Enhanced Efficiency of Dye-Sensitized Solar Cells Based on Polymer-Assisted Dispersion of Platinum Nanoparticles/Carbon Nanotubes Nanohybrid Films as FTO-Free Counter Electrodes
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of Polymer-Assisted Dispersants
2.3. Preparation of Photoanode
2.4. Preparation of the PtNPs/CNT Hybrid Material
2.5. Preparation of CNTs, PtNPs/CNT Dispersant, and Counter Electrode, and DSSC Packaging
2.6. Characterization and Instruments
3. Results and Discussion
3.1. Influence of Polymer-Assisted Dispersant on the Dispersion of CNTs
3.2. Preparation of Counter Electrodes with CNT/PIB-ED2003-PIB
3.3. Preparation of the Highly Conductive CNT/PtNP Counter Electrode
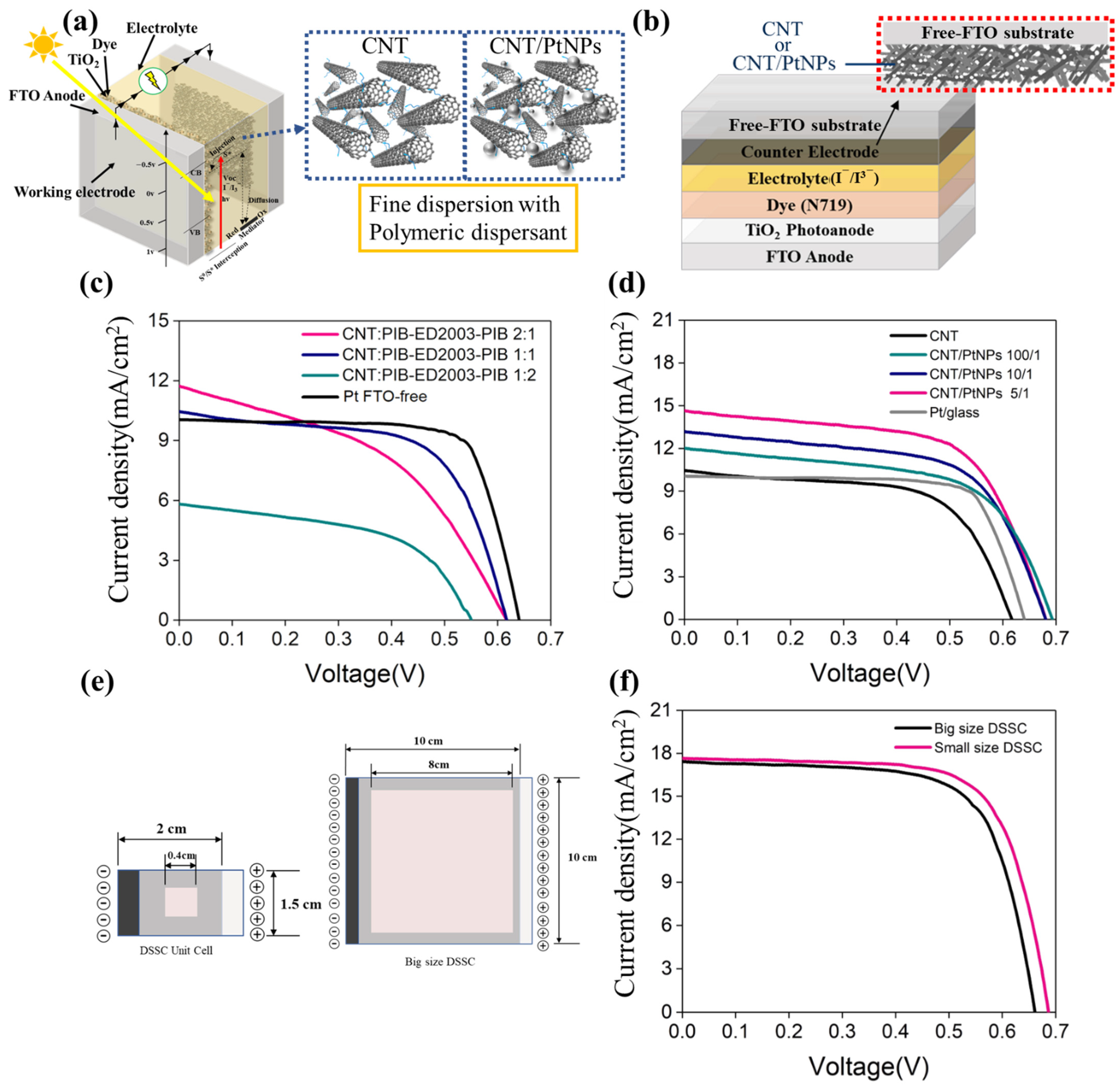
3.4. Fabrication and Photoelectric Conversion Efficiency of DSSCs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Storck, J.L.; Dotter, M.; Adabra, S.; Surjawidjaja, M.; Brockhagen, B.; Grothe, T. Long-term stability improvement of non-toxic dye-sensitized solar cells via poly(ethylene oxide) gel electrolytes for future textile-based solar cells. Polymers 2020, 12, 3035. [Google Scholar] [CrossRef] [PubMed]
- Pezzana, L.; Malmström, E.; Johansson, M.; Sangermano, M. UV-curable bio-based polymers derived from industrial pulp and paper processes. Polymers 2021, 13, 1530. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Chiu, C.W. Facile fabrication of a stretchable and flexible nanofiber carbon film-sensing electrode by electrospinning and its application in smart clothing for ECG and EMG monitoring. ACS Appl. Electron. Mater. 2021, 3, 676–686. [Google Scholar] [CrossRef]
- Soong, Y.C.; Chiu, C.W. Multilayered graphene/boron nitride/thermoplastic polyurethane composite films with high thermal conductivity, stretchability, and washability for adjustable-cooling smart clothes. J. Colloid Interface Sci. 2021, 599, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Ambapuram, M.; Ramireddy, R.; Maddala, G.; Godugunuru, S.; Yerva, P.V.S.; Mitty, R. Effective upconverter and light scattering dual function LiYF4:Er3+/Yb3+ assisted photoelectrode for high performance cosensitized dye sensitized solar cells. ACS Appl. Electron. Mater. 2020, 2, 962–970. [Google Scholar] [CrossRef]
- Manafi, P.; Nazockdast, H.; Karimi, M.; Sadighi, M.; Magagnin, L. Microstructural development and rheological study of a nanocomposite gel polymer electrolyte based on functionalized graphene for dye-sensitized solar cells. Polymers 2020, 12, 1443. [Google Scholar] [CrossRef]
- Selvanathan, V.; Yahya, R.; Ruslan, M.H.; Sopian, K.; Amin, N.; Nour, M.; Sindi, H.; Rawa, M.; Akhtaruzzaman, M. Organosoluble starch-cellulose binary polymer blend as a quasi-solid electrolyte in a dye-sensitized solar cell. Polymers 2020, 12, 516. [Google Scholar] [CrossRef] [Green Version]
- Nagaraj, P.; Sasidharan, A.; David, V.; Sambandam, A. Effect of cross-linking on the performances of starch-based biopolymer as gel electrolyte for dye-sensitized solar cell applications. Polymers 2017, 9, 667. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Wei, S.; Zhou, S.; Wei, X.; Xu, Z.; Wang, Z.; Wei, B.; Lu, X. Theoretical investigation on copper(I) complexes featuring a phosphonic acid anchor with asymmetric ligands for DSSC. ACS Appl. Electron. Mater. 2020, 2, 2141–2150. [Google Scholar] [CrossRef]
- Sudhakar, V.; Singh, A.K.; Chini, M.K. Nanoporous reduced graphene oxide and polymer composites as efficient counter electrodes in dye-sensitized solar cells. ACS Appl. Electron. Mater. 2020, 2, 626–634. [Google Scholar] [CrossRef]
- Ahn, H.J.; Lee, J.S.; Kim, H.S.; Hwang, I.T.; Hong, J.H.; Shin, J.; Jung, C.H. Fabrication of large Pt nanoparticles-decorated rGO counter electrode for highly efficient DSSCs. J. Ind. Eng. Chem. 2018, 65, 318–324. [Google Scholar] [CrossRef]
- Gurulakshmi, M.; Meenakshamma, A.; Susmitha, K.; Charanadhar, N.; Srikanth, V.V.S.S.; Babu, S.N.; Subbaiah, Y.P.V.; Venkateswarlu, K.; Raghavender, M. A Transparent and Pt-free all-carbon nanocomposite counter electrode catalyst for efficient dye sensitized solar cells. Sol. Energy 2019, 193, 568–575. [Google Scholar] [CrossRef]
- Cheng, R.; Gao, X.; Yue, G.; Fan, L.; Gao, Y.; Tan, F. Synthesis of a novel MoIn2S4 alloy film as efficient electrocatalyst for dye-sensitized solar cell. Sol. Energy 2020, 201, 116–121. [Google Scholar] [CrossRef]
- Wu, K.; Wu, Y.; Fu, P.; Yang, D.; Ruan, B.; Wu, M.; Wu, R. Composites of vanadium (III) oxide (V2O3) incorporating with amorphous C as Pt-free counter electrodes for low-cost and high-performance dye-sensitized solar cells. ACS Omega 2021, 6, 11183–11191. [Google Scholar] [CrossRef]
- Sarkar, A.; Chakraborty, A.K.; Bera, S. NiS/rGO nanohybrid: An excellent counter electrode for dye sensitized solar cell. Sol. Energy Mater. Sol. Cells 2018, 182, 314–320. [Google Scholar] [CrossRef]
- Xu, T.; Kong, D.; Tang, H.; Qin, X.; Li, X.; Gurung, A.; Kou, K.; Chen, L.; Qiao, Q.; Huang, W. Transparent MoS2/PEDOT composite counter electrodes for bifacial dye-sensitized solar cells. ACS Omega 2020, 5, 8687–8696. [Google Scholar] [CrossRef] [Green Version]
- Demir, E.; Savk, A.; Sen, B.; Sen, F. A novel monodisperse metal nanoparticles anchored graphene oxide as counter electrode for dye-sensitized solar cells. Nano-Struct. Nano-Objects 2017, 12, 41–45. [Google Scholar] [CrossRef]
- Sarkar, A.; Bera, S.; Chakraborty, A.K. CoNi2S4-reduced graphene oxide nanohybrid: An excellent counter electrode for Pt-free DSSC. Sol. Energy 2020, 208, 139–149. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, P.; Zhang, T.; Gou, B. High-efficiency dye-sensitized solar cells based on kesterite Cu2ZnSnSe4 inlaid on a flexible carbon fabric composite counter electrode. ACS Omega 2020, 5, 24898–24905. [Google Scholar] [CrossRef]
- Wang, T.; Li, Y.; Li, H.; Shi, D.; Jiao, Q.; Zhao, Y.; Su, P.; Wang, W.; Wu, Q. Rational design of hierarchical structural CoSe@NPC/CoSe@CNT nanocomposites derived from metal–organic frameworks as a robust Pt-free electrocatalyst for dye-sensitized solar cells. ACS Omega 2020, 5, 26253–26261. [Google Scholar] [CrossRef] [PubMed]
- O’regan, B.; Grätzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Mustafa, M.N.; Sulaiman, Y. Review on The effect of compact layers and light scattering layers on the enhancement of dye-sensitized solar cells. Sol. Energy 2021, 215, 26–43. [Google Scholar] [CrossRef]
- Kumar, D.K.; Kříž, J.; Bennett, N.; Chen, B.; Upadhayaya, H.; Reddy, K.R.; Sadhu, V. Functionalized metal oxide nanoparticles for efficient dye-sensitized solar cells (DSSCs): A review. Mater. Sci. Energy Technol. 2020, 3, 472–481. [Google Scholar] [CrossRef]
- Wang, C.; Jing, Y.; Zhou, X.; Li, Y.F. Sb2TeSe2 monolayers: Promising 2D semiconductors for highly efficient excitonic solar cells. ACS Omega 2021, 6, 20590–20597. [Google Scholar] [CrossRef]
- Ansari, S.A.; Goumri-Said, S.; Yadav, H.M.; Belarbi, M.; Aljaafari, A.; Kanoun, M.B. Directly grown of NiCo2S4 nanoparticles on a conducting substrate towards the high-performance counter electrode in dye-sensitized solar cell: A combined theoretical and experimental study. Sol. Energy Mater. Sol. Cells 2021, 225, 111064. [Google Scholar] [CrossRef]
- Suriani, A.B.; Mohamed, A.; Hashim, N.; Rosmi, M.S.; Mamat, M.H.; Malek, M.F.; Mamat, M.H.; Malek, M.F.; Salifairus, M.J.; Khalil, H.A. Reduced graphene oxide/platinum hybrid counter electrode assisted by custom-made triple-tail surfactant and zinc oxide/titanium dioxide bilayer nanocomposite photoanode for enhancement of DSSCs photovoltaic performance. Optik 2018, 161, 70–83. [Google Scholar] [CrossRef]
- Kim, G.H.; Park, S.H.; Birajdar, M.S.; Lee, J.; Hong, S.C. Core/shell structured carbon nanofiber/platinum nanoparticle hybrid web as a counter electrode for dye-sensitized solar cell. J. Ind. Eng. Chem. 2017, 52, 211–217. [Google Scholar] [CrossRef]
- Yeh, M.H.; Lin, L.Y.; Chang, L.Y.; Leu, Y.A.; Cheng, W.Y.; Lin, J.J.; Ho, K.C. Dye-sensitized solar cells with reduced graphene oxide as the counter electrode prepared by a green photothermal reduction process. ChemPhysChem 2014, 15, 1175–1181. [Google Scholar] [CrossRef] [PubMed]
- Chou, J.C.; Yang, C.M.; Chang, J.X.; Lai, C.H.; Nien, Y.H.; Kuo, P.Y.; Lin, Y.C.; Yong, Z.R. Performance of dye-sensitized solar cell based on AZO/TiO2 photoelectrode and RGO@TiO2/Pt counter electrode. J. Innov. Technol. 2021, 3, 27–33. [Google Scholar]
- Dang, C.L.T.; Van Le, C.; Nguyen, T.D.; Le, N.T.T.; Nguyen, K.D.; Nguyen, M.T.T.; Le, H.K.; Ho, D.H.; Nguyen, H.T.; Pham, C.T.L.; et al. Effect of synthesis conditions of platinum/reduced graphene oxide nanocomposites on the electrochemical behaviors of cathodes in dye-sensitized solar cells. J. Electron. Mater. 2021, 50, 1425–1433. [Google Scholar] [CrossRef]
- Shih, P.T.; Dong, R.X.; Shen, S.Y.; Vittal, R.; Lin, J.J.; Ho, K.C. Transparent graphene—Platinum nanohybrid films for counter electrodes in high efficiency dye-sensitized solar cells. J. Mater. Chem. A 2014, 2, 8742–8748. [Google Scholar] [CrossRef]
- Chen, P.W.; Lee, C.P.; Chang, L.Y.; Chang, J.; Yeh, M.H.; Lin, L.Y.; Vittal, R.; Lin, J.J.; Ho, K.C. Dye-sensitized solar cells with low-cost catalytic films of polymer-loaded carbon black on their counter electrode. RSC Adv. 2013, 3, 5871–5881. [Google Scholar] [CrossRef]
- Oh, W.C.; Areerob, Y. A new aspect for band gap energy of graphene-Mg2CuSnCoO6-gallic acid as a counter electrode for enhancing dye-sensitized solar cell performance. ACS Appl. Mater. Interfaces 2019, 11, 38859–38867. [Google Scholar] [CrossRef] [PubMed]
- Younas, M.; Gondal, M.A.; Dastageer, M.A.; Harrabi, K. Efficient and cost-effective dye-sensitized solar cells using MWCNT-TiO2 nanocomposite as photoanode and MWCNT as Pt-free counter electrode. Sol. Energy 2019, 188, 1178–1188. [Google Scholar] [CrossRef]
- Younas, M.; Gondal, M.A.; Dastageer, M.A.; Baig, U. Fabrication of cost effective and efficient dye sensitized solar cells with WO3-TiO2 nanocomposites as photoanode and MWCNT as Pt-free counter electrode. Ceram. Int. 2019, 45, 936–947. [Google Scholar] [CrossRef]
- Chen, H.; Wang, J.; Jia, C.; Mou, J.; Zhu, L. Highly efficient dye-sensitized solar cell with a novel nanohybrid film of Cu2ZnSnS4-MWCNTs as counter electrode. Appl. Surf. Sci. 2017, 422, 591–596. [Google Scholar] [CrossRef]
- Samantaray, M.R.; Mondal, A.K.; Murugadoss, G.; Pitchaimuthu, S.; Das, S.; Bahru, R.; Mohamed, M.A. Synergetic effects of hybrid carbon nanostructured counter electrodes for dye-sensitized solar cells: A review. Materials 2020, 13, 2779. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.C.; Wang, Y.C.; Dong, R.X.; Tsai, W.C.; Tsai, K.W.; Wang, C.C.; Chen, T.H.; Vittal, R.; Lin, J.J.; Ho, K.C. A high performance dye-sensitized solar cell with a novel nanocomposite film of PtNP/MWCNT on the counter electrode. J. Mater. Chem. 2010, 20, 4067–4073. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.T.; Wang, Y.C.; Dong, R.X.; Wang, C.C.; Huang, K.C.; Vittal, R.; Ho, K.C.; Lin, J.J. A dual-functional Pt/CNT TCO-free counter electrode for dye-sensitized solar cell. J. Mater. Chem. 2012, 22, 25311–25315. [Google Scholar] [CrossRef]
- Sun, J.T.; Li, J.W.; Tsou, C.H.; Pang, J.C.; Chung, R.J.; Chiu, C.W. Polyurethane/nanosilver-doped halloysite nanocomposites: Thermal, mechanical properties, and antibacterial properties. Polymers 2020, 12, 2729. [Google Scholar] [CrossRef]
- Chiu, C.W.; Lee, Y.C.; Ou, G.B.; Cheng, C.C. Controllable 3D hot-junctions of silver nanoparticles stabilized by amphiphilic tri-block copolymer/graphene oxide hybrid surfactants for use as surface-enhanced Raman scattering substrates. Ind. Eng. Chem. Res. 2017, 56, 2935–2942. [Google Scholar] [CrossRef]
- Chiu, C.W.; Li, J.W.; Huang, C.Y.; Yang, S.S.; Soong, Y.C.; Lin, C.L.; Lee, J.C.M.; Lee Sanchez, W.A.; Cheng, C.C.; Suen, M.C. Controlling the structures, flexibility, conductivity stability of three-dimensional conductive networks of silver nanoparticles/carbon-based nanomaterials with nanodispersion and their application in wearable electronic sensors. Nanomaterials 2020, 10, 1009. [Google Scholar] [CrossRef]
- Lee, Y.C.; Chiu, C.W. Immobilization and 3D hot-junction formation of gold nanoparticles on two-dimensional silicate nanoplatelets as substrates for high-efficiency surface-enhanced Raman scattering detection. Nanomaterials 2019, 9, 324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiu, C.W.; Lin, C.A.; Hong, P.D. Melt-spinning and thermal stability behavior of TiO2 nanoparticle/polypropylene nanocomposite fibers. J. Polym. Res. 2010, 18, 367–372. [Google Scholar] [CrossRef]
- Sarwar, S.; Park, S.; Dao, T.T.; Lee, M.S.; Ullah, A.; Hong, S.; Han, C.H. Scalable photoelectrochromic glass of high performance powered by ligand attached TiO2 photoactive layer. Sol. Energy Mater. Sol. Cells 2020, 210, 110498. [Google Scholar] [CrossRef]
- Li, Y.P.; Cao, H.B.; Liu, C.M.; Zhang, Y. Electrochemical reduction of nitrobenzene at carbon nanotube electrode. J. Hazard. Mater. 2007, 148, 158–163. [Google Scholar] [CrossRef]
- Liu, J.; Wen, Y.; van Aken, P.A.; Maier, J.; Yu, Y. In situ reduction and coating of SnS2 nanobelts for free-standing SnS@polypyrrole-nanobelt/carbon-nanotube paper electrodes with superior Li-ion storage. J. Mater. Chem. A 2015, 3, 5259–5265. [Google Scholar] [CrossRef]



| Counter Electrode | Epp (V) a | Ipc (mA cm−2) b | Sheet Resistance (ohm/sq.) |
|---|---|---|---|
| CNT:PIB-ED2003-PIB 2:1 | 0.93 | 6.09 | -- |
| CNT:PIB-ED2003-PIB 1:1 | 0.73 | 4.75 | 8.45 |
| CNT:PIB-ED2003-PIB 1:2 | 1.23 | 3.55 | -- |
| CNT-ED2003/PtNPs 100/1 c | 1.01 | 3.93 | 8.35 |
| CNT-ED2003/PtNPs 10/1 c | 0.89 | 4.97 | 7.59 |
| CNT-ED2003/PtNPs 5/1 c | 0.51 | 5.70 | 7.25 |
| Pt d | 0.58 | 4.40 | -- |
| Counter Electrodes a | VOC (V) | JSC (mA/cm2) | FF | η (%) |
|---|---|---|---|---|
| CNT:PIB-ED2003-PIB 2:1 | 0.62 | 11.82 | 0.44 | 3.22 |
| CNT:PIB-ED2003-PIB 1:1 | 0.62 | 10.40 | 0.63 | 4.03 |
| CNT:PIB-ED2003-PIB 1:2 | 0.55 | 5.84 | 0.57 | 1.83 |
| CNT-PIB-ED2003-PIB/PtNPs 100/1 b | 0.69 | 11.94 | 0.60 | 4.94 |
| CNT-PIB-ED2003-PIB/PtNPs 10/1 b | 0.68 | 13.32 | 0.61 | 5.52 |
| CNT-PIB-ED2003-PIB/PtNPs 5/1 b | 0.68 | 14.63 | 0.63 | 6.28 |
| Pt c | 0.64 | 10.04 | 0.68 | 4.37 |
| Counter Electrode a | VOC (V) | JSC (mA/cm2) | FF | η (%) |
|---|---|---|---|---|
| CNT:PIB-ED2003-PIB 1:1 | 0.65 | 10.82 | 0.64 | 4.52 |
| CNT-PIB-ED2003-PIB/PtNPs 100/1 b | 0.64 | 11.49 | 0.64 | 4.67 |
| CNT-PIB-ED2003-PIB/PtNPs 10/1 b | 0.66 | 15.78 | 0.69 | 7.19 |
| CNT-PIB-ED2003-PIB/PtNPs 5/1 b | 0.68 | 17.50 | 0.71 | 8.45 |
| Pt c | 0.68 | 18.65 | 0.63 | 7.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.-W.; Chen, Y.-S.; Chen, Y.-F.; Chen, J.-X.; Kuo, C.-F.J.; Chen, L.-Y.; Chiu, C.-W. Enhanced Efficiency of Dye-Sensitized Solar Cells Based on Polymer-Assisted Dispersion of Platinum Nanoparticles/Carbon Nanotubes Nanohybrid Films as FTO-Free Counter Electrodes. Polymers 2021, 13, 3103. https://doi.org/10.3390/polym13183103
Li J-W, Chen Y-S, Chen Y-F, Chen J-X, Kuo C-FJ, Chen L-Y, Chiu C-W. Enhanced Efficiency of Dye-Sensitized Solar Cells Based on Polymer-Assisted Dispersion of Platinum Nanoparticles/Carbon Nanotubes Nanohybrid Films as FTO-Free Counter Electrodes. Polymers. 2021; 13(18):3103. https://doi.org/10.3390/polym13183103
Chicago/Turabian StyleLi, Jia-Wun, Yu-Sheng Chen, Yan-Feng Chen, Jian-Xun Chen, Chung-Feng Jeffrey Kuo, Liang-Yih Chen, and Chih-Wei Chiu. 2021. "Enhanced Efficiency of Dye-Sensitized Solar Cells Based on Polymer-Assisted Dispersion of Platinum Nanoparticles/Carbon Nanotubes Nanohybrid Films as FTO-Free Counter Electrodes" Polymers 13, no. 18: 3103. https://doi.org/10.3390/polym13183103
APA StyleLi, J.-W., Chen, Y.-S., Chen, Y.-F., Chen, J.-X., Kuo, C.-F. J., Chen, L.-Y., & Chiu, C.-W. (2021). Enhanced Efficiency of Dye-Sensitized Solar Cells Based on Polymer-Assisted Dispersion of Platinum Nanoparticles/Carbon Nanotubes Nanohybrid Films as FTO-Free Counter Electrodes. Polymers, 13(18), 3103. https://doi.org/10.3390/polym13183103









