2.1. Bilayer Hydrogels for Skin Regeneration
A separate area is occupied by bilayer hydrogel dressings for the treatment of skin diseases. The biocompatibility of hydrogels is explained by their similarity to the macromolecular components of the body. An optimal hydrogel scaffold should have high degradation rates as well as reproducible and controllable characteristics such as (i) material composition; (ii) environment; (iii) structure; (iv) surface treatment; (v) external intervention; and (vi) physical loading, so that it can be used in wound healing, cell differentiation, angiogenesis, etc. [
88,
89].
Bilayer hydrogels have revealed greater biocompatibility with living organisms. In addition, the two layers, showing multifunctional properties, have different tasks: the upper layer protects against the influence of external factors and maintains a humid environment, while the lower layer promotes cell adhesion and proliferation [
90].
Skin lesions of a diverse nature of origin increase the risk of consequences for the body, even after years. In order to avoid the appearance of adhesions after surgery, a bilayer hydrogel dressing was developed consisting of gelatin and polyvinyl alcohol (PVA) with simultaneous non-reagent radiation-induced crosslinking by γ-rays and having both dressing and anti-adhesion properties [
91].
Wounds caused by diabetes differ from typical skin lesions and healing stages [
92]. In diabetic wounds, there are changes in keratinocyte function that occur due to the effect of insulin on the proliferation, differentiation, and migration of keratinocytes, which consequently leads to delayed wound healing [
93]. Combining approaches for the treatment of diabetic wounds have been embodied by the creation of bilayer wound dressings with different therapeutic properties [
94]. Gelatine methacrylamide (GelMa) was used as the base for the hydrogel double layer dressing. In addition, referring to previous studies, in order to impart and combine therapeutic properties in each of the layers, the authors of the work encapsulated the following additives: the top layer consisted of a layer of ε-poly-L-lysine (PLL) with strong antibacterial properties against infections and methacryloyl-substituted recombinant tropoelastin (MeTro), which helps to repair the wound and increase the rate of wound healing [
95,
96]. The low layer of the dressing with the addition of VEGF-mimetic peptides, which can enhance the angiogenesis process and collagen fiber regeneration, is porous. Initially, the two layers were injectable, but after exposure to blue light (405 nm) for 3 min, the liquid solidified on the wound without changing shape.
An in vivo study has proven the effectiveness of the use of a two-layer dressing, which deprives all of the necessary therapeutic methods for the treatment of a diabetic wound in comparison with single-layer component hydrogels (
Figure 3).
The sorption capacity of hydrogels is an important material parameter that determines their unique properties and drug release control from polymer networks [
97]. Hydrogels, as three-dimensional polymeric networks, can be chemically stable and able to reversibly swell and hold a large volume of liquid [
98]. A bioinspired hydrogel-forming scaffold was developed as a two-layer microneedle (MN) adhesive, consisting of a swelling shell based on mussel adhesive protein/hyaluronic acid (MAP/HA) and a non-swelling core based on silk fibroin (SF) with silkworm cocoon, the action of which is based on its use mainly in a humid environment [
99]. Endoparasites tightly fixed in the host’s intestines have also been investigated. The results of the ex vivo study showed that the resulting hydrogel composition had excellent wound sealing ability against luminal leaks (139.7 ± 14.1 mm Hg), which was comparable to the suturing (151.0 ± 23.3 mm Hg). The authors, referring to previous studies, stated that MAPs, which contain almost equal amounts of both aromatic and cationic residues, can efficiently attach to biological substrates through various chemical/physical interactions such as the cation–π interaction, π–π-stacking, hydrogen bonds, and electrostatic interactions [
100,
101,
102]. At the same time, the two-layer coating demonstrates adhesion to both semi-dry and wet skin, unlike a commercial medical plaster, when it was applied only to semi-dry surfaces. Drug delivery can be quite effective due to the excellent interfacial adhesion via various physical/chemical interactions (
Figure 4).
Antibacterial and Antioxidant Activity
The antimicrobial activity of hydrogels may be mediated either by encapsulation in the gel network and the subsequent release of drugs, or some gels are antimicrobial in the case when the hydrogel itself is polycationic and the network also exhibits antibacterial activity itself [
103,
104]. The choice of the antimicrobial component depends on the virulence, the type of patient and pathologies, the type and location of the wound, and the amount of exudate produced by the wound [
105]. Antibacterial drugs include antibiotics, some biological extracts, natural polymers, and some metal nanoparticles [
106]. Recently, hydrogels with sensitive components such as pH and are light- and temperature-sensitive have attracted scientific and practical interest.
Oustadi et al. developed a locally releasing ibuprofen bilayer dressing with membrane bactericidal activity. Initially, the layer with 1% ibuprofen was obtained in various ratios of a poly(vinyl alcohol) (PVA)/poly(vinyl pyrrolidone) (PVP) solution. The hydrogels were crosslinked using the repeated freeze–thaw cycles followed by lyophilization as the bottom layer [
107].
The loading efficiency and loading capacity of the ibuprofen of 88.50 ± 2.05% and 0.087 ± 0.003%, respectively, were achieved. The drug release percentage was 30% and 50% at the first and third hours, respectively. These results indicate that at the initial stage, the drug was burst released. The final release was approximately 90% and 100% after 10 and 15 h, respectively. The drug was completely released within 16 h, which underlines the useful property of antibacterial materials to prevent bacterial growth at the early stages of the wound process.
The study of the hydrogel mechanical properties revealed the decrease in the tensile strength with the increase in the PVP content. The authors suggest that the annular structure of PVP plays a role in this phenomenon. PVP creates more space between the polymer chains for greater mobility, and as a result, as the PVP content increases, the structure becomes less dense, resulting in the decrease in PVA/PVP tensile strength.
Various skin diseases require treatment with antibiotics. The aim of the research by Tamahkar et al. was the creation of a multilayer wound dressing with the controlled release of antibiotics [
108]. Hydrogels were prepared in four layers using carboxylated polyvinyl alcohol (PVA-C), gelatin (G), hyaluronic acid (HA), and gelatin, respectively, via a layer-by-layer self-assembly technique. The top layers (PVA-C and G) provided moisture control and a physical barrier to microorganisms. The HA-based middle layer was designed as the antibiotic loaded layer. The lower layer served as the control membrane for the release of the antibiotic and ensured the removal of excess exudate from the wound. The following models were used to describe the release of the antibiotic from the hydrogel matrix: Zero-order model (1); First-order model (2); Higuchi model (3); and the Korsmeyer–Peppas model (4) (
Table 3).
where Q
t is the drug released amount at time
t; Q
0 is the initial drug amount of the release medium; Q
eq is the amount of drug released at equilibrium;
k0 is the rate constant of the zero order model;
k1 is the rate constant of the first order model;
kH the rate constant of the Higuchi kinetic model;
kKP is the rate constant of the Korsmeyer–Peppas kinetic model;
t is the release time;
n is the release exponent.
The zero-order kinetic model was the most adequate parameter to describe the drug release because the drug release was only time dependent and independent from the drug concentration. Nearly 65% of ampicillin was released within 7 days, indicating that the long-term release profile could be achieved using the prepared multilayer hydrogels. The authors of the study developed the hydrogel dressing with prolonged time periods of drug release compared with the previously fast releasing kinetics in drug release studies [
109,
110,
111,
112].
The programmed release of an antibiotic over a specified period has been demonstrated by Contardi et al. Wound dressing production is an environmentally friendly water-based molding process where the first layer (for the direct contact with the wound) is polyvinylpyrrolidone (PVP) containing the commercial antiseptic Neomercurocromo
® (Neo), and the second layer is a mixture of hyaluronic acid (HA) and PVP containing ciprofloxacin [
113]. In vitro drug release measurements were collected from the following samples: ciprofloxacin and eosin release from BL (full bilayer), eosin release from BLN (bilayer without ciprofloxacin), ciprofloxacin release from SLHC (lower layer only), and eosin release from SLHC (lower layer only).
Glycerin was used as the plasticizer. Peel adhesion tests showed self-adhesion to both wet and dry human skin surfaces. This material showed high antibacterial activity against
Staphylococcus aureus,
Escherichia coli, and
Pseudomonas aeruginosa and reliable adhesion (
Figure 5).
The presence of silver nanoparticles (AgNP) endows the bilayer hydrogel with antibacterial properties and promotes an accelerated healing process.
The top layer of the material consists of carboxylated chitosan, which, due to its bacterial activity, is able to prevent the penetration of a bacterial infection. The basis of the bottom layer is polyvinyl alcohol (PVA) and polyethylene glycol (PEG) as a blowing agent. A layer with small pores is able to maintain a moist environment, and a layer with large pores absorbs the wound exudate and provides oxygen exchange. This material also exhibits high adhesive activity due to the catechin groups of PDA [
114] (
Figure 6).
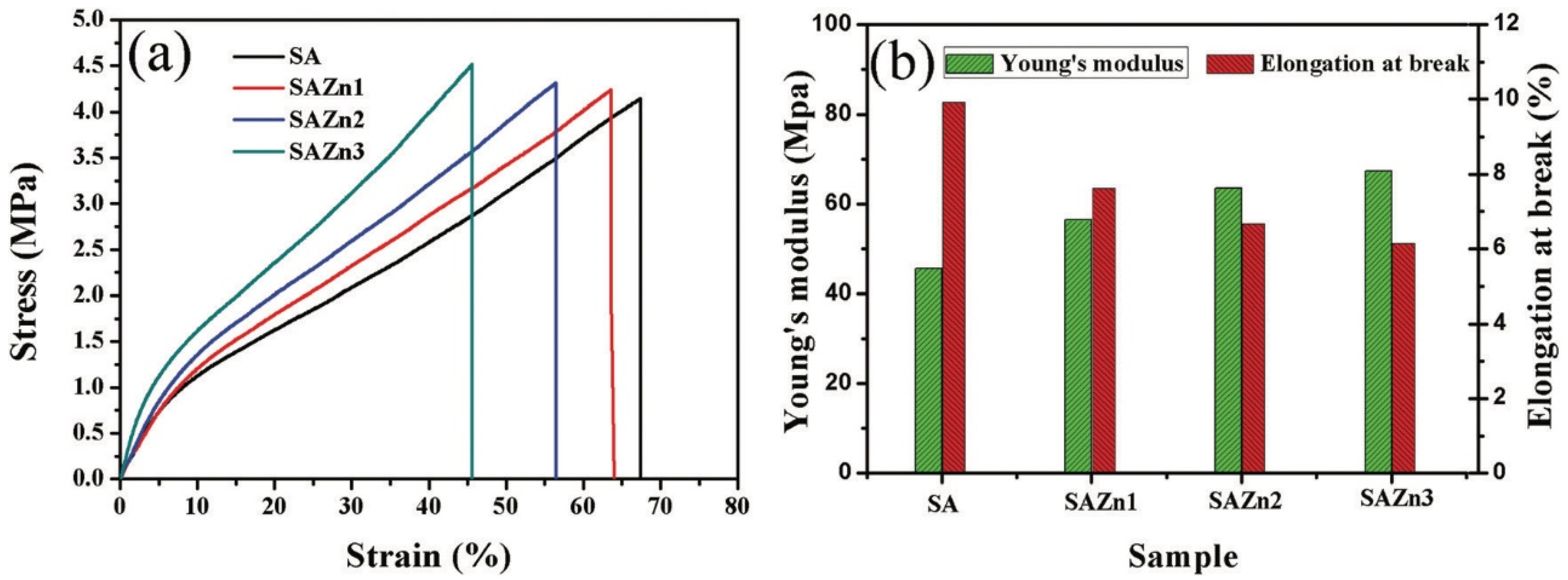
Another option for the use of alginate salts in obtaining an antibacterial two-layer dressing was described in [
115]. Zinc oxide (ZnO) nanoparticles have also been used as an antibacterial agent. ZnO shows significant growth inhibition of a wide range of bacteria [
116]. Hydrogels, in which metal oxide nanoparticles are encapsulated, also have good antibacterial properties, but unlike metal nanoparticles, the antibacterial mechanism of metal oxide nanoparticles is different [
117]. There are two common antibacterial mechanisms of the action of metal oxide nanoparticles in hydrogel materials: (1) antimicrobial toxicity arises from the production of metal ions by the nanoparticles; and (2) oxidative stress due to the formation of reactive oxygen species (ROS) on the surface of nanoparticles [
118]. The outer layer of the bilayer hydrogel material of the ZnO/SA film prevented the entry of bacteria, while the authors reported on the ability of oxygen to diffuse and maintain a favorable moist environment at the wound boundary; the inner layer was water-absorbent for the wound exudate. It is worth noting that the most well-known theory of alginate–metal binding is the “Egg-box” model. This model assumes the arrangement of the hydroxyl groups of polymeric α-L-guluronates (G) in such a way that special cavities for cations are formed, like an egg carton for eggs [
119]. The zinc oxide nanoparticle encapsulation not only led to the manifestation of antibacterial properties and an increase in the healing rate, but also improved the mechanical properties of the resulting material—the Young’s modulus increased by 10%, and the elongation at break decreased from 67.4 to 45.6%, respectively (
Figure 7) (
Table 4).
As a wound dressing for skin and the prevention soft tissue infections, a hybrid based on an antibacterial bilayer sponge scaffold made of silk fibroin/gelatin (SF/Gel) loaded with various concentrations of a cationic antimicrobial peptide (CM
11 peptide) was developed. The release of the CM
11 peptide after 500 min was observed from the samples, while the complete release of up to 80% was observed after 4000 min. The authors confirmed that compared to the blank (SF/Gel), the material modified with antimicrobial peptides demonstrated improved mechanical and antibacterial properties against
Staphylococcus aureus, Escherichia coli, and
Pseudomonas aeruginosa [
120].
In addition, the polymers used have to be able to provide adequate mechanical support and provide an environment for cell adhesion, proliferation, and differentiation [
121]. The creation of bilayer systems allows for the use of polymers with mutually complementary properties. Sodium alginate is biocompatible, non-toxic, economically available, but at the same time cannot prevent bacteria from entering into the wound. Kiti et al. obtained a two-layer structure containing sodium alginate with the addition of curcumin to accelerate wound healing and a separate layer of chitosan, which provided the necessary mechanical support and adhesion of the dressing to the skin, and also slowed down the growth of bacteria due to its antibacterial activity [
122]. Calcium chloride was used as the crosslinking agent, which improved the mechanical properties, and the results of the tensile tests showed that an increase in CaCl
2 led to an increase in the mechanical properties, but at the same time, moisture absorption, water swelling, and weight loss decreased. At the same time, the samples with the lowest content of the cross-linking agent showed the highest release rate (
Figure 8).
The release kinetics of CM from the CMx-loaded SA layer were characterized according to the following equation:
where
Mt is the cumulative amount of the CM released at an arbitrary time
t;
M∞ is the cumulative amount of the CM released at an infinite time;
n is an exponent characterizing the mechanism; and
k is the release rate of CM incorporated in the system (
Table 5).
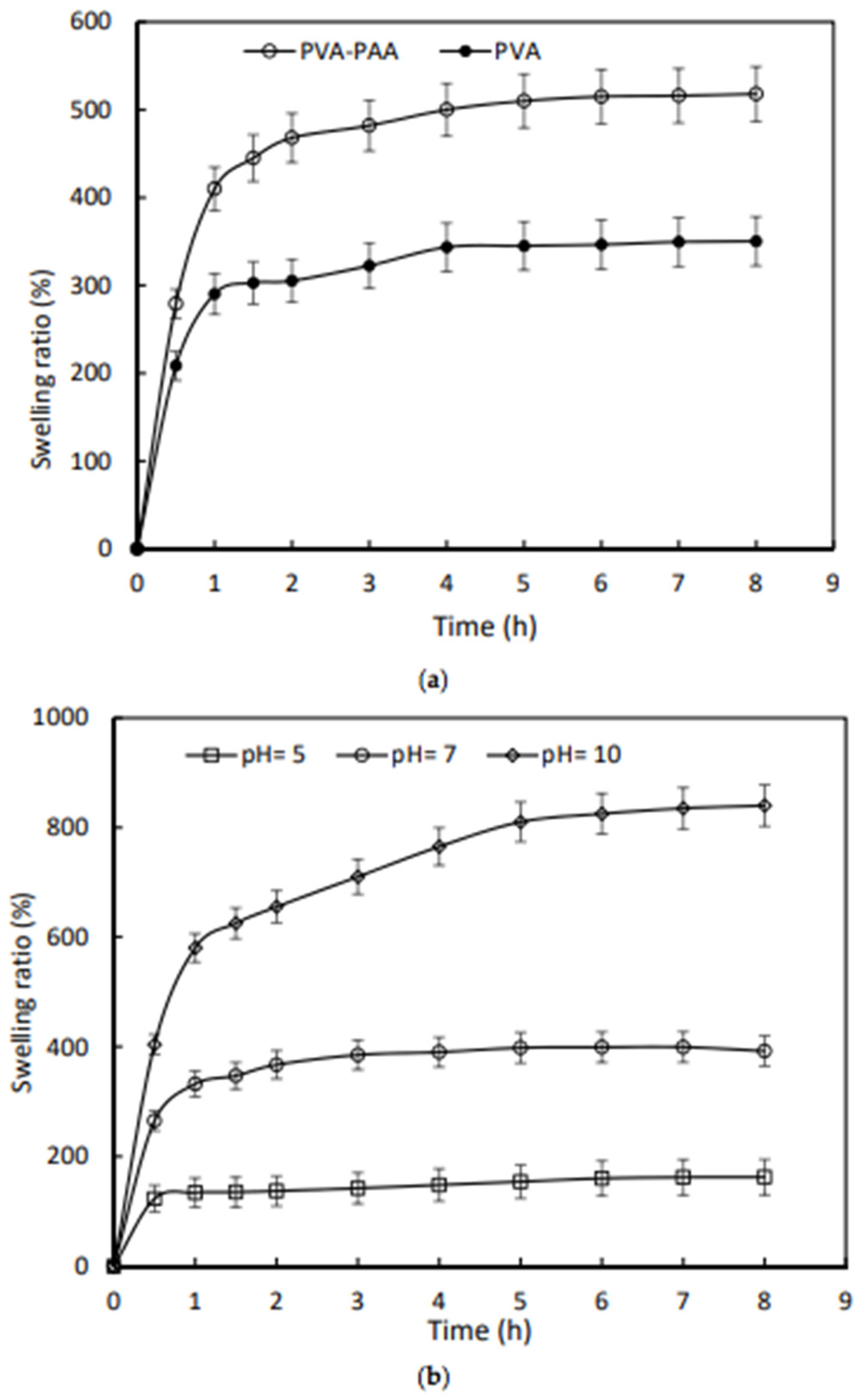
In contrast, by selecting synthetic polymers as materials for hydrogels, it is possible to control the mechanical properties of the dressing. Leading positions are occupied by polyacrylic acid-polyvinyl alcohol (PAA-PVA) mixtures as components for biomedical applications dressings due their biocompatibility and biodegradability [
121]. The encapsulation of polyacrylate layers into a bilayer hydrogel improves the performance of the resulting material such as moisture absorption, while minimizing the influence of interface-binding on the water uptake capacity of PVA [
123]. The equilibrium swelling coefficient of the PVA-PAA hydrogel increased by 48%. The PVA-PAA hydrogel was also sensitive to pH changes; at various pH values of 5 and 10, the degree of equilibrium swelling changed by 115% and −58%, respectively, compared with the index at pH = 7. The equilibrium swelling time was measured as 7, 3, and 1 h for the PVA-PAA hydrogel at various pH values of 10, 7, and 5, respectively. Moreover, it was possible to control the drug release by the pH increase of infected wounds due to the fact that the release rate for tetracycline was found at a higher pH = 8 compared to neutral pH (
Figure 9).
An example of a sensitive component of a bilayer material is a thermosensitive drug of Lactobacillus brevis (LB) [
124]. The bilayer hybrid material consists of a hydrogel layer filled with LB for the controlled release of the active compound, which has pro-angiogenic and antimicrobial activity for wound healing, and a hydrocolloid-type outer layer to improve the mechanical properties. The top layer of the hydrogel consisting of PVA and PVP was prepared by the freeze–thaw method. To compare the healing ability, the developed two-layer coating was compared with the effect of a commercial product (Duoderm
TM) on rat wounds infected with
P. aeruginosa, where the Lactobacillus brevis treated wound demonstrated faster healing (
Figure 10).
A hydrogel’s antibacterial resistance can be exhibited without drugs by the modification of polymers [
125]. A bilayer hydrogel material consisting of N-isopropylacrylamide and a chitosan-N-2-hydroxypropyltrimethylammonium chloride (HACC) inner layer (Hm-PNn) and a polyvinyl alcohol and acrylamide outer layer (PVAo-PAmp) was covalently crosslinked by the photoinduced electron/energy transfer polymerization-reversible addition-fragmentation with chain transfer (PET-RAFT). The antibacterial resistance of the material was manifested by
Staphylococcus aureus as a Gram-positive bacteria and
Escherichia coli as a Gram-negative bacteria due to the components of the lower layer. At the same time, the outer layer, exposed to the environment, had good extensibility and strength. The results of the in vivo tests demonstrated the improved collagen location and granulation tissue thickness, which proved the effect of the Hm-PNn/PVAo-PAmp hydrogel material on the acceleration of wound healing, which was demonstrated in a full-thickness skin defect model demonstrating the improved collagen location and granulation tissue thickness.
Hydrogels with tannic acid have antibacterial and antioxidant properties, which in turn facilitates the process of recovery and healing. The accelerated wound healing when using a hydrogel dressing with a two-layer structure with different pore sizes to prevent bacteria from entering into the wound and control moisture content and gas exchange, was proven [
126]. In addition, it is worth noting that the resulting hydrogel had shape memory and self-healing ability due to the hydrogen bonds that formed between TA and the bilayer hydrogel (
Figure 11). Hydrogel dressings are designed to control fluid exchange on the wound surface, accelerate regeneration and recovery, and prevent infection from the entering into the body [
127,
128,
129].
The reason for the adhesiveness of hydrogels is the continuous formation of catechin groups as a result of a dynamic redox system [
130]. The Fe
3+/TA-CN system triggers a dynamic redox system of catechin groups; the activated potassium persulfate initiator forms a large amount of free radicals for accelerated polymerization, even at low temperatures [
131,
132] (
Figure 12). The author confirmed the preservation of the adhesiveness of the material during storage under extreme conditions for a long time.
One of the most important processes in wound healing is the assessment of the wound state. In the treatment of skin diseases, bilayer hydrogels can act as a highly adhesive sensor in the form of an ultra-extensible wear-resistant sensor [
133].
The elastic upper layer, consisting mainly of synthetic polymers and integrated cellulose nanofibers with high extensibility and strength, plays the role of a sensor; and the lower layer, consisting of tannin and proline, has high biocompatibility, which accelerates wound healing and also increases the adhesion of the dressing to the skin [
134]. In real-time, the hydrogel strain sensor has the ability to monitor the physical activity of various human movements such as the opening of the mouth, curling fingers, smiling, and diagnosing health status. The strain sensor can also detect both large-scale and small movements of the human body. During flexion movements of the fingers, the strain sensor demonstrated a clear discrepancy in the changes in the relative resistance when bending the finger with different amplitudes, which indicates the possibility of distinguishing the flexion movements of different fingers. When applying a load cell near the mouth, a significant change in the current during a smile was noticed, which lays the foundation for the accurate recognition of facial expressions (
Figure 13).
To characterize the sensitivity of a strain gauge sensor, a strain gauge coefficient (GF) is introduced, which is calculated by the formula:
where
R0 is the initial resistance; Δ
R is the changes in resistance; and
ε is the applied strain.
2.2. Bilayer Hydrogels as Scaffold for Tissue Engineering Applications
Hydrogels act as scaffolds for the delivery system of cells and biochemical factors in tissue engineering, having the form of a three-dimensional substrate that allows cells to attach, proliferate, and grow, resulting in the regeneration of new tissue [
135,
136].
Cell therapy promotes additional therapeutic possibilities that contribute to faster wound healing and restoration of normal skin architecture.
Scaffolds made from natural polymers are biocompatible, biologically active, promote cell attachment to cell surface receptors, and provide a niche for cell function control [
137]. One example of such biopolymer is collagen. Attempts have been made to mimic the natural bilayer organization of the skin using porous scaffolds based on collagen (Coll) for the epidermis and sodium carboxymethyl cellulose (NaCMC) for the dermal layer [
138].
One of the main parameters of the hydrogel scaffold is the pore size. One example of a study of size variation for wound treatment was the biomatrix of a bilayer gelatin-CS-hyaluronic acid hydrogel [
139]. Chondroitin-6-sulfate and hyaluronic acid were included in the gelatin matrix to mimic the composition of the skin and to create a suitable microenvironment for cell proliferation, differentiation, and migration. The bottom layer was seeded with dermal fibroblasts and served as a feed layer for keratinocyte inoculation (pore size: 150 µm) while the upper layer was seeded with keratinocytes for epidermalization (pore size: 20–50 μm)
.The top layer seeded with keratinocytes developed into an epidermis-like structure, and the bottom layer seeded with dermal fibroblasts developed into a dermis-like structure after culturing the cells for 21 days. Histological studies and immunostaining have proven that keratinocytes form multi-layered layers of the epidermis within 21 days. The results showed that, in addition to the permanent coverage of histologically healthy and differentiated epithelial tissue, there was a well-defined dermo-epidermal junction and collagen network in the dermis. Importantly, pore size plays an important role in cell migration within the membrane. The micropores of the upper layer (pore size: 20–50 µm) not only allow for the migration of keratinocyte populations from the upper surface into the wound, but also serve as a substrate to prevent excessive spread of keratinocytes into the dermal layer, while seeded on the macroporous (pore size: 150 µm) on the top layer of the membrane part, dermal fibroblasts can act as a feeding layer for keratinocytes, and the porous structure of the hydrogel is used to remove the wound exudate.
Hydrogel combinations with materials of a different nature that exist in the form of foams, sponges, and nanofibers can serve as an alternative to bilayer materials based on hydrogels for skin regeneration. Most porous scaffolds seeded with keratinocytes or fibroblasts are based on collagen [
140]. The two-layer sponge structure has a top layer of non-porous collagen gel, on top of which keratinocytes are added, and a layer below it is a porous collagen sponge, where fibroblasts are seeded. In a non-toxicity and efficacy study, a bilayer cell matrix (OrCel™) from Ortec International, Inc. (New York, NY, USA) to facilitate the timely wound closure of varying thicknesses of donor skin in patients with severe burns was compared with a commercial single layer analogue Biobrane-L
® (Bertek Pharmaceuticals, Sugarland, TX, USA). Donor sites treated with OrCel
TM were examined for the severity of scarring. Investigators undertook assessment at weeks 12 and 24, and at subsequent visits twice a year using the Vancouver Scar Scale, which assesses pigmentation, vascularity, compliance, and height of the donor site, and the Hamilton Burn-Scar Rating Scale, which was conducted by blind viewing (
Figure 14).
The results showed that OrCel™ was more effective: it was recorded that the healing rate for the treated areas of OrCel™ was significantly faster.
Hybrids of a bilayer structure to accelerate wound healing can be made from keratin and chitosan nanofibers with a gelatin methylacrylate hydrogel layer [
141]. The synthesis technique makes it possible to obtain a two-layer multifunctional structure with a high water content to mimic tissue with human keratinocytes (hKC) and dermal fibroblasts (hDFb). To prepare the top layer of nanofibers, consisting of the solution of human hair keratin and chitosan (mixture ratio: 5/5), an electrospinning method was used with formic acid as a solvent in the presence of polyethylene glycol, followed by crosslinking with glutaraldehyde. The lower hydrogel layer was obtained by radical crosslinking of the solubilized gelatin derivatives. A bilayer structure was prepared by the photopolymerization of GelMA under a mat of crosslinked human hair keratin and chitosan nanofibers.
The GelMA hydrogel layer contained a huge amount of micropores with a diameter of 10–20 µm, while the shear modulus was about 0.5 kPa. The 30 µm thick nanofiber layer was firmly bonded to the bottom hydrogel layer without any separation (
Figure 15).
The inclusion of chitosan in the nanofibers improved the mechanical stability of the material. To assess the effectiveness of the scaffold, human fibroblasts were placed in the hydrogel layer and HaCaT cells were cultured on the layer of nanofibers and co-cultured for 10 days. As the result, encapsulated fibroblasts proliferated in the hydrogel matrix, and HaCaT cells formed a cell layer on top of the scaffold, imitating the dermis and epidermis of skin tissue.
The composition based on poly(ε-caprolactone-colactide)/poloxamer (PLCL/poloxamer) nanofibers as the upper layer and a hydrogel consisting of dextran and gelatin as the lower layer formed a material with increased mechanical stability and supported cell proliferation [
142] (
Figure 16).
The upper layer of the nanofibers demonstrated high biocompatibility and mechanical protection of the material, while the lower hydrogel dextran/gelatin (5/5) was a rapidly forming scaffold in situ, combining the ability to maintain cell viability and the mechanical strength necessary for skin scaffolds. The pore sizes of the hydrogel layer were about 50 to 200 microns. It is important to note that the porous structure of the scaffolds supports the diffusion of nutrients and gases, allows for growth cell, and maintains a high water level. Dry hydrogels can absorb large amounts of water from 19.47 to 43.45 g/g. At the same time, the degree of swelling changes nonmonotonically with the increase in the concentration of dextran in the hydrogel. The degree of swelling decreases rapidly as the dextran content increases from 30 to 50%, which can be explained by the increase in the crosslink density. With an increase in the dextran content from 50 to 70%, the crosslink density in hydrogels decreased, and the degree of swelling slightly increased from 19.47 to 25.94 g/g. In addition, the concentration of dextran and, accordingly, the density of crosslinking, played important roles in the controlled degradation of the material over a certain period of time, which, in turn, creates a comfortable environment for the formation of epidermal tissue. The mechanical properties of dextran/gelatin hydrogels are highly influenced by the mass ratio of monomers, which may be associated with pore size and water content.
The study results under a fluorescent microscope revealed no difference in the effect on the vital activity of the cells of the control group. The group with a bilayer PLCL/poloxamer nanofibers and dextran/gelatin hydrogels scaffold was not found, thereby confirming the biocompatibility of the degradation products.
Gelatin-based hydrogels are scaffolds for skin regeneration, with the inclusion of keratinocytes and fibroblasts [
143]. The two-layer skin substitute, “PG-1” or “first generation pullulan-gelatin hydrogel”, with the addition of human fibroblasts and keratinocytes in vitro, is characterized by low cost due to the use of pullulan—a polysaccharide with antioxidant properties and gelatin with a high sorption capacity. The investigation into the material demonstrated the optimal mechanical characteristics for a hydrogel as a skin substitute such as an average pore size of 61.69 μm with an ideal elastic modulus, swelling behavior, and biodegradability. Compared to the cell-free PG-1 study in vivo, which showed that cell encapsulation increased proliferation, the thickness of the neodermis layer was 204.00 ± 19.65 μm compared to acellular hydrogels of 115.63 ± 9.83 μm, and the controls, after the skin biopsy. Furthermore, PG-1 provided cell viability and angiogenesis and led to inflammation reduction and macrophage infiltration.
Crystallization is one example of the physical crosslinking of gels, involving a freeze/thaw process [
144]. A two-layer wound dressing made of polyvinyl alcohol/carboxymethyl cellulose/polyethylene glycol (PVA/CMC/PEG) with a gradual change in pore size was developed using freeze–thaw and phase separation methods [
145]. The study results showed increasing pore sizes from the upper to the low layer. The pore sizes were 20 μm and 100 μm in the upper dense layer of the wound dressing and the lower porous layer, respectively.
The freeze–thaw cycles did not affect the pore size, which confirmed the pore formation during the phase separation at room temperature, while as the exposure time increased, the pore sizes constantly increased. In addition, the concentration of polymers affected the pore size. As the PVA concentration increased (from 7% to 10%), the pore sizes first increased and then decreased. This phenomenon can be explained by the increase in the viscosity of the solution, which prevents the phase separation due to the decrease in fluidity. The increase in PEG concentration from 7.0% to 9.5% led to the increase in pore size, where PEG plays the role of a pore former. At a low PEG content, the degree of phase separation was weak. The increase in the CMC concentration led to a decrease in pore size, which the authors suggest is due to the nature of CMC, as it is a surfactant that can reduce the surface tension, thus making the dissolution of PEG easier and more uniform. The increase in the CMC concentration increases the viscosity of the system, which slows down the phase separation, while insufficient viscosity, on the other hand, cannot lead to the phase separation (
Figure 17).
Zonari et al. designed a polyhydroxybutyrate-co-hydroxyvalerate (PHBV) bilayer skin tissue scaffold considering the two main layers of the skin: the epidermis and dermis. The new material was a thin membrane and a highly porous structure that was constructed by the combination of both types of methodologies, created through membrane solvent casting and the 3D freeze-dried scaffold, respectively [
146]. The skin scaffold demonstrated high water retention capability and controlled stability against degradation by enzymes. One of the main disadvantages of PHBV is the hydrolytic degradation [
147,
148,
149]. Degradation studies revealed the resistance to the action of lysozyme within 8 weeks of the treatment, the action of lipase led to the change in the weight of the material by almost 90% over the same period, while the co-treatment of the two enzymes led to the complete destruction of the material (
Figure 18). Enzyme concentrations were approximately equal to the blood serum concentrations.
Kamali et al. presented the development of a bilayer skin scaffold consisting of a layer obtained by the electropressing of polycaprolactone and PVA, and a porous hydrogel layer made of chitosan and gelatin. The hydrogel layer was obtained using a lyophilization method to obtain a porous structure in the hydrogel [
150]. The authors compared the characteristics of the two-layer structure with a single-layer chitosan-gelatin hydrogel. The mechanical strength and modulus tests of the combination of the hydrogel layers and electrospinning resulted in strength increases of 110% and 133% compared to the single layer hydrogel sample (
Table 6).
It is important to note that the gelation step does not damage the electrospun layer of the bilayer structure during the manufacturing process. At the same time, significant superiority of the two-layer structure was not observed in the study of sorption capacity. The study results of the histopathology and histomorphometry showed that the wounds treated with a bilayer scaffold showed the fastest healing: epidermal proliferation and an increase in the thickness of the epidermal layer were observed 21 days after the treatment. A decrease in the inflammatory response and granulation tissue was also observed (
Figure 19).
Keratinocytes were co-cultivated with fibroblasts in the chitosan–gelatin–hyaluronic acid scaffold to construct an artificial bilayer [
151]. The study results of the physicochemical properties suggested that the porosity and pore size of the scaffolds could be modulated by the thermodynamic and kinetic parameters of the freeze-thawing process. The encapsulation of hyaluronic acid led to the increase in the sorption capacity of the material and to a long-term retention of water compared to the single-layer hydrogels. Fibroblasts cultured in chitosan–gelatin–hyaluronic acid scaffolds grew and proliferated well and showed high viability. Keratinocytes were co-cultivated with fibroblasts in chitosan–gelatin–hyaluronic acid scaffolds to create artificial bilayer skin in vitro.
A scaffold made of polycaprolactone electrospinning membrane and poly(lacto-glycolic acid) (PCL/PLGA) and cross-linked with glutaraldehyde (3.5% by volume) with a chitosan/gelatin hydrogel was fabricated using two methods: electrospinning of the membrane onto the lyophilized hydrogel (BS-1) and the membrane underlaying and casting method (BS-2) [
152,
153]. However, glutaraldehyde negatively affected cell proliferation [
154]. The bilayer scaffold consisted of galvanized polycaprolactone/poly(lacto-glycolic acid) (PCL/PLGA) and chitosan/gelatin hydrogel crosslinked with glutaraldehyde. The cytotoxicity of the bilayer scaffold and chitosan/gelatin hydrogel (CGH) was compared. The thickness of the hydrogel was 2.13 ± 0.22 mm at a density of 0.125 ± 0.002 g/cm
3, its porosity was 97.49%, and the average pore diameter was 290 ± 109 mm. The difference in the membrane layer was noted. The membrane deposition in BS-1 had a rough, variable microsurface while the membrane adhesion in BS-2 resulted in a flat and smooth appearance.
A membrane layer was added to maintain the mechanical stability of the scaffold during the implantation and to withstand suturing. However, it was noted that the hydrogel layer degraded much faster in vivo compared to the in vitro experiments. Thus, the authors of the study suggest that this material can be used as a temporary skin substitute or tissue bandage.
The results showed that while the PCL/PLGA membrane supported fibroblast cells, the decrease in cell viability was observed in the hydrogel–gelatin–chitosan–glutaraldehyde due to the remaining glutaraldehyde content. These results show that the addition of a membrane layer to the hydrogel reduces the rate of swelling and degradation, which in turn opens up prospects for implantation.
The method of creating artificial skin using 3D bioprinting has become an innovative method in the area of 3D printing development [
155]. A bilayer structure consisting of dermal fibroblasts, keratinocytes, and microvascular endothelial cells was designed and fabricated using an extrusion 3D printer to create micro vessels in skin grafts. Human dermal fibroblasts and microvascular endothelial cells were mixed with a gelatin–alginate composite hydrogel as the dermis, and human keratinocytes were mixed with the gel as the epithelium.
The material obtained by 3D printing increased cell survival by more than 90%. The results of the histological and immunohistochemical analyses showed that two-layer constructions contributed to the healing and contraction of skin wounds with the predominant regeneration of micro vessels (
Figure 20).
With the development of 3D skin printing technology, a bilayer membrane (BLM) scaffold was designed and printed consisting of a poly(lactic-glycolic acid) (PLGA) outer membrane and an alginate hydrogel bottom layer that respectively mimicked the epidermis and dermis of the skin [
156]. The multi-porous alginate hydrogel of the BLM scaffolds promoted cell adhesion and proliferation in vitro, while the PLGA prevented bacterial invasion and the membrane maintained the moisture content of the hydrogel.
A double-layer membrane showed excellent ability in promoting neovascularization and collagen I/III deposition after implantation in the wounds of dorsal rats, which ultimately accelerated the wound healing process compared to the PLGA and alginate hydrogel control groups.
The 3D printing possibilities have served in the fight against diabetic wounds, characterized by long-term chronic inflammation, and reduced granulation tissue formation and vascularization. A bio-layer skin substrate was created consisting of an upper layer made of a gelatin cryogel with silver and a lower layer of platelet-derived growth factor-BB (PDGF-BB) with a 3D printed gelatin scaffold [
157]. The bilayer scaffold application led to a high level of skin regeneration due to the increased neovascularization and collagen I/III deposition. The 3D printed BLM scaffolds promoted wound healing and so are suitable for a wide range of applications as wound dressings or skin prostheses. The substrate could not influence the proliferation of fibroblasts, keratinocytes, U937 cells, and HL60 cells. The CFU quantification results demonstrated that the scaffold showed good antibacterial ability, namely, the release of silver nanoparticles was able to significantly kill
Pseudomonas aeruginosa,
Staphylococcus aureus, and
E. coli. Moreover, the substrate was able to promote re-epithelialization, granulation tissue formation, collagen deposition, and angiogenesis in vivo. The PDGF-BB-loaded scaffolds, silver and PDGF-BB coloaded scaffolds were able to accelerate wound closure, re-epithelialization, granulation tissue formation, and angiogenesis compared to the scaffold and the silver-loaded scaffold groups in vivo at each indicated time point.
Problems of skin scarring and reduced wound contraction with accelerated wound healing were demonstrated using a gelatin/sodium alginate/gelatin methacrylate bioprinter supplemented with normal human dermal fibroblasts (NHDF) and normal human epidermal keratinocytes (NHEK) to print the dermis and epidermis. The results showed accelerated epithelialization as well as the absence of scarring [
158]. The multilayer hydrogel material gelatin/SA/GelMA had a high elongation at break, so the authors of the study suggest that the risks of tearing caused by the wound activity in the grafted skin are minimal (
Table 7).
Stem cells are formed at the stage of embryonic development and their main distinguishing ability is their differentiation into specialized cells that can participate in the formation of skin prostheses. The main sources of cells that can be used to model such prostheses are adult stem cells, embryonic stem cells (ESCs), and induced pluripotent stem cells (iPSCs) [
159].
It has been shown that stem cell therapy has the potential to treat chronic wounds, but has not been very successful in clinical practice. Research is currently underway to create suitable polymeric stem cell matrices to promote paracrine activity, which ultimately affects the healing process.
Natesan et al. developed a bilayer hydrogel based on collagen and pegylated fibrin to deliver adipose-derived mesenchymal stem cells (ADSCs) derived from burnt skin layers. This work demonstrated the successful differentiation and proliferation of ADSCs, which formed dense tubular microvascular networks based on PEGylated fibrin. The bilayered hydrogels’ influence on wound healing was demonstrated by considering the wound process of rats with a control group. Wounds treated with bilayer hydrogels showed less wound shrinkage and better dermal matrix deposition and margin progression. In addition, in the experimental rats, the significant increase in the formation of granulation tissue and re-epithelialization of the wound edges were observed [
160,
161]. Similar cells in a bilayer hydrogel based on polyethylene glycol and fibrin differentiated into an epithelial layer, a vascularized dermal layer, and a subcutaneous layer. Polytransretinoic acid and fenofibrate have also been used to differentiate dsASCs into epithelial-like cells (
Figure 21) [
162].