1. Introduction
The transition of the plastic industry from its traditional Linear Economy to a Circular Economy, a more valuable and sustainable model, is being spearheaded by the European Union (EU), where legislative measures are being introduced to eliminate excessive waste [
1]. To this end, it is first necessary to promote sustainable polymer technologies that decouple plastics from fossil feedstocks [
2]. Furthermore, it is also important to increase the quality and uptake of plastic recycling [
3]. In this context, the food packaging sector it is among the most heavily scrutinized, given the large production and the short life cycle of their products [
4]. Polyethylene terephthalate (PET) is a thermoplastic polyester that is widely used in the manufacture of bottles for water or beverages and also food trays due to its good mechanical properties, chemical resistance, clarity, thermal stability, barrier properties, and low production cost [
5]. PET is fully recyclable and it is, indeed, one of the most recycled plastics in the world. Since 2012, PET monomaterial packaging has showed recycling rates of approximately 52% in the EU and 31% in the United States (US) [
6]. Moreover, the recycling of PET articles is expected to increase, as exemplified by companies like Coca-Cola, which have already announced a full switch to recycled plastics for their beverage bottles in some EU countries, starting with at least 50% rates by the end of 2023 [
7].
In recent years, the replacement of conventional or petrochemical polymer materials with those obtained from natural and renewable sources is of great interest for research due to the depletion of fossil resources and the cost of extracting them. The main interest and advantage of using bio-based polymers reflects the concept of the so-called “biorefinery system design” due to its ability to improve the environmental impact of a product by reducing greenhouse gas emissions, economizing fossil resources, exploring the possibility of using a local resource, and the use of by-products or even wastes [
8]. Bio-based polymers, which can be either biodegradable or non-biodegradable, are certified according to international standards such as EN 16640:2015, ISO 16620-4:2016, ASTM 6866-18, and EN 16785-1:2015. In particular, EN 16640:2015, ISO 16620-4:2016, and ASTM 6866-18 measure the bio-based carbon content in a material through 14C measurements, while EN 16785-1:2015 measures the bio-based content of a material using radiocarbon and elemental analyses [
9]. Indeed, the pattern of production of biopolymers is shifting from biodegradable to bio-based. Bio-based but non-biodegradable polymers represented 57.1% of the total biopolymer production in 2018, whereas biodegradable polymers accounted for 42.9% [
10]. This is in contradiction to the public perception that most biopolymers are biodegradable. Among them, partially bio-based polyethylene terephthalate (bio-PET) is currently the most produced bioplastic, reaching ~27% of the total production in 2018, that is, 0.54 million tons per year. [
11]. This is based on the fact that this “green polyester” offers an almost identical chemical structure and properties to its petrochemical counterpart, that is, PET.
Several studies on PET recycling methods have indicated that mechanical recycling appears to be the most desirable method for the management of PET waste, as compared to chemical recycling and incineration [
12,
13,
14]. The chemical or tertiary recycling method involves depolymerization of the PET polymer by chemical agents (chemolysis) or temperature (pyrolysis) to obtain its constituent monomers, that is, monoethylene glycol (MEG) and terephthalic acid (TA), or their derivatives, which can be used for new polymerization processes [
15,
16]. In addition, the char from pyrolysis of washed PET wastes can successfully replace up to 50 wt% the epoxy resin used in the production of thermosets [
17]. In mechanical or secondary recycling, PET waste is subjected to mechanical processes including shredding, grinding, melting, and, when necessary, as in the case of contaminated articles, is combined with washing and/or drying [
18]. In this process, the polymers stay intact, which permits multiple reuses in the same or similar products. However, mechanical recycling is currently the preferred option for monomaterial packaging since it has the advantages of simplicity and low cost, requires little investment, uses established equipment, and has little adverse environmental impact. Despite these advantages, mechanical recycling of PET is difficult due to complexity and waste contamination [
12,
19,
20]. Another issue observed during the mechanical recycling of PET by “fusion reprocessing” is that the polymer is habitually subjected to chemical, mechanical, thermal, and oxidative degradation, which decreases the molar mass and, finally, causes the deterioration of the performance and transparency of PET articles [
21,
22].
Different studies have conventionally supported the notion that the use of recycled polymer streams in a mixture with virgin polymer of the same nature is a good solution for improving the properties of recycled polymer materials and achieving mechanical recycling. Elamri et al. [
23] investigated fibers from blends of recycled polyethylene terephthalate (r-PET) and virgin PET. An improvement in the melt processing of r-PET and its fibers, with similar mechanical characteristics to those obtained from virgin PET, was reported. In another study, Scarfato and La Mantia [
24] studied recycled and virgin mixtures of polyamide 6 (PA6), showing mechanical and rheological properties similar to those of the virgin polyamide. Moreover, Elamri et al. [
25] also investigated blends of recycled and virgin high-density polyethylene (HDPE), reporting a predictable linear behavior in the mechanical properties as a function of the recycled content. A novel, plausible solution to increase the properties of r-PET during melt reprocessing is the use of additives such as chain extenders [
26,
27,
28,
29]. This option represents a more economical and attractive strategy than chemical recycling processes for monomaterial packaging since these additives can be used during reprocessing cycles by extrusion or injection molding [
26,
27]. Chain extenders are additives containing at least two functional groups that are capable of reacting with the end groups of different macromolecular fragments, thereby creating new covalent bonds. During melt reprocessing, these additives are able to “reconnect” the previously broken polymer chains, leading to a polymer with higher molecular weight (M
W) and restored properties [
27,
28].
The use of chain extenders during PET reprocessing has been widely investigated. Some studies have reported the use of pyromellitic dianhydride (PMDA) [
30,
31], organic phosphites [
32,
33], bis-oxazolines [
34,
35,
36], bis-anhydrides [
30,
37], diisocyanates [
29,
34,
38], diepoxides [
34], epoxy-based oligomers [
34,
39], or oligomeric polyisocyanates [
40]. However, new commercial chain extenders based on random copolymers of poly(styrene-
co-glycidyl methacrylate) (PS-
co-GMA) have recently arisen. In particular, the glycidyl methacrylate (GMA) multi-functionality has excellent affinity with condensation polymers, not only PET, but also polybutylene terephthalate (PBT) or polylactide (PLA). It performs by connecting the polymer chains through a chemical reaction of the GMA groups with the hydroxyl (–OH), carboxyl (–COOH), and amine (–NH
2) terminal groups of the polycondensation polymers. This process can result in a branched structure that also offers higher melt strength for optimal processing conditions. Although some other block copolymers based on GMA groups have been previously used as a chain extenders in other types of polymer blends, such as poly(trimethylene terephthalate) (PTT)/polystyrene (PS) [
41] or PET/polypropylene (PP) [
42], their use in PET systems remains almost unexplored. Only Benvenuta et al. [
43] recently synthesized reactive tri-block copolymers of styrene glycidyl methacrylate (SGMA) and butyl acrylate (BA) (SGMA-
co-BA-
co-SGMA) by reversible addition-fragmentation chain transfer (RAFT) and used them as chain extenders of r-PET. These additives turned out to be very effective at increasing the molar mass and intrinsic viscosity, diminishing the melt flow, and improving the melt elasticity and processability of r-PET.
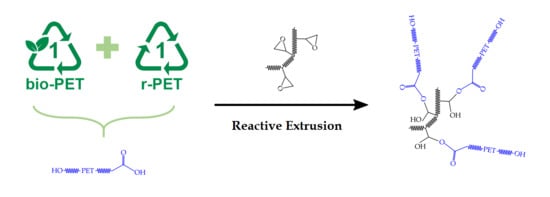
Despite the large amount of work that has been undertaken on the recycling of PET and r-PET blends, those based on bio-PET are barely beginning to be studied. Furthermore, while the price of virgin PET remains stable [
44], the use of r-PET is significantly increasing, and thus, novel and more sustainable technologies for PET recycling could encourage more competitive prices in the industry. In the present work, the properties of bio-PET and pre-consumer (uncontaminated) r-PET blends were analyzed to ascertain the potential for mechanical recycling of the next generation of PET materials. To this end, different amounts of r-PET were melt-mixed with bio-PET in a twin-screw industrial extruder and then shaped into pieces by injection molding. During melt compounding, PS-
co-GMA was added and the effect of the random copolymer on bio-PET/r-PET blends was assessed through a detailed characterization, including measurements of the mechanical, thermal, and thermomechanical properties as well as the rheological behavior.
4. Conclusions
PS-co-GMA, a multi-functional random copolymer, was melt-mixed with bio-PET/r-PET blends at contents of 1–5 phr of polyester blend and shaped into pieces by injection molding. The resultant pieces were characterized to ascertain the potential use of PS-co-GMA as a chain extender for the mechanical recycling of these polyester blends. The results showed that while the incorporation of 1 phr of PS-co-GMA had a slight influence on the mechanical and thermal performance of the bio-PET/r-PET blend pieces, the contents of 3 phr and 5 phr successfully yielded a significant increase in their ductility and toughness. In particular, εb increased from 10.8%, for the blend piece containing 45 wt% of r-PET, to 312.9% and 378.8%, for the same pieces that were melt-processed with 3 phr and 5 phr of PS-co-GMA, respectively. In addition, the impact strength values increased from 1.84 kJ·m−2 to 2.43 kJ·m−2 and 2.52 kJ·m−2, respectively. This mechanical improvement was ascribed to the chain extension mechanism of PET by reactive extrusion due to the reaction of the multiple GMA groups that are present in PS-co-GMA with the –OH and –COOH terminal groups of both bio-PET and r-PET. Furthermore, PS-co-GMA reduced the crystallinity of the PET blends, suggesting that the structural disorder introduced by the chain extender potentially hindered both the nucleation and crystallization of r-PET on the bio-PET chains, contributing to increase material flexibility. In addition, the influence of the reactive copolymer on the thermal stability of the bio-PET/r-PET blends was low, since the blends were already thermally stable up to nearly 400 °C. The thermomechanical properties of the bio-PET/r-PET blends also improved after the addition of PS-co-GMA due to the formation of a branched and larger macromolecule.
Bio-PET can be effectively mixed with its recycled petrochemical counterpart, that is, r-PET, and then mechanically recycled in existing recycling facilities by means of reactive extrusion with PS-co-GMA. Therefore, the original biopolymer properties can be successfully restored and the ultimate performance of bio-PET articles would be retained for a given number of reprocessing cycles. This will permit the recovery of upcoming bio-PET streams with current r-PET waste to manufacture the same or similar products. According to this scenario, mechanical recycling for bio-based but non-biodegradable polymers will be appropriate from both an economic and environmental point of view. This will potentially contribute to accelerating the transition of the plastic packaging industry from its traditional linear model to a more valuable and sustainable circular model.