1. Introduction
Additive manufacture technology (AM), commonly known as 3D printing, allows the creation of complex structures using different types of materials, such as polymers, photopolymer resins, ceramics, and metals [
1]. Its freedom of design, low cost, and speed are some of the advantages that make 3D printing attractive for many industrial sectors [
2,
3], especially in the biomedical field, where it can be used to produce patients’ customized implants [
4].
AM can be divided into seven technologies: material extrusion, material jetting, powder bed fusion, direct energy deposition, binder jetting, sheet addition, and vat polymerization, each with its own advantages and limitations [
5,
6]. Stereolithography, part of the vat polymerization technology, uses a combination of photosensitive liquids made of acrylates or epoxy-acrylates with a photo-initiator and a light emitter (wavelength between 360 and 405 nm) to produce highly accurate components, down to 5 microns of resolution [
7,
8]. The objects produced by this technique usually exhibit good thermal, mechanical, and chemical properties [
9]. However, most of the materials commonly used to produce the resins are derived from crude oil, which, in addition to being nonrenewable and having low biocompatibility, is a major cause of environmental pollution [
10].
In recent years, a limited number of academic studies started to address the lack of biocompatible and biobased materials for SLA applications. These new resins are based on renewable materials, such as vegetable oils (soybean, corn, sunflower, linseed, etc.) [
11,
12,
13] or clinically approved polymers such as poly(ethylene glycol) (PEG), poly(caprolactone) (PCL), poly(lactic acid) (PLA), and poly(glycolic acid) (PGA) [
14,
15,
16,
17]. Among these different polymers, PEG is one of the best candidates for 3D printing, since it can be easily functionalized and modified [
18,
19]. Kuo et al. [
20] used poly(ethylene glycol)diacrylate (
PEGDA) with a suitable photo-initiator (namely, Irgacure-819) to create a variety of complex 3D microfluidic devices [
20]. Seo et al. [
21] employed a similar resin composition to investigate its suitability for complex structures (such as tilted ratchets and ladders) [
21]. Unfortunately, the objects produced using pure
PEGDA and its derivatives via radical polymerization demonstrated poor mechanical properties [
22].
An alternative to improve PEG properties while maintaining its biocompatibility could be its combination with other types of resins; in this context, plant-based resins have attractive characteristics due to their renewability, biodegradability, and low pollutant emissions [
23]. Originating from renewable sources, they are environmentally friendly molecules, are relatively cheap to manufacture and employ, and can be modified to react with ultraviolet radiation [
24,
25]. Epoxy-acrylated soybean oil (
AESO), produced from soybean, has been shown to be a promising material [
25]. It was recently used to develop 4D scaffolds [
26], complex structures [
25], and high-performance parts [
27], where it demonstrated good biocompatibility, as well as thermal and mechanical properties. Lebedevaite et al. [
28] combined AESO with biobased materials to develop a resin with similar properties to petroleum-based counterparts. Their resins showed high renewable carbon content (between 75% and 82%), high crosslinking rate, and tunable mechanical properties.
Another method to improve the properties of SLA resins is through the addition of fibers, fillers, or nanoparticles [
29,
30]. These particles can be added directly to the resin or have their chemical structure modified to improve their dispersion and interfacial adhesion, i.e., by attaching a functional group onto the particle surface (“
grafting”) [
31]. Moreover, the functionalization of particles can help in preventing the phase separation between two polymeric components (an A/B mixture) [
32]. Among the many types of fillers, cellulose is an excellent reinforcement material. It is the most abundant natural polymer on Earth, and its reinforcing derivatives are environmentally friendly, are relatively cheap to produce, and show impressive mechanical properties [
33,
34]. Cellulose and its derivatives are also being tested for biomedical applications [
35]. Dutta et al. developed a hydrogel containing cellulose nanocrystal (CNC), alginate, and gelatin to be tested for bone regeneration. Their results showed that the scaffolds with CNC had an improvement in cell proliferation when compared to the control group [
36]. However, the biomolecular interaction of cellulose micro- and nanoparticles on cells and tissues, as well as their hazard potential, still needs to be further investigated [
37]. Nevertheless, for 3D printing, cellulose has already been used to reinforce different types of materials [
38,
39]. Murphy et al. used microcrystalline cellulose (MCC) to improve the mechanical properties of PLA bio-composites, exploiting its high Young’s modulus (25 ± 4 GPa) [
40]. Palaganas et al. successfully combined cellulose nanocrystals with
PEGDA. Their results showed increased glass transition temperature and tensile strength and decreased swelling when 0.3 wt.% CNCs were incorporated into the diacrylate [
15].
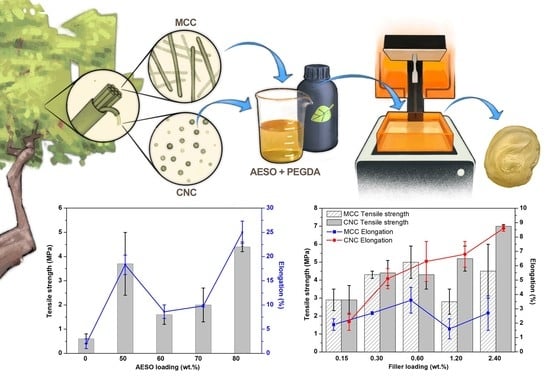
The main goal of the present work was to develop a high-quality and environmentally friendly biobased system, suitable for 3D printing. To this aim, PEGDA was combined with different concentrations of AESO, and their optimum ratio was determined. Then, micro- or nanocrystalline cellulose was incorporated into the optimized PEGDA/AESO formulation, and Fourier-transform infrared spectroscopy (FTIR) was exploited for monitoring the progress of the UV curing of the designed system during the SLA process. Furthermore, the rheological, thermal, mechanical, and 3D printing properties of the biobased composites were investigated. To the best of our knowledge, this is the first time that PEGDA, AESO, MCCs, and CNCs have been combined and tested for 3D-printing applications. The obtained results show the possible exploitation of cellulose-reinforced biobased materials in the 3D printing industry.
2. Materials and Methods
2.1. Materials
Poly(ethylene glycol) diacrylate (number average molecular weight: 575), epoxy-acrylate soybean oil (containing 4000 ppm of monomethyl ether hydroquinone as inhibitor), diphenyl(2,4,6-trimethylbenzoyl)phosphine oxide (TPO), and cellulose microcrystalline powder (20 μm) were purchased from Merck KGaA (Darmstadt, Germany). Cellulose nanocrystals were purchased from Sappi limited (Maastricht, the Netherlands). Isopropanol (purity: 99%) was purchased from Carlo Erba (Cornaredo, Italy). All products were used as received.
2.2. Preparation of the Photocurable Systems
Photocurable resins were prepared while avoiding any exposure to light, and the procedure is presented in
Scheme 1. First, using a lightproof beaker, different weight ratios (varying from 50 to 90 wt.%) of
AESO were mixed with poly(ethylene glycol). Then, 2 wt.% TPO was added to the mixtures, which were ultrasonicated for 10 min at room temperature to dissolve the photo-initiator, before vigorously stirring for 30 min. The resulting mixture had a yellowish appearance.
The bio content (
BC) of each
PEGDA/
AESO sample is described in
Table 1 and was calculated according to Equation (1).
where
w is the mass fraction,
i is the resin type, and bio-renewable carbon (
BRC) is the ratio of bio-sourced carbon to the sum of bio- and fossil-based carbons. The
BRC values of
AESO and
PEGDA are 86% and 0%, respectively [
41].
The mixture of PEGDA/AESO with a 20:80 ratio was selected for the present investigation since it had the best mechanical properties with the most significant bio-content percentage, equal to 68.8%. Then, different percentages of MCCs or CNCs (ranging from 0.15 to 2.4 wt.%) were incorporated into the selected mixture. The fillers were added in a small amount (maximum 0.2 g at a time) and ultrasonicated for 10 min at 30 °C to avoid the formation of aggregates.
2.3. 3D Printing Process
The samples were printed using a Peopoly moai 130 SLA 3D printer with an easy-to-level build plate. The machine was equipped with a solid-state laser with frequency conversion, emitting at 405 nm with a power of 150 mW. The power level was 58, and the initial exposure time was in the range between 40 and 60 s. The printed process was carried out maintaining the temperature constant at 25 °C. After the end of the printing, the excess of resin was drained and washed off with isopropanol; then, the samples were gently dipped into distilled water for 5 min. Finally, the samples were dried using a paper towel and transferred to a UV chamber equipped with a 405 nm UV lamp to post-cure for 40 min. The post-curing time was the same for all samples to ensure a complete photopolymerization, as assessed by FTIR spectroscopy. The 3D models were created using FreeCad software, and the printing layer height of the samples was set at 0.1 mm.
2.4. FTIR Spectroscopy
Fourier-transform infrared spectroscopy (FTIR) was used to confirm the formation of
PEGDA:
AESO copolymers at different ratios and to monitor the conversion of the acrylic double bonds. The infrared analysis was performed using a Thermo Avatar 370 spectrophotometer equipped with an attenuated total reflectance (ATR) device with a diamond crystal for solid analysis. Using Omnic 7.3 software, the spectra were collected in absorbance mode with a resolution of 4 cm
−1 and 32 scans per measurement, within the range of 2000 to 650 cm
−1, using 3D-printed dog-bone specimens. The conversion of double bonds was calculated from the decrease in the area of the double-bond absorption peak at 810 cm
−1, as shown by Equation (2).
where A
0(810) and A
t(810) are the areas of the peak at 810 cm
−1, before and after exposure to UV irradiation, respectively.
2.5. Rheological Analysis
Rheological measurements were performed using an ARES (TA Instrument, Waters LLC, New Castle, DE, USA) strain-controlled rheometer in parallel plate geometry (plate diameter: 50 mm; gap between the plates: 0.7 mm). The complex viscosity of the samples was measured through strain sweep measurements in a range of strain amplitude from 1% to 400%. In all tests, the frequency was fixed at 1 rad·s−1. All measurements were performed at room temperature.
2.6. Tensile Measurements
All tensile tests were performed following the ASTM D638 standard. Five samples were printed with a dog-bone shape with the dimensions of 63.5 mm × 9.53 mm × 3.2 mm (L × W × T) and with 3.2 mm width in the narrow section. The tests were performed at room temperature with an Instron 5966 dynamometer (Norwood, MA, USA), equipped with 5 kN load cell. Each test was performed at a crosshead speed of 10 mm/min. Tensile strength (MPa) and elongation at break (%) were determined using the average of the five tests. Then, various mechanical properties were calculated using the data from the tensile curves, such as Young’s modulus (MPa) and fracture energy (mJ).
2.7. Scanning Electron Microscopy
The morphology of the obtained 3D-printed systems was studied using an EVO 15 scanning electron microscope (SEM) from Zeiss (Oberkochen, Germany), coupled with an Ultim Max 40 energy dispersive X-ray (EDX) microanalyzer by Oxford Instruments (High Wycombe, UK); the samples were fractured in liquid nitrogen, fastened to a conductive adhesive tape, and finally gold-metallized. The samples were analyzed using a secondary electron detector, with energy set at 20.00 kV, at two magnifications (1000× and 2500×).
2.8. Thermogravimetric Analysis
The thermal and thermo-oxidative stability of all prepared systems was assessed by thermogravimetric (TG) analyses carried out on a Discovery apparatus (TA Instruments), from 50 to 700 °C, using a heating rate of 10 °C·min−1, under both nitrogen and air flow (35 and 25 mL·min−1, respectively). The experimental error was ±0.5% for the weight and ±1 °C for the temperature.
2.9. Swelling Behavior
Five rectangular samples with dimensions of 30 mm × 10 mm × 5 mm (L × W × H) were prepared with different
PEGDA:
AESO weight ratios and CNC or MCC loadings and then weighed (
M1). The samples were then dipped into deionized water for a total period of 30 days. Every 5 days, the samples were taken from the water, dried using a dry cloth, and weighed again (
M2). The swelling ratio,
Sw, was calculated using Equation (3).
2.10. Contact Angle Measurement
Static contact angle values of AESO/PEGDA samples were measured using a homemade instrument equipped with a high-speed CCD camera. The used equipment allows the determination of contact angle, with a precision of ±1°, by taking images at frequencies as high as 200 Hz, starting within a few tens of milliseconds after the deposition of the drop. All measurements were performed at room temperature and relative humidity RH = 40% ± 5%. Five rectangular-shaped parts (size: 50 mm × 25 mm × 1 mm) were 3D-printed for each formulation. A small drop of high-purity distilled water was slowly placed on the surface of the samples, and various photos of the droplet were recorded after 5 s. The volume of the water drop was 4 ± 0.5 μL. The contact angle (CA) values were determined using image analysis software.
2.11. Working Curve
Since the kinetics during the curing process is highly complex, simplified equations are commonly used to describe the kinetics during photopolymerization [
42]. In this study, an equation was adapted from the Beer–Lambert relationship, expressing the exponential decay of the light intensity as it passes through an absorbing medium i [
43,
44]. This semiempirical equation relates the thickness of the crosslinked layer (cure depth) to the irradiation dose through the following equation:
where
Cd is the cure depth of a single layer,
Dp is the penetration depth at which the intensity of the beam is reduced to 1/e
2 (13.5%) of its value at the surface,
Ec is the exposure per unit of area needed for the resin to reach its gel point, and
Emax is the laser exposure on the resin surface.
Dp and E
c were estimated by varying the resin exposure and measuring the cured layer depth. By plotting
Cd vs. ln(
Emax), a line known as the working curve was obtained.
Dp was calculated from the slope of the line, and
Ec was the X-intercept. To this aim, a square part composed of 25 equal-area square tiles (1 mm
2), based on the model described by Bassett et al. [
45], as shown in
Figure 1, was printed using the Peopoly moai 130 SLA printer.
The part had 25 layers in total, with the height of each layer being 0.05 mm. Each square was exposed to light in an arithmetic progression increasing from 1 to 25. Furthermore, to avoid the effect of light scattering, the squares were separated by a gap of 1 mm. The height of each tile was measured three times using a digital caliper with 0.01 mm of resolution (Preciva Digital Caliper) and a ±10% deviation was assumed [
46]. Finally, with the height of each tile, the maximum exposure of a tile was calculated using Equation (5).
where
n is the number of times the laser passed over the tile,
PL is the laser power,
W0 is the beam width, and
Vs is the scanning speed.
PL and
W0 values are specific to the 3D printer; in this case, they were 150 mW and 0.07 mm, respectively. Three parts were printed with scanning speeds of 80 mm·s
−1 and 200 mm·s
−1.
When the MCCs and CNCs were added to the mixture, the light scattering effect exerted by these particles needed to be taken into account [
44]. Thus,
Dp was adjusted to consider the scattering effect, as shown by Equation (6).
where
d is the filler mean size,
φ is the filler volume fraction, and
Q is the scattering efficiency.
Q was calculated according to Equation (7), in which
λ is the irradiation wavelength,
h is the interparticle distance, and Δ
n is the difference in refractive index between the filler (
nf) and the photosensitive material (
n0).
4. Discussion
The development of biobased resins for 3D printing is essential to reduce the impact of fossil-based materials in nature and to design new applications. This study focused on the development and reinforcement of a biocompatible system capable of printing complex structures. The resin system was first optimized in order to identify the best PEGDA-to-AESO ratio suitable for the 3D printing process, i.e., with an appropriate viscosity, good mechanical properties, high bio-content, and high reactivity; in particular, the mixture containing 80 wt.% AESO was the most performing and was, therefore, chosen as the reference matrix for dispersing micro- and nanocellulose crystals, assessing their potential as fillers for 3D-printed composites. The incorporation of the fillers at different loadings (up to 2.4 wt.%) into the resin formulation containing 80 wt.% AESO did not interfere with the 3D printing process, nor had a significant impact on the resin critical energy and depth penetration. Furthermore, the presence of the cellulose crystals, irrespective of their size, increased the wettability of the printed parts (with a lowering of the water contact angle values by about 34% and 43%, when 2.4 wt.% CNCs and MCCs were incorporated, respectively). This finding was attributed to the highly polar characteristics of the fillers, bearing several hydroxyl functionalities. Meanwhile, as assessed by mechanical tests, the tensile strength increased by about 59%, in the presence of 2.4 wt.% CNCs, and the elastic modulus increased by 890%, when the same MCC loading was employed. However, further research on the influence of AESO:PEGDA resin and its combination with CNCs and MCCs on cellular proliferation and tissue response is needed to better understand its impact on living cells. In conclusion, the composite system developed in this work may represent a sustainable solution to the increasing demand for new environmentally friendly materials for additive manufacturing processes.