Fructose-Based Production of Short-Chain-Length and Medium-Chain-Length Polyhydroxyalkanoate Copolymer by Arctic Pseudomonas sp. B14-6
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Whole-Genome Sequencing and BLAST Search for PHA Genes
2.3. Strain and Culture Conditions
2.4. PHA Analysis
2.5. Physical Properties of Produced PHA
3. Results and Discussion
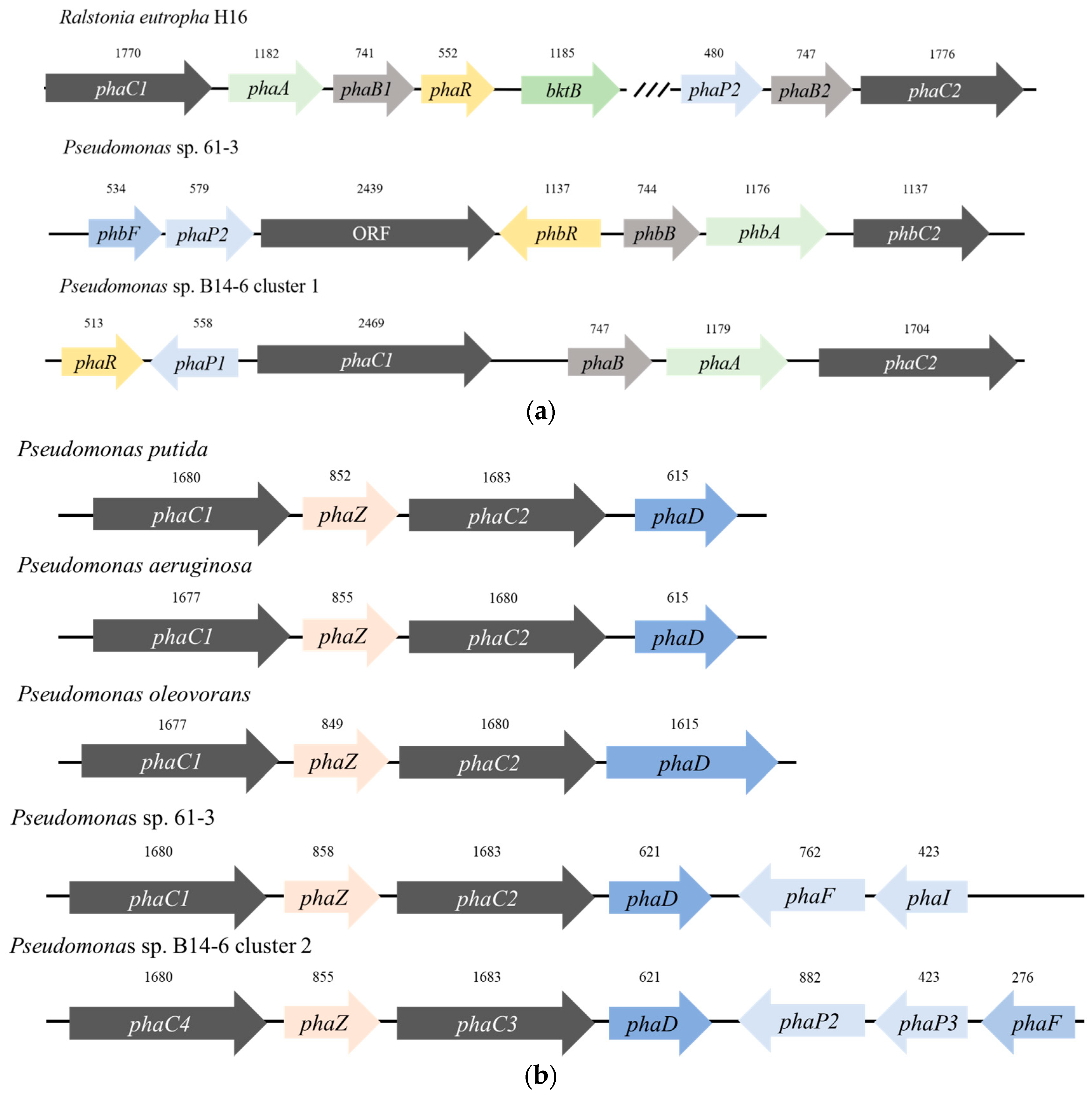
3.1. Screening of PHA-Related Genes in Pseudomonas sp. B14-6
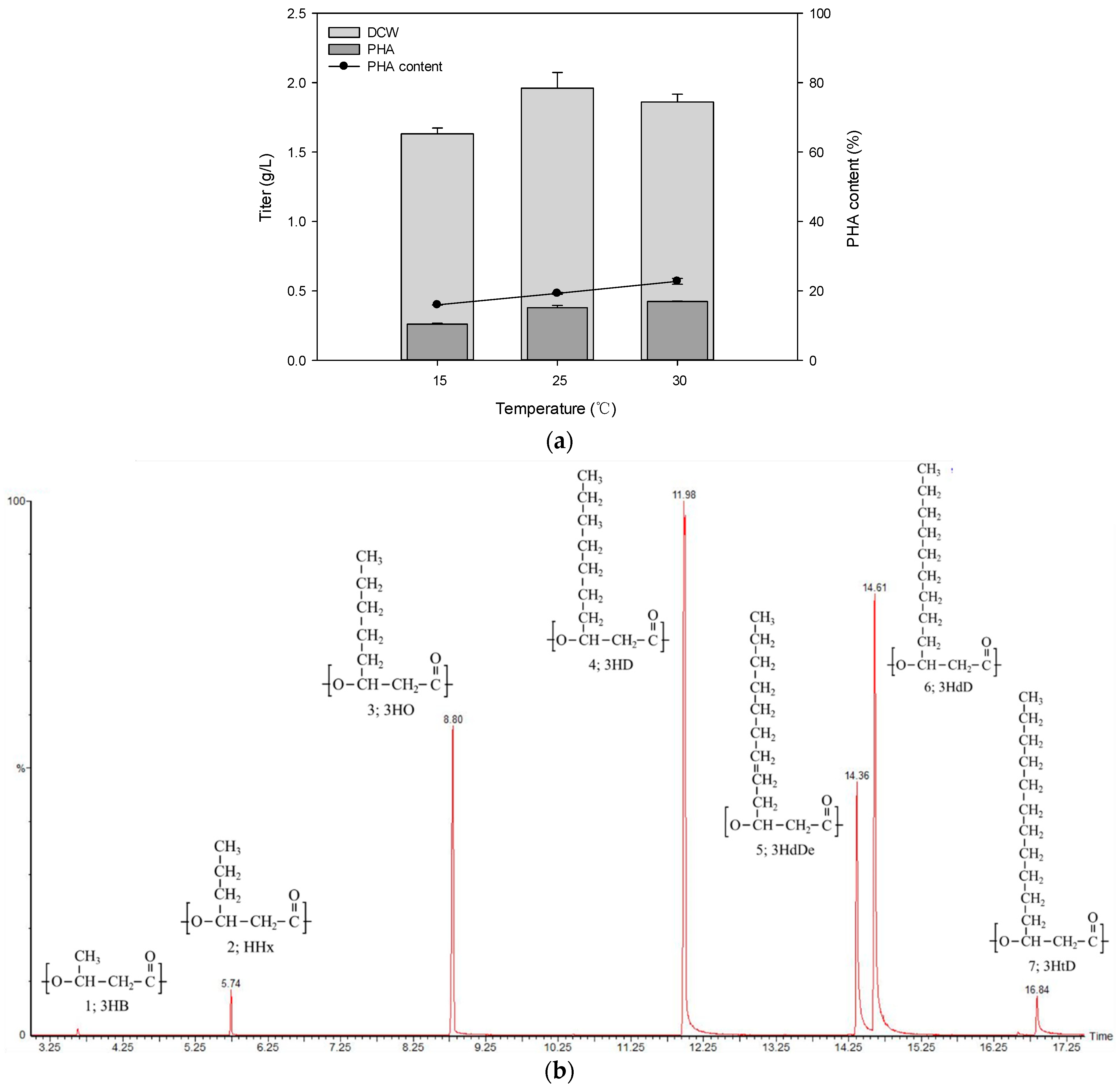
3.2. Analysis of PHA from Pseudomonas sp. B14-6
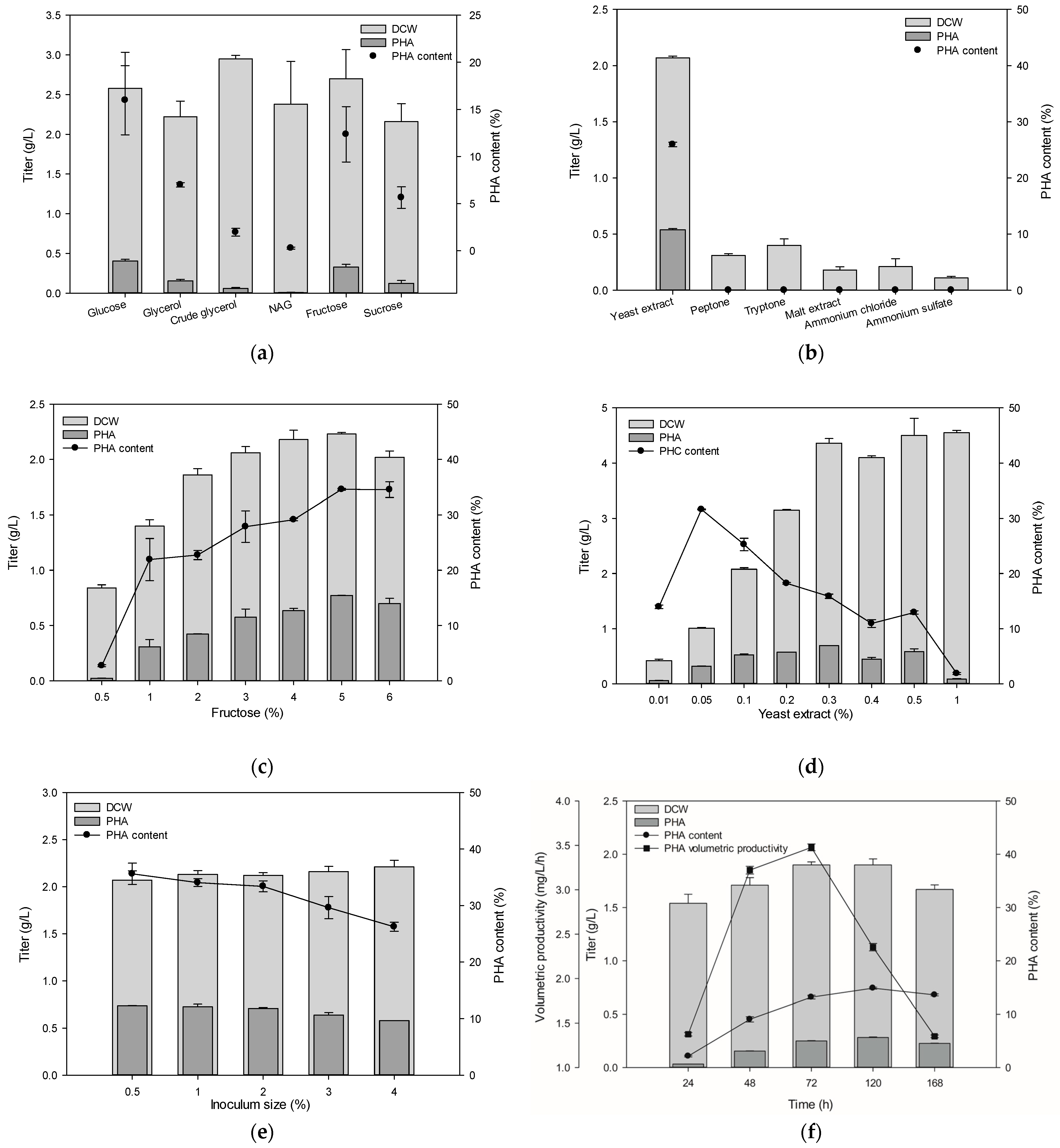
3.3. Optimization of mcl-PHA Production
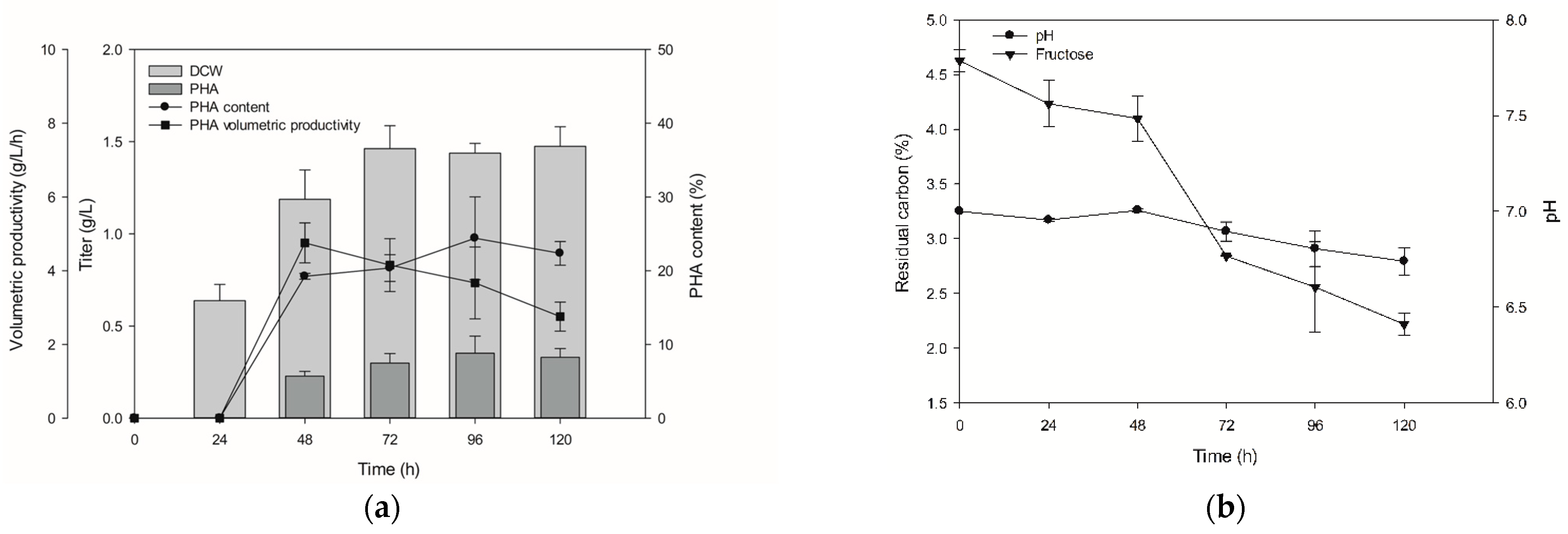
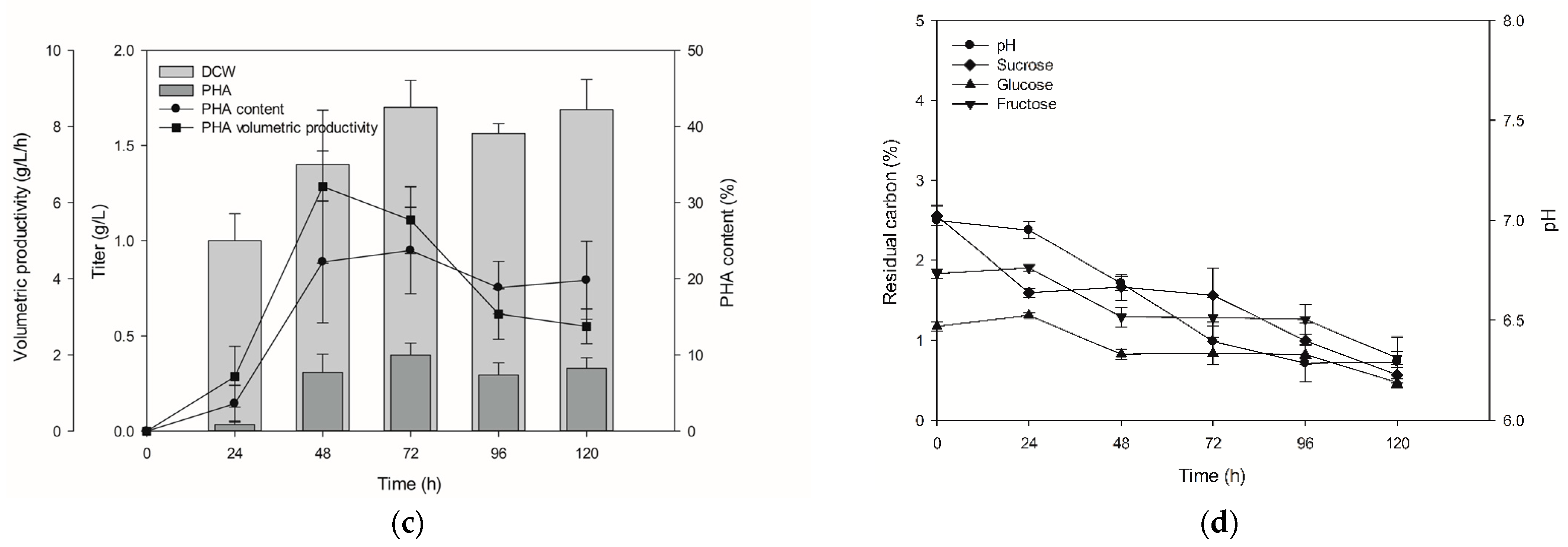
3.4. Monitoring Time-Dependent PHA Production and Fructose Syrup Application
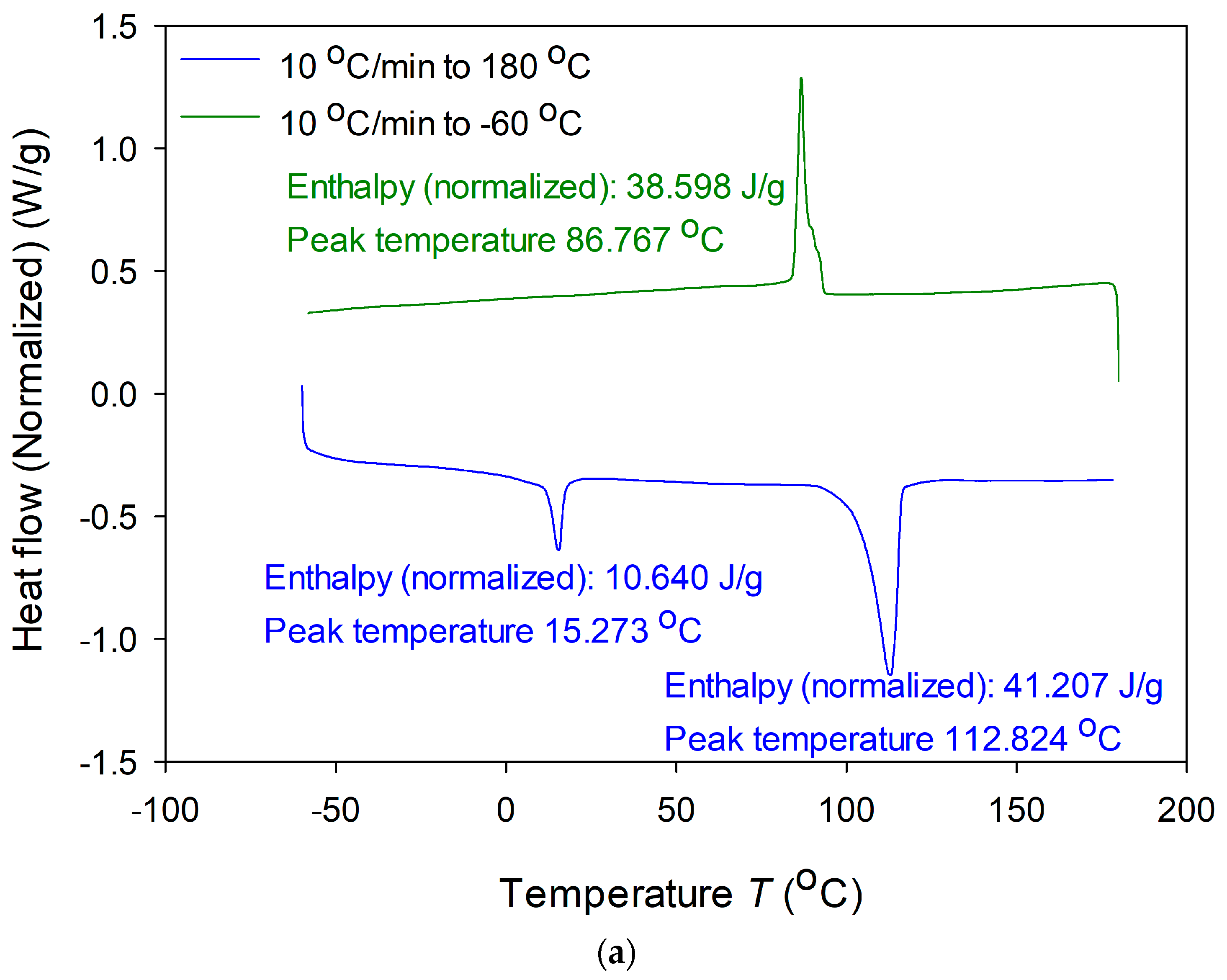
3.5. Physical Properties of Produced PHA
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Harding, T.; Jungblut, A.D.; Lovejoy, C.; Vincent, W.F. Microbes in High Arctic Snow and Implications for the Cold Biosphere. Appl. Environ. Microbiol. 2011, 77, 3234–3243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayub, N.D.; Pettinari, M.J.; Méndez, B.S.; López, N.I. The polyhydroxyalkanoate genes of a stress resistant Antarctic Pseudomonas are situated within a genomic island. Plasmid 2007, 58, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Deming, J.W. Psychrophiles and polar regions. Curr. Opin. Microbiol. 2002, 5, 301–309. [Google Scholar] [CrossRef]
- Lee, H.J.; Rho, J.K.; Yoon, S.C. Growth temperature-dependent conversion of de novo-synthesized unsaturated fatty acids into polyhydroxyalkanoic acid and membrane cyclopropane fatty acids in the psychrotrophic bacterium Pseudomonas fluorescens BM07. J. Microbiol. Biotechnol. 2004, 14, 1217–1226. [Google Scholar]
- Verlinden, R.A.J.; Hill, D.J.; Kenward, M.; Williams, C.D.; Radecka, I. Bacterial synthesis of biodegradable polyhydroxyalkanoates. J. Appl. Microbiol. 2007, 102, 1437–1449. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Sedlacek, P.; Krzyzanek, V.; Mravec, F.; Hrubanova, K.; Samek, O.; Kucera, D.; Benesova, P.; Marova, I. Accumulation of Poly(3-hydroxybutyrate) Helps Bacterial Cells to Survive Freezing. PLoS ONE 2016, 11, e0157778. [Google Scholar] [CrossRef] [Green Version]
- Ayub, N.D.; Tribelli, P.M.; López, N.I. Polyhydroxyalkanoates are essential for maintenance of redox state in the Antarctic bacterium Pseudomonas sp. 14-3 during low temperature adaptation. Extremophiles 2008, 13, 59–66. [Google Scholar] [CrossRef] [Green Version]
- Obruca, S.; Sedlacek, P.; Koller, M.; Kucera, D.; Pernicova, I. Involvement of polyhydroxyalkanoates in stress resistance of microbial cells: Biotechnological consequences and applications. Biotechnol. Adv. 2018, 36, 856–870. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Otari, S.V.; Jeon, J.-M.; Gurav, R.; Choi, Y.-K.; Bhatia, R.K.; Pugazhendhi, A.; Kumar, V.; Banu, J.R.; Yoon, J.-J.; et al. Biowaste-to-bioplastic (polyhydroxyalkanoates): Conversion technologies, strategies, challenges, and perspective. Bioresour. Technol. 2021, 326, 124733. [Google Scholar] [CrossRef]
- Luengo, J.M.; García, B.; Sandoval, A.; Naharro, G.; Olivera, E.R. Bioplastics from microorganisms. Curr. Opin. Microbiol. 2003, 6, 251–260. [Google Scholar] [CrossRef]
- Park, S.J.; Kim, T.W.; Kim, M.K.; Lee, S.Y.; Lim, S.-C. Advanced bacterial polyhydroxyalkanoates: Towards a versatile and sustainable platform for unnatural tailor-made polyesters. Biotechnol. Adv. 2012, 30, 1196–1206. [Google Scholar] [CrossRef]
- Jiang, G.; Hill, D.J.; Kowalczuk, M.; Johnston, B.; Adamus, G.; Irorere, V.; Radecka, I. Carbon Sources for Polyhydroxyalkanoates and an Integrated Biorefinery. Int. J. Mol. Sci. 2016, 17, 1157. [Google Scholar] [CrossRef] [Green Version]
- Danis, O.; Ogan, A.; Tatlican, P.; Attar, A.; Cakmakci, E.; Mertoglu, B.; Birbir, M. Preparation of poly(3-hydroxybutyrate-co-hydroxyvalerate) films from halophilic archaea and their potential use in drug delivery. Extremophiles 2015, 19, 515–524. [Google Scholar] [CrossRef]
- Chen, G.-Q.; Hajnal, I.; Wu, H.; Lv, L.; Ye, J. Engineering Biosynthesis Mechanisms for Diversifying Polyhydroxyalkanoates. Trends Biotechnol. 2015, 33, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Charles, T.C. Functional metagenomics using Pseudomonas putida expands the known diversity of polyhydroxyalkanoate synthases and enables the production of novel polyhydroxyalkanoate copolymers. BioRxiv 2016. [Google Scholar] [CrossRef] [Green Version]
- Bhatia, S.K.; Gurav, R.; Choi, T.-R.; Jung, H.-R.; Yang, S.-Y.; Song, H.-S.; Jeon, J.-M.; Kim, J.-S.; Lee, Y.-K.; Yang, Y.-H. Poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) production from engineered Ralstonia eutropha using synthetic and anaerobically digested food waste derived volatile fatty acids. Int. J. Biol. Macromol. 2019, 133, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Możejko-Ciesielska, J.; Kiewisz, R. Bacterial polyhydroxyalkanoates: Still fabulous? Microbiol. Res. 2016, 192, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Philip, S.; Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: Biodegradable polymers with a range of applications. J. Chem. Technol. Biotechnol. 2007, 82, 233–247. [Google Scholar] [CrossRef]
- Yang, T.H.; Jung, Y.K.; Kang, H.O.; Kim, T.W.; Park, S.J.; Lee, S.Y. Tailor-made type II Pseudomonas PHA synthases and their use for the biosynthesis of polylactic acid and its copolymer in recombinant Escherichia coli. Appl. Microbiol. Biotechnol. 2011, 90, 603–614. [Google Scholar] [CrossRef]
- Schweizer, H.P. Fatty Acid Biosynthesis and Biologically Significant Acyl Transfer Reactions in Pseudomonads; Metzler, J.B., Ed.; Springer: Boston, MA, USA, 2004; pp. 83–109. [Google Scholar]
- Kessler, B.; Palleroni, N.J. Taxonomic implications of synthesis of poly-beta-hydroxybutyrate and other poly-beta-hydroxyalkanoates by aerobic pseudomonads. Int. J. Syst. Evol. Microbiol. 2000, 50, 711–713. [Google Scholar] [CrossRef] [Green Version]
- Solaiman, D.K.; Ashby, R.D. Genetic Characterization of the Poly(hydroxyalkanoate) Synthases of Various Pseudomonas oleovorans Strains. Curr. Microbiol. 2005, 50, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, G.H.D.; Zaiat, M.; Rodrigues, J.A.D.; Ramsay, J.A.; Ramsay, B.A. Towards the Production of mcl-PHA with Enriched Dominant Monomer Content: Process Development for the Sugarcane Biorefinery Context. J. Polym. Environ. 2020, 28, 844–853. [Google Scholar] [CrossRef]
- Poblete-Castro, I.; Rodriguez, A.L.; Lam, C.M.C.; Kessler, W. Improved production of medium-chain-length Polyhydroxyalkanotes in glucose-based fed-batch cultivations of metabolically engineered Pseudomonas putida strains. J. Microbiol. Biotechnol. 2013, 24, 59–69. [Google Scholar] [CrossRef] [Green Version]
- Löwe, H.; Schmauder, L.; Hobmeier, K.; Kremling, A.; Pflüger-Grau, K. Metabolic engineering to expand the substrate spectrum of Pseudomonas putida toward sucrose. Microbiologyopen 2017, 6, e00473. [Google Scholar] [CrossRef]
- Sohn, Y.J.; Kim, H.T.; Baritugo, K.-A.; Song, H.M.; Ryu, M.H.; Kang, K.H.; Jo, S.Y.; Kim, H.; Kim, Y.J.; Choi, J.-I.; et al. Biosynthesis of polyhydroxyalkanoates from sucrose by metabolically engineered Escherichia coli strains. Int. J. Biol. Macromol. 2020, 149, 593–599. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Yoon, J.-J.; Kim, H.-J.; Hong, J.W.; Hong, Y.G.; Song, H.-S.; Moon, Y.-M.; Jeon, J.-M.; Kim, Y.-G.; Yang, Y.-H. Engineering of artificial microbial consortia of Ralstonia eutropha and Bacillus subtilis for poly(3-hydroxybutyrate-co-3-hydroxyvalerate) copolymer production from sugarcane sugar without precursor feeding. Bioresour. Technol. 2018, 257, 92–101. [Google Scholar] [CrossRef]
- Choi, T.-R.; Park, Y.-L.; Song, H.-S.; Lee, S.M.; Park, S.L.; Lee, H.S.; Kim, H.-J.; Bhatia, S.K.; Gurav, R.; Lee, Y.K.; et al. Effects of a Δ-9-fatty acid desaturase and a cyclopropane-fatty acid synthase from the novel psychrophile Pseudomonas sp. B14-6 on bacterial membrane properties. J. Ind. Microbiol. Biotechnol. 2020, 47, 1045–1057. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.-L.; Bhatia, S.K.; Gurav, R.; Choi, T.-R.; Kim, H.J.; Song, H.-S.; Park, J.-Y.; Han, Y.-H.; Lee, S.M.; Park, S.L.; et al. Fructose based hyper production of poly-3-hydroxybutyrate from Halomonas sp. YLGW01 and impact of carbon sources on bacteria morphologies. Int. J. Biol. Macromol. 2020, 154, 929–936. [Google Scholar] [CrossRef]
- Sathiyanarayanan, G.; Bhatia, S.K.; Song, H.-S.; Jeon, J.-M.; Kim, J.; Lee, Y.K.; Kim, Y.-G.; Yang, Y.-H. Production and characterization of medium-chain-length polyhydroxyalkanoate copolymer from Arctic psychrotrophic bacterium Pseudomonas sp. PAMC 28620. Int. J. Biol. Macromol. 2017, 97, 710–720. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Gurav, R.; Choi, T.-R.; Han, Y.H.; Park, Y.-L.; Park, J.Y.; Jung, H.-R.; Yang, S.-Y.; Song, H.-S.; Kim, S.-H.; et al. Bioconversion of barley straw lignin into biodiesel using Rhodococcus sp. YHY01. Bioresour. Technol. 2019, 289, 121704. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Kim, J.; Song, H.-S.; Kim, H.J.; Jeon, J.-M.; Sathiyanarayanan, G.; Yoon, J.-J.; Park, K.; Kim, Y.-G.; Yang, Y.-H. Microbial biodiesel production from oil palm biomass hydrolysate using marine Rhodococcus sp. YHY01. Bioresour. Technol. 2017, 233, 99–109. [Google Scholar] [CrossRef]
- Gumel, A.M.; Annuar, M.S.M.; Heidelberg, T. Biosynthesis and Characterization of Polyhydroxyalkanoates Copolymers Produced by Pseudomonas putida Bet001 Isolated from Palm Oil Mill Effluent. PLoS ONE 2012, 7, e45214. [Google Scholar] [CrossRef] [Green Version]
- Choi, T.-R.; Jeon, J.-M.; Bhatia, S.K.; Gurav, R.; Han, Y.H.; Park, Y.L.; Park, J.-Y.; Song, H.-S.; Park, H.Y.; Yoon, J.-J. Production of low molecular weight P (3HB-co-3HV) by butyrateacetoacetate CoA-transferase (cftAB) in Escherichia coli. Biotechnol. Bioprocess Eng. 2020, 25, 279–286. [Google Scholar] [CrossRef]
- Hokamura, A.; Fujino, K.; Isoda, Y.; Arizono, K.; Shiratsuchi, H.; Matsusaki, H. Characterization and identification of the proteins bound to two types of polyhydroxyalkanoate granules in Pseudomonas sp. 61-3. Biosci. Biotechnol. Biochem. 2015, 79, 1369–1377. [Google Scholar] [CrossRef] [Green Version]
- Choi, S.Y.; Cho, I.J.; Lee, Y.; Kim, Y.J.; Kim, K.J.; Lee, S.Y. Microbial Polyhydroxyalkanoates and Nonnatural Polyesters. Adv. Mater. 2020, 32, e1907138. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, N.; Orellana-Saez, M.; Pepczynska, M.; Enrione, J.; Bassas-Galia, M.; Acuña, J.M.B.-D.; Zacconi, F.C.; Marcoleta, A.E.; Poblete-Castro, I. Exploiting the natural poly(3-hydroxyalkanoates) production capacity of Antarctic Pseudomonas strains: From unique phenotypes to novel biopolymers. J. Ind. Microbiol. Biotechnol. 2019, 46, 1139–1153. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Bao, H.J.; Kang, C.-K.; Fukui, T.; Doi, Y. Production of a novel copolyester of 3-hydroxybutyric acid and medium-chain-length 3-hydroxyalkanoic acids by Pseudomonas sp. 61-3 from sugars. Appl. Microbiol. Biotechnol. 1996, 45, 363–370. [Google Scholar] [CrossRef]
- Matsusaki, H.; Manji, S.; Taguchi, K.; Kato, M.; Fukui, T.; Doi, Y. Cloning and Molecular Analysis of the Poly(3-hydroxybutyrate) and Poly(3-hydroxybutyrate-co-3-hydroxyalkanoate) Biosynthesis Genes in Pseudomonas sp. Strain 61-3. J. Bacteriol. 1998, 180, 6459–6467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orellana-Saez, M.; Pacheco, N.; Costa, J.I.; Mendez, K.N.; Miossec, M.J.; Meneses, C.; Castro-Nallar, E.; Marcoleta, A.E.; Poblete-Castro, I. In-Depth Genomic and Phenotypic Characterization of the Antarctic Psychrotolerant Strain Pseudomonas sp. MPC6 Reveals Unique Metabolic Features, Plasticity, and Biotechnological Potential. Front. Microbiol. 2019, 10, 1154. [Google Scholar] [CrossRef] [PubMed]
- Goh, Y.S.; Tan, I.K.P. Polyhydroxyalkanoate production by antarctic soil bacteria isolated from Casey Station and Signy Island. Microbiol. Res. 2012, 167, 211–219. [Google Scholar] [CrossRef]
- Li, R.; Jiang, Y.; Wang, X.; Yang, J.; Gao, Y.; Zi, X.; Zhang, X.; Gao, H.; Hu, N. Psychrotrophic Pseudomonas mandelii CBS-1 produces high levels of poly-β-hydroxybutyrate. SpringerPlus 2013, 2, 335. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.-J.; Choi, M.H.; Kim, T.-U.; Yoon, S.C. Accumulation of Polyhydroxyalkanoic Acid Containing Large Amounts of Unsaturated Monomers in Pseudomonas fluorescens BM07 Utilizing Saccharides and Its Inhibition by 2-Bromooctanoic Acid. Appl. Environ. Microbiol. 2001, 67, 4963–4974. [Google Scholar] [CrossRef] [Green Version]
- Choonut, A.; Prasertsan, P.; Klomklao, S.; Sangkharak, K. Study on mcl-PHA Production by Novel Thermotolerant Gram-Positive Isolate. J. Polym. Environ. 2020, 28. [Google Scholar] [CrossRef]
- Wellen, R.M.R.; Rabello, M.S.; Júnior, I.C.A.; Fechine, G.J.M.; Canedo, E.L. Melting and crystallization of poly(3-hydroxybutyrate): Effect of heating/cooling rates on phase transformation. Polímeros 2015, 25, 296–304. [Google Scholar] [CrossRef] [Green Version]
- Kong, Y.; Hay, J. The measurement of the crystallinity of polymers by DSC. Polymer 2002, 43, 3873–3878. [Google Scholar] [CrossRef]
- Matsusaki, H.; Abe, H.; Doi, Y. Biosynthesis and properties of poly(3-hydroxybutyrate-co-3-hydroxyalkanoates) by recombinant strains of Pseudomonas sp. 61-3. Biomacromolecules 2000, 1, 17–22. [Google Scholar] [CrossRef]
- Abe, H.; Doi, Y.; Kumagai, Y. Synthesis and Characterization of Poly[(R,S)-3-hydroxybutyrate-b-6-hydroxyhexanoate] as a Compatibilizer for a Biodegradable Blend of Poly[(R)-3-hydroxybutyrate] and Poly(6-hydroxyhexanoate). Macromole 1994, 27, 6012–6017. [Google Scholar] [CrossRef]
- Brandrup, J.; Immergut, E.H.; Grulke, E.A.; Abe, A.; Bloch, D.R. Polymer Handbook; Wiley: New York, NY, USA, 1999. [Google Scholar]
| Substrate | 3HB | HHx | 3HO | 3HD | 3HdDe | 3HdD | 3HtD |
|---|---|---|---|---|---|---|---|
| Glucose | 5.89 ± 0.07 | 8.77 ± 0.16 | 29.09 ± 0.14 | 37.27 ± 0.03 | 8.56 ± 0.15 | 9.82 ± 0.16 | 0.60 ± 0.03 |
| Glycerol | 5.99 ± 0.75 | 8.51 ± 0.28 | 39.80 ± 1.06 | 33.55 ± 0.19 | 5.58 ± 0.05 | 6.46 ± 0.33 | 0.12 ± 0.02 |
| Fructose | 5.04 ± 0.15 | 7.75 ± 0.10 | 25.60 ± 0.05 | 40.03 ± 0.31 | 9.88 ± 0.03 | 11.01 ± 0.20 | 0.68 ± 0.02 |
| Fructose syrup | 0.83 ± 0.20 | 9.30 ± 1.05 | 38.43 ± 0.49 | 30.36 ± 1.17 | 13.33 ± 0.32 | 0.83 ± 0.20 | ND |
| Strain | Substrate | Polymer Type | Content (%) | Volumetric Productivity (mg/L/h) | Reference |
|---|---|---|---|---|---|
| Pseudomonas sp. UMAB-40 | Glucose | mcl-PHA | 23 | 2.6 | [41] |
| Glycerol | mcl-PHA | 11 | 1.6 | [41] | |
| Sodium octanoate | mcl-PHA | 48 | 9.4 | [41] | |
| Pseudomonas sp. PAMC 28620 | Glycerol | mcl-PHA | 52 | 24 | [30] |
| Pseudomonas mandelii CBS-1 | Fructose | scl-PHA | 76 | 464 | [42] |
| Pseudomonas fluorescens BM07 | Fructose | mcl-PHA | 25 | 23 | [43] |
| Pseudomonas sp. B14-6 | Fructose | scl-co-mcl PHA | 35 | 4.6 | This study |
| Fructose syrup | scl-co-mcl PHA | 24 | 6.4 | This study |
| PHA Composition (mol %) | Molecular Weight | Thermal Properties | Reference | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Sample | 3HB (C4) | 3HA (C6-C12) | Mn (×104) | Mw (×104) | PD | Tg (°C) | Tm (°C) | ΔHm (J/g) | |
| P(3HA)B14-6 | 5 | 95 | 3.6 | 9.1 | 2.5 | 15.3 | 113 | 41.2 | This study |
| P(3HA)MPC6 | 89.5 | 10.5 | 118 | 490 | 4.1 | 2.3 | 164 | ND | [37] |
| P(3HA)1 | 44 | 56 | 4.0 | 139 | 4.4 | −43 | ND | 0 | [38] |
| P(3HA)2 | 88 | 12 | 34.9 | 48.9 | 1.4 | −13 | 106 | 38 | [47] |
| P(3HB) | 100 | 0 | 65.0 | 117 | 1.8 | 4 | 178 | 91 | [48] |
| LDPE | −30 | 130 | 220 | [49] | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, T.-R.; Park, Y.-L.; Song, H.-S.; Lee, S.M.; Park, S.L.; Lee, H.S.; Kim, H.-J.; Bhatia, S.K.; Gurav, R.; Choi, K.-Y.; et al. Fructose-Based Production of Short-Chain-Length and Medium-Chain-Length Polyhydroxyalkanoate Copolymer by Arctic Pseudomonas sp. B14-6. Polymers 2021, 13, 1398. https://doi.org/10.3390/polym13091398
Choi T-R, Park Y-L, Song H-S, Lee SM, Park SL, Lee HS, Kim H-J, Bhatia SK, Gurav R, Choi K-Y, et al. Fructose-Based Production of Short-Chain-Length and Medium-Chain-Length Polyhydroxyalkanoate Copolymer by Arctic Pseudomonas sp. B14-6. Polymers. 2021; 13(9):1398. https://doi.org/10.3390/polym13091398
Chicago/Turabian StyleChoi, Tae-Rim, Ye-Lim Park, Hun-Suk Song, Sun Mi Lee, Sol Lee Park, Hye Soo Lee, Hyun-Joong Kim, Shashi Kant Bhatia, Ranjit Gurav, Kwon-Young Choi, and et al. 2021. "Fructose-Based Production of Short-Chain-Length and Medium-Chain-Length Polyhydroxyalkanoate Copolymer by Arctic Pseudomonas sp. B14-6" Polymers 13, no. 9: 1398. https://doi.org/10.3390/polym13091398







