Dental Sealant Empowered by 1,3,5-Tri Acryloyl Hexahydro-1,3,5-Triazine and α-Tricalcium Phosphate for Anti-Caries Application
Abstract
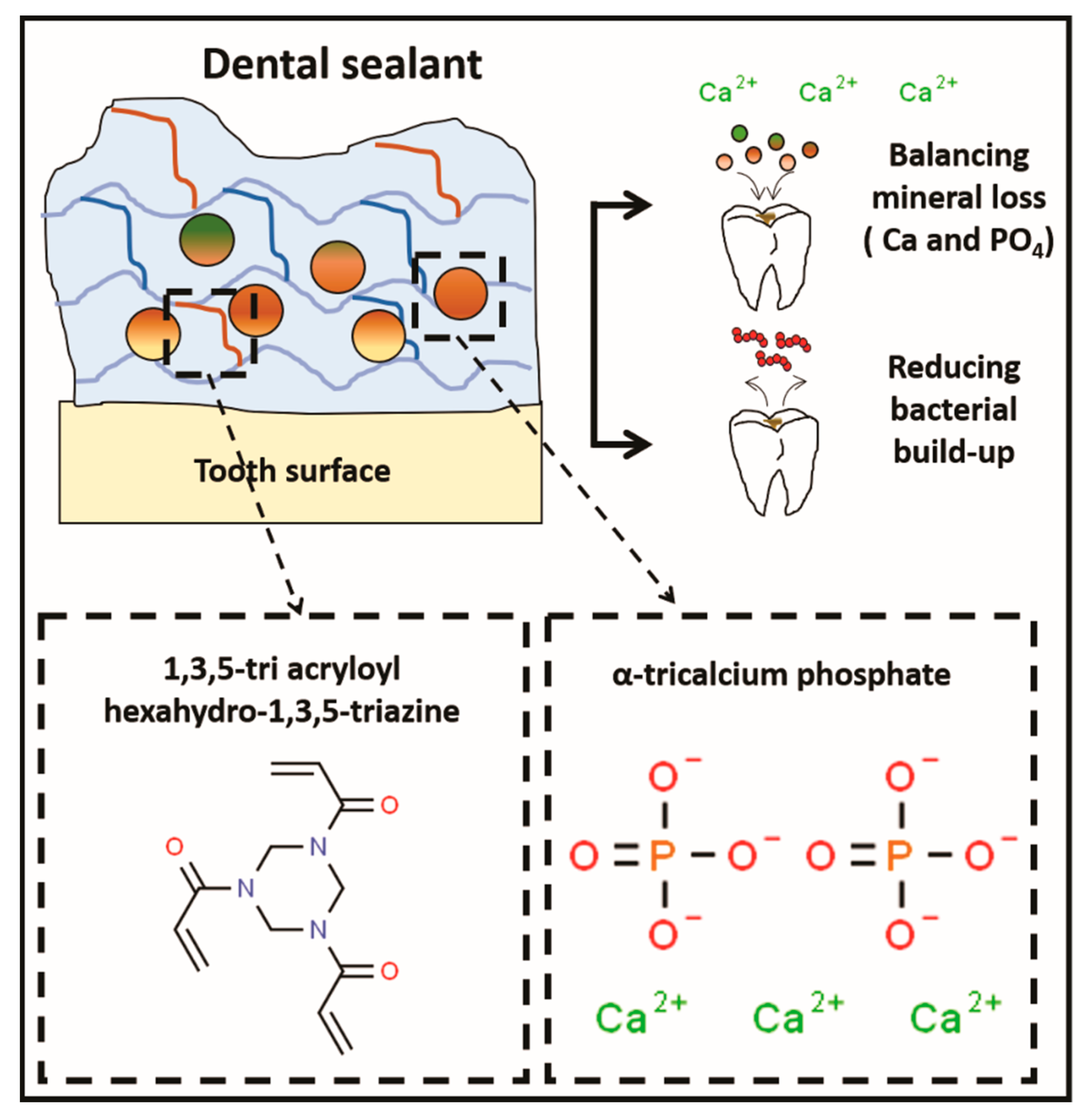
1. Introduction
2. Materials and Methods
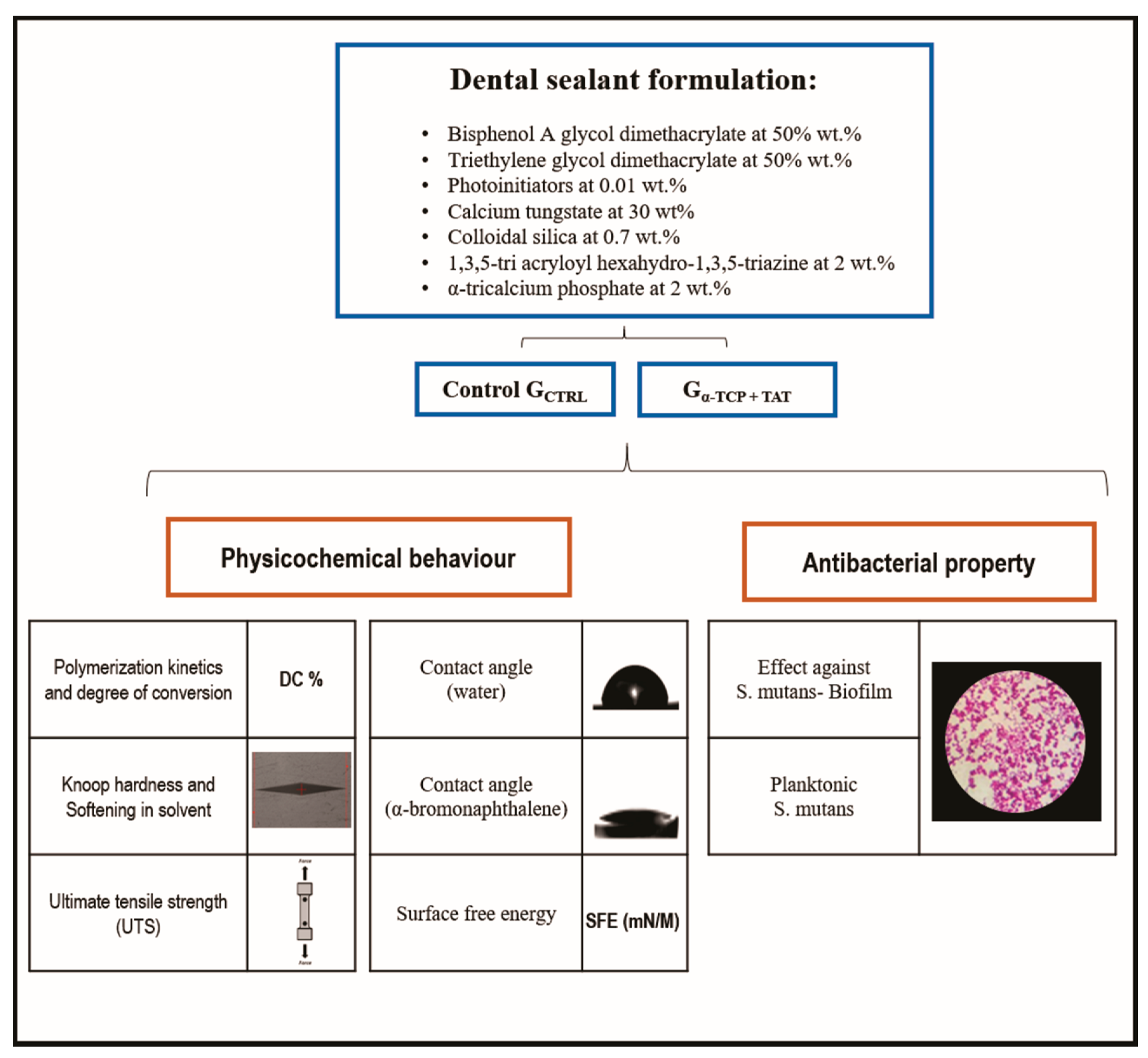
2.1. Resin Sealants Formulation
2.2. Polymerization Kinetics and Degree of Conversion (DC %)
2.3. Knoop Hardness and Softening in Solvent
2.4. Ultimate Tensile Strength (UTS)
2.5. Contact Angle and Surface Free Energy (SFE)
2.6. Activity against Streptococcus Mutans
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Petersen, P.E.; Bourgeois, D.; Ogawa, H.; Estupinan-Day, S.; Ndiaye, C. The global burden of oral diseases and risks to oral health. Bull. World Health Organ. 2005, 83, 661–669. [Google Scholar] [PubMed]
- Hicks, J.; Garcia-Godoy, F.; Flaitz, C. Biological factors in dental caries: Role of saliva and dental plaque in the dynamic process of demineralization and remineralization (part 1). J. Clin. Pediatric Dent. 2003, 28, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Ahovuo-Saloranta, A.; Forss, H.; Walsh, T.; Nordblad, A.; Makela, M.; Worthington, H.V. Pit and fissure sealants for preventing dental decay in permanent teeth. Cochrane Database Syst. Rev. 2017, 7, Cd001830. [Google Scholar] [CrossRef] [PubMed]
- Kuhnisch, J.; Mansmann, U.; Heinrich-Weltzien, R.; Hickel, R. Longevity of materials for pit and fissure sealing--results from a meta-analysis. Dent. Mater. 2012, 28, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.S.; AlQarni, F.D.; Al-Dulaijan, Y.A.; Weir, M.D.; Oates, T.W.; Xu, H.H.K.; Melo, M.A.S. Tuning Nano-Amorphous Calcium Phosphate Content in Novel Rechargeable Antibacterial Dental Sealant. Materials 2018, 11, 1544. [Google Scholar] [CrossRef]
- Zhou, C.; Min, J.; Liu, Z.; Young, A.; Deshazer, H.; Gao, T.; Chang, Y.T.; Kallenbach, N.R. Synthesis and biological evaluation of novel 1,3,5-triazine derivatives as antimicrobial agents. Bioorg. Med. Chem. Lett. 2008, 18, 1308–1311. [Google Scholar] [CrossRef]
- Schiroky, P.R.; Leitune, V.C.B.; Garcia, I.M.; Ogliari, F.A.; Samuel, S.M.W.; Collares, F.M. Triazine Compound as Copolymerized Antibacterial Agent in Adhesive Resins. Braz. Dent. J. 2017, 28, 196–200. [Google Scholar] [CrossRef]
- Altmann, A.S.; Collares, F.M.; Leitune, V.C.; Arthur, R.A.; Takimi, A.S.; Samuel, S.M. In vitro antibacterial and remineralizing effect of adhesive containing triazine and niobium pentoxide phosphate inverted glass. Clin. Oral Investig. 2017, 21, 93–103. [Google Scholar] [CrossRef]
- Altmann, A.S.; Collares, F.M.; Ogliari, F.A.; Samuel, S.M. Effect of methacrylated-based antibacterial monomer on orthodontic adhesive system properties. Am. J. Orthod. Dentofac. Orthop. 2015, 147, S82–S87. [Google Scholar] [CrossRef]
- Cocco, A.R.; Rosa, W.L.; Silva, A.F.; Lund, R.G.; Piva, E. A systematic review about antibacterial monomers used in dental adhesive systems: Current status and further prospects. Dent. Mater. 2015, 31, 1345–1362. [Google Scholar] [CrossRef]
- Khan, A.S.; Syed, M.R. A review of bioceramics-based dental restorative materials. Dent. Mater. J. 2019, 38, 163–176. [Google Scholar] [CrossRef]
- Garcia, I.M.; Leitune, V.C.B.; Samuel, S.M.W.; Collares, F.M. Influence of Different Calcium Phosphates on an Experimental Adhesive Resin. J. Adhes. Dent. 2017, 19, 379–384. [Google Scholar]
- Tezvergil-Mutluay, A.; Seseogullari-Dirihan, R.; Feitosa, V.P.; Cama, G.; Brauer, D.S.; Sauro, S. Effects of composites containing bioactive glassess on demineralized dentin. J. Dent. Res. 2017, 96, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Collares, F.M.; Portella, F.F.; Leitune, V.C.; Samuel, S.M. Discrepancies in degree of conversion measurements by FTIR. Braz. Oral Res. 2013, 27, 453–454. [Google Scholar] [PubMed]
- Owens, D.K.; Wends, R.C. Estimation of the surface free energy of polymers. J. Appl. Polym. Sci. 1969, 13, 1741–1747. [Google Scholar] [CrossRef]
- Collares, F.M.; Ogliari, F.A.; Zanchi, C.H.; Petzhold, C.L.; Piva, E.; Samuel, S.M. Influence of 2-hydroxyethyl methacrylate concentration on polymer network of adhesive resin. J. Adhes. Dent. 2011, 13, 125–129. [Google Scholar]
- Taira, M.; Suzuki, H.; Toyooka, H.; Yamaki, M. Refractive index of inorganic fillers in seven visible-light-cured dental composite resins. J. Mater. Sci. 1994, 13, 68–70. [Google Scholar] [CrossRef]
- Utneja, S.; Talwar, S.; Nawal, R.R.; Sapra, S.; Mittal, M.; Rajain, A.; Verma, M. Evaluation of remineralization potential and mechanical properties of pit and fissure sealants fortified with nano-hydroxyapatite and nano-amorphous calcium phosphate fillers: An in vitro study. J. Conserv. Dent. 2018, 21, 681–690. [Google Scholar] [CrossRef]
- Krumova, M.; Klingshirn, C.; Friedrich, K. Microhardness studies on functionally graded polymer composites. Compos. Sci. Technol. 2001, 61, 557–563. [Google Scholar] [CrossRef]
- Yadav, R.; Kumar, M. Dental restorative composite materials: A review. J. Oral Biosci. 2019, 61, 78–83. [Google Scholar] [CrossRef]
- Lohbauer, U.; Wagner, A.; Belli, R.; Stoetzel, C.; Hilpert, A.; Kurland, H.D.; Grabow, J.; Müller, F.A. Zirconia nanoparticles prepared by laser vaporization as fillers for dental adhesives. Acta Biomater. 2010, 6, 4539–4546. [Google Scholar] [CrossRef] [PubMed]
- Khosravani, M.R. Mechanical behavior of restorative dental composites under various loading conditions. J. Mech. Behav. Biomed. Mater. 2019, 93, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Degrazia, F.W.; Leitune, V.C.B.; Samuel, S.M.W.; Collares, F.M. Boron nitride nanotubes as novel fillers for improving the properties of dental adhesives. J. Dent. 2017, 62, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Paris, S.; Meyer-Lueckel, H.; Colfen, H.; Kielbassa, A.M. Penetration coefficients of commercially available and experimental composites intended to infiltrate enamel carious lesions. Dent. Mater. 2007, 23, 742–748. [Google Scholar] [CrossRef]
- Mena Silva, P.A.; Garcia, I.M.; Nunes, J.; Visioli, F. Myristyltrimethylammonium Bromide (MYTAB) as a Cationic Surface Agent to Inhibit Streptococcus mutans Grown over Dental Resins: An In Vitro Study. J. Funct. Biomater. 2020, 11, 9. [Google Scholar] [CrossRef]
- Ibrahim, M.S.; Ibrahim, A.S.; Balhaddad, A.A.; Weir, M.D.; Lin, N.J.; Tay, F.R.; Oates, T.W.; Xu, H.H.K.; Melo, M.A.S. A Novel Dental Sealant Containing Dimethylaminohexadecyl Methacrylate Suppresses the Cariogenic Pathogenicity of Streptococcus mutans Biofilms. Int. J. Mol. Sci. 2019, 20, 3491. [Google Scholar] [CrossRef]
- Imazato, S.; Kinomoto, Y.; Tarumi, H.; Ebisu, S.; Tay, F.R. Antibacterial activity and bonding characteristics of an adhesive resin containing antibacterial monomer MDPB. Dent. Mater. 2003, 19, 313–319. [Google Scholar] [CrossRef]
- Makvandi, P.; Jamaledin, R.; Jabbari, M.; Nikfarjam, N.; Borzacchiello, A. Antibacterial quaternary ammonium compounds in dental materials: A systematic review. Dent. Mater. 2018, 34, 851–867. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, K.; Zhou, X.; Xu, N.; Xu, H.H.; Weir, M.D.; Ge, Y.; Wang, S.; Li, M.; Li, Y.; et al. Antibacterial effect of dental adhesive containing dimethylaminododecyl methacrylate on the development of Streptococcus mutans biofilm. Int. J. Mol. Sci. 2014, 15, 12791–12806. [Google Scholar] [CrossRef]
- Kreth, J.; Ferracane, J.L.; Pfeifer, C.S.; Khajotia, S.; Merritt, J. At the Interface of Materials and Microbiology: A Call for the Development of Standardized Approaches to Assay Biomaterial-Biofilm Interactions. J. Dent. Res. 2019, 98, 850–852. [Google Scholar] [CrossRef]
- Kirstila, V.; Hakkinen, P.; Jentsch, H.; Vilja, P.; Tenovuo, J. Longitudinal analysis of the association of human salivary antimicrobial agents with caries increment and cariogenic micro-organisms: A two-year cohort study. J. Dent. Res. 1998, 77, 73–80. [Google Scholar] [CrossRef] [PubMed]

| Group | DC % | KHN1 | KHN2 | ΔKHN% | UTS (MPa) |
|---|---|---|---|---|---|
| GCTRL | 51.08 (±3.17) A | 16.41 (±0.97) Aa | 6.10 (±0.53) b | 62.64 (±4.98) A | 46.71 (±3.52) B |
| Gα-TCPTAT | 49.67 (±4.47) A | 18.41 (±2.22) Aa | 6.40 (±0.83) b | 65.02 (±4.83) A | 55.61 (±3.33) A |
| Group | Contact Angle (Water) | Contact Angle (α-Bromonaphthalene) | SFE (mN/M) |
|---|---|---|---|
| GCTRL | 67.71 (±3.32) A | 24.69 (±3.09) A | 48.68 (±1.40) A |
| Gα-TCPTAT | 66.49 (±10.9) A | 26.96 (±3.77) A | 49.08 (±6.05) A |
| Group | Effect against S. Mutans | |
|---|---|---|
| Biofilm | Planktonic Bacteria | |
| GCTRL | 06.38 (±0.57) A | 09.21 (±0.14) A |
| Gα-TCPTAT | 04.95 (±0.30) B | 07.73 (±0.56) B |
| Negative control | - | 09.14 (±0.10) A |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monteiro, J.C.; Stürmer, M.; Garcia, I.M.; Melo, M.A.; Sauro, S.; Leitune, V.C.B.; Collares, F.M. Dental Sealant Empowered by 1,3,5-Tri Acryloyl Hexahydro-1,3,5-Triazine and α-Tricalcium Phosphate for Anti-Caries Application. Polymers 2020, 12, 895. https://doi.org/10.3390/polym12040895
Monteiro JC, Stürmer M, Garcia IM, Melo MA, Sauro S, Leitune VCB, Collares FM. Dental Sealant Empowered by 1,3,5-Tri Acryloyl Hexahydro-1,3,5-Triazine and α-Tricalcium Phosphate for Anti-Caries Application. Polymers. 2020; 12(4):895. https://doi.org/10.3390/polym12040895
Chicago/Turabian StyleMonteiro, Juliana Caletti, Michele Stürmer, Isadora Martini Garcia, Mary Anne Melo, Salvatore Sauro, Vicente Castelo Branco Leitune, and Fabrício Mezzomo Collares. 2020. "Dental Sealant Empowered by 1,3,5-Tri Acryloyl Hexahydro-1,3,5-Triazine and α-Tricalcium Phosphate for Anti-Caries Application" Polymers 12, no. 4: 895. https://doi.org/10.3390/polym12040895
APA StyleMonteiro, J. C., Stürmer, M., Garcia, I. M., Melo, M. A., Sauro, S., Leitune, V. C. B., & Collares, F. M. (2020). Dental Sealant Empowered by 1,3,5-Tri Acryloyl Hexahydro-1,3,5-Triazine and α-Tricalcium Phosphate for Anti-Caries Application. Polymers, 12(4), 895. https://doi.org/10.3390/polym12040895







