1. Introduction
Severe acute respiratory syndrome coronavirus (SARS-CoV-2 or COVID-19 virus) pandemic attacked the world vigorously from the fourth quarter of 2019 until the present. Thus, the World Health Organization (WHO) [
1] announced a public health emergency due to the outbreak on 30 January 2020. As reported on 7 February 2021, there were 106 million active COVID-19 cases, with 2.3 million deaths calculated from 219 countries and regions affected by the global outbreak [
2]. Environmental pollution is one of the most worrying consequences due to this COVID-19 epidemic. As of 22 November 2020, a massive amount of COVID-19 medical waste (CMW), approximately 54,000 tons of waste, had been produced daily [
3]. In this context, the CMW is defined as medical waste that has been used in treating COVID-19 patients in the hospitals and healthcare facilities, which comprises personal protective equipment (PPE), such as face masks, non-woven polypropylene-based isolation gowns, gloves, goggles, and disinfectant containers. Specifically, the amount of medical waste generated during the COVID-19 crisis has increased dramatically to almost four times the usual situation in Hubei province. Furthermore, the CMW in Wuhan was recorded at a peak of roughly 240 t/day and indeed, the incineration capacity is around five times greater [
3]. Thus, this scenario leads to increasing global production of plastics waste.
Plastics are synthetic organic materials made up of polymers, consisting of long molecules built around chains of carbon atoms, generally hydrogen, oxygen, sulphur, and nitrogen filling. Production of plastic wastes increases every year, with 381 million tonnes as recorded in 2020 [
4,
5], which includes various types of plastics such as high-density polyethylene (HDPE), low-density polyethylene (LDPE), polyethylene terephthalate (PET), polyvinyl chloride (PVC), polystyrene (PS), and polypropylene (PP) [
5,
6]. It is noted that PPE and N95 masks utilised by HCWs and publics include plastics as major constitutes, representing 25% by weight [
7]; whereas widely used facemasks and other PPEs, including protective suits, isolation gowns and gloves, are mainly made up of PP (about 72%) [
8]. According to the Ministry of Health Malaysia (MOH) [
9], PPEs, e.g., isolation gowns and face masks that have been used widely by healthcare workers in Malaysia, are made from non-woven fabric PP polymer. In current situations, the practice used to decompose them is via burning and incineration methods, where various significant atmospheric pollutants were released. This is due to these plastics containing additives such as colorants, plasticisers, and stabilisers. These cadmium- and lead-containing materials are hazardous to the environment. Therefore, proper green technology is paramount in transforming these hazardous wastes into wealth; as the abnormal situations happened, the urgent requirement of the existing treatment system to cope with the significant changes must be prioritised.
Pyrolysis is a thermal degradation approach of plastic materials conversion into three major product forms, includes gaseous, liquid (oil), and solid (char), at elevated temperature under a deoxygenated environment [
10]. The products mentioned above are valuable for industries, especially green production and refineries. Generally, gas produced from the pyrolysis has significant calorific value as well as high potential to be utilised as a heating source in industrial pyrolysis plants, converted as chemical feedstock for polyolefins production, and used in combustion (gas) turbines to generate electricity [
11]; whereas liquid oil yielded via fast pyrolysis with a higher temperature has wide and multiple applications, including furnaces, boilers, diesel engines, and turbines [
12]. High-quality solid char formation in the slow pyrolysis approach is higher than in fast pyrolysis. Furthermore, its higher calorific value and low sulphur content has made it ideal for combustion with coal or other natural binders [
13,
14], used as fuel, filler in composite and building materials [
15,
16], and water adsorbent in water treatment [
17]. Temperatures, type of reactors, time of sample residence, pressure, heating rate, process flow rate, type of fluidising gas, and catalysts are the main parameters affecting the end product of the pyrolysis. From previous studies, pyrolysis of plastic is generally carried out at final temperature for about 500 °C (moderate temperature) with lower heating rate (~10 °C/min) using a batch reactor [
14,
18,
19,
20,
21,
22,
23,
24], with these requirements leading to higher char yields; whereas fast pyrolysis contributes to higher gaseous and fuel-like liquid yields [
13,
25,
26,
27]. Recently, there have been several review articles [
28,
29,
30] that summarised the proper plastic wastes treatment in detail.
Literature reviews on the application of slow pyrolysis in PP plastic conversion are few to the best of our knowledge [
23,
24,
31,
32,
33,
34,
35]. Demirbas et al. [
36] investigated the relationship between the yield distributions of the pyrolytic products collected from PP with the different pyrolysis temperatures. The authors observed that the increasing pyrolysis temperature led to lower total olefin fraction yields, while contributing to higher aromatics fraction yields. The pyrolysis experiment of the 3 mm-size PP powder was carried out in a batch reactor with a heating rate of 10 °C/min in deoxygenated conditions. At pyrolysis temperature of 740 °C of PP waste, the gas, oil, and solid residue collected were 49.6, 48.8, and 1.6 wt.%, respectively. From the results obtained, the authors had suggested practicing pyrolysis via lower temperature and reduced heating rate to optimise the solid residue yields. This statement is supported by the works of Ahmad et al. [
33], as they studied the product yields as a function of pyrolysis temperature. The total solid residue of 1.34 wt.% was achieved at 300 °C with a higher heating rate of 20 °C/min. Sogancioglu et al. [
37] slow pyrolysed pp plastic waste to yield chars for filler applications using a batch reactor. Approximately 2.67% of chars were obtained from low pyrolysis temperature and slow heating rate. Wong and Broadbelt [
38] investigated the influence of residence time by fixed two pyrolysis temperatures of 350 °C and 420 °C of PP and PE plastics for 2 h and 18 h, respectively. The authors found that longer residence time increased the consistency of the product with lighter and lower molecular products, which is suitable for fuel and chemical applications.
To optimise the pyrolysis parameters to maximise the char residue yields, an insight into the applicability of a detailed analysis considering temperature and reaction time may be useful. Char residue yielded from pyrolysis of disinfected PP-based isolation gown waste (PP-IG) has its own significant structural, mechanical, and surface area properties. In this study, a comprehensive analysis of the effect of temperature on the char yields and specific surface area values via the facile pyrolysis process to degrade PP plastic waste will be presented. As for novelties, these findings will contribute to understanding of the applicability and impediments of these hazardous waste materials generated from hospitals, PP-IG derived char as solid precursors for activated carbon/graphene production, and also fuel briquette applications while promoting environmentally friendly and more efficient waste-to-wealth conversion ways.
2. Materials and Methods
2.1. Polypropylene (PP) Isolation Gown Waste
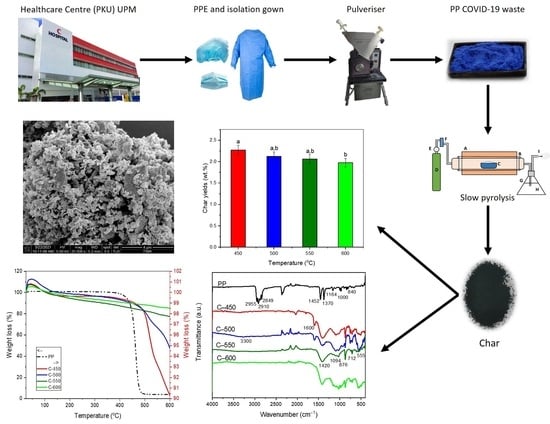
The pyrolysis experiments were performed with 0.25 mm PP-IG samples. The PP-IG samples were prepared from used PPEs, including isolation gowns. Next, the collected materials were shredded using FRITSCH Universal Cutting Mill PULVERISETTE 19, Idar-Oberstein, Germany, with 0.25 mm perforation sieve cassette into 0.25 mm-size PP-IG samples. The samples then were directly used for sample characterisations and experiments of laboratory-scale pyrolysis with specified parameters.
For a comprehensive overview, the PPEs, include the isolation gowns, hairnets, shoe covers, and face masks, were collected from Universiti Putra Malaysia (UPM) Healthcare Centre (PKU), UPM Serdang, Selangor, Malaysia. Priors to shredding steps, the collected waste PPEs were disinfected thoroughly under the supervision of HCWs from UPM PKU staff. Materials collected were cleaned and disinfected using normal soap meticulously. The materials were dried under direct sunlight and shredded using the abovementioned cutting mill into small powder of size ~0.25 mm for better pyrolysis experiment. Twenty g of segregated PP-IG samples were filled into a quartz boat and placed at the centre of the furnace. The elemental composition of PP plastic is as shown in
Table 1.
2.2. Preparation of Char
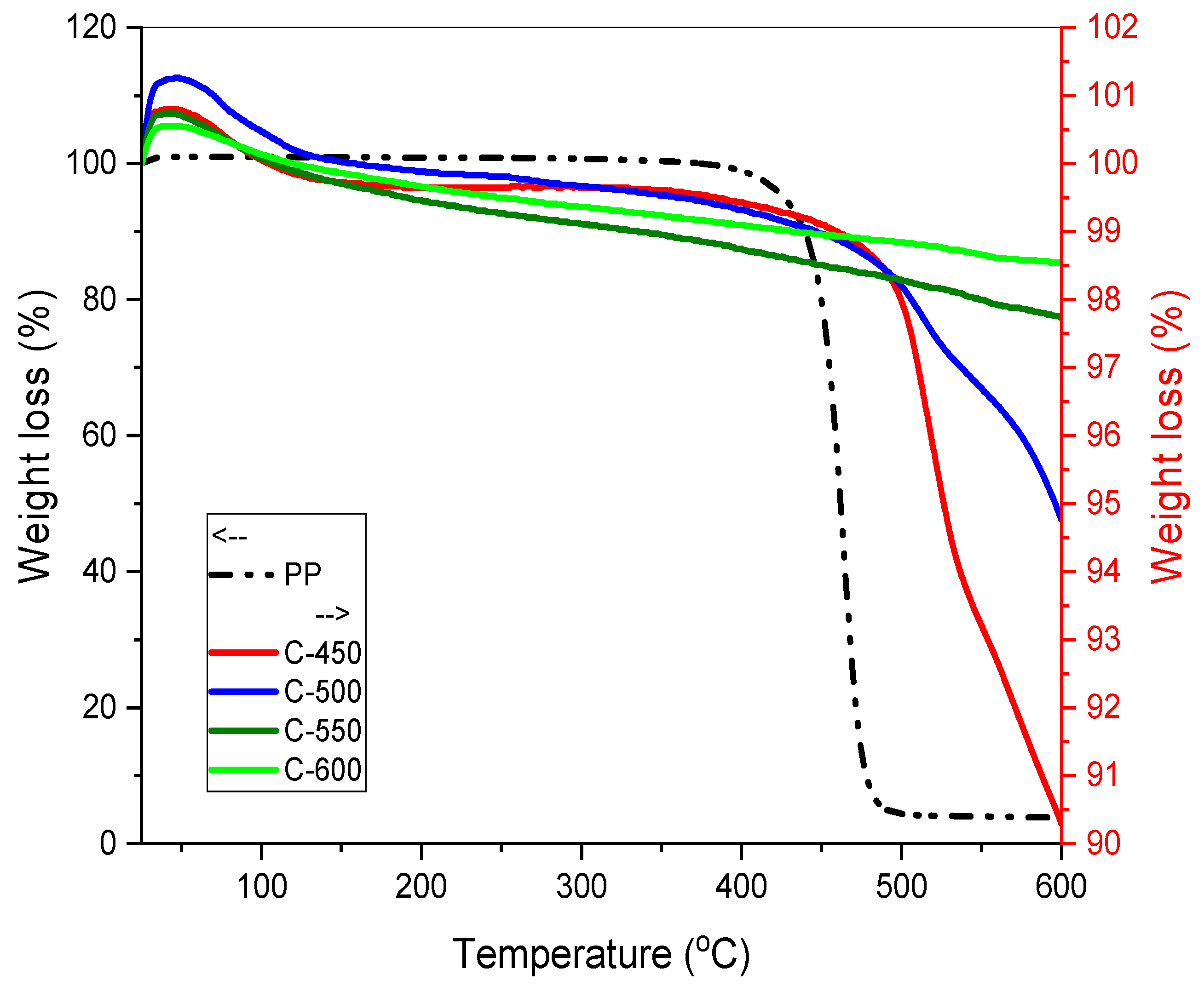
A slow pyrolysis study was executed for PP-IG samples using a tube furnace batch reactor (Carbolite) at a fixed heating rate of 3 °C/min with 1.5 atm pressure under nitrogen gas environment (99.9992% purified N2 gas). Prior to the thermal degradation study, thermal-gravimetric analysis (TGA) and derivative thermogravimetry (DTG) analysis were performed in order to determine the optimum temperature in which degradation of PP-IG samples occurs.
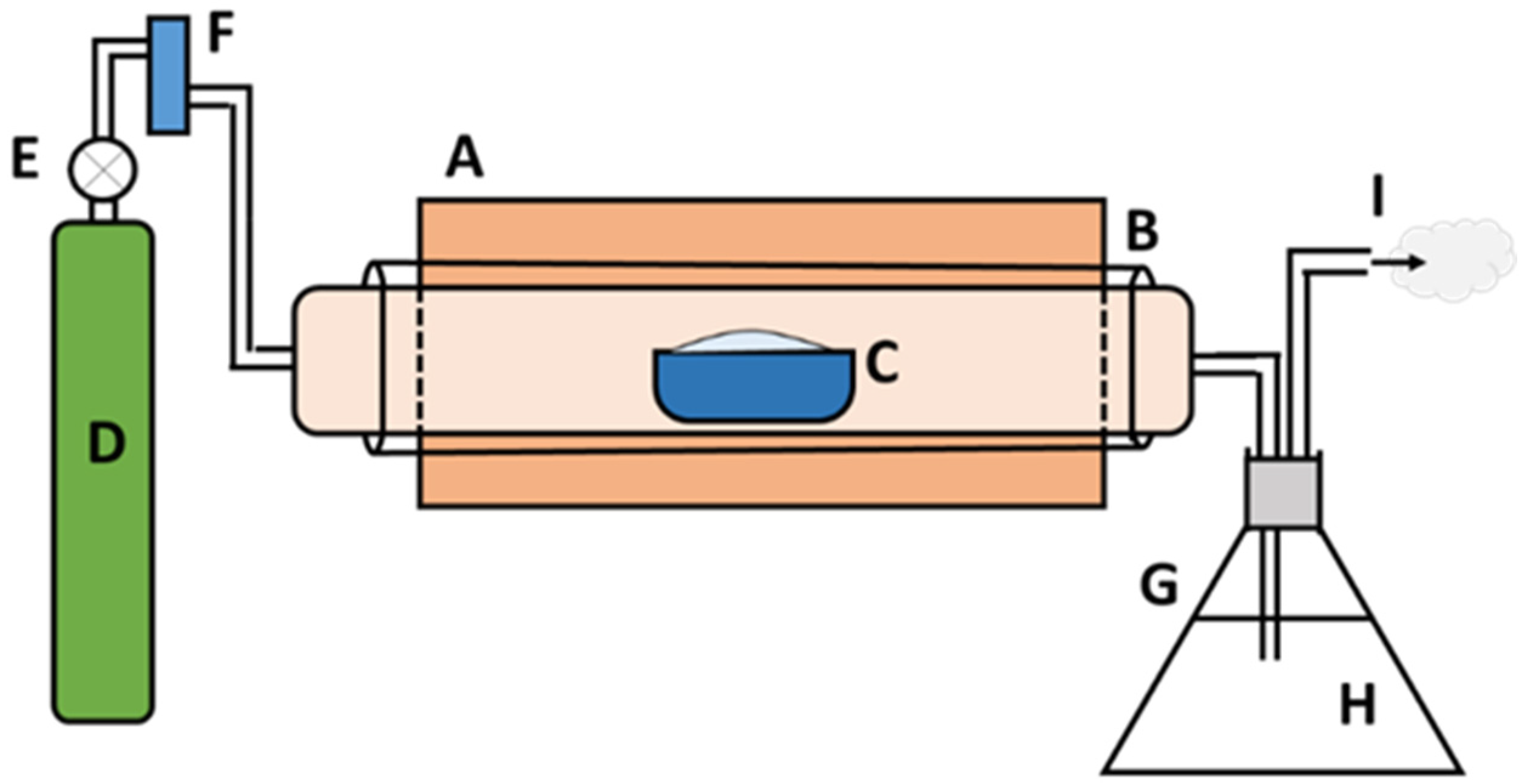
Based on the TGA and DSC data, five pyrolysis temperatures (450, 500, 550, 600, and 650 °C) were used as the degradation range of PP-IG samples was from 400–550 °C. For a better understanding, the following processing procedure was applied: 20 g PP-IG samples were filled into alumina boats for each run and then inserted into the ceramic tube furnace. N
2 gas with a 1.5 cc/min flow rate was purged continuously in order to produce an inert environment inside the isolated reactor. Thus, it also helped to expunge the unwanted pyrolysis vapour products. The gas outlet pipe was then passed through a conical flask filled with water, which was used to detect any leakage throughout the system. The schematic diagram of the experimental setup is represented in
Figure 1. A fixed lower heating rate with a residence time of 5 h was used to produce the maximum amount of char yields. Next, the amount of char (by weight) was collected and measured using an electronic balance. The char yields (wt.%) were calculated with Equation (1) and recorded, which was then analysed.
where
is the amount of char and
is the pyrolysed waste amount.
2.3. Characterisation of Char
Qualitative and quantitative characterisation of char was carried out. Morphological and structural analyses were carried out using analytical techniques for the yielded char like field emission scanning electron microscopy (FESEM, FEI, Sydney, NSW, Australia) and energy dispersive X-ray (EDX, Oxford Instruments, Oxford, UK); whereas the compositional analysis was conducted via the analytical approach of Fourier transform infrared spectroscopy (FTIR, Thermo Fisher Scientific, Waltham, MA, USA) and Raman spectroscopy. Next, the physical properties of specific surface area were measured using Brunauer–Emmett–Teller (BET, Micromeritics Instrument, Norcross, GA, USA) analysis, while its thermal properties were analysed via thermal-gravimetric analysis (TGA, Mettler Toledo, Shah Alam, Selangor, Malaysia) and derivative thermogravimetry (DTG, Mettler Toledo, Shah Alam, Selangor, Malaysia).
2.3.1. Proximate Analysis
The proximate analysis was performed using TGA to obtain the moisture, ash, volatile matter, and fixed carbon contents; whereas Equation (2) was used for calculating the fixed carbon. The moisture content has been overlooked since the result of TGA depends on a dry basis [
39].
2.3.2. FESEM
Nova NanoSEM 230 FESEM (FEI, Sydney, NSW, Australia) is used to view the specified area of the samples via 1 nm resolution at 15 kV. Secondary electron (SE) imaging can be undertaken in field-free and immersion mode (TLD) for comprehensive low-to-high resolution imaging (100,000× magnification). The FESEM service has been accredited by the Quality System of MS ISO/IEC 17,025 Certification.
2.3.3. EDX
Max 20 EDX (Oxford Instruments, Oxford, UK) was used to determine the elemental composition of specific points or to map out the lateral distribution of elements from the imaged area. EDX has combined with FESEM above in order to provide the elemental analysis on a specified area.
2.3.4. FTIR
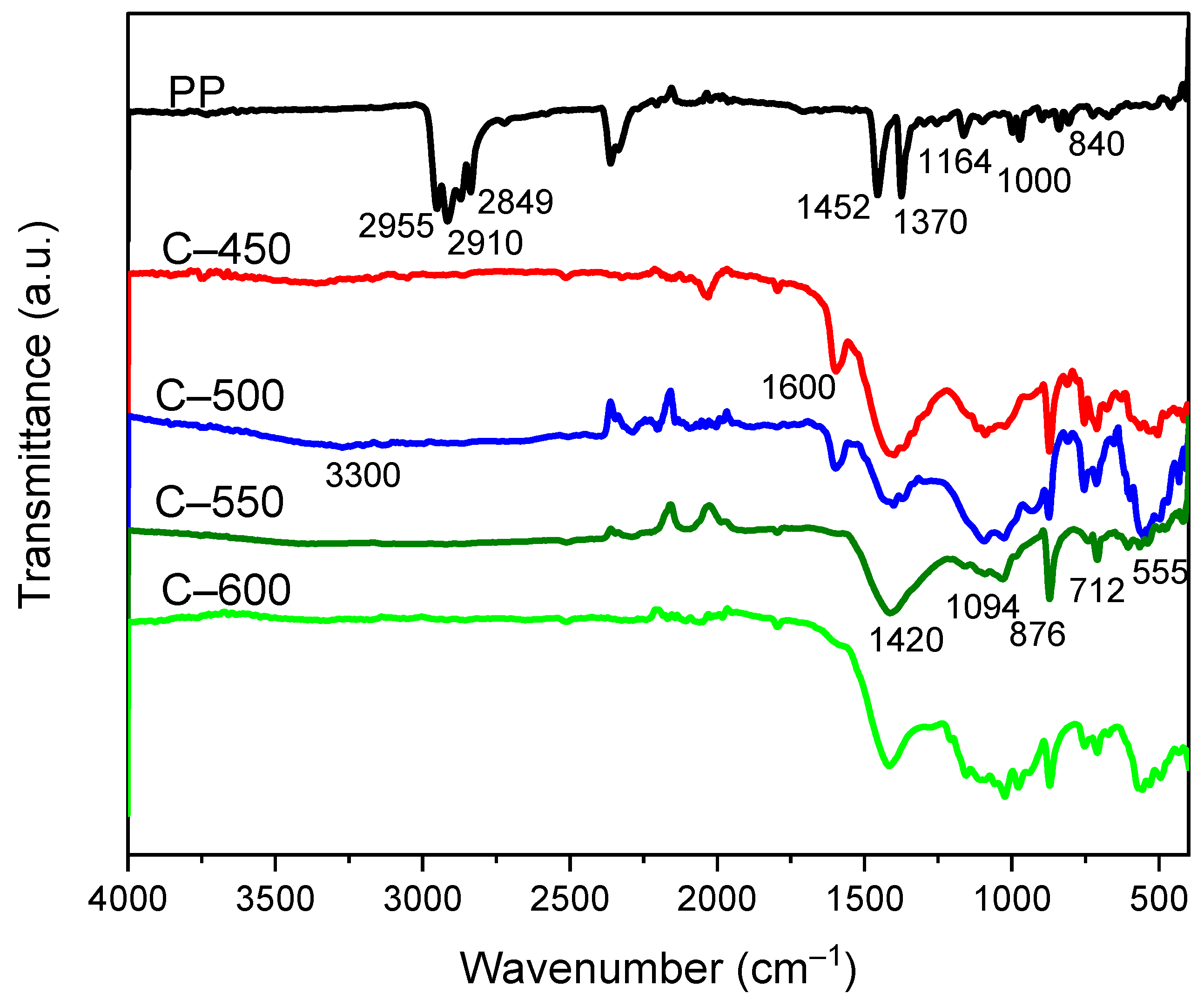
The FTIR analysis adopted an attenuated total reflective (ATR) technique with a wavelength range of 400 to 4000 cm−1 to identify functional groups in the char materials. The solid chars collected from various pyrolytic temperatures were characterised by using a Nicolet 6700 FTIR Spectrometer (Thermo Fisher Scientific, Waltham, MA, USA). In addition, the quality or consistency of the samples could be determined.
2.3.5. Raman Spectroscopy
WITec Alpha 300R Raman Spectroscopy (Ulm, Germany) was used to determine the chemical structure of the samples and to identify the compounds present by measuring their molecular vibrations. A 488 nm laser excitation with a single spectrum type of analysis was used to characterise the char samples.
2.3.6. BET
Micromeritics’ Tristar II Plus BET equipment (Micromeritics Instrument, Norcross, GA, USA) was used to carry out BET surface area analysis, which provides the value of the specific surface area, pore volume, and pore size distribution of solid char via nitrogen adsorption measured as a function of relative pressure. For a comprehensive understanding, a calculation of the volume of adsorbed gas corresponding to a monomolecular layer on the surface of the samples shall decide the surface area. This technique, therefore, covers essentially external area and pores to assess the total surface area. Thus, the BET surface area was used to determine the material porosity, from solid microporous materials to mesoporous materials.
2.3.7. TGA-DTG
Thermal degradation was employed to measure the changes in characteristics of the char with increased temperatures using Mettler Toledo’s TGA-DSC HT 3 equipment (Mettler Toledo, Shah Alam, Selangor, Malaysia). TGA was performed at a range of temperatures, 25 to 600 °C, with a heating rate of 10 °C/min.
2.4. Statistical Analysis
SPSS software, a statistical tool, was used to do a variance analysis (ANOVA) on the weight percentage of yielded chars. Duncan’s test was used for mean comparison at a significance level of 0.05 (p ≤ 0.05).
4. Conclusions and Future Outlooks
This study investigated the slow pyrolysis of PP-IG plastic waste using a batch reactor. The thermal decomposition of the pyrolysis model was established with a slow heating rate, long residence time, and lower pyrolytic temperature. It was used to enhance the char yields as the experimental results. The main conclusions of this study are:
- (i)
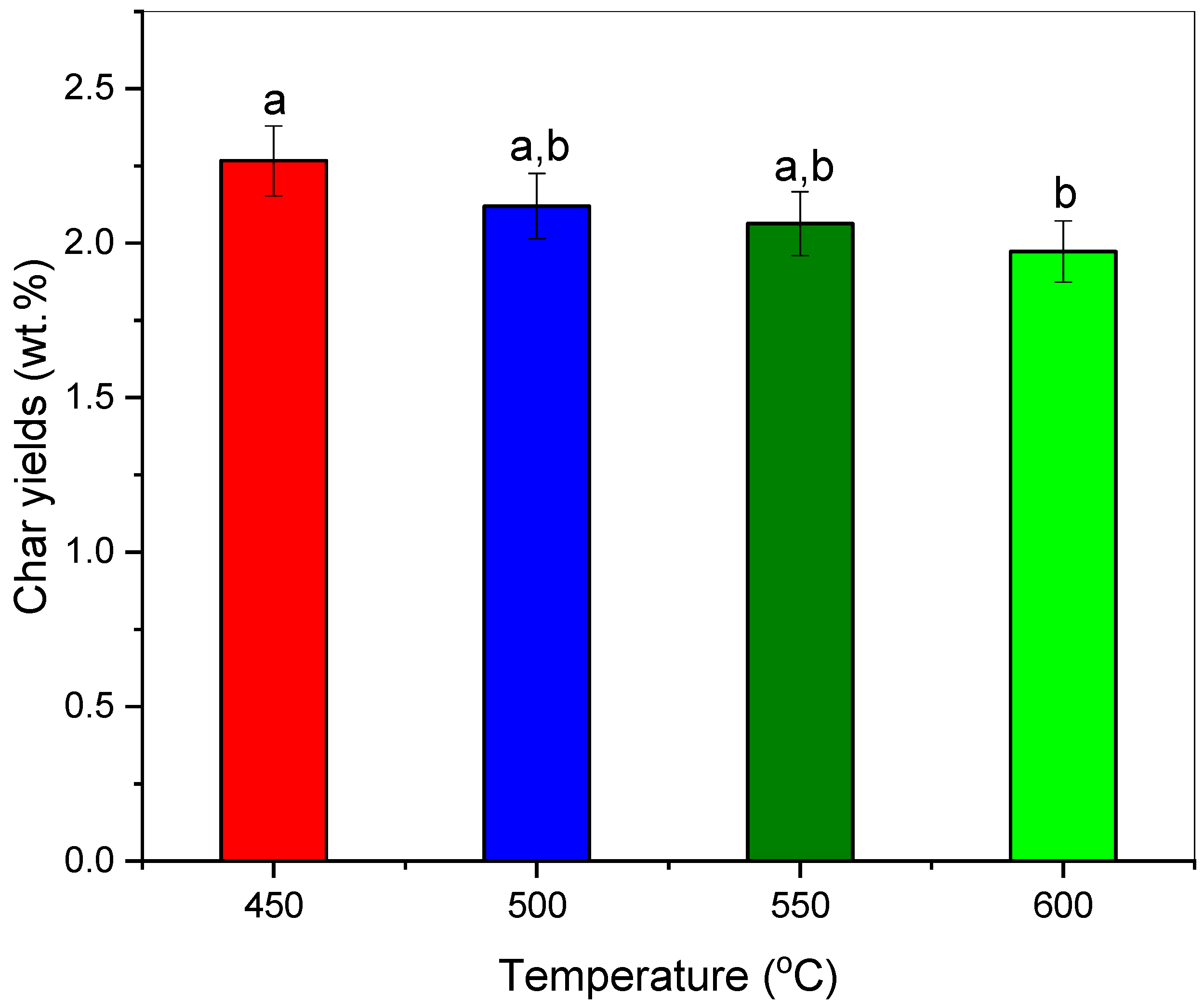
The low–temperature pyrolysis of polypropylene-based COVID-19 isolation gown waste (PP-IG) resulted in improved char yields (2.27 wt.%), which mostly comprises aliphatic and carbonaceous alkene structures.
- (ii)
On the other hand, increasing the temperature decreased the yield of char products but produced char with higher surface area value and enhanced pore volumes, ~24 m2g−1 and ~0.08 cm3g−1, respectively.
- (iii)
The char obtained at higher temperatures (C-550 and C-600) comprised more aromatics C=C and carbonaceous C–H structures due to volatilisation and carbonisation.
- (iv)
The low temperature (450 °C) and long residence time (5 h) degradation process supported the reaction from the polymer scission and led to a lighter hydrocarbon with reduced microporosity due to lower carbon structure deterioration.
- (v)
In order to generate unique char products from PP-IG waste, data collected during this study are valuable for the use of pyrolysis. At the same time, further studies are needed in order to optimise the pyrolysis parameters, design, and catalyst support for better pyrolysis outputs.
- (vi)
In addition, the isothermal process used less energy and not an isothermal process, making the present work more reliable and practical in terms of transforming COVID-19 related plastic waste into energy.
Char yielded under low-temperature pyrolysis from PP-IG waste can fulfill the demand for alternate fuel such as barbeque application and also utilization of plastic waste, especially during the COVID-19 pandemic. Even after the end of the pandemic, this study is still practical to be used to treat other PP-based plastic waste, that of PP being one of the highest produced plastics globally.
In terms of waste treatment, the practice of slow pyrolysis of PP-based plastic waste has shown numerous positive impacts. This is due to the fact that this study has successfully demonstrated how easy the process design is, with pyrolysis and heating rate optimisations, to convert PP plastic waste originated from used PPEs into multi–functional carbonaceous products. Furthermore, this research may also add to the effort to uncover the potential of plastic-derived char as solid precursors in developing green char fuel briquettes products and graphene/activated carbon production, as there are such abundant plastic waste resources were underutilized.
However, to be employed for domestic use, health considerations must be taken into account. Thus, toxicology characteristics and further effects from its applications towards the environment and human health must be studied extensively. These considerations will provide a better practicality point for this study for applications even after the end of the pandemic. The utilization of hazardous PP plastic waste will contribute to plastic waste reduction and better environmental health.