FTIR, Weight, and Surface Morphology of Poly(vinyl chloride) Doped with Tin Complexes Containing Aromatic and Heterocyclic Moieties
Abstract
:1. Introduction
2. Materials and Methods
2.1. General
2.2. Synthesis of Complex 1
2.3. Synthesis of Complexes 2 and 3
2.4. Preparation of PVC Films
2.5. Irradiation of PVC Films
2.6. Measurement of PVC Functional Group Indices
2.7. Measurement of PVC Weight Loss
3. Results and Discussion
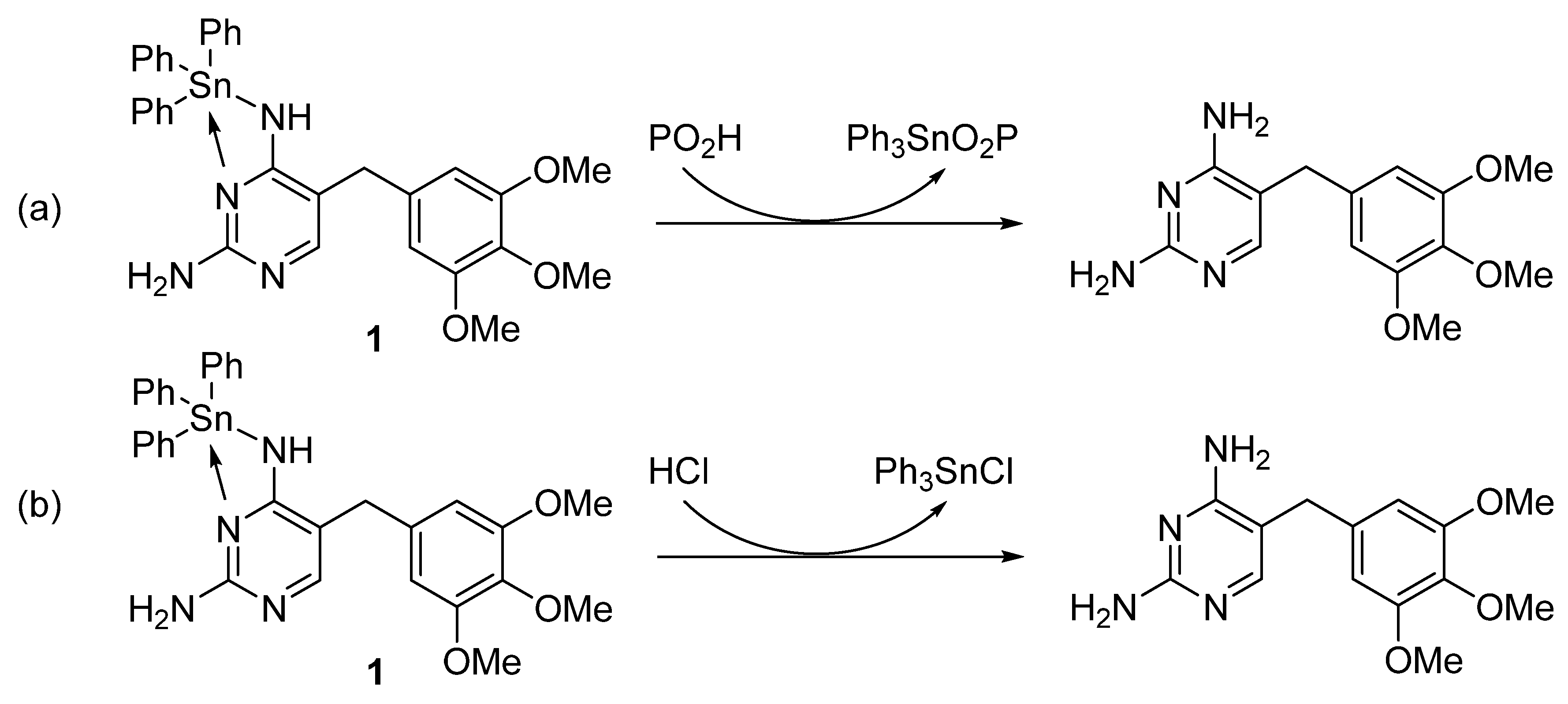
3.1. Synthesis of Trimethoprim–tin Complexes 1–3
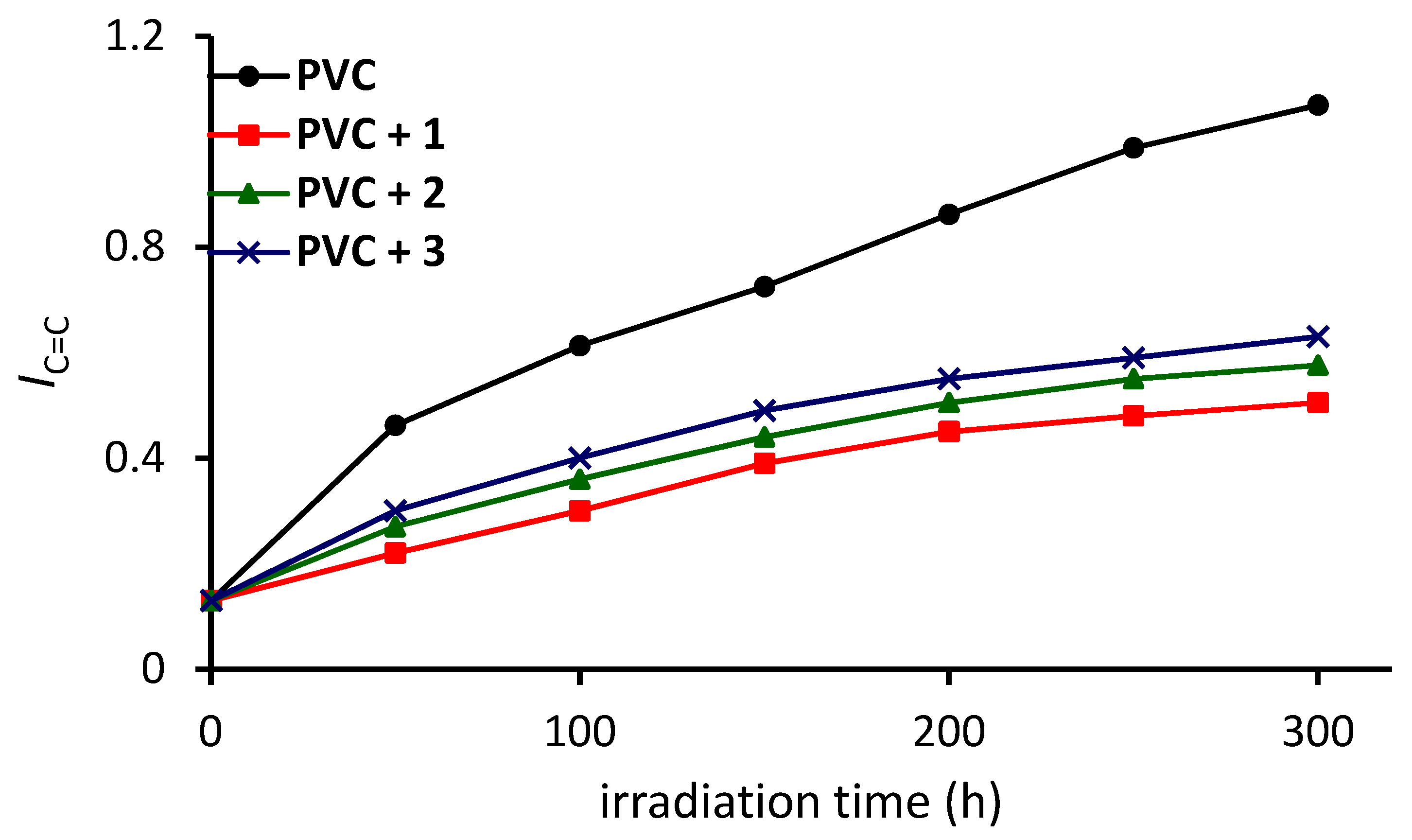
3.2. Investigation of PVC Photodegradation through FTIR Spectrophotometry
3.3. Investigation of PVC Photodegradation through Weight Loss
3.4. Investigation of PVC Photodegradation through Surface Morphology
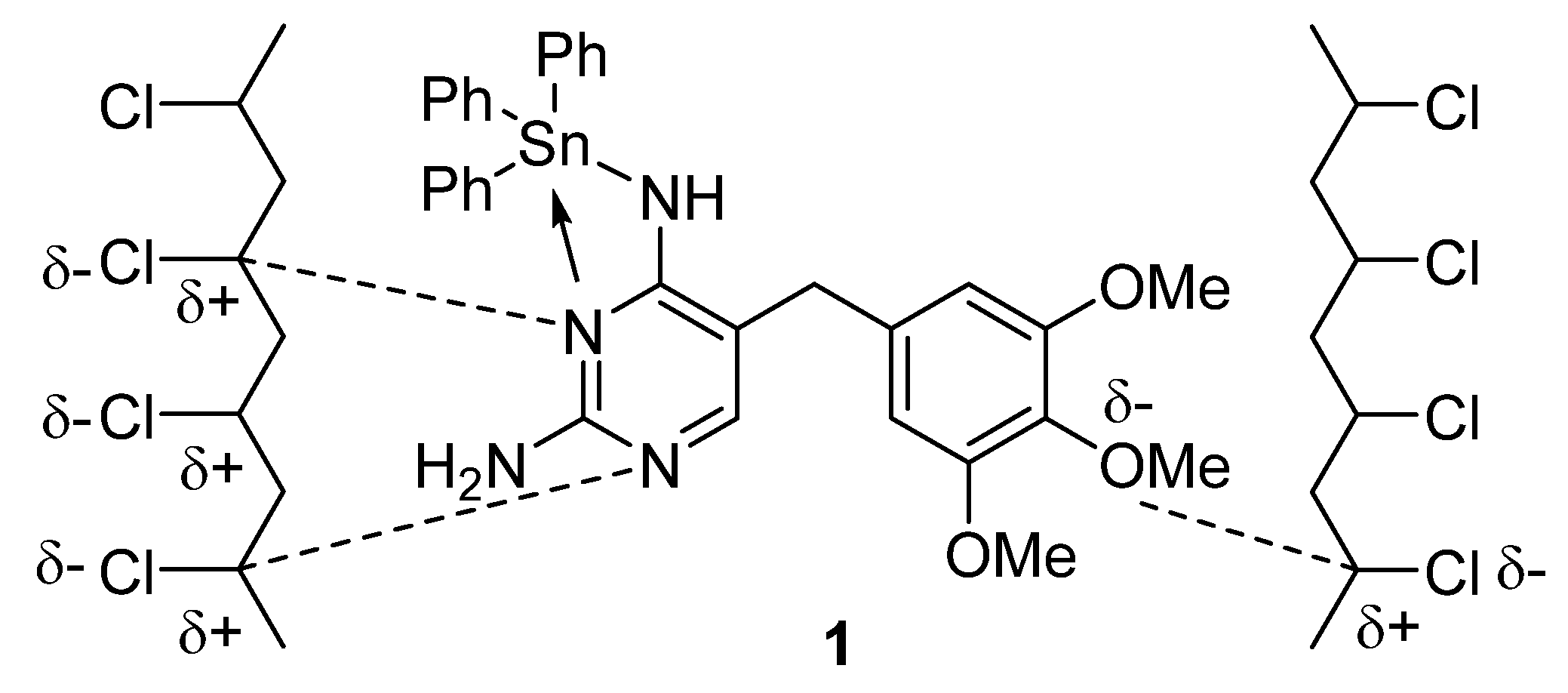
3.5. PVC Photostabilization Mechanisms
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Millet, H.; Vangheluwe, P.; Block, C.; Sevenster, A.; Garcia, L.; Antonopoulos, R. The Nature of Plastics and Their Societal Usage. In Plastics and the Environment; Royal Society of Chemistry: Cambridge, UK, 2018; pp. 1–20. [Google Scholar] [CrossRef]
- Andrady, A.L.; Neal, M.A. Applications and societal benefits of plastics. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 1977–1984. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [Green Version]
- Plastics—The Facts 2019. An Analysis of European Plastics Production, Demand and Waste Data. Available online: https://www.plasticseurope.org/application/files/9715/7129/9584/FINAL_web_version_Plastics_the_facts2019_14102019.pdf (accessed on 24 August 2021).
- Ma, Y.-F.; Liao, S.-L.; Li, Q.-G.; Guan, Q.; Jia, P.-Y.; Zhou, Y.-H. Physical and chemical modifications of poly(vinyl chloride) materials to prevent plasticizer migration—Still on the run. React. Funct. Polym. 2019, 147, 104458. [Google Scholar] [CrossRef]
- Patrick, S.G. Practical Guide to Polyvinyl Chloride; Rapra Technology Limited: Shrewsbury, UK, 2005. [Google Scholar]
- Guo, Y.; Leroux, F.; Tian, W.; Li, D.; Tang, P.; Feng, Y. Layered double hydroxides as thermal stabilizers for poly(vinyl chloride): A review. Appl. Clay Sci. 2021, 211, 106198. [Google Scholar] [CrossRef]
- Walters, P.; Cadogan, D.F.; Howick, C.J. Plasticizers. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2020. [Google Scholar] [CrossRef]
- Maeda, Y.; Paul, D.R. Effect of antiplasticization on gas sorption and transport. I. Polysulfone. J. Polym. Sci. Part B Polym. Phys. 1987, 25, 957–980. [Google Scholar] [CrossRef]
- Maeda, Y.; Paul, D.R. Effect of antiplasticization on gas sorption and transport. III. Free volume interpretation. J. Polym. Sci. Part B Polym. Phys. 1987, 25, 1005–1016. [Google Scholar] [CrossRef]
- Wypych, G. PVC Degradation and Stabilization, 3rd ed.; ChemTec Publishing: Scarborough, ON, Canada, 2015. [Google Scholar] [CrossRef]
- Huang, Z.; Ding, A.; Guo, H.; Lu, G.; Huang, X. Construction of nontoxic polymeric UV-absorber with great resistance to UV-photoaging. Sci. Rep. 2016, 6, 25508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabek, J.F. Polymer Photodegradation—Mechanisms and Experimental Methods; Chapman & Hall: London, UK, 1995. [Google Scholar]
- Veronelli, M.; Mauro, M.; Bresadpla, S. Influence of thermal dehydrochlorination on the photooxidation kinetics of PVC samples. Polym. Degrad. Stab. 1999, 66, 349–357. [Google Scholar] [CrossRef]
- Yu, J.; Sun, L.; Ma, C.; Qiao, Y.; Yao, H. Thermal degradation of PVC: A review. Waste Manag. 2016, 48, 300–314. [Google Scholar] [CrossRef]
- Lucarini, M.; Pedulli, G.F. Free radical intermediates in the inhibition of the autoxidation reaction. Chem. Soc. Rev. 2010, 39, 2106–2119. [Google Scholar] [CrossRef]
- Gryn’ova, G.; Hodgson, J.L.; Coote, M.L. Revising the mechanism of polymer autooxidation. Org. Biomol. Chem. 2011, 9, 480–490. [Google Scholar] [CrossRef]
- Karayıldırım, T.; Yanık, J.; Yüksel, M.; Saglam, M.; Haussmann, M. Degradation of PVC containing mixtures in the presence of HCl fixators. J. Polym. Environ. 2005, 13, 365–379. [Google Scholar] [CrossRef]
- Mohamed, N.A.; Yassin, A.A.; Khalil, K.D.; Sabaa, M.W. Organic thermal stabilizers for rigid poly(vinyl chloride) I. Barbituric and thiobarbituric acids. Polym. Degrad. Stab. 2000, 70, 5–10. [Google Scholar] [CrossRef]
- Grossman, R.F. Mixed metal vinyl stabilizer synergism. II: Reactions with zinc replacing cadmium. J. Vinyl Addit. Technol. 1990, 12, 142–145. [Google Scholar] [CrossRef]
- Porta, M.; Zumeta, E. Implementing the Stockholm treaty on persistent organic pollutants. Occup. Environ. Med. 2002, 59, 651–652. [Google Scholar] [CrossRef] [Green Version]
- Li, D.; Xie, L.; Fu, M.; Zhang, J.; Indrawirawan, S.; Zhang, Y.; Tang, S. Synergistic effects of lanthanum-pentaerythritol alkoxide with zinc stearates and with beta-diketone on the thermal stability of poly(vinyl chloride). Polym. Degd. Stab. 2015, 114, 52–59. [Google Scholar] [CrossRef]
- Fu, M.; Li, D.; Liu, H.; Ai, H.; Zhang, Y.; Zhang, L. Synergistic effects of zinc-mannitol alkoxide with calcium/zinc stearates and with β-diketone on thermal stability of rigid poly(vinyl chloride). J. Polym. Res. 2016, 23, 13. [Google Scholar] [CrossRef]
- El-Hiti, G.A.; Ahmed, D.S.; Yousif, E.; Alotaibi, M.H.; Star, H.A.; Ahmed, A.A. Influence of polyphosphates on the physicochemical properties of poly(vinyl chloride) after irradiation with ultraviolet light. Polymers 2020, 12, 193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, D.; Khanal, S.; Zhang, C.; Xu, S. Photodegradation of polybenzimidazole/polyvinyl chloride composites and polybenzimidazole: Density functional theory and experimental study. J. Appl. Polym. Sci. 2021, 138, 49693. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Chaudhuri, B.; Bhattacharjee, S.; Das, P.; Dutta, B.K. Degradation mechanism and kinetic model for photocatalytic oxidation of PVC-ZnO composite film in presence of a sensitizing dye and UV radiation. J. Hazard. Mater. 2008, 154, 230–236. [Google Scholar] [CrossRef]
- Yang, T.C.; Noguchi, T.; Isshiki, M.; Wu, J.H. Effect of titanium dioxide particles on the surface morphology and the mechanical properties of PVC composites during QUV accelerated weathering. Polym. Compos. 2016, 37, 3391–3397. [Google Scholar] [CrossRef]
- Yang, T.C.; Noguchi, T.; Isshiki, M.; Wu, J.H. Effect of titanium dioxide on chemical and molecular changes in PVC sidings during QUV accelerated weathering. Polym. Degrad. Stab. 2014, 104, 33–39. [Google Scholar] [CrossRef]
- Schiller, M. PVC Additives: Performance, Chemistry, Developments, and Sustainability; Carl Hanser Verlag: Munich, Germany, 2015. [Google Scholar]
- Ahmed, A.A.; Ahmed, D.S.; El-Hiti, G.A.; Alotaibi, M.H.; Hashim, H.; Yousif, E. SEM morphological analysis of irradiated polystyrene film doped by a Schiff base containing a 1,2,4-triazole ring system. Appl. Petrochem. Res. 2019, 9, 169–177. [Google Scholar] [CrossRef] [Green Version]
- Hashim, H.; El-Hiti, G.A.; Alotaibi, M.H.; Ahmed, D.S.; Yousif, E. Fabrication of ordered honeycomb porous poly(vinyl chloride) thin film doped with a Schiff base and nickel(II) chloride. Heliyon 2018, 4, e00743. [Google Scholar] [CrossRef] [Green Version]
- Shaalan, N.; Laftah, N.; El-Hiti, G.A.; Alotaibi, M.H.; Muslih, R.; Ahmed, D.S.; Yousif, E. Poly(vinyl chloride) photostabilization in the presence of Schiff bases containing a thiadiazole moiety. Molecules 2018, 23, 913. [Google Scholar] [CrossRef] [Green Version]
- Ali, G.Q.; El-Hiti, G.A.; Tomi, I.H.R.; Haddad, R.; Al-Qaisi, A.J.; Yousif, E. Photostability and performance of polystyrene films containing 1,2,4-triazole-3-thiol ring system Schiff bases. Molecules 2016, 21, 1699. [Google Scholar] [CrossRef] [Green Version]
- Balakit, A.A.; Ahmed, A.; El-Hiti, G.A.; Smith, K.; Yousif, E. Synthesis of new thiophene derivatives and their use as photostabilizers for rigid poly(vinyl chloride). Int. J. Polym. Sci. 2015, 2015, 510390. [Google Scholar] [CrossRef]
- Ahmed, A.; El-Hiti, G.A.; Hadi, A.G.; Ahmed, D.S.; Baashen, M.A.; Hashim, H.; Yousif, E. Photostabilization of poly(vinyl chloride) films blended with organotin complexes of mefenamic acid for outdoor applications. Appl. Sci. 2021, 11, 2853. [Google Scholar] [CrossRef]
- Mousa, O.G.; El-Hiti, G.A.; Baashen, M.A.; Bufaroosha, M.; Ahmed, A.; Ahmed, A.A.; Ahmed, D.S.; Yousif, E. Synthesis of carvedilol-organotin complexes and their effects on reducing photodegradation of poly(vinyl chloride). Polymers 2021, 13, 500. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, Z.N.; Yousif, E.; Alias, M.; El-Hiti, G.A.; Ahmed, D.S. Synthesis, characterization, properties, and use of new fusidate organotin complexes as additives to inhibit poly(vinyl chloride) photodegradation. J. Polym. Res. 2020, 27, 267. [Google Scholar] [CrossRef]
- Jasem, H.; Hadi, A.G.; El-Hiti, G.A.; Baashen, M.A.; Hashim, H.; Ahmed, A.A.; Ahmed, D.S.; Yousif, E. Tin-naphthalene sulfonic acid complexes as photostabilizers for poly(vinyl chloride). Molecules 2021, 26, 3629. [Google Scholar] [CrossRef]
- Crellin, E.; Mansfield, K.E.; Leyrat, C.; Nitsch, D.; Douglas, I.J.; Root, A.; Williamson, E.; Smeeth, L.; Tomlinson, L.A. Trimethoprim use for urinary tract infection and risk of adverse outcomes in older patients: Cohort study. BMJ 2018, 360, k341. [Google Scholar] [CrossRef] [Green Version]
- Gaumet, S.; Gardette, J.-L. Photo-oxidation of poly(vinyl chloride): Part 2—A comparative study of the carbonylated products in photo-chemical and thermal oxidations. Polym. Degrad. Stab. 1991, 33, 17–34. [Google Scholar] [CrossRef]
- Pospíšil, J.; Nešpurek, S. Photostabilization of coatings. Mechanisms and performance. Prog. Polym. Sci. 2000, 25, 1261–1335. [Google Scholar] [CrossRef]
- Chaochanchaikul, K.; Rosarpitak, V.; Sombatsompop, N. Photodegradation profiles of PVC compound and wood/PVC composites under UV weathering. Express Polym. Lett. 2013, 7, 146–160. [Google Scholar] [CrossRef] [Green Version]
- Nief, O.A. Photostabilization of polyvinyl chloride by some new thiadiazole derivatives. Eur. J. Chem. 2015, 6, 242–247. [Google Scholar] [CrossRef] [Green Version]
- Jafari, A.J.; Donaldson, J.D. Determination of HCl and VOC emission from thermal degradation of PVC in the absence and presence of copper, copper(II) oxide and copper(II) chloride. J. Chem. 2009, 6, 685–692. [Google Scholar] [CrossRef] [Green Version]
- Valko, L.; Klein, E.; Kovařík, P.; Bleha, T.; Šimon, P. Kinetic study of thermal dehydrochlorination of poly(vinyl chloride) in the presence of oxygen: III. Statistical thermodynamic interpretation of the oxygen catalytic activity. Eur. Polym. J. 2001, 37, 1123–1132. [Google Scholar] [CrossRef]
- Nikafshar, S.; Zabihi, O.; Ahmadi, M.; Mirmohseni, A.; Taseidifar, M.; Naebe, M. The effects of UV light on the chemical and mechanical properties of a transparent epoxy-diamine system in the presence of an organic UV absorber. Materials 2017, 10, 180. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Zhang, J.; Shi, X.-M.; Jiang, G.-D. Different photo-degradation processes of PVC with different average degrees of polymerization. J. Appl. Polym. Sci. 2008, 107, 528–540. [Google Scholar] [CrossRef]
- Yousif, E.; Ahmed, D.S.; El-Hiti, G.A.; Alotaibi, M.H.; Hashim, H.; Hameed, A.S.; Ahmed, A. Fabrication of novel ball-like polystyrene films containing Schiff base microspheres as photostabilizers. Polymers 2018, 10, 1185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alotaibi, M.H.; El-Hiti, G.A.; Yousif, E.; Ahmed, D.S.; Hashim, H.; Hameed, A.S.; Ahmed, A. Evaluation of the use of polyphosphates as photostabilizers and in the formation of ball-like polystyrene materials. J. Polym. Res. 2019, 26, 161. [Google Scholar] [CrossRef]
- Hadi, A.G.; Yousif, E.; El-Hiti, G.A.; Ahmed, D.S.; Jawad, K.; Alotaibi, M.H.; Hashim, H. Long-term effect of ultraviolet irradiation on poly(vinyl chloride) films containing naproxen diorganotin(IV) complexes. Molecules 2019, 24, 2396. [Google Scholar] [CrossRef] [Green Version]
- Mohammed, R.; El-Hiti, G.A.; Ahmed, A.; Yousif, E. Poly(vinyl chloride) doped by 2-(4-isobutylphenyl)propanoate metal complexes: Enhanced resistance to UV irradiation. Arab. J. Sci. Eng. 2017, 42, 4307–4315. [Google Scholar] [CrossRef]
- Ali, M.M.; El-Hiti, G.A.; Yousif, E. Photostabilizing efficiency of poly(vinyl chloride) in the presence of organotin(IV) complexes as photostabilizers. Molecules 2016, 21, 1151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohammed, A.; El-Hiti, G.A.; Yousif, E.; Ahmed, A.A.; Ahmed, D.S.; Alotaibi, M.H. Protection of poly(vinyl vhloride) films against photodegradation using various valsartan tin complexes. Polymers 2020, 12, 969. [Google Scholar] [CrossRef] [Green Version]
- Hadi, A.G.; Jawad, K.; El-Hiti, G.A.; Alotaibi, M.H.; Ahmed, A.A.; Ahmed, D.S.; Yousif, E. Photostabilization of poly(vinyl chloride) by organotin(IV) compounds against photodegradation. Molecules 2019, 24, 3557. [Google Scholar] [CrossRef] [Green Version]
- Ghazi, D.; El-Hiti, G.A.; Yousif, E.; Ahmed, D.S.; Alotaibi, M.H. The effect of ultraviolet irradiation on the physicochemical properties of poly(vinyl chloride) films containing organotin(IV) complexes as photostabilizers. Molecules 2018, 23, 254. [Google Scholar] [CrossRef] [Green Version]
- Ghani, H.; Yousif, E.; Ahmed, D.S.; Kariuki, B.M.; El-Hiti, G.A. Tin complexes of 4-(benzylideneamino)benzenesulfonamide: Synthesis, structure elucidation and their efficiency as PVC photostabilizers. Polymers 2021, 13, 2434. [Google Scholar] [CrossRef]
- Sabaa, M.W.; Oraby, E.H.; Abdel Naby, A.S.; Mohamed, R.R. Anthraquinone derivatives as organic stabilizers for rigid poly(vinyl chloride) against photo-degradation. Eur. Polym. J. 2005, 41, 2530–2543. [Google Scholar] [CrossRef]
- Zheng, X.-G.; Tang, L.-H.; Zhang, N.; Gao, Q.-H.; Zhang, C.-F.; Zhu, Z.-B. Dehydrochlorination of PVC materials at high temperature. Energy Fuels 2003, 17, 896–900. [Google Scholar] [CrossRef]









| Complex | Color | M.P. (°C) | Yield (%) | Elemental Analyses (%) Calculated (Found) | |||
|---|---|---|---|---|---|---|---|
| C | H | N | Sn | ||||
| 1 | White | 267–269 | 75 | 60.12 (60.25) | 5.05 (5.10) | 8.76 (8.78) | 18.57 (18.48) |
| 2 | Off white | 217–218 | 79 | 56.42 (56.36) | 5.21 (5.25) | 13.16 (13.23) | 13.94 (14.01) |
| 3 | White | 245–246 | 82 | 53.28 (53.27) | 6.46 (6.39) | 13.81 (13.89) | 14.63 (14.76) |
| Complex | FTIR, Wave Number (ν, cm−1) | ||||
|---|---|---|---|---|---|
| NH2 | NH | C=N | Sn–C | Sn–N | |
| 1 | 3406 | 3176 | 1680 | 605 | 497 |
| 2 | 3408 | 3180 | 1680 | 607 | 586 |
| 3 | 3406 | 3163 | 1682 | 605 | 516 |
| Complex | 1H NMR (DMSO-d6: δ, ppm, J in Hz) | 13C NMR (DMSO-d6: δ, ppm) |
|---|---|---|
| 1 | 3.16 (s, exch., 1H, NH), 3.61 (s, 2H, CH2), 3.64 (s, 3H, OMe), 3.76 (s, 6H, 2 OMe), 6.64 (s, 2H, 3,4,5-(OMe)3C6H2), 7.47–7.53 (m, 17H, 3 Ph and NH2), 7.95 (s, 1H, pyrimidinyl) | 32.7 (CH2), 56.4 (OMe), 60.4 (OMe), 106.7 (C2/C6 of 3,4,5-(OMe)3C6H2), 109.2 (C5 of pyrimidinyl), 128.5 (C3/C5 of Ph), 128.2 (C4 of Ph), 133.7 (C1 of 3,4,5-(OMe)3C6H2), 136.6 (C4 of 3,4,5-(OMe)3C6H2), 137.0 (C2/C6 of Ph), 137.5 (C1 of Ph), 141.0 (C3/C5 of 3,4,5-(OMe)3C6H2), 153.3 (C4 of pyrimidinyl), 161.1 (C2 of pyrimidinyl), 164.3 (C1 of pyrimidinyl) |
| 2 | 3.46 (s, exch., 2H, 2 NH), 3.56 (s, 4H, 2 CH2), 3.64 (s, 6H, 2 OMe), 3.74 (s, 12H, 4 OMe), 6.28 (s, exch., 4H, 2 NH2), 6.59 (s, 4H, 3,4,5-(OMe)3C6H2), 7.26–7.36 (10H, 2 Ph), 7.96 (s, 2H, pyrimidinyl) | 33.2 (CH2), 56.3 (OMe), 60.4 (OMe), 106.4 (C2/C6 of 3,4,5-(OMe)3C6H2), 107.2 (C5 of pyrimidinyl), 127.4 (C3/C5 of Ph), 127.6 (C4 of Ph), 133.1 (C1 of 3,4,5-(OMe)3C6H2), 135.3 (C4 of 3,4,5-(OMe)3C6H2), 135.5 (C2/C6 of Ph), 136.3 (C1 of Ph), 151.5 (C3/C5 of 3,4,5-(OMe)3C6H2), 153.2 (C4 of pyrimidinyl), 160.4 (C2 of pyrimidinyl), 163.2 (C1 of pyrimidinyl) |
| 3 | 0.89 (t, J = 7.6 Hz, 6H, 2 Me), 1.30 (sextet, J = 7.6 Hz, 4H, 2 MeCH2), 1.49 (quintet, J = 7.6 Hz, 4H, 2 MeCH2CH2), 1.69 (t, J = 7.6 Hz, 4H, 2 MeCH2CH2CH2), 3.30 (s, exch., 2H, 2 NH), 3.54 (s, 4H, 2 CH2), 3.63 (s, 6H, 2 OMe), 3.74 (s, 12H, 4 OMe), 6.11 (s, exch., 4H, 2 NH2), 6.58 (s, 4H, 3,4,5-(OMe)3C6H2), 7.51 (s, 2H, pyrimidinyl) | 14.2 (Me), 16.5 (SnCH2), 26.1 (SnCH2CH2), 28.1 (MeCH2), 33.3 (CH2), 56.3 (OMe), 60.4 (OMe), 106.4 C2/C6 of 3,4,5-(OMe)3C6H2), 107.0 (C5 of pyrimidinyl), 135.8 (C1 of 3,4,5-(OMe)3C6H2), 136.3 (C4 of 3,4,5-(OMe)3C6H2), 150.0 (C3/C5 of 3,4,5-(OMe)3C6H2), 153.2 (C4 of pyrimidinyl), 161.2 (C2 of pyrimidinyl), 163.1 (C1 of pyrimidinyl) |
| PVC Additive | Rq Reduction (Fold) | Reference |
|---|---|---|
| Trimethoprim–tin complexes | 11.3 | [current work] |
| Naproxen–tin complexes | 5.2 | [50] |
| 2-(4-Isobutylphenyl)propanoate-nickel complexes | 6.3 | [51] |
| Carvedilol–tin complexes | 6.4 | [36] |
| Furosemide–tin complexes | 6.6 | [52] |
| Valsartan–tin complexes | 7.4 | [53] |
| Telmisartan–tin complexes | 9.4 | [54] |
| Ciprofloxacin–tin complexes | 16.6 | [55] |
| 4-(Benzylideneamino)benzenesulfonamide–tin complexes | 18.4 | [56] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yaseen, A.A.; Yousif, E.; Al-Tikrity, E.T.B.; El-Hiti, G.A.; Kariuki, B.M.; Ahmed, D.S.; Bufaroosha, M. FTIR, Weight, and Surface Morphology of Poly(vinyl chloride) Doped with Tin Complexes Containing Aromatic and Heterocyclic Moieties. Polymers 2021, 13, 3264. https://doi.org/10.3390/polym13193264
Yaseen AA, Yousif E, Al-Tikrity ETB, El-Hiti GA, Kariuki BM, Ahmed DS, Bufaroosha M. FTIR, Weight, and Surface Morphology of Poly(vinyl chloride) Doped with Tin Complexes Containing Aromatic and Heterocyclic Moieties. Polymers. 2021; 13(19):3264. https://doi.org/10.3390/polym13193264
Chicago/Turabian StyleYaseen, Anaheed A., Emad Yousif, Emaad T. B. Al-Tikrity, Gamal A. El-Hiti, Benson M. Kariuki, Dina S. Ahmed, and Muna Bufaroosha. 2021. "FTIR, Weight, and Surface Morphology of Poly(vinyl chloride) Doped with Tin Complexes Containing Aromatic and Heterocyclic Moieties" Polymers 13, no. 19: 3264. https://doi.org/10.3390/polym13193264










