
). For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.
 ), giant cells and elongated cells with protrusions from the plasma membrane and cytoplasmic bridges between cells (marked by the yellow arrows
), giant cells and elongated cells with protrusions from the plasma membrane and cytoplasmic bridges between cells (marked by the yellow arrows  ). For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.
). For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.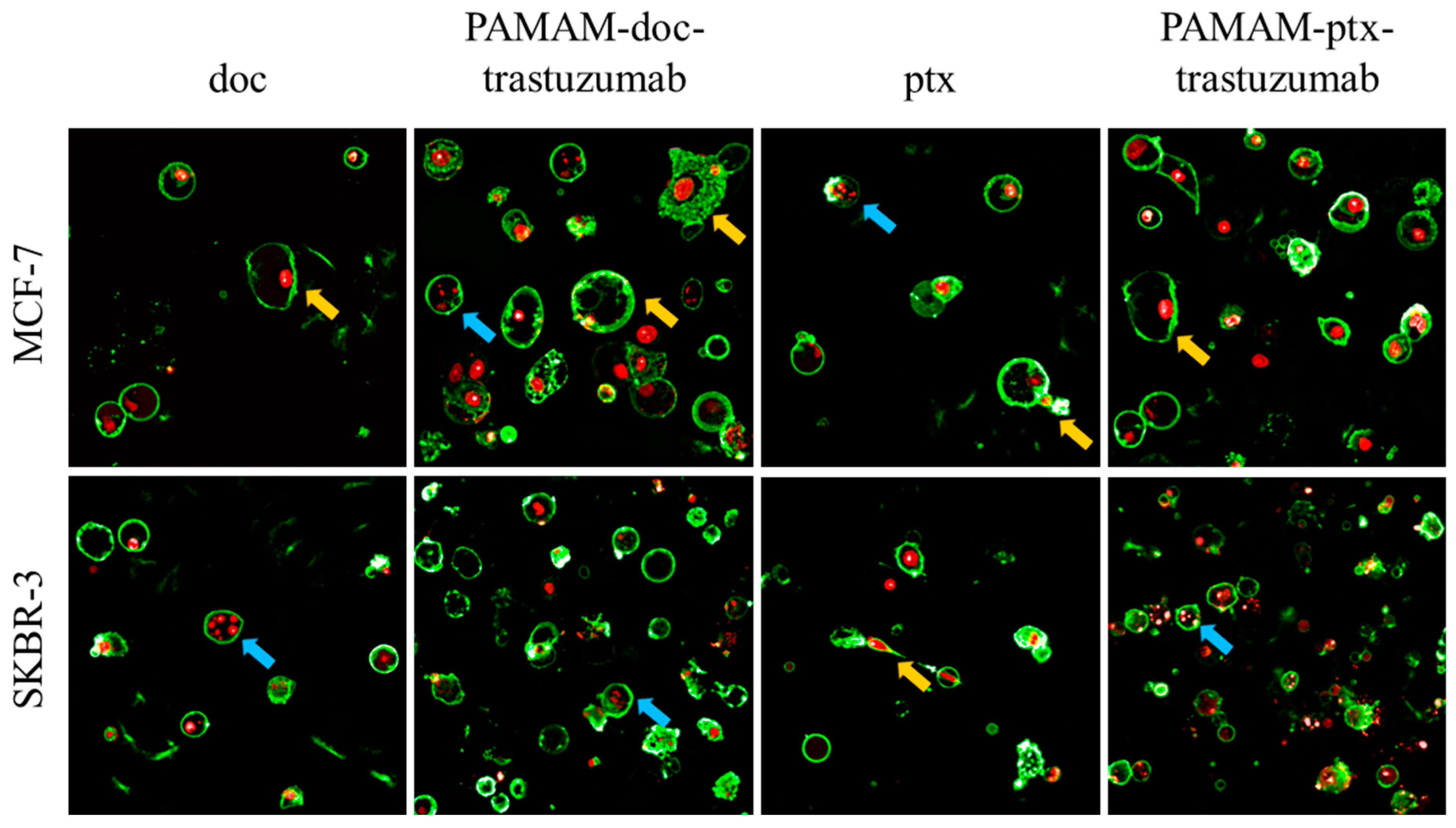

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcinkowska, M.; Stanczyk, M.; Janaszewska, A.; Gajek, A.; Ksiezak, M.; Dzialak, P.; Klajnert-Maculewicz, B. Molecular Mechanisms of Antitumor Activity of PAMAM Dendrimer Conjugates with Anticancer Drugs and a Monoclonal Antibody. Polymers 2019, 11, 1422. https://doi.org/10.3390/polym11091422
Marcinkowska M, Stanczyk M, Janaszewska A, Gajek A, Ksiezak M, Dzialak P, Klajnert-Maculewicz B. Molecular Mechanisms of Antitumor Activity of PAMAM Dendrimer Conjugates with Anticancer Drugs and a Monoclonal Antibody. Polymers. 2019; 11(9):1422. https://doi.org/10.3390/polym11091422
Chicago/Turabian StyleMarcinkowska, Monika, Maciej Stanczyk, Anna Janaszewska, Arkadiusz Gajek, Malgorzata Ksiezak, Paula Dzialak, and Barbara Klajnert-Maculewicz. 2019. "Molecular Mechanisms of Antitumor Activity of PAMAM Dendrimer Conjugates with Anticancer Drugs and a Monoclonal Antibody" Polymers 11, no. 9: 1422. https://doi.org/10.3390/polym11091422






