A Brief Review of Poly(Vinyl Chloride) (PVC) Recycling
Abstract
:1. Introduction
2. PVC Recycling
2.1. Mechanical Recycling
2.1.1. Post-Production PVC Recycling
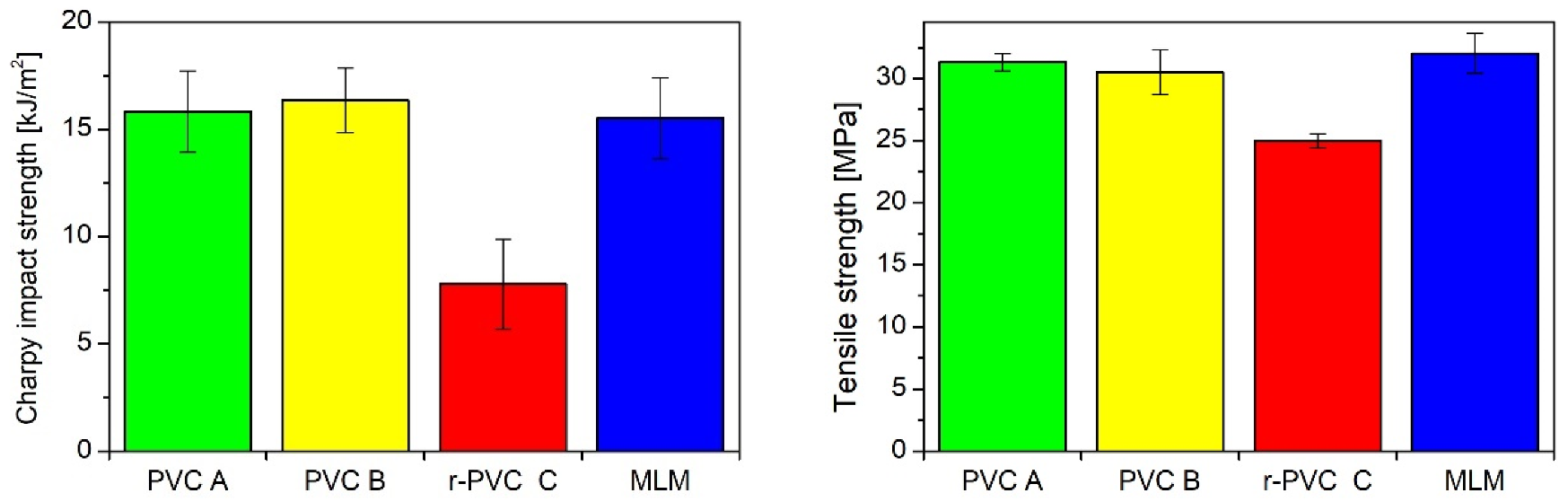
2.1.2. Post-Consumer PVC Recycling
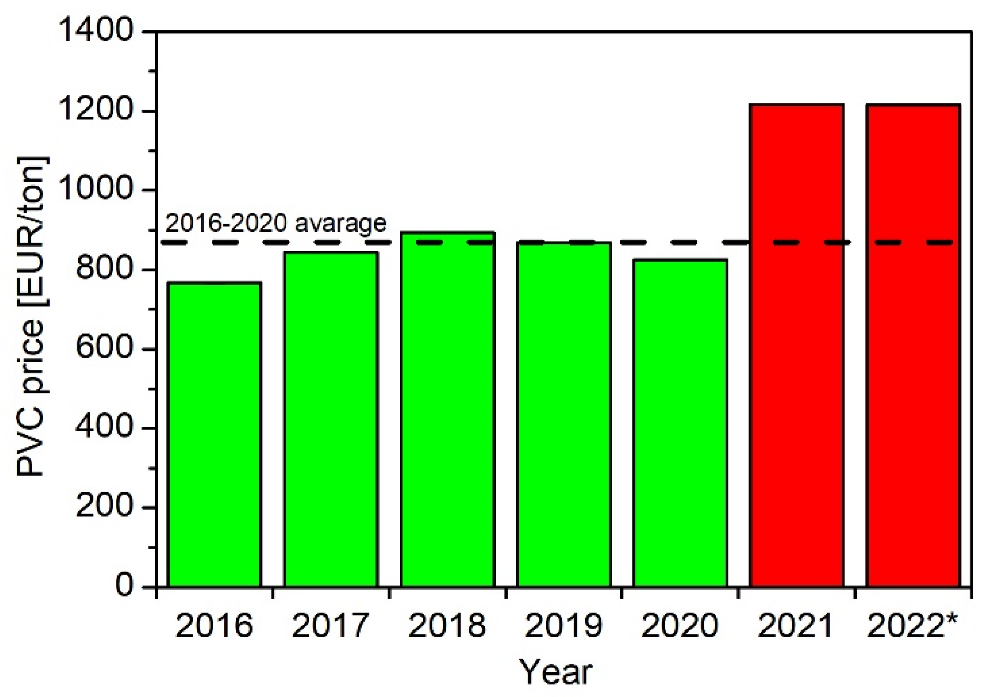
2.2. Feedstock Recycling
3. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- PVC—A Circular Material for the Future. Available online: https://pvc.org (accessed on 1 July 2022).
- Plastics Europe. Plastics the Facts, An Analysis of European Plastics Production, Demand and Waste Data; Plastics Europe: Brussels, Belgium, 2021. [Google Scholar]
- Alsadi, A. Evaluation of Carbon Footprint during the Life-Cycle of Four Different Pipe Materials. Ph.D. Thesis, Louisiana Tech University, Ruston, LA, USA, 2019. Available online: https://digitalcommons.latech.edu/dissertations/37 (accessed on 1 July 2022).
- Alsabri, A.; Al-Ghamdi, S.G. Carbon Footprint and Embodied Energy of PVC, PE, and PP Piping: Perspective on Environmental Performance. Energy Rep. 2020, 6, 364–370. [Google Scholar] [CrossRef]
- Jones, M.P.; Archodoulaki, V.-M.; Köck, B.-M. The Power of Good Decisions: Promoting Eco-Informed Design Attitudes in Plastic Selection and Use. Resour. Conserv. Recycl. 2022, 182, 106324. [Google Scholar] [CrossRef]
- Wang, M.; Ma, D. Reaction: Direct Chlorination of Ethane to Dichloroethane. Chem 2022, 8, 886–887. [Google Scholar] [CrossRef]
- Zichittella, G.; Ceruti, A.; Guillén-Gosálbez, G.; Pérez-Ramírez, J. Catalyst: A Step Forward for PVC Manufacture from Natural Gas. Chem 2022, 8, 883–885. [Google Scholar] [CrossRef]
- IPEX. Chemical Resistance Guide, Polyvinyl Chloride (PVC), 1st ed.; IPEX: Oakville, ON, Canada, 2020. [Google Scholar]
- Wypych, G. PVC Degradation and Stabilization; ChemTec Publishing: Toronto, ON, Canada, 2020. [Google Scholar]
- Brockmann, W.; Geiß, P.L.; Klingen, J.; Schröder, K.B. Adhesive Bonding: Materials, Applications and Technology; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009. [Google Scholar]
- James, N.R.; Jayakrishnan, A. Surface Thiocyanation of Plasticized Poly(Vinyl Chloride) and Its Effect on Bacterial Adhesion. Biomaterials 2003, 24, 2205–2212. [Google Scholar] [CrossRef]
- Mark, J.E. (Ed.) Physical Properties of Polymers Handbook; Springer: New York, NY, USA, 2007. [Google Scholar]
- Wypych, G. PVC Formulary; ChemTec Publishing: Toronto, ON, Canada, 2020. [Google Scholar]
- Cousins, K. Polymers in Building and Construction; Rapra Technology LTD: Shawbury, UK, 2002. [Google Scholar]
- PVC Applications. Available online: https://pvc.org/pvc-applications (accessed on 1 July 2022).
- Abdel-Monem, R.A.; Rabie, S.T.; El-Liethy, M.A.; Hemdan, B.A.; El-Nazer, H.A.; Gaballah, S.T. Chitosan- PVC Conjugates/Metal Nanoparticles for Biomedical Applications. Polym. Adv. Technol. 2022, 33, 514–523. [Google Scholar] [CrossRef]
- Zhong, R.; Wang, H.; Wu, X.; Cao, Y.; He, Z.; He, Y.; Liu, J. In Vitro Investigation of the Effect of Plasticizers on the Blood Compatibility of Medical Grade Plasticized Poly (Vinyl Chloride). J. Mater. Sci. Mater. Med. 2013, 24, 1985–1992. [Google Scholar] [CrossRef]
- Asadinezhad, A.; Novák, I.; Lehocký, M.; Bílek, F.; Vesel, A.; Junkar, I.; Sáha, P.; Popelka, A. Polysaccharides Coatings on Medical-Grade PVC: A Probe into Surface Characteristics and the Extent of Bacterial Adhesion. Molecules 2010, 15, 1007–1027. [Google Scholar] [CrossRef] [Green Version]
- Why PVC Floor Tiles (Vinyl Floor Tiles) Is the Best Choice for Hospital Flooring? Available online: https://www.accessfloorstore.com/news/110--why-pvc-floor-tiles-vinyl-floor-tiles-is-the-best-choice-for-hospital-flooring (accessed on 1 July 2022).
- Marsh, K.; Bugusu, B. Food Packaging Roles, Materials, and Environmental Issues. J. Food Sci. 2007, 72, R39–R55. [Google Scholar] [CrossRef]
- Pedrosa de Oliveira, D.; Costa, J.S.R.; Oliveira-Nascimento, L. Sustainability of Blisters for Medicines in Tablet Form. Sustain. Chem. Pharm. 2021, 21, 100423. [Google Scholar] [CrossRef]
- Meyer, M.; Dietrich, S.; Schulz, H.; Mondschein, A. Comparison of the Technical Performance of Leather, Artificial Leather, and Trendy Alternatives. Coatings 2021, 11, 226. [Google Scholar] [CrossRef]
- Cao, H.; Wool, R.; Sidoriak, E.; Dan, Q. Evaluating Mechanical Properties of Environmentally Friendly Leather Substitute (Eco-Leather). In Proceedings of the International Textile and Apparel Association Annual Conference, New Orleans, LA, USA, 13–18 October 2013; Volume 70. Available online: https://dr.lib.iastate.edu/handle/20.500.12876/503 (accessed on 1 July 2022).
- Zhang, W.; Zhang, J.; Li, H. Study on Abnormal Noise of Leather of Car Shift Mask Based on FAT. J. Phys. Conf. Ser. 2020, 1635, 012086. [Google Scholar] [CrossRef]
- Defonseka, C. Polymeric Coating Systems for Artificial Leather: Standard and Latest Technologies; Walter de Gruyter GmbH & Co KG: Berlin, Germany; Boston, MA, USA, 2022. [Google Scholar]
- Braun, D. Recycling of PVC. Prog. Polym. Sci. 2002, 27, 2171–2195. [Google Scholar] [CrossRef]
- Allen, N.S.; Edge, M. Perspectives on Additives for Polymers. 1. Aspects of Stabilization. J. Vinyl Addit. Technol. 2021, 27, 5–27. [Google Scholar] [CrossRef]
- Titow, M.V. PVC Plastics: Properties, Processing, and Applications; Elsevier Applied Science: London, UK; New York, NY, USA, 2012. [Google Scholar]
- Unar, I.N.; Soomro, S.A.; Aziz, S. Effect of Various Additives on the Physical Properties of Polyvinylchloride Resin. Pak. J. Anal. Environ. Chem. 2010, 11, 44–50. [Google Scholar]
- Ciacci, L.; Passarini, F.; Vassura, I. The European PVC Cycle: In-Use Stock and Flows. Resour. Conserv. Recycl. 2017, 123, 108–116. [Google Scholar] [CrossRef]
- Arnold, J.C.; Maund, B. The Properties of Recycled PVC Bottle Compounds. 2: Reprocessing Stability. Polym. Eng. Sci. 1990, 39, 1242–1250. [Google Scholar] [CrossRef]
- Arnold, J.C.; Maund, B. The Properties of Recycled PVC Bottle Compounds. 1: Mechanical Performance. Polym. Eng. Sci. 1999, 39, 1234–1241. [Google Scholar] [CrossRef]
- Hamos WRS Recycling Systems for PVC Window Profiles. Available online: https://www.hamos.com/products/recycling-equipment/recycling-systems-for-pvc-windows,37,eng,45 (accessed on 30 May 2022).
- METAL-PLAST No. 1 in the Recycling of PVC Windows, a Producer of Profiles for the Transport and Installation of Windows. Available online: https://www.youtube.com/watch?v=mrUKiWgUaXs (accessed on 30 May 2022).
- Recycling PVC (Vernie, France) SUEZ. Available online: https://www.youtube.com/watch?v=WS6pgbTmqYQ (accessed on 30 May 2022).
- MIXED PLASTICS SORTING LINE. Available online: https://disan.com.tr/mixed-plastic-sorting-plant/?lang=en (accessed on 30 May 2022).
- Tomaszewska, J.; Zajchowski, S. Właściwości Mechaniczne i Struktura Mieszanin Recyklatów Polietylenu i Poli(Chlorku Winylu) Napełnionych Ma̧czka̧ Drzewna̧. Polimery 2013, 58, 106–113. [Google Scholar] [CrossRef]
- Tomaszewska, J.; Wrześniewska-Tosik, K.; Zajchowski, S.; Partyka, A.; Mik, T. Tri-Component Recycled PVC/PE-HD Material Composite with Keratin Fibres. Fibres Text. East. Eur. 2016, 24, 73–80. [Google Scholar] [CrossRef]
- Andričić, B.; Kovačić, T.; Klarić, I. Properties of Recycled Material Containing Poly(Vinyl Chloride), Polypropylene, and Calcium Carbonate Nanofiller Branka. Polym. Eng. Sci. 2008, 48, 572–577. [Google Scholar] [CrossRef]
- Lewandowski, K.; Skórczewska, K.; Piszczek, K.; Manikowski, M. Modyfikacja Nieplastyfikowanego Poli(Chlorku Winylu) Do Aplikacji w Trójwarstwowych Rurach Paszociągowych. Polimery 2020, 65, 304–310. [Google Scholar] [CrossRef]
- Commission Regulation (EU) No 494/2011 of 20 May 2011. Available online: http://data.europa.eu/eli/reg/2011/494/2011-06-10 (accessed on 30 May 2022).
- Commission Regulation (EU) No 10/2011 of 14 January 2011. Available online: http://data.europa.eu/eli/reg/2011/10/2020-09-23 (accessed on 30 May 2022).
- 2005/84/EC, Annex XVII (Restrictions) of the REACH Chemicals Regulation 1907/2006. Available online: http://data.europa.eu/eli/dir/2005/84/2009-06-01 (accessed on 30 May 2022).
- Baitz, M.; Kreibig, J.; Byrne, E.; Makishi, C.; Kupfer, T.; Frees, N.; Bey, N.; Hansen, M.S.; Hansen, A.; Bosch, T.; et al. Final Report—Life Cycle Assessment (LCA) of PVC and of Principal Competing Materials; European Commission: Brussels, Belgium, 2004. [Google Scholar]
- Choi, W.Z. Removal of PVC from Mixed Plastic Waste by Gravity Separation Process. Geosyst. Eng. 2004, 7, 57–62. [Google Scholar] [CrossRef]
- Luciani, V.; Bonifazi, G.; Rem, P.; Serranti, S. Upgrading of PVC Rich Wastes by Magnetic Density Separation and Hyperspectral Imaging Quality Control. Waste Manag. 2015, 45, 118–125. [Google Scholar] [CrossRef]
- Gent, M.R.; Menendez, M.; Toraño, J.; Diego, I. Recycling of Plastic Waste by Density Separation: Prospects for Optimization. Waste Manag. Res. 2009, 27, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Pongstabodee, S.; Kunachitpimol, N.; Damronglerd, S. Combination of Three-Stage Sink-Float Method and Selective Flotation Technique for Separation of Mixed Post-Consumer Plastic Waste. Waste Manag. 2008, 28, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Mallampati, S.R.; Lee, C.H.; Park, M.H.; Lee, B.K. Processing Plastics from ASR/ESR Waste: Separation of Poly Vinyl Chloride (PVC) by Froth Flotation after Microwave-Assisted Surface Modification. J. Mater. Cycles Waste Manag. 2018, 20, 91–99. [Google Scholar] [CrossRef]
- Marques, G.A.; Tenório, J.A.S. Use of Froth Flotation to Separate PVC/PET Mixtures. Waste Manag. 2000, 20, 265–269. [Google Scholar] [CrossRef]
- Park, C.H.; Jeon, H.S.; Park, J.K. PVC Removal from Mixed Plastics by Triboelectrostatic Separation. J. Hazard. Mater. 2007, 144, 470–476. [Google Scholar] [CrossRef]
- Mallampati, S.R.; Lee, B.H.; Mitoma, Y.; Simion, C. Selective Sequential Separation of ABS/HIPS and PVC from Automobile and Electronic Waste Shredder Residue by Hybrid Nano-Fe/Ca/CaO Assisted Ozonisation Process. Waste Manag. 2017, 60, 428–438. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, H.; Wang, K.; Wang, H.; Wang, C. Green Flotation of Polyethylene Terephthalate and Polyvinyl Chloride Assisted by Surface Modification of Selective CaCO3 Coating. J. Clean. Prod. 2020, 242, 118441. [Google Scholar] [CrossRef]
- Królikowski, K.; Piszczek, K. Separation of High-Density Polyethylene/Poly(Ethylene Terephthalate)/Poly(Vinyl Chloride) Mixtures Based on Differences in Their Hardness. Polimery 2017, 62, 599–602. [Google Scholar] [CrossRef]
- Rodrigues, B.M.; Saron, C. Electrostatic Separation of Polymer Waste by Tribocharging System Based on Friction with PVC. Int. J. Environ. Sci. Technol. 2022, 19, 1293–1300. [Google Scholar] [CrossRef]
- Yuan, H.; Fu, S.; Tan, W.; He, J.; Wu, K. Study on the Hydrocyclonic Separation of Waste Plastics with Different Density. Waste Manag. 2014, 45, 108–111. [Google Scholar] [CrossRef]
- Mahmood, H.; Nart, F.; Pegoretti, A. Effective Recycling of End-of-life Polyvinyl Chloride Foams in Ethylene–Propylene Diene Monomers Rubber. J. Vinyl Addit. Technol. 2022, 1. [Google Scholar] [CrossRef]
- Lakhdar, A.; Moumen, A.; Mansouri, K. Experimental Study of the Mechanical Effect of Bio-Loads on PVC Recycling. J. Appl. Eng. Sci. 2022, 20, 221–229. [Google Scholar] [CrossRef]
- Youssef, A.M.; Abd El-Aziz, M.E.; Abouzeid, R.E. A Morphological and Mechanical Analysis of Composites from Modified Bagasse Fibers and Recycled Polyvinyl Chloride. Polym. Compos. 2022, 43, 2878–2887. [Google Scholar] [CrossRef]
- Awad, S.; Hamouda, T.; Midani, M.; Zhou, Y.; Katsou, E.; Fan, M. Date Palm Fibre Geometry and Its Effect on the Physical and Mechanical Properties of Recycled Polyvinyl Chloride Composite. Ind. Crops Prod. 2021, 174, 114172. [Google Scholar] [CrossRef]
- Dobrotă, D.; Petrescu, V.; Dimulescu, C.S.; Oleksik, M. Preparation and Characterization of Composites Materials with Rubber Matrix and with Polyvinyl Chloride Addition (PVC). Polymers 2020, 12, 1978. [Google Scholar] [CrossRef]
- Kumar, H.; Kumagai, S.; Kameda, T.; Saito, Y.; Yoshioka, T. Simultaneous Recovery of High-Purity Cu and Poly(Vinyl Chloride) from Waste Wire Harness via Swelling Followed by Ball Milling. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Bedeković, G.; Trbović, R. Electrostatic Separation of Aluminium from Residue of Electric Cables Recycling Process. Waste Manag. 2020, 108, 21–27. [Google Scholar] [CrossRef]
- Kumar, H.; Kumagai, S.; Kameda, T.; Saito, Y.; Yoshioka, T. One-Pot Wet Ball-Milling for Waste Wire-Harness Recycling. J. Mater. Cycles Waste Manag. 2021, 23, 461–469. [Google Scholar] [CrossRef]
- Barbakadze, K.; Brostow, W.; Granowski, G.; Hnatchuk, N.; Lohse, S.; Osmanson, A.T. Separation of Metal and Plastic Wastes from Wire and Cable Manufacturing for Effective Recycling. Resour. Conserv. Recycl. 2018, 139, 251–258. [Google Scholar] [CrossRef]
- Blinová, L.; Godovčin, P. Importance of Recycling the Waste-Cables Containing Copper and PVC. Res. Pap. Fac. Mater. Sci. Technol. Slovak Univ. Technol. 2021, 29, 1–21. [Google Scholar] [CrossRef]
- Sheih, S.W.; Tsai, M.S. Hot Water Separation Process for Copper and Insulating Material Recovery from Electric Cable Waste. Waste Manag. Res. 2000, 18, 478–484. [Google Scholar] [CrossRef]
- Xu, J.; Lu, J.; Kumagai, S.; Kameda, T.; Saito, Y.; Takahashi, K.; Hayashi, H.; Yoshioka, T. Validation of a Deplasticizer–Ball Milling Method for Separating Cu and PVC from Thin Electric Cables: A Simulation and Experimental Approach. Waste Manag. 2018, 82, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Xu, J.; Kumagai, S.; Kameda, T.; Saito, Y.; Yoshioka, T. Separation Mechanism of Polyvinyl Chloride and Copper Components from Swollen Electric Cables by Mechanical Agitation. Waste Manag. 2019, 93, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Suresh, S.S.; Mohanty, S.; Nayak, S.K. Composition Analysis and Characterization of Waste Polyvinyl Chloride (PVC) Recovered from Data Cables. Waste Manag. 2017, 60, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Boss, A.; Ab, S.I.V.F.; Nilsson, P.; Chlorvinyls, I.; Eriksson, A.; Iko, N.; Svenningsson, E.; Kabel, D.; Ab, S. New Technology for Recycling of Plastics from Cable Waste. In Proceedings of the 8th International Conference on Insulated Power Cables, Versailles, France, 19–23 June 2011; pp. 6–11. [Google Scholar]
- Markarian, J. Choosing a Melt Filtration System. Plast. Addit. Compd. 2008, 10, 32–35. [Google Scholar] [CrossRef]
- Hannemann, A. Compounding with Process-Constant Melt Filtration. Plast. Addit. Compd. 2006, 8, 34–38. [Google Scholar] [CrossRef]
- Grigorescu, R.M.; Ghioca, P.; Iancu, L.; David, M.E.; Ion, R.M.; Nicolae, C.A.; Gabor, R.A.; Radu, E.R.; Ganciarov, M.; Spurcaciu, B.; et al. Influence of Non-Metallic Fraction of Printed Circuit Boards Waste on Recycled Polyvinyl Chloride from Waste Wires. J. Appl. Polym. Sci. 2022, 139, e51469. [Google Scholar] [CrossRef]
- Gohatre, O.K.; Biswal, M.; Mohanty, S.; Nayak, S.K. Effect of Silane Treated Fly Ash on Physico-Mechanical, Morphological, and Thermal Properties of Recycled Poly(Vinyl Chloride) Composites. J. Appl. Polym. Sci. 2021, 138. [Google Scholar] [CrossRef]
- Gohatre, O.K.; Biswal, M.; Mohanty, S.; Nayak, S.K. Study on Thermal, Mechanical and Morphological Properties of Recycled Poly(Vinyl Chloride)/Fly Ash Composites. Polym. Int. 2020, 69, 552–563. [Google Scholar] [CrossRef]
- Manjunatha, M.; Seth, D.; Balaji, K.V.G.D.; Chilukoti, S. Influence of PVC Waste Powder and Silica Fume on Strength and Microstructure Properties of Concrete: An Experimental Study. Case Stud. Constr. Mater. 2021, 15, e00610. [Google Scholar] [CrossRef]
- Manjunatha, M.; Seth, D.; Balaji, K.V.G.D. Role of Engineered Fibers on Fresh and Mechanical Properties of Concrete Prepared with GGBS and PVC Waste Powder—An Experimental Study. Mater. Today Proc. 2021, 47, 3683–3693. [Google Scholar] [CrossRef]
- Bolat, H.; Erkus, P. Use of Polyvinyl Chloride (PVC) Powder and Granules as Aggregate Replacement in Concrete Mixtures. Sci. Eng. Compos. Mater. 2016, 23, 209–216. [Google Scholar] [CrossRef]
- Gregorova, V.; Ledererova, M.; Stefunkova, Z. Investigation of Influence of Recycled Plastics from Cable, Ethylene Vinyl Acetate and Polystyrene Waste on Lightweight Concrete Properties. Procedia Eng. 2017, 195, 127–133. [Google Scholar] [CrossRef]
- Ruiz-Herrero, J.L.; Velasco Nieto, D.; López-Gil, A.; Arranz, A.; Fernández, A.; Lorenzana, A.; Merino, S.; De Saja, J.A.; Rodríguez-Pérez, M.Á. Mechanical and Thermal Performance of Concrete and Mortar Cellular Materials Containing Plastic Waste. Constr. Build. Mater. 2016, 104, 298–310. [Google Scholar] [CrossRef]
- Wu, S.; Montalvo, L. Repurposing Waste Plastics into Cleaner Asphalt Pavement Materials: A Critical Literature Review. J. Clean. Prod. 2021, 280, 124355. [Google Scholar] [CrossRef]
- Fakhri, M.; Shahryari, E.; Ahmadi, T. Investigate the Use of Recycled Polyvinyl Chloride (PVC) Particles in Improving the Mechanical Properties of Stone Mastic Asphalt (SMA). Constr. Build. Mater. 2022, 326, 126780. [Google Scholar] [CrossRef]
- Joseph, B.; James, J.; Kalarikkal, N.; Thomas, S. Recycling of Medical Plastics. Adv. Ind. Eng. Polym. Res. 2021, 4, 199–208. [Google Scholar] [CrossRef]
- What Is PVC Recycling in Hospitals? Available online: http://recyclinginhospitals.com.au/ (accessed on 1 July 2022).
- Chantreux, M.; Ricard, D.; Asia, L.; Rossignol, S.; Wong-Wah-Chung, P. Additives as a Major Source of Radiolytic Organic Byproducts of Polyvinyl Chloride (PVC). Radiat. Phys. Chem. 2021, 188. [Google Scholar] [CrossRef]
- Kumagai, S.; Lu, J.; Fukushima, Y.; Ohno, H.; Kameda, T.; Yoshioka, T. Diagnosing Chlorine Industrial Metabolism by Evaluating the Potential of Chlorine Recovery from Polyvinyl Chloride Wastes—A Case Study in Japan. Resour. Conserv. Recycl. 2018, 133, 354–361. [Google Scholar] [CrossRef]
- Xu, X.; Zhu, D.; Wang, X.; Deng, L.; Fan, X.; Ding, Z.; Zhang, A.; Xue, G.; Liu, Y.; Xuan, W.; et al. Transformation of Polyvinyl Chloride (PVC) into a Versatile and Efficient Adsorbent of Cu(II) Cations and Cr(VI) Anions through Hydrothermal Treatment and Sulfonation. J. Hazard. Mater. 2022, 423, 126973. [Google Scholar] [CrossRef] [PubMed]
- Evode, N.; Qamar, S.A.; Bilal, M.; Barceló, D.; Iqbal, H.M.N. Plastic Waste and Its Management Strategies for Environmental Sustainability. Case Stud. Chem. Environ. Eng. 2021, 4, 100142. [Google Scholar] [CrossRef]
- Miliute-Plepiene, J.; Fråne, A.; Almasi, A.M. Overview of Polyvinyl Chloride (PVC) Waste Management Practices in the Nordic Countries. Clean. Eng. Technol. 2021, 4, 100246. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, S.; Han, X.; Zhang, X.; Yi, M.; Yang, S.; Yu, D.; Liu, W. Catalytic Dechlorination and Charring Reaction of Polyvinyl Chloride by CuAl Layered Double Hydroxide. Energy Fuels 2018, 32, 2407–2413. [Google Scholar] [CrossRef]
- Yuan, Z.; Zhang, J.; Zhao, P.; Wang, Z.; Cui, X.; Gao, L.; Guo, Q.; Tian, H. Synergistic Effect and Chlorine-Release Behaviors during Co-Pyrolysis of LLDPE, PP, and PVC. ACS Omega 2020, 5, 11291–11298. [Google Scholar] [CrossRef]
- Poerschmann, J.; Weiner, B.; Woszidlo, S.; Koehler, R.; Kopinke, F.D. Hydrothermal Carbonization of Poly(Vinyl Chloride). Chemosphere 2015, 119, 682–689. [Google Scholar] [CrossRef]
- Miandad, R.; Rehan, M.; Nizami, A.-S.; El-Fetouh Barakat, M.A.; Ismail, I.M. The Energy and Value-Added Products from Pyrolysis of Waste Plastics. In Environmental Footprints and Eco-Design of Products and Processes; Karthikeyan, O., Heimann, K., Muthu, S., Eds.; Springer: Singapore, 2016; pp. 333–355. [Google Scholar] [CrossRef]
- Hubáček, J.; Lederer, J.; Kuráň, P.; Koutník, P.; Gholami, Z.; Zbuzek, M.; Bačiak, M. Dechlorination during Pyrolysis of Plastics: The Potential of Stepwise Pyrolysis in Combination with Metal Sorbents. Fuel Process. Technol. 2022, 231, 107226. [Google Scholar] [CrossRef]
- Wang, G.; Liu, S.; Zhang, H.; Wang, J.; Xue, Q. Pulverization of Waste Polyvinyl Chloride (PVC) Film by Low Temperature Heat Treatment and Properties of Pulverized Product for Blast Furnace Injection as Alternative Fuel. Polymers 2022, 14, 1689. [Google Scholar] [CrossRef]
- Kameda, T.; Ono, M.; Grause, G.; Mizoguchi, T.; Yoshioka, T. Ball Mill-Assisted Dechlorination of Flexible and Rigid Poly (Vinyl Chloride) in NaOH/EG Solution. Ind. Eng. Chem. Res. 2008, 47, 8619–8624. [Google Scholar] [CrossRef]
- Lu, J.; Borjigin, S.; Kumagai, S.; Kameda, T.; Saito, Y.; Yoshioka, T. Practical Dechlorination of Polyvinyl Chloride Wastes in NaOH/Ethylene Glycol Using an up-Scale Ball Mill Reactor and Validation by Discrete Element Method Simulations. Waste Manag. 2019, 99, 31–41. [Google Scholar] [CrossRef]
- Kameda, T.; Fukushima, S.; Shoji, C.; Grause, G.; Yoshioka, T. Electrodialysis for NaCl/EG Solution Using Ion-Exchange Membranes. J. Mater. Cycles Waste Manag. 2013, 15, 111–114. [Google Scholar] [CrossRef]
- Zhao, P.; Li, T.; Yan, W.; Yuan, L. Dechlorination of PVC Wastes by Hydrothermal Treatment Using Alkaline Additives. Environ. Technol. 2018, 39, 977–985. [Google Scholar] [CrossRef]
- Jiang, G.; Sanchez Monsalve, D.A.; Clough, P.; Jiang, Y.; Leeke, G.A. Understanding the Dechlorination of Chlorinated Hydrocarbons in the Pyrolysis of Mixed Plastics. ACS Sustain. Chem. Eng. 2021, 9, 1576–1589. [Google Scholar] [CrossRef]
- Li, T.; Zhao, P.; Lei, M.; Li, Z. Understanding Hydrothermal Dechlorination of PVC by Focusing on the Operating Conditions and Hydrochar Characteristics. Appl. Sci. 2017, 7, 256. [Google Scholar] [CrossRef] [Green Version]
- Yang, M.; Zhao, P.; Cui, X.; Geng, F.; Guo, Q. Kinetics Study on Hydrothermal Dechlorination of Poly(Vinyl Chloride) by in-Situ Sampling. Environ. Technol. Innov. 2021, 23, 101703. [Google Scholar] [CrossRef]
- Ephraim, A.; Pham Minh, D.; Lebonnois, D.; Peregrina, C.; Sharrock, P.; Nzihou, A. Co-Pyrolysis of Wood and Plastics: Influence of Plastic Type and Content on Product Yield, Gas Composition and Quality. Fuel 2018, 231, 110–117. [Google Scholar] [CrossRef] [Green Version]
- Kuramochi, H.; Nakajima, D.; Goto, S.; Sugita, K.; Wu, W.; Kawamoto, K. HCl Emission during Co-Pyrolysis of Demolition Wood with a Small Amount of PVC Film and the Effect of Wood Constituents on HCl Emission Reduction. Fuel 2008, 87, 3155–3157. [Google Scholar] [CrossRef]
- Matsuzawa, Y.; Ayabe, M.; Nishino, J. Acceleration of Cellulose Co-Pyrolysis with Polymer. Polym. Degrad. Stab. 2001, 71, 435–444. [Google Scholar] [CrossRef]
- Li, H.Z.; Zhang, Y.N.; Guo, J.Z.; Lv, J.Q.; Huan, W.W.; Li, B. Preparation of Hydrochar with High Adsorption Performance for Methylene Blue by Co-Hydrothermal Carbonization of Polyvinyl Chloride and Bamboo. Bioresour. Technol. 2021, 337, 125442. [Google Scholar] [CrossRef]
- Lu, P.; Huang, Q.; (Thanos) Bourtsalas, A.C.; Chi, Y.; Yan, J. Synergistic Effects on Char and Oil Produced by the Co-Pyrolysis of Pine Wood, Polyethylene and Polyvinyl Chloride. Fuel 2018, 230, 359–367. [Google Scholar] [CrossRef]
- Yuan, R.; Shen, Y. Catalytic Pyrolysis of Biomass-Plastic Wastes in the Presence of MgO and MgCO3 for Hydrocarbon-Rich Oils Production. Bioresour. Technol. 2019, 293, 122076. [Google Scholar] [CrossRef]
- Maraveas, C. Environmental Sustainability of Greenhouse Covering Materials. Sustainability 2019, 11, 6129. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.P.; Zhang, C.C.; Zhang, F.S. A Novel Process for Waste Polyvinyl Chloride Recycling: Plant Growth Substrate Development. J. Environ. Chem. Eng. 2021, 9, 105475. [Google Scholar] [CrossRef]
- Chen, X.; Bai, X. Co-Conversion of Wood and Polyvinyl Chloride to Valuable Chemicals and High-Quality Solid Fuel. Waste Manag. 2022, 144, 376–386. [Google Scholar] [CrossRef]
- Zhang, N.; Shen, Y. One-Step Pyrolysis of Lignin and Polyvinyl Chloride for Synthesis of Porous Carbon and Its Application for Toluene Sorption. Bioresour. Technol. 2019, 284, 325–332. [Google Scholar] [CrossRef]
- Xu, Y.; Luo, G.; He, S.; Deng, F.; Pang, Q.; Xu, Y.; Yao, H. Efficient Removal of Elemental Mercury by Magnetic Chlorinated Biochars Derived from Co-Pyrolysis of Fe(NO3)3-Laden Wood and Polyvinyl Chloride Waste. Fuel 2019, 239, 982–990. [Google Scholar] [CrossRef]
- Xu, Y.; Zeng, X.; Luo, G.; Zhang, B.; Xu, P.; Xu, M.; Yao, H. Chlorine-Char Composite Synthesized by Co-Pyrolysis of Biomass Wastes and Polyvinyl Chloride for Elemental Mercury Removal. Fuel 2016, 183, 73–79. [Google Scholar] [CrossRef]
- Wang, J.; Wang, F.; Duan, H.; Li, Y.; Xu, J.; Huang, Y.; Liu, B.; Zhang, T. Polyvinyl Chloride-Derived Carbon Spheres for CO2 Adsorption. ChemSusChem 2020, 13, 6426–6432. [Google Scholar] [CrossRef]
- Khan, M.I.H.; Rana, M.; Nshizirungu, T.; Jo, Y.T.; Park, J.H. Recovery of Valuable and Hazardous Metals (Ni, Co, and Cd) from Spent Ni-Cd Batteries Using Polyvinyl Chloride (PVC) in Subcritical Water. ACS Sustain. Chem. Eng. 2022, 10, 2368–2379. [Google Scholar] [CrossRef]
- Baláž, M.; Boldyreva, E.V.; Rybin, D.; Pavlović, S.; Rodríguez-Padrón, D.; Mudrinić, T.; Luque, R. State-of-the-Art of Eggshell Waste in Materials Science: Recent Advances in Catalysis, Pharmaceutical Applications, and Mechanochemistry. Front. Bioeng. Biotechnol. 2021, 8. [Google Scholar] [CrossRef]
- Zhao, P.; Li, Z.; Li, T.; Yan, W.; Ge, S. The Study of Nickel Effect on the Hydrothermal Dechlorination of PVC. J. Clean. Prod. 2017, 152, 38–46. [Google Scholar] [CrossRef]
- Ma, D.; Liang, L.; Hu, E.; Chen, H.; Wang, D.; He, C.; Feng, Q. Dechlorination of Polyvinyl Chloride by Hydrothermal Treatment with Cupric Ion. Process Saf. Environ. Prot. 2021, 146, 108–117. [Google Scholar] [CrossRef]
- Zakharyan, E.M.; Petrukhina, N.N.; Maksimov, A.L. Pathways of Chemical Recycling of Polyvinyl Chloride: Part 1. Russ. J. Appl. Chem. 2020, 93, 1271–1313. [Google Scholar] [CrossRef]
- Ye, L.; Li, T.; Hong, L. Co-Pyrolysis of Fe3O4-Poly(Vinyl Chloride) (PVC) Mixtures: Mitigation of Chlorine Emissions during PVC Recycling. Waste Manag. 2021, 126, 832–842. [Google Scholar] [CrossRef]
- Kumagai, S.; Hirahashi, S.; Grause, G.; Kameda, T.; Toyoda, H.; Yoshioka, T. Alkaline Hydrolysis of PVC-Coated PET Fibers for Simultaneous Recycling of PET and PVC. J. Mater. Cycles Waste Manag. 2018, 20, 439–449. [Google Scholar] [CrossRef]
- Zakharyan, E.M.; Petrukhina, N.N.; Dzhabarov, E.G.; Maksimov, A.L. Pathways of Chemical Recycling of Polyvinyl Chloride. Part 2. Russ. J. Appl. Chem. 2020, 93, 1445–1490. [Google Scholar] [CrossRef]
- Nishibata, H.; Uddin, M.A.; Kato, Y. Simultaneous Degradation and Dechlorination of Poly (Vinyl Chloride) by a Combination of Superheated Steam and CaO Catalyst/Adsorbent. Polym. Degrad. Stab. 2020, 179, 109225. [Google Scholar] [CrossRef]
- Kurashima, K.; Kumagai, S.; Kameda, T.; Saito, Y.; Yoshioka, T. Heavy Metal Removal from Municipal Solid Waste Fly Ash through Chloride Volatilization Using Poly(Vinyl Chloride) as Chlorinating Agent. J. Mater. Cycles Waste Manag. 2020, 22, 1270–1283. [Google Scholar] [CrossRef]
- Slapak, M.J.P.; Van Kasteren, J.M.N.; Drinkenburg, A.A.H. Design of a Process for Steam Gasification of PVC Waste. Resour. Conserv. Recycl. 2000, 30, 81–93. [Google Scholar] [CrossRef]
- Baláž, M.; Bujňáková, Z.; Achimovičová, M.; Tešinský, M.; Baláž, P. Simultaneous Valorization of Polyvinyl Chloride and Eggshell Wastes by a Semi-Industrial Mechanochemical Approach. Environ. Res. 2019, 170, 332–336. [Google Scholar] [CrossRef]
- Polyvinyl Chloride Futures (DPVc1). Available online: in.investing.com/commodities/pvc-com-futures (accessed on 1 July 2022).
- EUR/CNY Euro Chinese Yuan. Available online: www.investing.com/currencies/eur-cny-historical-data (accessed on 1 July 2022).
- Our History. Available online: www.vinylplus.eu/about-us/our-history/ (accessed on 1 July 2022).





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewandowski, K.; Skórczewska, K. A Brief Review of Poly(Vinyl Chloride) (PVC) Recycling. Polymers 2022, 14, 3035. https://doi.org/10.3390/polym14153035
Lewandowski K, Skórczewska K. A Brief Review of Poly(Vinyl Chloride) (PVC) Recycling. Polymers. 2022; 14(15):3035. https://doi.org/10.3390/polym14153035
Chicago/Turabian StyleLewandowski, Krzysztof, and Katarzyna Skórczewska. 2022. "A Brief Review of Poly(Vinyl Chloride) (PVC) Recycling" Polymers 14, no. 15: 3035. https://doi.org/10.3390/polym14153035
APA StyleLewandowski, K., & Skórczewska, K. (2022). A Brief Review of Poly(Vinyl Chloride) (PVC) Recycling. Polymers, 14(15), 3035. https://doi.org/10.3390/polym14153035





