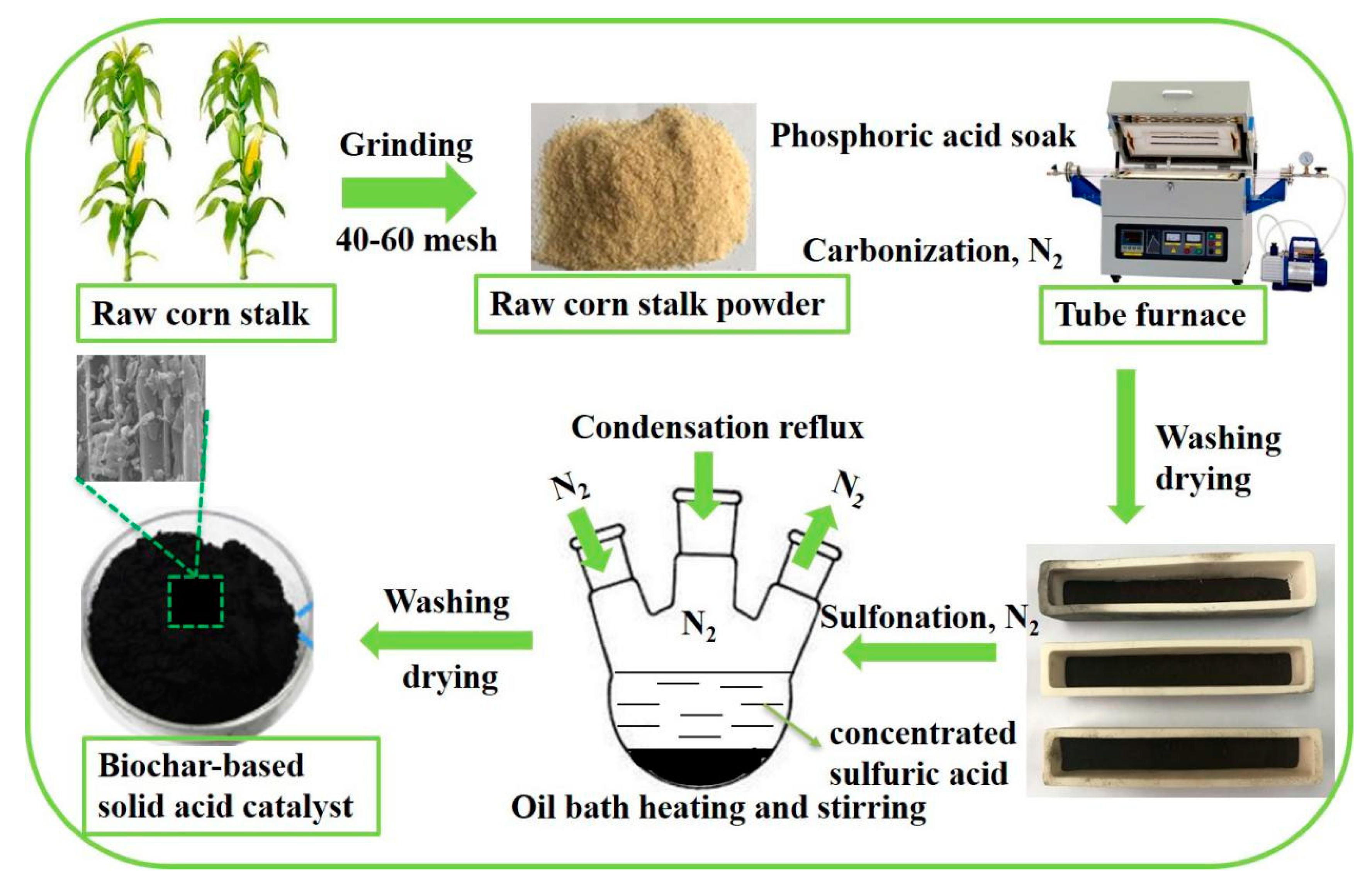
3.2. Acid Amount and Specific Surface Area Analysis
The acid amount and specific surface area analysis of solid acid catalysts are presented in
Table 2. It is observed that the acid amount in these catalysts varied between 1.2 and 2.4 mmol/g. In addition, the specific surface area of these catalysts varied from 1120 to 1640 m
2/g. The acid amount and specific surface area were higher than most of solid acid catalysts [
13,
14,
15]. In comparison with this solid acid catalyst, the products in this study had higher sulfonate content and larger specific surface area. Phosphoric acid activation could provide the conditions to form more pores and holes in the carbonization process [
16,
17]. It is also observed in
Table 2 that the catalyst (SAC-3) produced at a higher carbonization temperature and a longer carbonization time had a larger specific surface area and more sulfonate group (compared to other samples). The larger specific surface area of the sample was due to its larger pore volume and pore opening (
Table 2). In addition, the destruction of the massive structure and the formation of small broken pieces were also conducive to the increase in specific surface area, as confirmed in SEM images that SAC-3 showed more small broken pieces. Therefore, the solid acid catalyst prepared by a higher carbonization temperature and longer carbonization time could immobilize more sulfonate groups in the next sulfonation process, because the porous structures were in favor of the infiltration and diffusion of acid liquor [
18]. These results illustrate that carbonation temperature and time played a crucial role in generating products with a great specific surface area and high acid amount.
The FT-IR spectra of raw corn stalk, carbonized corn stalk and SAC-3 are shown in
Figure 3A. Compared to the raw corn stalk, carbonized corn stalk produced at 800 °C contained both aromatic and aliphatic hydrocarbons, which was indicated by the peaks at approximately 2899 (aliphatic C–H stretching) and 894 cm
−1 (aromatic C–H bending). After sulfonation of the carbonized corn stalk, the most obvious changes are the stretching vibration bands of SAC-3 at around 1210 and 1029 cm
−1 assigning to the O=S=O stretching of –SO
3H, which indicates the successful load of the –SO
3H group on the biochar-based solid acid catalyst [
13]. In addition, the stretching vibration band at 1650 cm
−1 is indicative of the vibration of C=O in the –COOH group [
19]. Moreover, the peak at 3428 cm
−1 was ascribed to the C–OH bending and –OH bending in the –COOH group. This suggests that incomplete carbonization reactions occurred in the carbonization process, resulting in structural changes of the carbon precursor.
In
Figure 3B, the XRD pattern of the synthesized solid acid catalysts exhibited a weak but broad diffraction peak at 2θ = 10°–30°, which was assigned to the amorphous carbon and was interpreted as the reflection of aromatic carbon. Moreover, the presence of the plane (100) also indicated a further carbonization during the sulfonation process, which resulted in the growth of the size of aromatic carbon. Different carbonization temperatures even affected the thermal stability of different solid acid catalysts as shown in
Figure 3C. From the TG curve, all synthesized solid acid catalysts degraded in two stages. The first stage began at about 25 °C, which lost the weight of about 14.98%, 12.75% and 15.64%, respectively, due to the release of residual and bound water inside the internal structure. The second stage degradation started at about 220 °C. In this stage, the weight loss was about 27.74%, 27.41% and 11.18%, respectively, because of the structural degradation of the solid acid catalysts. In general, SAC-3 showed the lowest degradation rate compared to the other two samples due to the high temperature (800 °C) in the preparing process having carbonized the whole structure.
Figure 3D–F shows the XPS spectra and the surface elemental composition of synthesized solid acid catalysts. As expected in
Figure 3D, three solid acid catalysts were dominated by carbon and oxygen peaks, which were the basic elements that made up the constituents of the carbon material. In
Figure 3E, the high-resolution spectrum of C1s peak of SAC-3 shows the presence of different chemical states of carbon on the solid acid catalyst surface. The C1s spectrum was fitted into three separate peaks at 284.9, 286.3, 289.0 eV, which are assigned to the C–C/C–H bond, C–O bond, C=O/O–C=O bond, respectively. The O1s spectrum in
Figure 3F was fitted into two separate peaks at 533.2 and 531.7 eV, which are associated with C–OH and C=O, respectively. This analysis further confirmed the existence of the –COOH and –OH groups.
The elemental components of raw corn stalk and biochar-based solid acid catalysts are listed in
Table 3. It is generally shown that the carbon and hydrogen contents of biochar-based solid acid catalysts dropped compared with the raw corn stalk, while the oxygen and sulfur contents of the samples increased continually from SAC-1 to SAC-3. The drops in carbon and hydrogen are probably attributed to the partial combustion of the material in the carbonation stage, and the increases in the sulfur and oxygen contents are ascribed to the introduction of the sulfonate group on the samples in the sulfonation process. The total concentration of acidic sites determined by acid-based titration was 1.2, 1.8 and 2.4 mmol/g for SAC-1, SAC-2 and SAC-3, respectively (
Table 2). In addition, the concentration of –SO
3H was 0.25, 0.34 and 0.46 mmol/g for SAC-1, SAC-2 and SAC-3, respectively (
Table 3).
To evaluate the catalytic performance of the prepared solid acid catalysts, hydrothermal degradation of lignin was performed for producing various degradation compounds. With the above relative analysis, the sample of SAC-3 was proven to be the most suitable choice to verify the catalytic ability of solid acid. The GC-MS chromatogram of liquid product produced from degradation of lignin under two different conditions are shown in
Figure 4. The chromatograms of a and b were the products of lignin hydrothermal degradation in the absence and presence of biochar-based solid acid catalyst (SAC-3), respectively. Different compounds would appear in different retention times in this analysis. The phenolic compounds, furfural, ester, acetate, benzene, toluene, heterocyclic compounds and long-chain alkanes were detected at 22.5, 30, 32, 18, 15, 20, 27.5 and 12 min, respectively [
20,
21,
22]. In the presence of the catalyst (
Figure 4B), the hydrothermal reaction process was enhanced and the product appeared rapidly after 13 min because of the catalytic effect of the solid acid catalyst. When the degradation of lignin happened, the sulfonic acid group of solid acid catalyst played an important role on activated localization reaction groups and promoted the reaction rate [
23,
24,
25]. Therefore, the products yield and rate were enhanced under the catalytic action of the solid acid catalyst.
Table 4 also lists the mass of products generated from the hydrothermal degradation of lignin with and without the solid acid catalyst. The primary products were aromatic and heterocyclic compounds when the catalyst was used, but without the catalyst under the same conditions, furfural and long-chain alkanes were produced more greatly [
21,
26]. As a contrast, phenolic and guaiacol compounds from lignin-derived oxygenated compounds were produced by using Ni/ZrO
2-SiO
2 catalysts and all of the phenol and guaiacol could be effectively converted into oxygen-free products (cyclohexanol, benzene, methyl-cyclohexane) [
27]. This shows the contents of lignin-derived degradation products are various under different catalytic effects of catalysts.
The stability and recyclability of solid acid catalyst are crucial for its practical application. After the first hydrothermal reaction, the solid residue containing catalyst was recovered from the hydrolytic solution by filtration and washing with hot water. After drying, the catalyst was used for the next run at identical conditions. This recycling process proceeded repeatedly for four times.
Table 5 lists the properties of SAC-3 after 4 runs of reutilization. Interestingly, the specific surface area of the catalyst hardly changed, but its sulfonate group dropped significantly. This change resulted in a slight reduction in the yield of reaction from 96% after the first run to 85% after the fifth run. These results show that the catalyst can be reused reasonably, but the reuse yield went down gradually because of the recovery loss. The yield stability of solid acid catalyst remained acceptable after three runs; this is because our biochar-based materials have relatively large specific surface area and can continuously immobilize more sulfonic acid groups on the surface and inside. After many runs, there were also enough effective functional groups to continue to participate in catalytic reactions. The results in
Table 5 showed that the catalyst was still active in each recycling run, which indicated the stability of the –SO
3H groups on the catalyst. In addition, the sulfonate group and yield of reaction of SAC-1 and SAC-2 were also determined as shown in
Table 6. The sulfonate group of SAC-1 and SAC-2, however, decreased significantly with the increase in the number of recycling times. Therefore, the two samples were not suitable for hydrothermal degradation of lignin. The SAC-3 was chosen to catalyze the hydrothermal degradation of lignin as described in
Figure 4 and
Table 4.