Abstract
The environmental damage caused by plastic packaging and the need to reduce pollution requires actions to substitute plastic materials for more sustainable and biodegradable materials. Starch, gelatin, and bacterial cellulose films are three potential biodegradable polymeric films for use in packaging. However, these materials need improvements in their physical, chemical, and mechanical properties to be used in packaging. In this work, these films were treated with cold plasma to evaluate the effects of treatment conditions on several physical, chemical, and mechanical properties. The dielectric barrier discharge plasma technology was applied with varying treatment times (0 to 20 min) and excitation frequencies (50 to 900 Hz) at 20 kV. The optimal excitation frequency for starch films (50 Hz) was different from the optimal frequency for gelatin and bacterial cellulose films (900 Hz), indicating a high dependency on the treatment in this variable that is often neglected. Plasma treatment improved the hydrophobicity, surface morphology, water resistance, and mechanical properties of all three films, with the advantage of not recurring to chemical or biological additives.
1. Introduction
Environmental damage caused by the accumulation of non-biodegradable plastic packaging waste has generated high interest in developing possible biodegradable substitutes. Natural biodegradable polymers-based packaging is especially demanded by the food industry, which is one of the largest consumers of plastic packaging [1].
Biomolecules such as starch, bacterial cellulose, and gelatin have been used to develop packaging materials [2]. These packaging materials are considered biodegradable, sustainable, and environmentally friendly [3,4]. However, some disadvantages must be overcome, such as their high hydrophilicity, low water vapor, gas, and light barrier, and low mechanical resistance [1,5].
Starch can be obtained from several vegetable sources, such as maize, seed kernels, cereal grains, roots, and fruits. Starches are biodegradable and usually have low costs. It consists of two fractions: amylose (straight chain) and amylopectin (branched chain). Starches have good film-forming properties but present some disadvantages, including low mechanical performance and high hydrophilicity, which result in rapid moisture absorption during use and storage [6].
Bacterial cellulose is a natural nanostructured polymer produced by several bacteria. It has good thermal stability, high porosity, good mechanical properties, amphiphilic character, and a high liquid sorption capacity [7]. However, it has a high permeability to water vapor, which limits its application [8].
Gelatin is a linear protein resulting from collagen denaturation of bovine, swine, or fish origin. It has a good ability to form films; however, it has a limited moisture barrier property [9].
Improvements on polymeric films can be carried out by several techniques, such as cold plasma [10], ultrasonics [11], doping with additives [12], production of composites [13], and other procedures. In this study, we have applied cold plasma technology to induce changes to starch, gelatin, and bacterial cellulose films.
Cold plasma is a technology that can be applied for surface treatment, including modification or functionalization of polymers. Recently, it has emerged as an innovative, effective, and sustainable alternative to traditional chemical treatments. One of the plasma generation methods is the dielectric barrier discharge (DBD), which operates at room temperature and atmospheric pressure and does not involve using petroleum-based solvents. Cold plasma generates ultraviolet light, high-energy electrons, and reactive particles, which can initiate several physical-chemical reactions on the surface of polymers [14,15].
Several studies have investigated the effect of DBD plasma on polymers, varying the exposure time, voltage, and excitation frequency. Wu et al. [15] showed that cold plasma improved casein-based films’ barrier and mechanical properties by increasing the plasma generation voltage. Films from corn starch with high amylose content and treated with high-voltage cold plasma (70 and 80 kV) increased the film roughness and hydrophilicity [16]. New functional groups containing oxygen were introduced to the sodium caseinate film surface, improving its hydrophilicity by applying short plasma exposure times (1 to 5 min, at 70 kV). At the same time, no changes occurred in the helical structure of the protein [17].
Studies on plasma treatment of bacterial cellulose films are still scarce and more work is needed to understand the changes induced by plasma species on the properties of bacterial cellulose films. Studies have been carried out on the degradation of toxic compounds from the raw material used as a carbon source for bacterial cellulose production [18,19] and for the deposition of oxides and other materials on the surface of bacterial cellulose [20]. However, there are no studies on plasma treatment of bacterial cellulose films aiming toward the improvement of its physical, chemical, and mechanical properties.
Modification of starch using plasma technology has been successfully addressed. Plasma increases carbonyl and carboxyl groups, resulting in improved paste viscosity, hydration properties, and digestibility [21,22]. These studies showed that the changes in starch properties were greater at excitation frequencies between 100 and 300 Hz and not at 50 Hz as commonly applied [10,21]. Plasma treatment in starch films also indicated that the optimal excitation frequency range was between 200 and 300 Hz [10,23].
Most studies on cold plasma treatment on polymers were carried out at a single plasma excitation frequency, usually at 50 Hz. However, the excitation frequency has been proven to be an essential factor in cold plasma applications [24,25,26]. The excitation frequency influences the concentration and type of ions and free radicals generated in the cold plasma, inducing different chemical transformations in the treated product. Plasma treatments on bacterial cellulose and gelatin films at 50 Hz have not resulted in good improvements in film properties. Due to the importance of the excitation frequency in the generation of plasma in many applications, such as starch modification [10,21], aroma modulation [24], phenolics content [27], and mitigation of off-flavors [28], this work has addressed the significance of the excitation frequency in three different biodegradable polymer films.
This study applied DBD plasma at three different plasma excitation frequencies to three films produced from natural polymers (gelatin, starch, and bacterial cellulose). The effects of cold plasma treatment on the film water solubility, hydrophobicity, chemical group composition, mechanical properties, and surface morphology were evaluated.
2. Materials and Methods
2.1. Materials
Commercial corn starch (Unilever, brand Maizena™, Garanhuns, Brazil) and commercial fish gelatin (Fitoway Laboratório Nutricional Ltd. a, brand São Pedro™, Tarumã, Brazil) were purchased in the local supermarket. Bacterial cellulose membranes were supplied by Seven Indústria de Produtos Biotecnológicas Ltd. (Ibiporã, Brazil). Glycerol was purchased from Dinâmica (São Paulo, Brazil).
2.2. Elaboration of the Biopolymer Films
The casting technique was used in the production of the films. Glycerol acted as a plasticizer. The degassing step of the filmogenic dispersions to remove the air bubbles formed during the process, common to the three films, was carried out before the final drying. The process took place in a vacuum system with a Vacuubrand-1C pump.
The starch film was produced following the methodology proposed by Oliveira et al. [29]. In brief, 5 g of corn starch was dissolved in 100 mL of distilled water (5% w/v) and heated to 95 °C under magnetic stirring for 30 min for complete starch gelatinization. Glycerol was added to the starch solution in a 25% w/w proportion and maintained at 60 °C for 15 min under magnetic stirring. Afterward, the dispersion was homogenized in an Ultra-Turrax (IKA model T25) at 10,000 rpm for 15 min. The degassed filmogenic dispersion was poured onto glass plates coated with a polyester film (Mylar™) and allowed to dry under ambient conditions (25 °C) for 24 h.
Bacterial cellulose membranes were oven-dried for 48 h at 50 °C and ground in an analytical mill (IKA model A11). The dried bacterial cellulose underwent an oxidation process following the method presented by Saito et al. [30]. After oxidation, a mechanical treatment took place in a colloid mill (Meteor model Rex Inox I-V-N) for 10 min, obtaining nano-fibrillated bacterial cellulose (NFBC). The NFBC was freeze-dried (Liotop model LP510). The methodology proposed by Nascimento et al. [31] was used to obtain the filmogenic dispersion of bacterial cellulose. For this purpose, an aqueous dispersion was prepared to contain 1% (w/v) of lyophilized NFBC and 50% (w/w) of glycerol on a dry basis of the matrix. The dispersion was homogenized in an Ultra-Turrax (IKA model T25) at 13,000 rpm for 15 min, sonicated for 2 min at 19 kHz (Unique model DES500) for degassing, and dried in stainless-steel trays coated with Mylar™ in an oven at 50 °C for 48 h.
Gelatin films were prepared following the procedure described by Santos et al. [32]. An aqueous solution was prepared with 9.6% w/v gelatin and 25% w/v glycerol (on a dry basis). The gelatin was hydrated and heated to 50 °C for 15 min, adding the glycerol in the last 5 min, under constant agitation. The solution was homogenized in an Ultra-Turrax (IKA model T25) at 10,000 rpm for 10 min. The degassed film-forming solution was poured onto glass plates covered with Mylar™ and allowed to dry at 25 °C for 24 h.
2.3. Dielectric Barrier Discharge Plasma Treatment
The films were treated with cold plasma in dielectric barrier discharge equipment consisting of a power source (Inergiae model PLS0130) and two 8 cm-diameter aluminum electrodes separated by 2 mm acrylic plates. The films were placed within a 3 cm gap between the dielectric barriers centered with the electrodes.
Cold plasma was generated at 20 kV (maximal voltage achieved with the equipment) and at excitation frequencies of 50, 400, and 900 Hz. The films were treated for 5 min. The films were treated adhered to the polyester support (MylarTM) and without the polyester support. All assays were carried out in triplicates.
The optimal excitation frequency for plasma treatment was defined as the frequency at which greater changes were observed in the chemical composition, hydrophobicity, and film solubility. The best films regarding hydrophilicity and solubility were plasma-treated for 10, 20, and 30 min under the optimal excitation frequency for further analysis of the films’ water solubility, hydrophobicity, and chemical group composition. The films with the best results for hydrophobicity, water solubility, and chemical composition were further tested for changes in surface morphology and mechanical properties.
2.4. Physical and Chemical Analysis
2.4.1. Thickness and Humidity
The thickness of the films was measured using a digital micrometer (Mitutoyo model IP65). Ten replicates were taken.
The film humidity was measured in an infrared moisture balance (Marte model ID50) operating at 100 °C. The humidity was based on 1 g of sample. Three replicates were taken.
2.4.2. Hydrophobicity
Changes in the hydrophobicity of the films were determined through the water contact angle (GBX Instrumentation Specifique), attained according to ASTM D-5725-99 [33]. Film samples (2 × 2 cm) were fixed to a glass holder, and an image was captured (Pixe Link Nikon camera) when the drop touched the film surface. The test was carried out at room temperature (23 ± 2 °C). All assays were performed in quadruplicate.
2.4.3. Water Solubility
The water solubility was determined following the method described by Pena-Serna and Lopes-Filho [34]. The film samples (2 cm-diameter discs) were dried in an oven at 105 °C for 24 h, weighed (W0), and immersed in 50 mL of distilled water at 25 ± 2 °C for 24 h under agitation in an orbital shaker (Tecnal model TE-142) at 100 rpm. After this period, the samples were removed and dried in an oven (105 °C for 24 h) to determine the mass of the material that was not solubilized (W). The solubility was calculated using Equation (1):
2.4.4. FTIR Analysis
The absence or formation of chemical groups on the surfaces of the films was evaluated using Fourier transform infrared (FTIR) spectra. A Perkin-Elmer Spectrum Two equipment (Perkin-Elmer, Waltham, MA, USA) was used in attenuated total reflection mode (ATR). The spectra were collected in wavelengths between 4000 and 650 cm−1, using 32 scans and a 4 cm−1 resolution.
2.5. Surface Morphology
The surface morphology was characterized using a Quanta FEG SEM at an accelerating voltage of 10 kV. The films were cut into 5 × 5 mm squares, fixed on the sample table with conductive tape, and covered with a thin layer of gold.
2.6. Mechanical Properties
The mechanical properties of the films were determined using an EMIC DL-3000 equipment, following the standard method D882-18 [35]. The tensile strength (TS), elongation at break (EB), and elasticity module (EM) were determined. The films were cut into 4 × 75 mm dumbbell shapes. The mechanical tests were performed with an initial clamping distance of 50 mm and a tensile rate of 50 mm/s. All measurements were carried out in quintuplicate.
2.7. Selection of the Best Films and Cold Plasma Operating Conditions
The selection of the best biodegradable polymeric films and their respective cold plasma operating conditions was based on a desirability function that considered the highest change in functional groups measured by FTIR, highest hydrophobicity, and lowest water solubility (Equation (1)). These three factors were chosen because the goal of cold plasma processing is to induce physical and chemical changes in the film surface and chemical structure, and because films with high hydrophobicity and low water solubility are desired for food applications. The weight of the hydrophobicity in the desirability function was higher than the other two factors due to the importance of this property in food packaging. The contact angle divided by 90° was used to address the hydrophobicity, measuring how close the contact angle was from the value for which a film is considered hydrophobic. For the changes in the chemical structure, the absorbance that presented the largest change was used in the function.
The starch, gelatin, and bacterial cellulose with the highest scores were considered the best biodegradable films. Surface analysis and mechanical properties’ analysis were carried out only with the best films.
2.8. Statistical Analysis
The results were presented as mean ± standard deviation. The statistical analysis was performed using analysis of variance (ANOVA), followed by the Tukey’s test, with the Statistica version 7 software.
3. Results
The films were produced by the casting method, which enabled attaining films with low thickness and humidity (Table 1). The starch and gelatin films were thinner than the bacterial cellulose film, while their humidity was not significantly different.

Table 1.
Width and humidity (mass basis) of the starch, bacterial cellulose, and gelatin films. For each property, values followed by at least one common letter are not different from each other (p > 0.05).
3.1. Hydrophobicity
Table 2 presents the effects of the excitation frequency on the hydrophobic character and water solubility of the films subjected to plasma treatment.

Table 2.
The angle of contact and water solubility of the starch, bacterial cellulose, and gelatin films treated with dielectric barrier discharge plasma: with and without Mylar™ support. For each property, values followed by at least one common letter are not different from each other (p > 0.05).
The hydrophobicity of the film is an essential feature in deciding the packaging application of any polymeric film [16]. A totally hydrophobic film is characterized by contact angles equal to or greater than 90° [14]. Among the films tested, the bacterial cellulose film had the highest hydrophobicity, while the starch film had the lowest hydrophobicity.
The Mylar support acted as an extra barrier between the electrodes and the films, reducing the effects of plasma treatment. The hydrophobicity of the films did not change significantly or decrease when the Mylar support was used. Thus, its use is not recommended when applying cold plasma.
Plasma treatment increased the hydrophobicity of the bacterial cellulose and gelatin films, while no significant changes were observed in starch films when the Mylar support was not used. The highest increase in hydrophobicity was observed at a plasma excitation frequency of 900 Hz. At 50 Hz, no significant change was observed. Thus, proper setting of the excitation frequency is critical to increasing the hydrophobicity of the films.
The films were subjected to plasma treatment for an extended period at the optimal excitation frequency of each film (Table 3). The increase in processing time increased the contact angle of all films, indicating the production of films with higher hydrophobicity. Thus, the processing time was a significant variable for the process (p < 0.05). Plasma application increased the hydrophobicity of the films by 15%, 6%, and 20%, respectively, for the starch, bacterial cellulose, and gelatin films.

Table 3.
The contact angle of the starch, bacterial cellulose, and gelatin films treated with dielectric barrier discharge plasma at an optimal excitation frequency (50 Hz for the starch film and 900 Hz for the bacterial cellulose and gelatin films) without Mylar™ support. For each property, values followed by at least one common letter are not different from each other (p > 0.05).
The increase in hydrophobicity has been attributed to changes in the surface morphology of the films due to an increase in surface roughness. This will be discussed in Section 3.4.
Table 4 presents the contact angles of several films reported in the literature. Starch films have a low contact angle (between 20 and 56°) and are distant from the recommended 90° contact angle for hydrophobic surfaces. Plasma processing and high-pressure processing increase the hydrophobicity of starch films, but these physical changes have limited effects on the films’ hydrophobicity. Chemical additives such as cinnamaldehyde have a higher impact on the contact angle and the hydrophilic nature of starch films.

Table 4.
The contact angles of several starches, bacterial cellulose, and gelatin films reported in the literature.
Plasma treatment increased the contact angle of gelatin films by 17%, while the production of a gelatin + chitosan composite reduced the contact angle [38].
The use of plasma treatment was shown to increase and decrease the contact angle. Proper setting of the excitation frequency (900 Hz) improved the contact angle, while low excitation frequencies (50 Hz) decreased the contact angle, worsening the wettability properties of gelatin films [39]. This comparison evidences the importance of the excitation frequency in plasma treatment of bacterial cellulose films.
3.2. Solubility in Water
The solubility of the films is an important property that can indicate the presence of hydrophilic groups, in addition to assessing the resistance in aqueous media [41]. Overall, the starch film is the most soluble film, and the bacterial cellulose film is the least soluble film (Table 2).
The Mylar support has also attenuated the effects of plasma treatment regarding the solubility of the films. The solubility of the bacterial cellulose and gelatin films increased when the support was used, which is not a desirable result.
On the other hand, the solubility of all films decreased when the Mylar support was not used. The highest decrease in solubility was observed for the bacterial cellulose film, which was already the least soluble film.
The excitation frequency affected each film differently. A lower excitation frequency (50 Hz) decreased the solubility of the starch film, while a higher excitation frequency (1000 Hz) decreased the bacterial cellulose and the gelatin film solubilities.
The films were subjected to plasma treatment for an extended period at the optimal excitation frequency of each film (Table 5). The increase in processing time decreased the solubility, improving its potential application in packaging. Processing time was also a significant variable for solubility (p < 0.05). Plasma application reduced the solubility of the films by 6%, 40%, and 18%, respectively, for the starch, bacterial cellulose, and gelatin films.

Table 5.
Solubility of the starch, bacterial cellulose, and gelatin films treated with dielectric barrier discharge plasma at an optimal excitation frequency (50 Hz for the starch film and 900 Hz for the bacterial cellulose and gelatin films) without Mylar™ support. For each property, values followed by at least one common letter are not different from each other (p > 0.05).
Although an 80% increase was observed in the insoluble matter of the starch film after plasma treatment, this film remained with an insoluble matter below that recommended for food packaging applications [42]. The potential application of a film in the food industry requires water-insoluble packaging to increase product integrity and water resistance [42].
Table 6 presents the water solubility of several films reported in the literature. Starch films have an extensive range of solubility (between 15% and 98%), which is usually related to the amylose–amylopectin ratio in the starch. Plasma processing decreased the water solubility of starch films more than adding chemical additives such as cinnamaldehyde and orange peel, while having a similar effect as the starch/date palm pits composite.

Table 6.
Solubility of several starches, bacterial cellulose, and gelatin films reported in the literature.
Plasma treatment decreased the solubility of bacterial cellulose by 40%, which was a similar result attained by chemical treatment to convert bacterial cellulose into nanocellulose (48%) and nanocrystalline cellulose (38%). However, the decrease achieved with plasma treatment reduced the bacterial cellulose to the lowest level reported in the literature. The physical and chemical treatments of bacterial cellulose reduced the solubility more than the formation of composites, for example, the whey protein + bacterial cellulose composite [46].
Gelatin films were less affected by plasma treatment than by enzymatic treatment and incorporation of chemical additives. Enzymatic treatment of gelatin films with transglutaminase [48] showed a slightly superior effect in decreasing the film solubility (25%) compared to the plasma treatment (18%). The addition of polyvinyl alcohol was reported as a good alternative to reduce the gelatin film solubility [47].
3.3. FTIR Analysis
The FTIR spectra of the plasma-treated films are shown in Figure 1. The spectra of the starch film presented the characteristic peaks at 3284 cm−1 (-OH vibration), 2900 cm−1 (-CH vibration), 1150 cm−1 (C-O stretching), 1076 cm−1 (C-H bending), and 929 cm−1 (C-H out-of-plane bending).
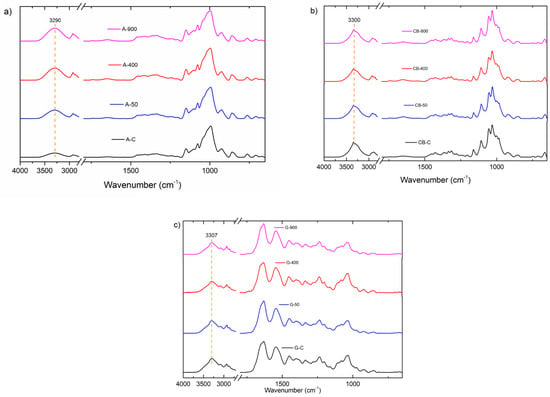
Figure 1.
FTIR spectra of (a) starch films, (b) bacterial cellulose films, and (c) gelatin films subjected to plasma treatment.
The spectra of the bacterial cellulose film presented the characteristic peaks at 3300 cm−1 (-OH vibration), 2900 cm−1 (-CH vibration), 1160 cm−1 (C-O-C asymmetric stretching vibration), and 1030 cm−1 (C-O stretching). The spectra of the gelatin film presented the characteristic peaks at 3300 cm−1 (-OH vibration), 2900 cm−1 (-CH vibration), 1635 cm−1 (C = O stretching vibration), 1550 cm−1 (N-H bending vibration), 1160 cm−1 (C-O-C asymmetric stretching and NH2 oscillation vibrations), and 1030 cm−1 (C-O stretching).
No new bands corresponding to the formation of chemical clusters were observed in the plasma-treated starch, bacterial cellulose, and gelatin films. Thus, plasma treatment promoted the interaction between the reactive plasma species and the functional groups already in the films [49].
The hydroxyl band (3284 cm−1) showed a slight reduction in intensity in all three films. The most significant reductions occurred at 50 Hz for the starch film and 900 Hz for the bacterial cellulose and gelatin films. All other characteristic bands of the films were preserved. The hydroxyl band further reduced its intensity when treated for an extended period (20 min). This reduction can be correlated to the higher hydrophobicity of the material plasma-treated for 15 and 20 min.
3.4. Best Biodegradable Films and Plasma Operating Conditions
The results obtained for hydrophobicity, water solubility, and FTIR analysis for the films processed at different cold plasma frequencies were evaluated using the desirability function to determine the best plasma conditions for each biodegradable film. Table 7 presents the scores obtained for the desirability function.

Table 7.
Scores obtained for the desirability function used to select the best starches, bacterial cellulose, and gelatin films’ processing conditions.
The scores of the desirability function have pointed out that the best starch film was produced at 50 Hz, while the best bacterial cellulose and gelatin films were produced at 900 Hz. These scores corroborate to point out that the cold plasma excitation frequency is an important variable when applying plasma to polymeric films.
The highest scores for each film agreed with the highest changes observed in hydrophobicity, water solubility, and chemical changes. Among the three films tested herein, the bacterial cellulose film presented the highest scores, especially due to its highest hydrophobicity, which is an important property of biodegradable films for food packaging.
3.5. Surface Morphology
The starch film showed a rough surface with no cracks (Figure 2). Plasma processing significantly changed the surface of the film. The surface became smoother than the untreated film, but many microscopic cracks appeared on the surface. Tiny white dots appeared scattered throughout the film surface, which could be related to the higher amount of insoluble particles in the film [44].

Figure 2.
Scanning electronic micrographs of: (a) untreated starch film, (b) plasma-treated starch film (20 min at 50 Hz), (c) untreated bacterial cellulose film, (d) plasma-treated bacterial cellulose film (20 min at 900 Hz), (e) untreated gelatin film, and (f) plasma-treated gelatin film (20 min at 900 Hz).
The bacterial cellulose film presented a reasonable roughness, which increased with plasma treatment. The treated film showed a more rugged structure.
The gelatin film presented a flat structure with several small dots attributed to insoluble matter. The plasma-treated film presented a different aspect, with a smoother surface. The small dots were drastically reduced, which could be attributed to a better homogenization and interaction of the soluble and insoluble matter.
The films’ hydrophobicity increased more in the films that presented a higher smoothness (starch and gelatin) than in the film (bacterial cellulose) where plasma treatment increased the roughness. The hydrophobicity is linked with the availability and exposure of hydroxyl groups, which are reduced during plasma treatment. Thus, the reduction in hydrophobicity may be associated with the decrease in hydroxyl groups and the changes in surface morphology, which may have reduced the exposure of the remaining hydroxyl groups.
3.6. Mechanical Properties
The mechanical properties of the films were evaluated at the best operating conditions for each film (20 min, 50 Hz for starch, 900 Hz for bacterial cellulose and gelatin) and compared to the control (untreated films). Table 8 presents the values obtained for the tensile strength (TS), elongation at break (ER), and elasticity module (EM) for all films.

Table 8.
Mechanical properties of the starch, bacterial cellulose, and gelatin films treated for 20 min with dielectric barrier discharge plasma at an optimal excitation frequency (50 Hz for the starch film and 900 Hz for the bacterial cellulose and gelatin films) without Mylar™ support. For each property, values followed by at least one common letter are not different from each other (p > 0.05).
Plasma treatment increased the mean values of the tensile strength of the films. However, due to the high standard deviation of this kind of test, it is not possible to affirm that plasma treatment has statistically increased the films’ tensile strength. The same trend was observed with the elasticity module, where the mean value increased after plasma treatment. The elongation at break decreased for the starch and gelatin films, while it increased for the bacterial cellulose film. Overall, it is possible to notice that plasma treatment tended to make the films more rigid.
The tensile strength is related to the mechanical resistance of the film. Plasma processing increased the average tensile strength of the films by 41%, 1%, and 23%, respectively, for starch, bacterial cellulose, and gelatin films. Although the mechanical resistances of the films were statistically similar, there was a positive trend in the average value, leading to more resistant starch and gelatin films.
The elongation at break is related to film flexibility. Plasma treatment only increased the flexibility of the bacterial cellulose (by 25%), while a reduction of 29% and 56% was observed in the starch and gelatin films, respectively.
Table 9 presents the mechanical properties of several films reported in the literature. Compared to other techniques, plasma treatment considerably increased the tensile strength of corn starch films. The increase in mechanical resistance attained by plasma treatment was higher than the increase gained with adding natural additives and similar to the increase achieved by high-pressure processing in potato starch films. Thus, mechanical resistance seems to increase more with physical treatments than chemical treatments.

Table 9.
Mechanical properties of several starches, bacterial cellulose, and gelatin films reported in the literature.
However, the flexibility of starch films decreased with the physical treatment and improved when specific natural products, such as fireweed extract, were incorporated into the film. In most cases, chemical treatments decreased the flexibility of starch films more than physical treatments.
The tensile strength and elongation at break of bacterial cellulose and its composites reported in the literature have a broad range of values (from 1 to 70 MPa for the tensile strength and from 7% to 80% for the elongation at break), which are affected by the formation methodology of the film and other parameters. The methods used herein produced a film with high tensile strength and low elongation at break, while the methods used by Efthymiou et al. [45] and Papadaki et al. [46] produced a film with low tensile strength and high elongation at break. Plasma treatment increased both tensile strength and elongation at break, as occurred with the chemical treatment to convert bacterial cellulose to nanocrystalline cellulose. The amplitude of change of the tensile strength was similar between the plasma and chemical treatments. However, the chemical treatment appears to better affect the elongation at break. The production of composites such as whey protein + bacterial cellulose increases the tensile strength but decreases the elongation at break.
Plasma treatment showed a superior effect in increasing the tensile strength of gelatin films compared to the addition of natural extracts, such as extracts from B. gymnorhiza and S. alba [53]. However, the effect was lower than that attained by producing a gelatin and polyvinyl alcohol composite [47]. Regarding the elongation at break, plasma treatment reduced this property, making a more rigid film. On the other hand, the addition of natural compounds [53], enzymatic treatment [48], and the composition of polyvinyl alcohol [47] increased the flexibility of gelatin films.
4. Conclusions
Plasma treatment improved the hydrophobicity, surface morphology, tensile strength, and elasticity module and reduced the water solubility of starch, bacterial cellulose, and gelatin films. The applied excitation frequency during plasma generation significantly affected the films’ properties and requires optimization for better results. A low excitation frequency (50 Hz) resulted in greater changes in starch films, while a high excitation frequency (900 Hz) induced greater changes in gelatin and bacterial cellulose films.
Films produced on Mylar™ support should have the support removed before subjecting the film to plasma treatment. Treating the films with the support resulted in less properties’ modifications because the changes occurred on a single surface instead of both surfaces.
Plasma treatment is an alternative to improve the hydrophobicity, surface morphology, water resistance, and mechanical properties of biodegradable films. Its performance can be superior, similar, or lower than other options, such as chemical treatment, enzymatic treatment, the addition of chemical compounds, and composites. It has the advantage of not recurring to chemical or biological additives, and no harmful compound remains in the film after plasma application. The treatment’s disadvantage is its limited effect on the mechanical properties of the films since bigger changes could be achieved by producing composites or by chemical addition.
Author Contributions
Conceptualization, M.d.F.R. and F.A.N.F.; methodology, H.M.C.d.A., M.d.F.R., and F.A.N.F.; formal analysis, M.L.G. and F.A.N.F.; investigation, M.L.G. and A.L.A.M.; writing—original draft preparation, M.L.G. and F.A.N.F.; writing—review and editing, F.A.N.F.; funding acquisition, M.d.F.R. and F.A.N.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP), grant number PR2-0101-00023.01.00/15, and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), a scholarship awarded to M.L.G.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Romani, V.P.; Olsen, B.; Pinto Collares, M.; Meireles Oliveira, J.R.; Prentice, C.; Martins, V.G. Cold Plasma and Carnauba Wax as Strategies to Produce Improved Bi-Layer Films for Sustainable Food Packaging. Food Hydrocoll. 2020, 108, 106087. [Google Scholar] [CrossRef]
- Gandhi, N.; Khurana, S.; Mathur, R.; Bansal, U.; Nair, R. Trends in Food Packaging: A Comprehensive Review. Asian J. Chem. 2022, 34, 2499–2510. [Google Scholar] [CrossRef]
- Choi, S.M.; Rao, K.M.; Zo, S.M.; Shin, E.J.; Han, S.S. Bacterial Cellulose and Its Applications. Polymers 2022, 14, 1080. [Google Scholar] [CrossRef] [PubMed]
- Onyeaka, H.; Obileke, K.; Makaka, G.; Nwokolo, N. Current Research and Applications of Starch-Based Biodegradable Films for Food Packaging. Polymers 2022, 14, 1126. [Google Scholar] [CrossRef]
- Moosavi, M.H.; Khani, M.R.; Shokri, B.; Hosseini, S.M.; Shojaee-Aliabadi, S.; Mirmoghtadaie, L. Modifications of Protein-Based Films Using Cold Plasma. Int. J. Biol. Macromol. 2020, 142, 769–777. [Google Scholar] [CrossRef]
- Sifuentes-Nieves, I.; Hernández-Hernández, E.; Neira-Velázquez, G.; Morales-Sánchez, E.; Mendez-Montealvo, G.; Velazquez, G. Hexamethyldisiloxane Cold Plasma Treatment and Amylose Content Determine the Structural, Barrier and Mechanical Properties of Starch-Based Films. Int. J. Biol. Macromol. 2019, 124, 651–658. [Google Scholar] [CrossRef]
- Azeredo, H.M.C.; Barud, H.; Farinas, C.S.; Vasconcellos, V.M.; Claro, A.M. Bacterial Cellulose as a Raw Material for Food and Food Packaging Applications. Front. Sustain. Food Syst. 2019, 3, 7. [Google Scholar] [CrossRef]
- Díaz-Calderón, P.; MacNaughtan, B.; Hill, S.; Foster, T.; Enrione, J.; Mitchell, J. Changes in Gelatinisation and Pasting Properties of Various Starches (Wheat, Maize and Waxy Maize) by the Addition of Bacterial Cellulose Fibrils. Food Hydrocoll. 2018, 80, 274–280. [Google Scholar] [CrossRef]
- Romani, V.P.; Olsen, B.; Pinto Collares, M.; Meireles Oliveira, J.R.; Prentice-Hernández, C.; Guimarães Martins, V. Improvement of Fish Protein Films Properties for Food Packaging through Glow Discharge Plasma Application. Food Hydrocoll. 2019, 87, 970–976. [Google Scholar] [CrossRef]
- Marenco-Orozco, G.A.; Rosa, M.F.; Fernandes, F.A.N. Effects of Multiple-step Cold Plasma Processing on Banana (Musa Sapientum) Starch-based Films. Packag. Technol. Sci. 2022, 35, 589–601. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, K.; Yao, X.; Jiang, J.; Wang, M.; Yuan, S. Ultrasound-Assisted Preparation of Fe(OH)3@bacterial Cellulose Aerogel for Efficient Removal of Organic Contamination in Water. Appl. Surf. Sci. 2023, 607, 154959. [Google Scholar] [CrossRef]
- Šafranko, S.; Stanković, A.; Hajra, S.; Kim, H.-J.; Strelec, I.; Dutour-Sikirić, M.; Weber, I.; Bosnar, M.H.; Grbčić, P.; Pavelić, S.K.; et al. Preparation of Multifunctional N-Doped Carbon Quantum Dots from Citrus Clementina Peel: Investigating Targeted Pharmacological Activities and the Potential Application for Fe3+ Sensing. Pharmaceuticals 2021, 14, 857. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.; Hajra, S.; Jeong, H.; Panigrahi, B.K.; Pakawanit, P.; Dubal, D.; Hong, S.; Kim, H.J. Biocompatible CaTiO3-PVDF Composite-Based Piezoelectric Nanogenerator for Exercise Evaluation and Energy Harvesting. Nano Energy 2022, 102, 107682. [Google Scholar] [CrossRef]
- Chen, G.; Chen, Y.; Jin, N.; Li, J.; Dong, S.; Li, S.; Zhang, Z.; Chen, Y. Zein Films with Porous Polylactic Acid Coatings via Cold Plasma Pre-Treatment. Ind. Crops Prod. 2020, 150, 112382. [Google Scholar] [CrossRef]
- Wu, X.; Liu, Q.; Luo, Y.; Murad, M.S.; Zhu, L.; Mu, G. Improved Packing Performance and Structure-Stability of Casein Edible Films by Dielectric Barrier Discharges (DBD) Cold Plasma. Food Packag. Shelf Life 2020, 24, 100471. [Google Scholar] [CrossRef]
- Pankaj, S.K.K.; Bueno-Ferrer, C.; Misra, N.N.N.; O’Neill, L.; Tiwari, B.K.K.; Bourke, P.; Cullen, P.J.J. Dielectric Barrier Discharge Atmospheric Air Plasma Treatment of High Amylose Corn Starch Films. LWT-Food Sci. Technol. 2015, 63, 1076–1082. [Google Scholar] [CrossRef]
- Pankaj, S.K.; Bueno-Ferrer, C.; Misra, N.N.; O’Neill, L.; Tiwari, B.K.; Bourke, P.; Cullen, P.J. Physicochemical Characterization of Plasma-Treated Sodium Caseinate Film. Food Res. Int. 2014, 66, 438–444. [Google Scholar] [CrossRef]
- Lin, S.-P.; Huang, S.-H.; Ting, Y.; Hsu, H.-Y.; Cheng, K.-C. Evaluation of Detoxified Sugarcane Bagasse Hydrolysate by Atmospheric Cold Plasma for Bacterial Cellulose Production. Int. J. Biol. Macromol. 2022, 204, 136–143. [Google Scholar] [CrossRef]
- Santoso, S.P.; Lin, S.-P.; Wang, T.-Y.; Ting, Y.; Hsieh, C.-W.; Yu, R.-C.; Angkawijaya, A.E.; Soetaredjo, F.E.; Hsu, H.-Y.; Cheng, K.-C. Atmospheric Cold Plasma-Assisted Pineapple Peel Waste Hydrolysate Detoxification for the Production of Bacterial Cellulose. Int. J. Biol. Macromol. 2021, 175, 526–534. [Google Scholar] [CrossRef]
- Janpetch, N.; Saito, N.; Rujiravanit, R. Fabrication of Bacterial Cellulose-ZnO Composite via Solution Plasma Process for Antibacterial Applications. Carbohydr. Polym. 2016, 148, 335–344. [Google Scholar] [CrossRef]
- Carvalho, A.P.M.G.; Barros, D.R.; da Silva, L.S.; Sanches, E.A.; da Costa Pinto, C.; de Souza, S.M.; Clerici, M.T.P.S.; Rodrigues, S.; Fernandes, F.A.N.; Campelo, P.H. Dielectric Barrier Atmospheric Cold Plasma Applied to the Modification of Ariá (Goeppertia Allouia) Starch: Effect of Plasma Generation Voltage. Int. J. Biol. Macromol. 2021, 182, 1618–1627. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Gou, Q.; Yang, L.; Yu, Q.; Han, L. Dielectric Barrier Discharge Plasma: A Green Method to Change Structure of Potato Starch and Improve Physicochemical Properties of Potato Starch Films. Food Chem. 2022, 370, 130992. [Google Scholar] [CrossRef] [PubMed]
- Goiana, M.L.; de Brito, E.S.; Alves Filho, E.G.; de Castro Miguel, E.; Fernandes, F.A.N.; de Azeredo, H.M.C.; de Freitas Rosa, M. Corn Starch Based Films Treated by Dielectric Barrier Discharge Plasma. Int. J. Biol. Macromol. 2021, 183, 2009–2016. [Google Scholar] [CrossRef] [PubMed]
- Campelo, P.H.; Alves Filho, E.G.; Silva, L.M.A.; de Brito, E.S.; Rodrigues, S.; Fernandes, F.A.N. Modulation of Aroma and Flavor Using Glow Discharge Plasma Technology. Innov. Food Sci. Emerg. Technol. 2020, 62, 102363. [Google Scholar] [CrossRef]
- Alves Filho, E.G.; Silva, L.M.A.; Oiram Filho, F.; Rodrigues, S.; Fernandes, F.A.N.; Gallão, M.I.; Mattison, C.P.; de Brito, E.S. Cold Plasma Processing Effect on Cashew Nuts Composition and Allergenicity. Food Res. Int. 2019, 125, 108621. [Google Scholar] [CrossRef]
- Farias, T.R.B.; Rodrigues, S.; Fernandes, F.A.N. Effect of Dielectric Barrier Discharge Plasma Excitation Frequency on the Enzymatic Activity, Antioxidant Capacity and Phenolic Content of Apple Cubes and Apple Juice. Food Res. Int. 2020, 136, 109617. [Google Scholar] [CrossRef]
- Almeida, F.D.L.; Cavalcante, R.S.; Cullen, P.J.; Frias, J.M.; Bourke, P.; Fernandes, F.A.N.N.; Rodrigues, S. Effects of Atmospheric Cold Plasma and Ozone on Prebiotic Orange Juice. Innov. Food Sci. Emerg. Technol. 2015, 32, 127–135. [Google Scholar] [CrossRef]
- Rodrigues, S.; Fernandes, F.A.N. Glow Discharge Plasma Processing for the Improvement of Pasteurized Orange Juice’s Aroma and Off-Flavor. Processes 2022, 10, 1812. [Google Scholar] [CrossRef]
- Oliveira, A.V.; da Silva, A.P.M.; Barros, M.O.; de sá M. Souza Filho, M.; Rosa, M.F.; Azeredo, H.M.C. Nanocomposite Films from Mango Kernel or Corn Starch with Starch Nanocrystals. Starch-Stärke 2018, 70, 1800028. [Google Scholar] [CrossRef]
- Saito, T.; Kimura, S.; Nishiyama, Y.; Isogai, A. Cellulose Nanofibers Prepared by TEMPO-Mediated Oxidation of Native Cellulose. Biomacromolecules 2007, 8, 2485–2491. [Google Scholar] [CrossRef]
- do Nascimento, E.S.; Pereira, A.L.S.; de Oliveira Barros, M.; de Aaraújo Barroso, M.K.; Lima, H.L.S.; de Fatima Borges, M.; de Andrade Feitosa, J.P.; de Azeredo, H.M.C.; de Freitas Rosa, M. TEMPO Oxidation and High-Speed Blending as a Combined Approach to Disassemble Bacterial Cellulose. Cellulose 2019, 26, 2291–2302. [Google Scholar] [CrossRef]
- Santos, T.M.; Men de Sá Filho, M.S.; Caceres, C.A.; Rosa, M.F.; Morais, J.P.S.; Pinto, A.M.B.; Azeredo, H.M.C. Fish Gelatin Films as Affected by Cellulose Whiskers and Sonication. Food Hydrocoll. 2014, 41, 113–118. [Google Scholar] [CrossRef]
- ASTM D 5725-99; Standard Test Method for Surface Wettability and Adsorbency of Sheeted Materials Using an Automated Contact Angle Tester. ASTM: West Conshohocken, PA, USA, 2008.
- Pena-Serna, C.; Lopes-Filho, J.F. Influence of Ethanol and Glycerol Concentration over Functional and Structural Properties of Zein–Oleic Acid Films. Mater. Chem. Phys. 2013, 142, 580–585. [Google Scholar] [CrossRef]
- ASTM D882-12; Standard Test Method for Tensile Properties of Thin Plastic Sheeting. ASTM International: West Conshohocken, PA, USA, 2018.
- Wan, S.; Liu, Q.; Yang, D.; Guo, P.; Gao, Y.; Mo, R.; Zhang, Y. Characterization of High Amylose Corn Starch-Cinnamaldehyde Inclusion Films for Food Packaging. Food Chem. 2022, 403, 134219. [Google Scholar] [CrossRef]
- Guo, S.; Fu, Z.; Yao, X.; Wang, X.; Wu, M. Impact of High Pressure Homogenization on the Properties of Potato Flour Film-Forming Dispersions and the Resulting Films. J. Food Eng. 2023, 339, 111281. [Google Scholar] [CrossRef]
- Fu, B.; Mei, S.; Su, X.; Chen, H.; Zhu, J.; Zheng, Z.; Lin, H.; Dai, C.; Luque, R.; Yang, D.-P. Integrating Waste Fish Scale-Derived Gelatin and Chitosan into Edible Nanocomposite Film for Perishable Fruits. Int. J. Biol. Macromol. 2021, 191, 1164–1174. [Google Scholar] [CrossRef]
- Cubas, A.L.V.; Bianchet, R.T.; de Oliveira, D.; Leonarski, E.; Cesca, K. Application of Non-Thermal Plasma as an Alternative for Purification of Bacterial Cellulose Membranes. Sustain. Chem. Pharm. 2022, 29, 100800. [Google Scholar] [CrossRef]
- Wasim, M.; Shi, F.; Liu, J.; Zhang, H.; Zhu, K.; Tian, Z. Synthesis and Characterization of Curcumin/MMT-Clay-Treated Bacterial Cellulose as an Antistatic and Ultraviolet-Resistive Bioscaffold. J. Polym. Res. 2022, 29, 423. [Google Scholar] [CrossRef]
- Mir, S.A.; Dar, B.N.; Wani, A.A.; Shah, M.A. Effect of Plant Extracts on the Techno-Functional Properties of Biodegradable Packaging Films. Trends Food Sci. Technol. 2018, 80, 141–154. [Google Scholar] [CrossRef]
- Pająk, P.; Przetaczek-Rożnowska, I.; Juszczak, L. Development and Physicochemical, Thermal and Mechanical Properties of Edible Films Based on Pumpkin, Lentil and Quinoa Starches. Int. J. Biol. Macromol. 2019, 138, 441–449. [Google Scholar] [CrossRef]
- Chhatariya, H.F.; Srinivasan, S.; Choudhary, P.M.; Begum, S.S. Corn Starch Biofilm Reinforced with Orange Peel Powder: Characterization of Physicochemical and Mechanical Properties. Mater. Today Proc. 2022, 59, 884–892. [Google Scholar] [CrossRef]
- Alqahtani, N.; Alnemr, T.; Ali, S. Development of Low-Cost Biodegradable Films from Corn Starch and Date Palm Pits (Phoenix Dactylifera). Food Biosci. 2021, 42, 101199. [Google Scholar] [CrossRef]
- Efthymiou, M.-N.; Tsouko, E.; Papagiannopoulos, A.; Athanasoulia, I.-G.; Georgiadou, M.; Pispas, S.; Briassoulis, D.; Tsironi, T.; Koutinas, A. Development of Biodegradable Films Using Sunflower Protein Isolates and Bacterial Nanocellulose as Innovative Food Packaging Materials for Fresh Fruit Preservation. Sci. Rep. 2022, 12, 6935. [Google Scholar] [CrossRef] [PubMed]
- Papadaki, A.; Manikas, A.C.; Papazoglou, E.; Kachrimanidou, V.; Lappa, I.; Galiotis, C.; Mandala, I.; Kopsahelis, N. Whey Protein Films Reinforced with Bacterial Cellulose Nanowhiskers: Improving Edible Film Properties via a Circular Economy Approach. Food Chem. 2022, 385, 132604. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, G.D.J.C.; Campelo, P.H.; de Abreu Figueiredo, J.; Barbosa de Souza, H.J.; Peixoto Joele, M.R.S.; Yoshida, M.I.; Henriques Lourenço, L.D.F. Effect of Polyvinyl Alcohol and Carboxymethylcellulose on the Technological Properties of Fish Gelatin Films. Sci. Rep. 2022, 12, 10497. [Google Scholar] [CrossRef]
- Baggio, E.; Scopel, B.S.; Rosseto, M.; Rigueto, C.V.T.; Dettmer, A.; Baldasso, C. Transglutaminase Effect on the Gelatin-Films Properties. Polym. Bull. 2022, 79, 7347–7361. [Google Scholar] [CrossRef]
- Hernandez-Perez, P.; Flores-Silva, P.C.; Velazquez, G.; Morales-Sanchez, E.; Rodríguez-Fernández, O.; Hernández-Hernández, E.; Mendez-Montealvo, G.; Sifuentes-Nieves, I. Rheological Performance of Film-Forming Solutions Made from Plasma-Modified Starches with Different Amylose/Amylopectin Content. Carbohydr. Polym. 2021, 255, 117349. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Szymanowska, U.; Skrzypek, T.; Basiura-Cembala, M.; Materska, M.; Łupina, K. Corn Starch and Methylcellulose Edible Films Incorporated with Fireweed (Chamaenerion Angustifolium L.) Extract: Comparison of Physicochemical and Antioxidant Properties. Int. J. Biol. Macromol. 2021, 190, 969–977. [Google Scholar] [CrossRef]
- Prachayawarakorn, J.; Kansanthia, P. Characterization and Properties of Singly and Dually Modified Hydrogen Peroxide Oxidized and Glutaraldehyde Crosslinked Biodegradable Starch Films. Int. J. Biol. Macromol. 2022, 194, 331–337. [Google Scholar] [CrossRef]
- Martins, P.C.; Latorres, J.M.; Martins, V.G. Impact of Starch Nanocrystals on the Physicochemical, Thermal and Structural Characteristics of Starch-Based Films. LWT 2022, 156, 113041. [Google Scholar] [CrossRef]
- Nurdiani, R.; Ma’rifah, R.D.A.; Busyro, I.K.; Jaziri, A.A.; Prihanto, A.A.; Firdaus, M.; Talib, R.A.; Huda, N. Physical and Functional Properties of Fish Gelatin-Based Film Incorporated with Mangrove Extracts. PeerJ 2022, 10, e13062. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).


