Biomaterials as Implants in the Orthopedic Field for Regenerative Medicine: Metal versus Synthetic Polymers
Abstract
1. Introduction
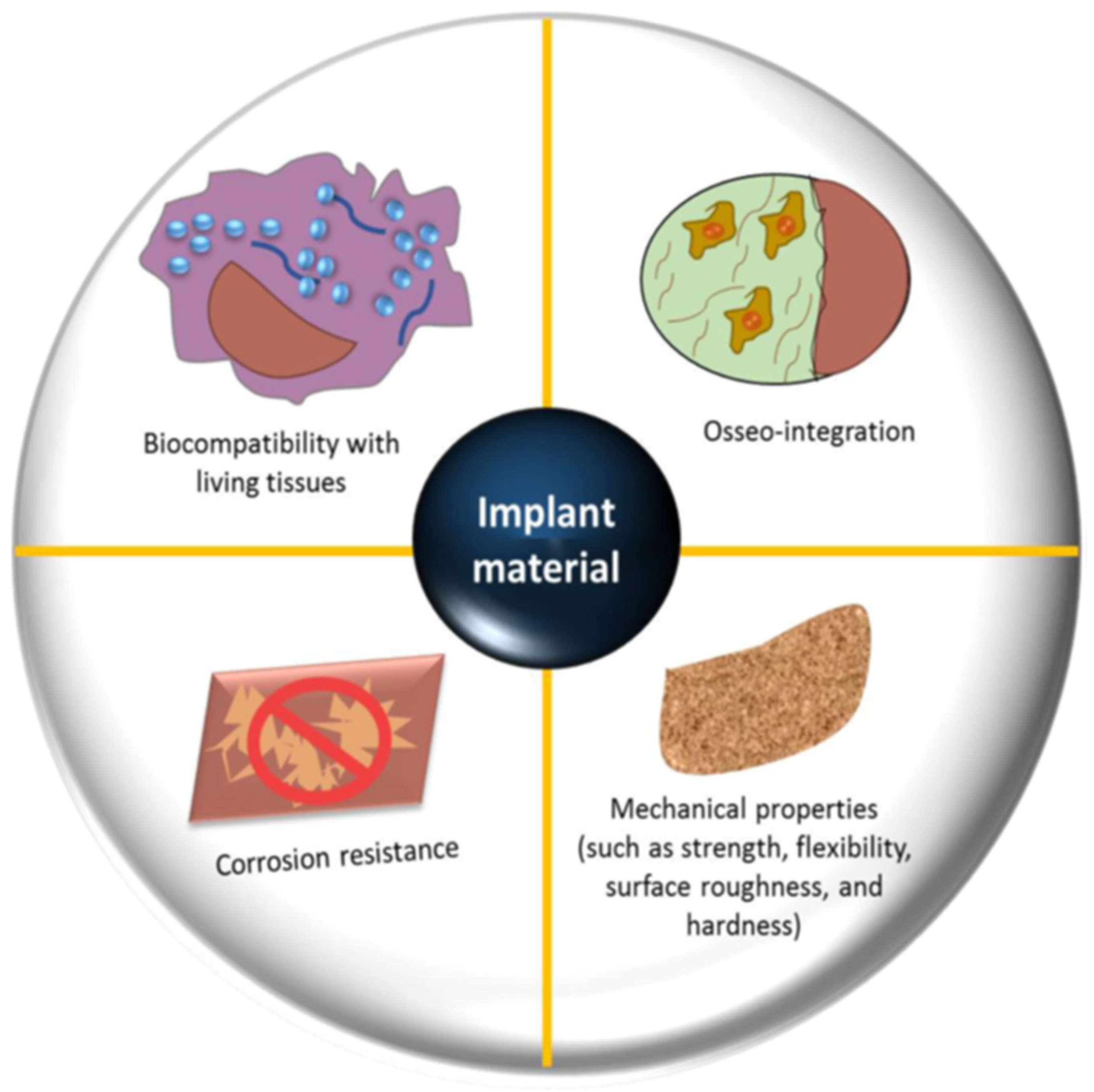
2. Biomaterials in Orthopedics
2.1. Metals and Alloys
2.1.1. Non-Biodegradable Metals
Stainless Steel and Its Alloys
Co–Cr Alloys
Titanium (Ti) and Its Alloys
2.1.2. Biodegradable Metals
Magnesium and Its Alloys
Fe and Its Alloys
Zinc and Its Alloys
2.1.3. Nanocrystalline Metallic Materials
2.2. Polymers
2.2.1. Natural Polymer
2.2.2. Synthetic Polymer
Non-Biodegradable Synthetic Polymer
- i.
- Poly (Methyl Methacrylate) (PMMA)
- ii.
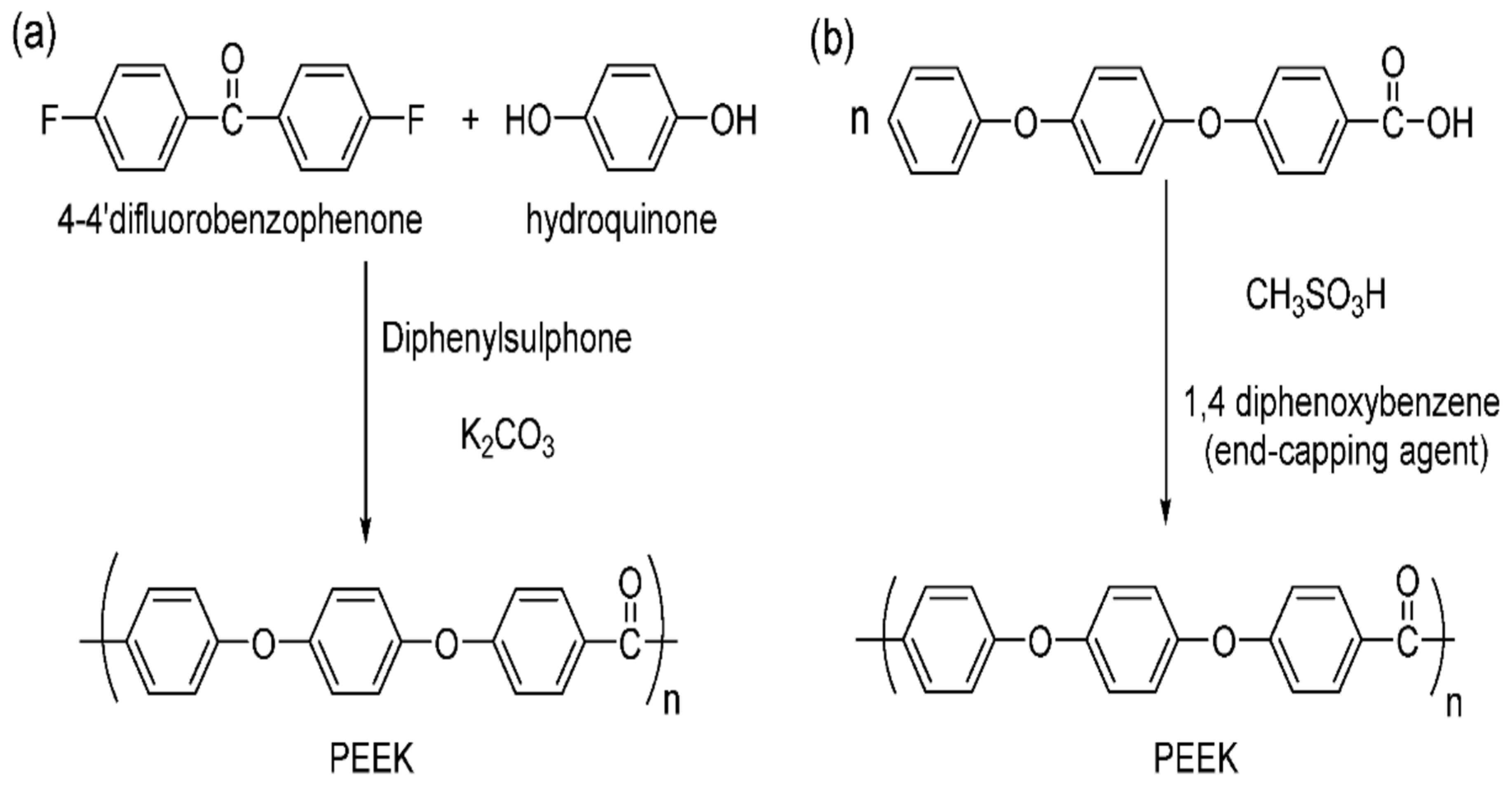
- Polyetheretherketone (PEEK)
- iii.
- Polythene (PE)
Biodegradable Synthetic Polymer
- i.
- Poly (Glycolic Acid) (PGA)
- ii.
- Poly (Lactic Acid) (PLA)
- iii.
- Poly (Lactide-Co-Glycolide)
- iv.
- Poly (Caprolactone)
3. Enhanced Bone–Implant Biocompatibility due to Osteogenic Factors
4. Additive Manufacturing (AM)
4.1. Additive Manufacturing of Metallic Implants
4.2. Additive Manufacturing of Polymeric Implants
5. Future Direction
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marin, E.; Boschetto, F.; Pezzotti, G. Biomaterials and biocompatibility: An historical overview. J. Biomed. Mater. Res. A 2020, 108, 1617–1633. [Google Scholar] [CrossRef]
- Al-Shalawi, F.D.; Hanim, M.A.; Ariffin, M.; Kim, C.L.S.; Brabazon, D.; Calin, R.; Al-Osaimi, M.O. Biodegradable synthetic polymer in orthopaedic application: A review. Mater. Today Proc. 2023, 74, 540–546. [Google Scholar] [CrossRef]
- Yousef, J.; Leow, S.K.H.; Morrison, W. Plastic surgery in antiquity: An examination of ancient documents. Eur. J. Plast. Surg. 2021, 44, 419–428. [Google Scholar] [CrossRef]
- Shahar, F.S.; Sultan, M.T.H.; Lee, S.H.; Jawaid, M.; Shah, A.U.M.; Safri, S.N.A.; Sivasankaran, P.N. A review on the orthotics and prosthetics and the potential of kenaf composites as alternative materials for ankle-foot orthosis. J. Mech. Behav. Biomed. Mater. 2019, 99, 169–185. [Google Scholar] [CrossRef]
- Eliaz, N. Corrosion of metallic biomaterials: A review. Materials 2019, 12, 407. [Google Scholar] [CrossRef] [PubMed]
- Bharadwaj, A. An Overview on Biomaterials and Its Applications in Medical Science. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1116, 012178. [Google Scholar] [CrossRef]
- Ali, S.; Rani, A.M.A.; Baig, Z.; Ahmed, S.W.; Hussain, G.; Subramaniam, K.; Hastuty, S.; Rao, T.V. Biocompatibility and corrosion resistance of metallic biomaterials. Corros. Rev. 2020, 38, 381–402. [Google Scholar] [CrossRef]
- Im, G.I. Biomaterials in orthopaedics: The past and future with immune modulation. Biomater. Res. 2020, 24, 7–10. [Google Scholar] [CrossRef]
- Williams, D.F. Challenges with the Development of Biomaterials for Sustainable Tissue Engineering. Front. Bioeng. Biotechnol. 2019, 7, 127. [Google Scholar] [CrossRef]
- Manivasagam, G.; Dhinasekaran, D.; Rajamanickam, A. Biomedical Implants: Corrosion and its Prevention—A Review. Recent Pat. Corros. Sci. 2010, 2, 40–54. [Google Scholar] [CrossRef]
- Winkler, T.; Sass, F.A.; Duda, G.N.; Schmidt-Bleek, K. A review of biomaterials in bone defect healing, remaining shortcomings and future opportunities for bone tissue engineering: The unsolved challenge. Bone Jt. Res. 2018, 7, 232–243. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi-Mobarakeh, L.; Kolahreez, D.; Ramakrishna, S.; Williams, D. Key terminology in biomaterials and biocompatibility. Curr. Opin. Biomed. Eng. 2019, 10, 45–50. [Google Scholar] [CrossRef]
- Thomas, S.; Balakrishnan, P.; Sreekala, M.S. Fundamental Biomaterials: Ceramics; Woodhead Publishing: Sawston, UK, 2018. [Google Scholar]
- Bairagi, D.; Mandal, S. A comprehensive review on biocompatible Mg-based alloys as temporary orthopaedic implants: Current status, challenges, and future prospects. J. Magnes. Alloy. 2022, 10, 627–669. [Google Scholar] [CrossRef]
- Seal, C.K.; Vince, K.; Hodgson, M.A. Biodegradable surgical implants based on magnesium alloys—A review of current research. IOP Conf. Ser. Mater. Sci. Eng. 2009, 4, 012011. [Google Scholar] [CrossRef]
- Sumner, D.R. Long-term implant fixation and stress-shielding in total hip replacement. J. Biomech. 2015, 48, 797–800. [Google Scholar] [CrossRef]
- Hermawan, H.; Dubé, D.; Mantovani, D. Developments in metallic biodegradable stents. Acta Biomater. 2010, 6, 1693–1697. [Google Scholar] [CrossRef] [PubMed]
- Hermawan, H.; Dubé, D.; Mantovani, D. Degradable metallic biomaterials: Design and development of Fe–Mn alloys for stents. J. Biomed. Mater. Res. Part A Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. Korean Soc. Biomater. 2010, 93, 1–11. [Google Scholar] [CrossRef]
- Mani, G.; Feldman, M.D.; Patel, D.; Agrawal, C.M. Coronary stents: A materials perspective. Biomaterials 2007, 28, 1689–1710. [Google Scholar] [CrossRef]
- Alizadeh-Osgouei, M.; Li, Y.; Wen, C. A comprehensive review of biodegradable synthetic polymer-ceramic composites and their manufacture for biomedical applications. Bioact. Mater. 2019, 4, 22–36. [Google Scholar] [CrossRef]
- Acklin, Y.P.; Michelitsch, C.; Sommer, C. Elective implant removal in symptomatic patients after internal fixation of proximal humerus fractures improves clinical outcome. BMC Musculoskelet. Disord. 2016, 17, 119. [Google Scholar] [CrossRef]
- Matusiewicz, H.; Richter, M. Local release of metal ions from endovascular metallic implants in the human biological specimens: An overview of in vivo clinical implications. World J. Adv. Res. Rev. 2021, 11, 91–102. [Google Scholar] [CrossRef]
- Chin, P.Y.Y.; Cheok, Q.; Glowacz, A.; Caesarendra, W. A review of in-vivo and in-vitro real-time corrosion monitoring systems of biodegradable metal implants. Appl. Sci. 2020, 10, 3141. [Google Scholar] [CrossRef]
- Matusiewicz, H.; Richter, M. Potentially toxic metallic wear nanoparticles and trace metal ions release from metal-on-metal orthopedic implants in the human biological specimens: An Overview of in vivo and ex vivo clinical studies. World J. Adv. Res. Rev. 2020, 8, 242–292. [Google Scholar] [CrossRef]
- Gómez-Gil, V.; Pascual, G.; Bellón, J.M. Biomaterial implants in abdominal wall Hernia Repair: A review on the importance of the peritoneal interface. Processes 2019, 7, 105. [Google Scholar] [CrossRef]
- Hudecki, A.; Kiryczyński, G.; Łos, M.J. Biomaterials, definition, overview. Stem Cells Biomater. Regen. Med. 2018, 7, 85–98. [Google Scholar] [CrossRef]
- Munir, K.S.; Wen, C.; Li, Y. Carbon Nanotubes and Graphene as Nanoreinforcements in Metallic Biomaterials: A Review. Adv. Biosyst. 2019, 3, 1800212. [Google Scholar] [CrossRef]
- Yang, L. Nanotechnology-Enhanced Orthopedic Materials: Fabrications, Applications and Future Trends; Woodhead Publishing: Sawston, UK, 2015. [Google Scholar]
- Hulbert, S.F. The use of alumina and zirconia in surgical implants. In An Introduction to Bioceramics; World Scientific: Singapore, 1993; pp. 25–40. [Google Scholar]
- Kumar, S.; Nehra, M.; Kedia, D.; Dilbaghi, N.; Tankeshwar, K.; Kim, K.H. Nanotechnology-based biomaterials for orthopaedic applications: Recent advances and future prospects. Mater. Sci. Eng. C 2020, 106, 110154. [Google Scholar] [CrossRef] [PubMed]
- Bommala, V.K.; Krishna, M.G.; Rao, C.T. Magnesium matrix composites for biomedical applications: A review. J. Magnes. Alloy. 2019, 7, 72–79. [Google Scholar] [CrossRef]
- Pajarinen, J.; Lin, T.; Gibon, E.; Kohno, Y.; Maruyama, M.; Nathan, K.; Lu, L.; Yao, Z.; Goodman, S.B. Mesenchymal stem cell-macrophage crosstalk and bone healing. Biomaterials 2019, 196, 80–89. [Google Scholar] [CrossRef]
- Chen, M.Q. Recent Advances and Perspective of Nanotechnology-Based Implants for Orthopedic Applications. Front. Bioeng. Biotechnol. 2022, 10, 878257. [Google Scholar] [CrossRef]
- Li, J.W.; Du, C.F.; Yuchi, C.X.; Zhang, C.Q. Application of Biodegradable Materials in Orthopedics. J. Med. Biol. Eng. 2019, 39, 633–645. [Google Scholar] [CrossRef]
- Pfeiffenberger, M.; Damerau, A.; Lang, A.; Buttgereit, F.; Hoff, P.; Gaber, T. Fracture Healing Research-Shift towards In Vitro Modeling? Biomedicines 2021, 9, 748. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, S.; Chen, W.; Hu, Y.; Geng, Z.; Su, J. Bone/cartilage targeted hydrogel: Strategies and applications. Bioact. Mater. 2023, 23, 156–169. [Google Scholar] [CrossRef]
- Jin, W.; Chu, P.K. Orthopedic implants. Encycl. Biomed. Eng. 2019, 1, 425–436. [Google Scholar] [CrossRef]
- Tian, L.; Tang, N.; Ngai, T.; Wu, C.; Ruan, Y.C.; Huang, L.; Qin, L. Hybrid fracture fixation systems developed for orthopaedic applications: A general review. J. Orthop. Transl. 2019, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bartolomeu, F.; Dourado, N.; Pereira, F.; Alves, N.; Miranda, G.; Silva, F.S. Additive manufactured porous biomaterials targeting orthopedic implants: A suitable combination of mechanical, physical and topological properties. Mater. Sci. Eng. C 2020, 107, 110342. [Google Scholar] [CrossRef]
- Dhandapani, R.; Krishnan, P.D.; Zennifer, A.; Kannan, V.; Manigandan, A.; Arul, M.R.; Jaiswal, D.; Subramanian, A.; Kumbar, S.G.; Sethuraman, S. Additive manufacturing of biodegradable porous orthopaedic screw. Bioact. Mater. 2020, 5, 458–467. [Google Scholar] [CrossRef]
- Wang, J.L.; Xu, J.K.; Hopkins, C.; Chow, D.H.K.; Qin, L. Biodegradable Magnesium-Based Implants in Orthopedics—A General Review and Perspectives. Adv. Sci. 2020, 7, 1902443. [Google Scholar] [CrossRef]
- Huzum, B.; Puha, B.; Necoara, R.M.; Gheorghevici, S.; Puha, G.; Filip, A.; Sirbu, P.D.; Alexa, O. Biocompatibility assessment of biomaterials used in orthopedic devices: An overview (Review). Exp. Ther. Med. 2021, 22, 1315. [Google Scholar] [CrossRef]
- Cvrček, L.; Horáková, M. Plasma modified polymeric materials for implant applications. In Non-Thermal Plasma Technology for Polymeric Materials; Elsevier: Amsterdam, The Netherlands, 2019; pp. 367–407. [Google Scholar]
- Filip, N.; Radu, I.; Veliceasa, B.; Filip, C.; Pertea, M.; Clim, A.; Pinzariu, A.C.; Drochioi, I.C.; Hilitanu, R.L.; Serban, I.L. Biomaterials in Orthopedic Devices: Current Issues and Future Perspectives. Coatings 2022, 12, 1544. [Google Scholar] [CrossRef]
- Silver, F.H.; Shah, R. Measurement of mechanical properties of natural and engineered implants. Adv. Tissue Eng. Regen. Med. 2016, 1, 20–25. [Google Scholar] [CrossRef]
- Xiao, D.; Zhang, J.; Zhang, C.; Barbieri, D.; Yuan, H.; Moroni, L.; Feng, G. The role of calcium phosphate surface structure in osteogenesis and the mechanisms involved. Acta Biomater. 2020, 106, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Wooley, P.H.; Hallab, N.J. Wound healing, chronic inflammation, and immune responses. Met. Met. Bear. A Clin. Pract. 2014, 109–133. [Google Scholar]
- Nouri, A.; Shirvan, A.R.; Li, Y.; Wen, C. Additive manufacturing of metallic and polymeric load-bearing biomaterials using laser powder bed fusion: A review. J. Mater. Sci. Technol. 2021, 94, 196–215. [Google Scholar] [CrossRef]
- Mourey, T.H.; Schunk, T.C. Synthetic Polymers; Elsevier Inc.: Amsterdam, The Netherlands, 1992. [Google Scholar] [CrossRef]
- Al-Amin, M.; Abdul-Rani, A.M.; Danish, M.; Rubaiee, S.; Mahfouz, A.B.; Thompson, H.M.; Ali, S.; Unune, D.R.; Sulaiman, M.H. Investigation of coatings, corrosion and wear characteristics of machined biomaterials through hydroxyapatite mixed-edm process: A review. Materials 2021, 14, 3597. [Google Scholar] [CrossRef]
- Majumdar, T.; Eisenstein, N.; Frith, J.E.; Cox, S.C.; Birbilis, N. Additive Manufacturing of Titanium Alloys for Orthopedic Applications: A Materials Science Viewpoint. Adv. Eng. Mater. 2018, 20, 1800172. [Google Scholar] [CrossRef]
- Bai, L.; Gong, C.; Chen, X.; Sun, Y.; Zhang, J.; Cai, L.; Zhu, S.; Xie, S.Q. Additive manufacturing of customized metallic orthopedic implants: Materials, structures, and surface modifications. Metals 2019, 9, 1004. [Google Scholar] [CrossRef]
- Al-Amin, M.; Rani, A.M.A.; Aliyu, A.A.A.; Razak, M.A.A.; Hastuty, S.; Bryant, M.G. Powder mixed-EDM for potential biomedical applications: A critical review. Mater. Manuf. Process. 2020, 35, 1789–1811. [Google Scholar] [CrossRef]
- Material Properties of UHMW Polyethylene—Thermoplastic. 25 July 2022. Available online: https://dielectricmfg.com/knowledge-base/uhmw/ (accessed on 21 February 2023).
- Qu, S.; Liu, Y.; Gong, K. Drug-Loaded UHMWPE to Inhibit Wear Particle-Induced Osteolysis: Processing, Characterization, and Biological Evaluation. In UHMWPE Biomaterials for Joint Implants: Structures, Properties and Clinical Performance; Springer: Singapore, 2019; pp. 151–190. [Google Scholar]
- Jin, W.; Hao, Q.; Peng, X.; Chu, P.K. Enhanced corrosion resistance and biocompatibilty of PMMA-coated ZK60 magnesium alloy. Mater. Lett. 2016, 173, 178–181. [Google Scholar] [CrossRef]
- Szczęsny, G.; Kopec, M.; Politis, D.J.; Kowalewski, Z.L.; Łazarski, A.; Szolc, T. A Review on Biomaterials for Orthopaedic Surgery and Traumatology: From Past to Present. Materials 2022, 15, 3622. [Google Scholar] [CrossRef]
- Trentin, A.; Gasparini, A.D.L.; Faria, F.A.; Harb, S.V.; dos Santos, F.C.; Pulcinelli, S.H.; Santilli, C.V. Barrier properties of high performance PMMA-silica anticorrosion coatings. Prog. Org. Coat. 2020, 138, 105398. [Google Scholar] [CrossRef]
- Harb, S.V.; Trentin, A.; de Souza, T.A.C.; Magnani, M.; Pulcinelli, S.H.; Santilli, C.V.; Hammer, P. Effective corrosion protection by eco-friendly self-healing PMMA-cerium oxide coatings. Chem. Eng. J. 2020, 383, 123219. [Google Scholar] [CrossRef]
- Harb, S.V.; Bassous, N.J.; de Souza, T.A.; Trentin, A.; Pulcinelli, S.H.; Santilli, C.V.; Webster, T.J.; Lobo, A.O.; Hammer, P. Hydroxyapatite and β-TCP modified PMMA-TiO2 and PMMA-ZrO2 coatings for bioactive corrosion protection of Ti6Al4V implants. Mater. Sci. Eng. C 2020, 116, 111149. [Google Scholar] [CrossRef] [PubMed]
- Material Properties of Thermoplastic PEEK—Polyetheretherketone. 25 July 2022. Available online: https://dielectricmfg.com/knowledge-base/peek/ (accessed on 21 February 2023).
- Zhao, Y.; Zhao, K.; Li, Y.; Chen, F. Mechanical characterization of biocompatible PEEK by FDM. J. Manuf. Process. 2020, 56, 28–42. [Google Scholar] [CrossRef]
- Ma, H.; Suonan, A.; Zhou, J.; Yuan, Q.; Liu, L.; Zhao, X.; Lou, X.; Yang, C.; Li, D.; Zhang, Y.-G. PEEK (Polyether-ether-ketone) and its composite materials in orthopedic implantation. Arab. J. Chem. 2021, 14, 102977. [Google Scholar] [CrossRef]
- Wang, J.; Sun, Y.; Bi, W.; Jiang, Z.; Zhang, M.; Pang, J. High-strength corrosion resistant membranes for the separation of oil/water mixtures and immiscible oil mixtures based on PEEK. J. Memb. Sci. 2020, 616, 118418. [Google Scholar] [CrossRef]
- Travieso-Rodriguez, J.A.; Jerez-Mesa, R.; Llumà, J.; Traver-Ramos, O.; Gomez-Gras, G.; Rovira, J.J.R. Mechanical Properties of 3D-Printing Polylactic Acid Parts subjected to Bending Stress and Fatigue Testing. Materials 2019, 12, 3859. [Google Scholar] [CrossRef]
- Mozafari, M.; Bordbar-Khiabani, A.; Yarmand, B. Emerging magnesium-based biomaterials for orthopedic implantation. Emerg. Mater. Res. 2020, 8, 305–319. [Google Scholar] [CrossRef]
- Li, J.; Qin, L.; Yang, K.; Ma, Z.; Wang, Y.; Cheng, L.; Zhao, D. Materials evolution of bone plates for internal fixation of bone fractures: A review. J. Mater. Sci. Technol. 2020, 36, 190–208. [Google Scholar] [CrossRef]
- Balestriere, M.A.; Schuhladen, K.; Seitz, K.H.; Boccaccini, A.R.; Cere, S.M.; Ballarre, J. Sol-gel coatings incorporating borosilicate bioactive glass enhance anti corrosive and surface performance of stainless steel implants. J. Electroanal. Chem. 2020, 876, 114735. [Google Scholar] [CrossRef]
- Gawad, S.A.; Nasr, A.; Fekry, A.M.; Filippov, L.O. Electrochemical and hydrogen evolution behaviour of a novel nano-cobalt/nano-chitosan composite coating on a surgical 316L stainless steel alloy as an implant. Int. J. Hydrog. Energy 2021, 46, 18233–18241. [Google Scholar] [CrossRef]
- Komatsu, A.; Fujinami, M.; Hatano, M.; Matsumoto, K.; Sugeoi, M.; Chiari, L. Straining-temperature dependence of vacancy behavior in hydrogen-charged austenitic stainless steel 316L. Int. J. Hydrog. Energy 2021, 46, 6960–6969. [Google Scholar] [CrossRef]
- Kwak, T.; Yang, J.; Heo, Y.; Kim, S.; Kwon, S.; Kim, W.; Lim, D. Additive manufacturing of a porous titanium layer structure Ti on a Co-Cr alloy for manufacturing cementless implants. J. Mater. Res. Technol. 2021, 10, 250–267. [Google Scholar] [CrossRef]
- Xiang, D.; Wang, P.; Tan, X.; Chandra, S.; Wang, C.; Nai, M.; Tor, S.; Liu, W.; Liu, E. Anisotropic microstructure and mechanical properties of additively manufactured Co–Cr–Mo alloy using selective electron beam melting for orthopedic implants. Mater. Sci. Eng. A 2019, 765, 138270. [Google Scholar] [CrossRef]
- Duan, J.; Yang, Y.; Zhang, E.; Wang, H. Co-Cr-Mo-Cu alloys for clinical implants with osteogenic effect by increasing bone induction, formation and development in a rabbit model. Burn. Trauma 2020, 8, tkaa036. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Li, Y.; Nie, Y.; Huang, Z.; Liu, F.; Huang, L.; Jiang, L.; Mei, H. Corrosion behaviour of CoCrMo alloy fabricated by electron beam melting. Corros. Sci. 2018, 139, 68–75. [Google Scholar] [CrossRef]
- He, X.; Reichl, F.-X.; Milz, S.; Michalke, B.; Wu, X.; Sprecher, C.M.; Yang, Y.; Gahlert, M.; Röhling, S.; Kniha, H.; et al. Titanium and zirconium release from titanium- and zirconia implants in mini pig maxillae and their toxicity in vitro. Dent. Mater. 2020, 36, 402–412. [Google Scholar] [CrossRef]
- Hourmand, M.; Sarhan, A.A.D.; Sayuti, M.; Hamdi, M. A comprehensive review on machining of titanium alloys. Arab. J. Sci. Eng. 2021, 46, 7087–7123. [Google Scholar] [CrossRef]
- Ren, B.; Wan, Y.; Liu, C.; Wang, H.; Yu, M.; Zhang, X.; Huang, Y. Improved osseointegration of 3D printed Ti-6Al-4V implant with a hierarchical micro/nano surface topography: An in vitro and in vivo study. Mater. Sci. Eng. C 2021, 118, 111505. [Google Scholar] [CrossRef]
- Tshephe, T.S.; Akinwamide, S.O.; Olevsky, E.; Olubambi, P.A. Additive manufacturing of titanium-based alloys- A review of methods, properties, challenges, and prospects. Heliyon 2022, 8, e09041. [Google Scholar] [CrossRef]
- Liang, L.; Huang, Q.; Wu, H.; Ouyang, Z.; Liu, T.; He, H.; Xiao, J.; Lei, G.; Zhou, K. Stimulation of in vitro and in vivo osteogenesis by Ti-Mg composite materials with the sustained-release function of magnesium ions. Colloids Surf. B Biointerfaces 2021, 197, 111360. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, J.M.; Nagay, B.E.; Ribeiro, A.L.R.; da Cruz, N.C.; Rangel, E.C.; Fais, L.M.; Vaz, L.G.; Barão, V.A. Functionalization of an experimental Ti-Nb-Zr-Ta alloy with a biomimetic coating produced by plasma electrolytic oxidation. J. Alloys Compd. 2019, 770, 1038–1048. [Google Scholar] [CrossRef]
- Zwolak, I. Protective Effects of Dietary Antioxidants against Vanadium-Induced Toxicity: A Review. Oxid. Med. Cell Longev. 2020, 2020, 1490316. [Google Scholar] [CrossRef] [PubMed]
- Engwa, G.A.; Ferdinand, P.U.; Nwalo, F.N.; Unachukwu, M.N. Mechanism and health effects of heavy metal toxicity in humans. In Poisoning in the Modern World-New Tricks for an Old Dog? IntechOpen: London, UK, 2019; Volume 5, pp. 77–100. [Google Scholar]
- Verma, R.P. Titanium based biomaterial for bone implants: A mini review. Mater. Today Proc. 2020, 26, 3148–3151. [Google Scholar] [CrossRef]
- Çaha, I.; Alves, A.C.; Rocha, L.A.; Toptan, F. A Review on Bio-functionalization of β-Ti Alloys. J. Bio-Tribo-Corros. 2020, 6, 135. [Google Scholar] [CrossRef]
- Hussein, M.A.; Mohammed, A.S.; Al-Aqeeli, N. Wear Characteristics of Metallic Biomaterials: A Review. Materials 2015, 8, 2749–2768. [Google Scholar] [CrossRef]
- Shuai, C.; Li, S.; Peng, S.; Feng, P.; Lai, Y.; Gao, C. Biodegradable metallic bone implants. Mater. Chem. Front. 2019, 3, 544–562. [Google Scholar] [CrossRef]
- Kraus, T.; Moszner, F.; Fischerauer, S.; Fiedler, M.; Martinelli, E.; Eichler, J.; Witte, F.; Willbold, E.; Schinhammer, M.; Meischel, M.; et al. Biodegradable Fe-based alloys for use in osteosynthesis: Outcome of an in vivo study after 52 weeks. Acta Biomater. 2014, 10, 3346–3353. [Google Scholar] [CrossRef]
- Ulum, M.F.; Nasution, A.K.; Yusop, A.H.; Arafat, A.; Kadir, M.R.A.; Juniantito, V.; Noviana, D.; Hermawan, H. Evidences of in vivo bioactivity of Fe-bioceramic composites for temporary bone implants. J. Biomed. Mater. Res. B Appl. Biomater. 2015, 103, 1354–1365. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, M.; He, C.; Qi, F.; Wang, D.; Peng, S.; Shuai, C. Rare earth improves strength and creep resistance of additively manufactured Zn implants. Compos. B Eng. 2021, 216, 108882. [Google Scholar] [CrossRef]
- Yang, Y.; Cheng, Y.; Peng, S.; Xu, L.; He, C.; Qi, F.; Zhao, M.; Shuai, C. Microstructure evolution and texture tailoring of reduced graphene oxide reinforced Zn scaffold. Bioact. Mater. 2021, 6, 1230–1241. [Google Scholar] [CrossRef]
- Li, H.F.; Shi, Z.Z.; Wang, L.N. Opportunities and challenges of biodegradable Zn-based alloys. J. Mater. Sci. Technol. 2020, 46, 136–138. [Google Scholar] [CrossRef]
- Yang, Y.; Cheng, Y.; Yang, M.; Qian, G.; Peng, S.; Qi, F.; Shuai, C. Semicoherent strengthens graphene/zinc scaffolds. Mater. Today Nano. 2022, 17, 100163. [Google Scholar] [CrossRef]
- Liu, L.; Ma, H.; Gao, C.; Shuai, C.; Peng, S. Island-to-acicular alteration of second phase enhances the degradation resistance of biomedical AZ61 alloy. J. Alloys Compd. 2020, 835, 155397. [Google Scholar] [CrossRef]
- Kang, M.-H.; Lee, H.; Jang, T.-S.; Seong, Y.-J.; Kim, H.-E.; Koh, Y.-H.; Song, J.; Jung, H.-D. Biomimetic porous Mg with tunable mechanical properties and biodegradation rates for bone regeneration. Acta Biomater. 2019, 84, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Cockerill, I.; Su, Y.; Zhang, Z.; Fu, J.; Lee, K.-W.; Ma, J.; Okpokwasili, C.; Tang, L.; Zheng, Y.; et al. Mechanical strength, biodegradation, and in vitro and in vivo biocompatibility of Zn biomaterials. ACS Appl. Mater. Interfaces 2019, 11, 6809–6819. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Mo, Y.; Gao, C.; Shuai, C.; Peng, S. A dual redox system for enhancing the biodegradability of Fe-C-Cu composite scaffold. Colloids Surf. B Biointerfaces 2022, 213, 112431. [Google Scholar] [CrossRef]
- Prokoshkin, S.; Pustov, Y.; Zhukova, Y.; Kadirov, P.; Dubinskiy, S.; Sheremetyev, V.; Karavaeva, M. Effect of Thermomechanical Treatment on Functional Properties of Biodegradable Fe-30Mn-5Si Shape Memory Alloy. Met. Mater. Trans. A Phys. Met. Mater. Sci. 2021, 52, 2024–2032. [Google Scholar] [CrossRef]
- Banerjee, P.C.; Al-Saadi, S.; Choudhary, L.; Harandi, S.E.; Singh, R. Magnesium implants: Prospects and challenges. Materials 2019, 12, 136. [Google Scholar] [CrossRef] [PubMed]
- Kamrani, S.; Fleck, C. Biodegradable magnesium alloys as temporary orthopaedic implants: A review. BioMetals 2019, 32, 185–193. [Google Scholar] [CrossRef]
- Tsakiris, V.; Tardei, C.; Clicinschi, F.M. Biodegradable Mg alloys for orthopedic implants—A review. J. Magnes. Alloy 2021, 9, 1884–1905. [Google Scholar] [CrossRef]
- Gao, X.; Dai, C.Y.; Jia, Q.; Zhai, C.; Shi, H.; Yang, Y.; Zhao, B.C.; Cai, H.; Lee, E.-S.; Jiang, H.B. In vivo corrosion behavior of biodegradable magnesium alloy by MAF treatment. Scanning 2021, 2021, 5530788. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ren, J.; Wang, X.; Ding, Y.; Li, P.; Hu, Y.; Yang, Y. Dilemmas and countermeasures of Fe-based biomaterials for next-generation bone implants. J. Mater. Res. Technol. 2022, 20, 2034–2050. [Google Scholar] [CrossRef]
- Xu, C.; Yu, S.; Wu, W.; Liu, Q.; Ren, L. Direct ink writing of Fe bone implants with independently adjustable structural porosity and mechanical properties. Addit. Manuf. 2022, 51, 102589. [Google Scholar] [CrossRef]
- Heiden, M.; Walker, E.; Stanciu, L. Magnesium, iron and zinc alloys, the trifecta of bioresorbable orthopaedic and vascular implantation-a review. J. Biotechnol. Biomater. 2015, 5, 1. [Google Scholar]
- Purnama, A.; Hermawan, H.; Couet, J.; Mantovani, D. Assessing the biocompatibility of degradable metallic materials: State-of-the-art and focus on the potential of genetic regulation. Acta Biomater. 2010, 6, 1800–1807. [Google Scholar] [CrossRef] [PubMed]
- Hermawan, H. Biodegradable metals: State of the art. In Biodegradable Metals; Springer: Berlin/Heidelberg, Germany, 2012; pp. 13–22. [Google Scholar]
- Wegener, B.; Sievers, B.; Utzschneider, S.; Müller, P.; Jansson, V.; Rößler, S.; Nies, B.; Stephani, G.; Kieback, B.; Quadbeck, P. Microstructure, cytotoxicity and corrosion of powder-metallurgical iron alloys for biodegradable bone replacement materials. Mater. Sci. Eng. B 2011, 176, 1789–1796. [Google Scholar] [CrossRef]
- Gorejová, R.; Haverová, L.; Oriňaková, R.; Oriňak, A.; Oriňak, M. Recent advancements in Fe-based biodegradable materials for bone repair. J. Mater. Sci. 2019, 54, 1913–1947. [Google Scholar] [CrossRef]
- Liu, B.; Zheng, Y.F.; Ruan, L. In vitro investigation of Fe30Mn6Si shape memory alloy as potential biodegradable metallic material. Mater. Lett. 2011, 65, 540–543. [Google Scholar] [CrossRef]
- Vojtech, D.; Kubasek, J.; Capek, J.; Pospisilova, I. Comparative mechanical and corrosion studies on magnesium, zinc and iron alloys as biodegradable metals. Mater. Tehnol. 2015, 49, 877–882. [Google Scholar] [CrossRef]
- Cheng, J.; Liu, B.; Wu, Y.H.; Zheng, Y.F. Comparative in vitro study on pure metals (Fe, Mn, Mg, Zn and W) as biodegradable metals. J. Mater Sci. Technol. 2013, 29, 619–627. [Google Scholar] [CrossRef]
- Zhang, E.; Chen, H.; Shen, F. Biocorrosion properties and blood and cell compatibility of pure iron as a biodegradable biomaterial. J. Mater. Sci. Mater. Med. 2010, 21, 2151–2163. [Google Scholar] [CrossRef] [PubMed]
- Jia, P.; Wang, Z.; Zhang, Y.; Zhang, D.; Gao, W.; Su, Y.; Li, Y.; Yang, C. Selective sensing of Fe3+ ions in aqueous solution by a biodegradable platform based lanthanide metal organic framework. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 230, 118084. [Google Scholar] [CrossRef] [PubMed]
- Kabir, H.; Munir, K.; Wen, C.; Li, Y. Recent research and progress of biodegradable zinc alloys and composites for biomedical applications: Biomechanical and biocorrosion perspectives. Bioact. Mater. 2021, 6, 836–879. [Google Scholar] [CrossRef] [PubMed]
- Ulum, M.; Arafat, A.; Noviana, D.; Yusop, A.; Nasution, A.; Kadir, M.A.; Hermawan, H. In vitro and in vivo degradation evaluation of novel iron-bioceramic composites for bone implant applications. Mater. Sci. Eng. C 2014, 36, 336–344. [Google Scholar] [CrossRef]
- Carluccio, D.; Xu, C.; Venezuela, J.; Cao, Y.; Kent, D.; Bermingham, M.; Demir, A.G.; Previtali, B.; Ye, Q.; Dargusch, M. Additively manufactured iron-manganese for biodegradable porous load-bearing bone scaffold applications. Acta Biomater. 2020, 103, 346–360. [Google Scholar] [CrossRef]
- Schinhammer, M.; Hänzi, A.C.; Löffler, J.F.; Uggowitzer, P.J. Design strategy for biodegradable Fe-based alloys for medical applications. Acta Biomater. 2010, 6, 1705–1713. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zheng, Y.F. Effects of alloying elements (Mn, Co, Al, W, Sn, B, C and S) on biodegradability and in vitro biocompatibility of pure iron. Acta Biomater. 2011, 7, 1407–1420. [Google Scholar] [CrossRef]
- Sezer, N.; Evis, Z.; Kayhan, S.M.; Tahmasebifar, A.; Koç, M. Review of magnesium-based biomaterials and their applications. J. Magnes. Alloy 2018, 6, 23–43. [Google Scholar] [CrossRef]
- Wang, S.; Xu, Y.; Zhou, J.; Li, H.; Chang, J.; Huan, Z. In vitro degradation and surface bioactivity of iron-matrix composites containing silicate-based bioceramic. Bioact. Mater. 2017, 2, 10–18. [Google Scholar] [CrossRef]
- Feng, Q.; Zhang, D.; Xin, C.; Liu, X.; Lin, W.; Zhang, W.; Chen, S.; Sun, K. Characterization and in vivo evaluation of a bio-corrodible nitrided iron stent. J. Mater. Sci. Mater. Med. 2013, 24, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Xia, D.; Wu, S.; Zheng, Y.; Guan, Z.; Rau, J.V. A review on current research status of the surface modification of Zn-based biodegradable metals. Bioact. Mater. 2022, 7, 192–216. [Google Scholar] [CrossRef]
- Pierson, D.; Edick, J.; Tauscher, A.; Pokorney, E.; Bowen, P.; Gelbaugh, J.; Stinson, J.; Getty, H.; Lee, C.H.; Drelich, J.; et al. A simplified in vivo approach for evaluating the bioabsorbable behavior of candidate stent materials. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Dunne, C.F.; Levy, G.K.; Hakimi, O.; Aghion, E.; Twomey, B.; Stanton, K.T. Corrosion behaviour of biodegradable magnesium alloys with hydroxyapatite coatings. Surf. Coat. Technol. 2016, 289, 37–44. [Google Scholar] [CrossRef]
- Levy, G.; Aghion, E. Effect of diffusion coating of Nd on the corrosion resistance of biodegradable Mg implants in simulated physiological electrolyte. Acta Biomater. 2013, 9, 8624–8630. [Google Scholar] [CrossRef] [PubMed]
- Song, G. Control of biodegradation of biocompatable magnesium alloys. Corros. Sci. 2007, 49, 1696–1701. [Google Scholar] [CrossRef]
- Aghion, E.; Levy, G. The effect of Ca on the in vitro corrosion performance of biodegradable Mg-Nd-Y-Zr alloy. J. Mater. Sci. 2010, 45, 3096–3101. [Google Scholar] [CrossRef]
- Hennig, B.; Toborek, M.; Mcclain, C.J. Antiatherogenic Properties of Zinc: Implications in Endothelial Cell Metabolism. Nutrition 1996, 12, 711–717. [Google Scholar] [CrossRef]
- Plum, L.M.; Rink, L.; Hajo, H. The essential toxin: Impact of zinc on human health. Int. J. Environ. Res. Public Health 2010, 7, 1342–1365. [Google Scholar] [CrossRef]
- Hambidge, K.M.; Krebs, N.F. Zinc Deficiency: A Special Challenge. J. Nutr. 2007, 137, 1101–1105. [Google Scholar] [CrossRef]
- Jia, B.; Yang, H.; Han, Y.; Zhang, Z.; Qu, X.; Zhuang, Y.; Wu, Q.; Zheng, Y.; Dai, K. In vitro and in vivo studies of Zn-Mn biodegradable metals designed for orthopedic applications. Acta Biomater. 2020, 108, 358–372. [Google Scholar] [CrossRef] [PubMed]
- Piao, F.; Yokoyama, K.; Ma, N.; Yamauchi, T. Subacute toxic effects of zinc on various tissues and organs of rats. Toxicol. Lett. 2003, 145, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Drelich, A.J.; Bowen, P.K.; LaLonde, L.; Goldman, J.; Drelich, J.W. Importance of oxide film in endovascular biodegradable zinc stents. Surf. Innov. 2016, 4, 133–140. [Google Scholar] [CrossRef]
- Levy, G.K.; Leon, A.; Kafri, A.; Ventura, Y.; Drelich, J.W.; Goldman, J.; Vago, R.; Aghion, E. Evaluation of biodegradable Zn-1%Mg and Zn-1%Mg-0.5%Ca alloys for biomedical applications. J. Mater Sci. Mater. Med. 2017, 28, 174. [Google Scholar] [CrossRef] [PubMed]
- Levy, G.K.; Goldman, J.; Aghion, E. The prospects of zinc as a structural material for biodegradable implants—A review paper. Metals 2017, 7, 402. [Google Scholar] [CrossRef]
- Bakhsheshi-Rad, H.R.; Hamzah, E.; Low, H.T.; Cho, M.H.; Kasiri-Asgarani, M.; Farahany, S.; Mostafa, A.; Medraj, M. Thermal characteristics, mechanical properties, in vitro degradation and cytotoxicity of novel biodegradable Zn-Al-Mg and Zn-Al-Mg-xBi alloys. Acta Metall. Sin. 2017, 30, 201–211. [Google Scholar] [CrossRef]
- Zhao, S.; Seitz, J.-M.; Eifler, R.; Maier, H.J.; Guillory, R.J.; Earley, E.J.; Drelich, A.; Goldman, J.; Drelich, J.W. Zn-Li alloy after extrusion and drawing: Structural, mechanical characterization, and biodegradation in abdominal aorta of rat. Mater. Sci. Eng. C 2017, 76, 301–312. [Google Scholar] [CrossRef]
- Li, Y.T.; Fang, Q.; Zhang, L.I.; Tao, H.W. Spatial Asymmetry and Short-Term Suppression Underlie Direction Selectivity of Synaptic Excitation in the Mouse Visual Cortex. Cereb. Cortex vol. 2018, 28, 2059–2070. [Google Scholar] [CrossRef]
- Bakhsheshi-Rad, H.; Hamzah, E.; Low, H.; Kasiri-Asgarani, M.; Farahany, S.; Akbari, E.; Cho, M. Fabrication of biodegradable Zn-Al-Mg alloy: Mechanical properties, corrosion behavior, cytotoxicity and antibacterial activities. Mater. Sci. Eng. C 2017, 73, 215–219. [Google Scholar] [CrossRef]
- El-Khair, M.T.A.; Daoud, A.; Ismail, A. Effect of different Al contents on the microstructure, tensile and wear properties of Zn-based alloy. Mater. Lett. 2004, 58, 1754–1760. [Google Scholar] [CrossRef]
- Fernández-Lizárraga, M.; García-López, J.; Rodil, S.E.; Ribas-Aparicio, R.M.; Silva-Bermudez, P. Evaluation of the biocompatibility and osteogenic properties of metal oxide coatings applied by magnetron sputtering as potential biofunctional surface modifications for orthopedic implants. Materials 2022, 15, 5240. [Google Scholar] [CrossRef]
- Sarraf, M.; Nasiri-Tabrizi, B.; Yeong, C.H.; Hosseini, H.R.M.; Saber-Samandari, S.; Basirun, W.J.; Tsuzuki, T. Mixed oxide nanotubes in nanomedicine: A dead-end or a bridge to the future? Ceram. Int. 2021, 47, 2917–2948. [Google Scholar] [CrossRef]
- Tipan, N.; Pandey, A.; Mishra, P. Selection and preparation strategies of Mg-alloys and other biodegradable materials for orthopaedic applications: A review. Mater. Today Commun. 2022, 31, 103658. [Google Scholar] [CrossRef]
- Wang, L.; Ding, X.; Feng, W.; Gao, Y.; Zhao, S.; Fan, Y. Biomechanical study on implantable and interventional medical devices. Acta Mech. Sin. 2021, 37, 875–894. [Google Scholar] [CrossRef]
- Yadav, R.; Meena, A.; Patnaik, A. Biomaterials for dental composite applications: A comprehensive review of physical, chemical, mechanical, thermal, tribological, and biological properties. Polym. Adv. Technol. 2022, 33, 1762–1781. [Google Scholar] [CrossRef]
- Pesode, P.; Barve, S. Surface modification of titanium and titanium alloy by plasma electrolytic oxidation process for biomedical applications: A review. Mater. Today Proc. 2021, 46, 594–602. [Google Scholar] [CrossRef]
- Sanchez, A.G.; Katunar, M.R.; Pastore, J.I.; de la Hoz, M.F.T.; Ceré, S. Evaluation of annealed titanium oxide nanotubes on titanium: From surface characterization to in vivo assays. J. Biomed. Mater. Res. A 2021, 109, 1088–1100. [Google Scholar] [CrossRef] [PubMed]
- Blendinger, F.; Seitz, D.; Ottenschlager, A.; Fleischer, M.; Bucher, V. Atomic layer deposition of bioactive TiO2 thin films on polyetheretherketone for orthopedic implants. ACS Appl. Mater Interfaces 2021, 13, 3536–3546. [Google Scholar] [CrossRef]
- Hazwani, M.R.S.N.; Lim, L.X.; Lockman, Z.; Zuhailawati, H. Fabrication of titanium-based alloys with bioactive surface oxide layer as biomedical implants: Opportunity and challenges. Trans. Nonferrous Met. Soc. China 2022, 32, 1–44. [Google Scholar] [CrossRef]
- Prestat, M.; Thierry, D. Corrosion of titanium under simulated inflammation conditions: Clinical context and in vitro investigations. Acta Biomater. 2021, 136, 72–87. [Google Scholar] [CrossRef] [PubMed]
- Vrchovecká, K.; Weiser, A.; Pribyl, J.; Kuta, J.; Holzer, J.; Pavkova-Goldbergova, M.; Sobola, D.; Dlouhy, A. A release of Ti-ions from nanostructured titanium oxide surfaces. Surf. Interfaces 2022, 29, 101699. [Google Scholar] [CrossRef]
- Xu, A.; Alhamad, M.; Ramachandran, R.A.; Shukla, A.; Barão, V.A.; Sukotjo, C.; Mathew, M.T. Peri-Implantitis in Relation to Titanium Corrosion: Current Status and Future Perspectives. J. Bio-Tribo-Corros. 2022, 8, 46. [Google Scholar] [CrossRef]
- Comino-Garayoa, R.; Brinkmann, J.C.-B.; Peláez, J.; López-Suárez, C.; Martínez-González, J.M.; Suárez, M.J. Allergies to titanium dental implants: What do we really know about them? A scoping review. Biology 2020, 9, 404. [Google Scholar] [CrossRef]
- Ding, Z.; Tang, Y.; Liu, L.; Ding, Z.; Tan, Y.; He, Q. Improving the adhesive, mechanical, tribological properties and corrosion resistance of reactive sputtered tantalum oxide coating on Ti6Al4V alloy via introducing multiple interlayers. Ceram. Int. 2022, 48, 5983–5994. [Google Scholar] [CrossRef]
- Mani, G.; Porter, D.; Grove, K.; Collins, S.; Ornberg, A.; Shulfer, R. A comprehensive review of biological and materials properties of Tantalum and its alloys. J. Biomed. Mater. Res. A 2022, 110, 1291–1306. [Google Scholar] [CrossRef] [PubMed]
- Fomina, M.; Koshuro, V.; Shumilin, A.; Voyko, A.; Zakharevich, A.; Skaptsov, A.; Steinhauer, A.; Fomin, A. Functionally graded ‘Ti-base+(Ta, Ta2O5)-coatings’ structure and its production using induction heat treatment. Compos. Struct. 2020, 234, 111688. [Google Scholar] [CrossRef]
- de Almeida Bino, M.C.; Eurídice, W.A.; Gelamo, R.V.; Leite, N.B.; da Silva, M.V.; de Siervo, A.; Pinto, M.R.; Buranello, P.A.D.A.; Moreto, J.A. Structural and morphological characterization of Ti6Al4V alloy surface functionalization based on Nb2O5 thin film for biomedical applications. Appl. Surf. Sci. 2021, 557, 149739. [Google Scholar] [CrossRef]
- Huang, H.-L.; Tsai, M.-T.; Chang, Y.-Y.; Lin, Y.-J.; Hsu, J.-T. Fabrication of a Novel Ta (Zn) O Thin Film on Titanium by Magnetron Sputtering and Plasma Electrolytic Oxidation for Cell Biocompatibilities and Antibacterial Applications. Metals 2020, 10, 649. [Google Scholar] [CrossRef]
- Horandghadim, N.; Khalil-Allafi, J.; Urgen, M. Effect of Ta2O5 content on the osseointegration and cytotoxicity behaviors in hydroxyapatite-Ta2O5 coatings applied by EPD on superelastic NiTi alloys. Mater. Sci. Eng. C 2019, 102, 683–695. [Google Scholar] [CrossRef]
- Fialho, L.; Grenho, L.; Fernandes, M.H.; Carvalho, S. Porous tantalum oxide with osteoconductive elements and antibacterial core-shell nanoparticles: A new generation of materials for dental implants. Mater. Sci. Eng. C 2021, 120, 111761. [Google Scholar] [CrossRef]
- Alves, C.F.A.; Fialho, L.; Marques, S.; Pires, S.; Rico, P.; Palacio, C.; Carvalho, S. MC3T3-E1 cell response to microporous tantalum oxide surfaces enriched with Ca, P and Mg. Mater. Sci. Eng. C 2021, 124, 112008. [Google Scholar] [CrossRef] [PubMed]
- Peron, M.; Cogo, S.; Bjelland, M.; Bin Afif, A.; Dadlani, A.; Greggio, E.; Berto, F.; Torgersen, J. On the evaluation of ALD TiO2, ZrO2 and HfO2 coatings on corrosion and cytotoxicity performances. J. Magnes. Alloy. 2021, 9, 1806–1819. [Google Scholar] [CrossRef]
- Peron, M.; Bertolini, R.; Cogo, S. On the corrosion, stress corrosion and cytocompatibility performances of ALD TiO2 and ZrO2 coated magnesium alloys. J. Mech. Behav. Biomed. Mater. 2022, 125, 104945. [Google Scholar] [CrossRef] [PubMed]
- Yenagolla, P.; Sandeep, K.; Sharma, J.V.S. EPRA International Journal of Research and Development (IJRD) Natural Polymers and Its Applications—A Review. Epra Int. J. Res. Dev. (IJRD) 2022, 7838, 6–15. [Google Scholar]
- Kumar, V.; Jule, L.T.; Ramaswamy, K. Conducting Polymers for Organic Solar Cell Application. In Conducting Polymers for Advanced Energy Applications; CRC Press: Boca Raton, FL, USA, 2021; p. 139. [Google Scholar]
- Ilyas, R.A.; Sapuan, S.M. The Preparation Methods and Processing of Natural Fibre Bio-polymer Composites. Curr. Org. Synth. 2020, 16, 1068–1070. [Google Scholar] [CrossRef] [PubMed]
- Chahal, S.; Kumar, A.; Hussian, F.S.J. Development of biomimetic electrospun polymeric biomaterials for bone tissue engineering. A review. J. Biomater. Sci. Polym. Ed. 2019, 30, 1308–1355. [Google Scholar] [CrossRef]
- James, D.M.B. Remodeling Life and Living—A Review of Advanced Polymeric Materials. Int. J. Res. Appl. Sci. Eng. Technol. 2020, 8, 396–400. [Google Scholar] [CrossRef]
- John, C.B. Sir Harold Ridley and the intraocular foreign body that made history. Kerala J. Ophthalmol. 2020, 32, 315–320. [Google Scholar] [CrossRef]
- Fitzgerald, J.K. Silicone Contact Lenses. In Advances in Biomaterials; CRC Press: Boca Raton, FL, USA, 2021; pp. 209–216. [Google Scholar]
- Wijesinghe, W.P.S.L.; Mantilaka, M.M.M.G.P.G.; Karunarathne, T.S.E.F.; Rajapakse, R.M.G. Synthesis of a hydroxyapatite/poly(methyl methacrylate) nanocomposite using dolomite. Nanoscale Adv. 2019, 1, 86–88. [Google Scholar] [CrossRef]
- Sedlak, J.; Vocilka, O.; Slany, M.; Chladil, J.; Polzer, A.; Varhanik, M. Design and production of eye prosthesis using 3D printing. MM Sci. J. 2020, 2020, 3806–3812. [Google Scholar] [CrossRef]
- Matsuda, Y.; Karino, M.; Okui, T.; Kanno, T. Complications of Poly-l-Lactic Acid and Polyglycolic Acid (PLLA/PGA) Osteosynthesis Systems for Maxillofacial Surgery: A Retrospective Clinical Investigation. Polymers 2021, 13, 889. [Google Scholar] [CrossRef]
- Reddy, M.S.B.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A comparative review of natural and synthetic biopolymer composite scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef]
- Hassan, M.E.; Bai, J.; Dou, D.Q. Biopolymers; Definition, classification and applications. Egypt. J. Chem. 2019, 62, 1725–1737. [Google Scholar] [CrossRef]
- Hacker, M.C.; Krieghoff, J.; Mikos, A.G. Synthetic polymers. In Principles of Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2019; pp. 559–590. [Google Scholar]
- Rodríguez, G.R.; Patrício, T.; López, J.D. Natural Polymers for Bone Repair, 2nd ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2019. [Google Scholar] [CrossRef]
- Hench, L.L.; Polak, J.M. Third-generation biomedical materials. Science 2002, 295, 1014–1017. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Pal, S. Mechanical properties of bone cement: A review. J. Biomed. Mater. Res. 1984, 18, 435–462. [Google Scholar] [CrossRef]
- Kenny, S.M.; Buggy, M. Bone cements and fillers: A review. J. Mater. Sci. Mater. Med. 2003, 14, 923–938. [Google Scholar] [CrossRef]
- Bettencourt, A.; Almeida, A.J. Poly (methyl methacrylate) particulate carriers in drug delivery. J. Microencapsul. 2012, 29, 353–367. [Google Scholar] [CrossRef] [PubMed]
- Webb, J.C.J.; Spencer, R.F. The role of polymethylmethacrylate bone cement in modern orthopaedic surgery. J. Bone Jt. Surg. Br. 2007, 89, 851–857. [Google Scholar] [CrossRef]
- Senra, M.R.; Marques, M.d.F.V. Synthetic polymeric materials for bone replacement. J. Compos. Sci. 2020, 4, 191. [Google Scholar] [CrossRef]
- Bistolfi, A.; Ferracini, R.; Albanese, C.; Vernè, E.; Miola, M. PMMA-based bone cements and the problem of joint arthroplasty infections: Status and new perspectives. Materials 2019, 12, 4002. [Google Scholar] [CrossRef]
- Bistolfi, A.; Massazza, G.; Verné, E.; Massè, A.; Deledda, D.; Ferraris, S.; Miola, M.; Galetto, F.; Crova, M. Antibiotic-loaded cement in orthopedic surgery: A review. Int. Sch. Res. Not. 2011, 2011, 290851. [Google Scholar] [CrossRef]
- Ferraris, S.; Miola, M.; Bistolfi, A.; Fucale, G.; Crova, M.; Masse’, A.; Verné, E. In vitro comparison between commercially and manually mixed antibiotic-loaded bone cements. J. Appl. Biomater. Biomech. 2010, 8, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Wang, J.; Yan, F. Comparative investigation on the tribological behaviors of CF/PEEK composites under sea water lubrication. Tribol. Int. 2012, 52, 170–177. [Google Scholar] [CrossRef]
- Patel, P.; Stec, A.A.; Hull, T.R.; Naffakh, M.; Diez-Pascual, A.M.; Ellis, G.; Safronava, N.; Lyon, R.E. Flammability properties of PEEK and carbon nanotube composites. Polym. Degrad. Stab. 2012, 97, 2492–2502. [Google Scholar] [CrossRef]
- Li, E.Z.; Guo, W.L.; Wang, H.D.; Xu, B.S.; Liu, X.T. Research on Tribological Behavior of PEEK and Glass Fiber Reinforced PEEK Composite. Phys. Procedia 2013, 50, 453–460. [Google Scholar] [CrossRef]
- Kurtz, S.M. Chemical and radiation stability of PEEK. In PEEK Biomaterials Handbook; Elsevier: Amsterdam, The Netherlands, 2012; pp. 75–79. [Google Scholar]
- Normand, B.; Takenouti, H.; Keddam, M.; Liao, H.; Monteil, G.; Coddet, C. Electrochemical impedance spectroscopy and dielectric properties of polymer: Application to PEEK thermally sprayed coating. Electrochim. Acta 2004, 49, 2981–2986. [Google Scholar] [CrossRef]
- Zhang, G.; Li, W.-Y.; Cherigui, M.; Zhang, C.; Liao, H.; Bordes, J.-M.; Coddet, C. Structures and tribological performances of PEEK (poly-ether-ether-ketone)-based coatings designed for tribological application. Prog. Org. Coat. 2007, 60, 39–44. [Google Scholar] [CrossRef]
- Kurtz, S.M. Synthesis and processing of PEEK for surgical implants. In PEEK Biomaterials Handbook; Elsevier: Amsterdam, The Netherlands, 2012; pp. 9–22. [Google Scholar]
- Zheng, Y.; Liu, L.; Ma, Y.; Xiao, L.; Liu, Y. Enhanced osteoblasts responses to surface-sulfonated polyetheretherketone via a single-step ultraviolet-initiated graft polymerization. Ind. Eng. Chem. Res. 2018, 57, 10403–10410. [Google Scholar] [CrossRef]
- Ma, R.; Guo, D. Evaluating the bioactivity of a hydroxyapatite-incorporated polyetheretherketone biocomposite. J. Orthop. Surg. Res. 2019, 14, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Johansson, P.; Jimbo, R.; Naito, Y.; Kjellin, P.; Currie, F.; Wennerberg, A. Polyether ether ketone implants achieve increased bone fusion when coated with nano-sized hydroxyapatite: A histomorphometric study in rabbit bone. Int. J. Nanomed. 2016, 11, 1435. [Google Scholar] [CrossRef]
- Khoury, J.; Selezneva, I.; Pestov, S.; Tarassov, V.; Ermakov, A.; Mikheev, A.; Lazov, M.; Kirkpatrick, S.R.; Shashkov, D.; Smolkov, A. Surface bioactivation of PEEK by neutral atom beam technology. Bioact. Mater. 2019, 4, 132–141. [Google Scholar] [CrossRef]
- Terpiłowski, K.; Wiącek, A.E.; Jurak, M. Influence of nitrogen plasma treatment on the wettability of polyetheretherketone and deposited chitosan layers. Adv. Polym. Technol. 2018, 37, 1557–1569. [Google Scholar] [CrossRef]
- Lu, T.; Qian, S.; Meng, F.; Ning, C.; Liu, X. Enhanced osteogenic activity of poly ether ether ketone using calcium plasma immersion ion implantation. Colloids Surf. B Biointerfaces 2016, 142, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wei, W.; Yang, L.; Pan, Y.; Wang, X.; Wang, T.; Tang, S.; Yao, Y.; Hong, H.; Wei, J. Stimulation of cell responses and bone ingrowth into macro-microporous implants of nano-bioglass/polyetheretherketone composite and enhanced antibacterial activity by release of hinokitiol. Colloids Surf. B Biointerfaces 2018, 164, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Liu, L.; Xiao, L.; Zhang, Q.; Liu, Y. Enhanced osteogenic activity of phosphorylated polyetheretherketone via surface-initiated grafting polymerization of vinylphosphonic acid. Colloids Surf. B Biointerfaces 2019, 173, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Tang, T. Current strategies to improve the bioactivity of PEEK. Int. J. Mol. Sci. 2014, 15, 5426–5445. [Google Scholar] [CrossRef]
- Kasemo, B. Biological surface science. Surf. Sci. 2002, 500, 656–677. [Google Scholar] [CrossRef]
- Almasi, D.; Iqbal, N.; Sadeghi, M.; Sudin, I.; Kadir, M.R.A.; Kamarul, T. Preparation methods for improving PEEK’s bioactivity for orthopedic and dental application: A review. Int. J. Biomater. 2016, 2016, 8202653. [Google Scholar] [CrossRef]
- Poulsson, A.H.C.; Eglin, D.; Richards, R.G. Surface modification techniques of PEEK, including plasma surface treatment. In PEEK Biomaterials Handbook; Elsevier: Amsterdam, The Netherlands, 2019; pp. 179–201. [Google Scholar]
- Poulsson, A.H.C.; Richards, R.G. Surface modification techniques of polyetheretherketone, including plasma surface treatment. In PEEK Biomaterials Handbook; Elsevier: Amsterdam, The Netherlands, 2012; pp. 145–161. [Google Scholar]
- Wang, H.; Lu, T.; Meng, F.; Zhu, H.; Liu, X. Enhanced osteoblast responses to poly ether ether ketone surface modified by water plasma immersion ion implantation. Colloids Surf. B Biointerfaces 2014, 117, 89–97. [Google Scholar] [CrossRef]
- Visakh, P.M. Polyaniline-Based Blends, Composites, and Nanocomposites. In Polyaniline Blends, Composites, and Nanocomposites; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–22. [Google Scholar] [CrossRef]
- Paxton, N.C.; Allenby, M.C.; Lewis, P.M.; Woodruff, M.A. Biomedical applications of polyethylene. Eur. Polym. J. 2019, 118, 412–428. [Google Scholar] [CrossRef]
- Sobieraj, M.C.; Rimnac, C.M. Ultra high molecular weight polyethylene: Mechanics, morphology, and clinical behavior. J. Mech. Behav. Biomed. Mater. 2009, 2, 433–443. [Google Scholar] [CrossRef]
- Kurtz, S.M. 1-A primer on UHMWPE. UHMWPE Biomaterials Handbook: Ultra-High Molecular Weight Polyethylene in Total Joint Replacement and Medical Devices; ELSEVIER: London, UK, 2016; Volume 1, pp. 1–6. [Google Scholar]
- Budak, K.; Sogut, O.; Sezer, U.A. A review on synthesis and biomedical applications of polyglycolic acid. J. Polym. Res. 2020, 27, 208. [Google Scholar] [CrossRef]
- On, S.W.; Cho, S.W.; Byun, S.H.; Yang, B.E. Bioabsorbable osteofixation materials for maxillofacial bone surgery: A review on polymers and magnesium-based materials. Biomedicines 2020, 8, 300. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cao, H.; Ye, L.; Coates, P.; Caton-Rose, F.; Zhao, X. Long-chain branched poly (lactic acid)-b-poly (lactide-co-caprolactone): Structure, viscoelastic behavior, and triple-shape memory effect as smart bone fixation material. Ind. Eng. Chem. Res. 2020, 59, 4524–4532. [Google Scholar] [CrossRef]
- Siakeng, R.; Jawaid, M.; Ariffin, H.; Sapuan, S.M.; Asim, M.; Saba, N. Natural fiber reinforced polylactic acid composites: A review. Polym. Compos. 2019, 40, 446–463. [Google Scholar] [CrossRef]
- DeStefano, V.; Khan, S.; Tabada, A. Applications of PLA in modern medicine. Eng. Regen. 2020, 1, 76–87. [Google Scholar] [CrossRef]
- Naseem, R.; Tzivelekis, C.; German, M.J.; Gentile, P.; Ferreira, A.M.; Dalgarno, K. Strategies for enhancing polyester-based materials for bone fixation applications. Molecules 2021, 26, 992. [Google Scholar] [CrossRef]
- Shirazi, R.N.; Aldabbagh, F.; Ronan, W.; Erxleben, A.; Rochev, Y.; McHugh, P. Effects of material thickness and processing method on poly (lactic-co-glycolic acid) degradation and mechanical performance. J. Mater. Sci. Mater. Med. 2016, 27, 154. [Google Scholar] [CrossRef] [PubMed]
- Ramos, D.M.; Dhandapani, R.; Subramanian, A.; Sethuraman, S.; Kumbar, S.G. Clinical complications of biodegradable screws for ligament injuries. Mater. Sci. Eng. C 2020, 109, 110423. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef]
- Lopes, M.S.; Jardini, A.L.; Filho, R.M. Poly (lactic acid) production for tissue engineering applications. Procedia Eng. 2012, 42, 1402–1413. [Google Scholar] [CrossRef]
- Middleton, J.C.; Tipton, A.J. Synthetic biodegradable polymers as orthopedic devices. Biomaterials 2000, 21, 2335–2346. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, M.A.; Hutmacher, D.W. The return of a forgotten polymer—Polycaprolactone in the 21st century. Prog. Polym. Sci. 2010, 35, 1217–1256. [Google Scholar] [CrossRef]
- Chocholata, P.; Kulda, V.; Babuska, V. Fabrication of scaffolds for bone-tissue regeneration. Materials 2019, 12, 568. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, D.; Kumar, V.; Sharma, S. Drug-loaded biomaterials for orthopedic applications: A review. J. Control. Release 2022, 344, 113–133. [Google Scholar] [CrossRef]
- Lishchynskyi, O.; Stetsyshyn, Y.; Raczkowska, J.; Awsiuk, K.; Orzechowska, B.; Abalymov, A.; Skirtach, A.G.; Bernasik, A.; Nastyshyn, S.; Budkowski, A. Fabrication and impact of fouling-reducing temperature-responsive poegma coatings with embedded caco3 nanoparticles on different cell lines. Materials 2021, 14, 1417. [Google Scholar] [CrossRef]
- Sebbe, N.P.V.; Fernandes, F.; Sousa, V.F.C.; Silva, F.J.G. Hybrid Manufacturing Processes Used in the Production of Complex Parts: A Comprehensive Review. Metals 2022, 12, 1874. [Google Scholar] [CrossRef]
- Kim, H.M.; Lee, D.; Kim, S.Y. Biocompatibility and osteogenic potential of calcium silicate-based cement combined with enamel matrix derivative: Effects on human bone marrow-derived stem cells. Materials 2021, 14, 7750. [Google Scholar] [CrossRef]
- Rochira, A.; Siculella, L.; Damiano, F.; Palermo, A.; Ferrante, F.; Carluccio, M.A.; Calabriso, N.; Giannotti, L.; Stanca, E. Concentrated growth factors (CGF) induce osteogenic differentiation in human bone marrow stem cells. Biology 2020, 9, 370. [Google Scholar] [CrossRef]
- Zha, Y.; Li, Y.; Lin, T.; Chen, J.; Zhang, S.; Wang, J. Progenitor cell-derived exosomes endowed with VEGF plasmids enhance osteogenic induction and vascular remodeling in large segmental bone defects. Theranostics 2020, 11, 397–409. [Google Scholar] [CrossRef]
- Jiao, X.; Sun, X.; Li, W.; Chu, W.; Zhang, Y.; Li, Y.; Wang, Z.; Zhou, X.; Ma, J.; Xu, C.; et al. 3D-Printed β-Tricalcium Phosphate Scaffolds Promote Osteogenic Differentiation of Bone Marrow-Deprived Mesenchymal Stem Cells in an N6-methyladenosine-Dependent Manner. Int. J. Bioprint. 2022, 8, 31–44. [Google Scholar] [CrossRef]
- Zhao, Y.; Xing, Y.; Wang, M.; Huang, Y.; Xu, H.; Su, Y.; Zhao, Y.; Shang, Y. Supramolecular Hydrogel Based on an Osteogenic Growth Peptide Promotes Bone Defect Repair. ACS Omega 2022, 7, 11395–11404. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, A.; Gualtieri, T.; Heer, B.; Bose, S. Introduction to Additive Manufacturing. In Additive Manufacturing; CRC Press: Boca Raton, FL, USA, 2019; pp. 1–23. [Google Scholar]
- Eckert, J.; Grieße, T. Additive Manufacturing at Montanuniversität Leoben. Adv. Eng. Mater. 2023, 25, 2300179. [Google Scholar] [CrossRef]
- Muhindo, D.; Elkanayati, R.; Srinivasan, P.; Repka, M.A.; Ashour, E.A. Recent advances in the applications of additive manufacturing (3D printing) in drug delivery: A comprehensive review. AAPS PharmSciTech 2023, 24, 57. [Google Scholar] [CrossRef] [PubMed]
- Appel, J.; Ho, D.; Dobyns, B.M.; Reichert, W.M.M.; Duranty, E.R. Additive Manufacturing of Biopolymers Via Modified FDM 3D Printing Enabled By the Dissolution Properties of Hydrophilic Ionic Liquids. In Electrochemical Society Meeting Abstracts 242; The Electrochemical Society, Inc.: Philadelphia, PA, USA, 2022; p. 2068. [Google Scholar]
- Pugliese, R.; Beltrami, B.; Regondi, S.; Lunetta, C. Polymeric biomaterials for 3D printing in medicine: An overview. Ann. 3d Print. Med. 2021, 2, 100011. [Google Scholar] [CrossRef]
- Skylar-Scott, M.A.; Uzel, S.G.; Nam, L.L.; Ahrens, J.H.; Truby, R.L.; Damaraju, S.; Lewis, J.A. Biomanufacturing of organ-specific tissues with high cellular density and embedded vascular channels. Sci. Adv. 2019, 5, eaaw2459. [Google Scholar] [CrossRef]
- Zhang, B.; Pei, X.; Zhou, C.; Fan, Y.; Jiang, Q.; Ronca, A.; D’Amora, U.; Chen, Y.; Li, H.; Sun, Y.; et al. The biomimetic design and 3D printing of customized mechanical properties porous Ti6Al4V scaffold for load-bearing bone reconstruction. Mater. Des. 2018, 152, 30–39. [Google Scholar] [CrossRef]
- Söhling, N.; Neijhoft, J.; Nienhaus, V.; Acker, V.; Harbig, J.; Menz, F.; Ochs, J.; Verboket, R.D.; Ritz, U.; Blaeser, A.; et al. 3D-printing of hierarchically designed and osteoconductive bone tissue engineering scaffolds. Materials 2020, 13, 1836. [Google Scholar] [CrossRef]
- Pei, X.; Ma, L.; Zhang, B.; Sun, J.; Sun, Y.; Fan, Y.; Gou, Z.; Zhou, C.; Zhang, X. Creating hierarchical porosity hydroxyapatite scaffolds with osteoinduction by three-dimensional printing and microwave sintering. Biofabrication 2017, 9, 045008. [Google Scholar] [CrossRef]
- Weems, A.C.; Pérez-Madrigal, M.M.; Arno, M.C.; Dove, A.P. 3D Printing for the Clinic: Examining Contemporary Polymeric Biomaterials and their Clinical Utility. Biomacromolecules 2020, 21, 1037–1059. [Google Scholar] [CrossRef]
- Attaran, M. The rise of 3-D printing: The advantages of additive manufacturing over traditional manufacturing. Bus. Horiz. 2017, 60, 677–688. [Google Scholar] [CrossRef]
- Stepniak, K.; Ursani, A.; Paul, N.; Naguib, H. Novel 3D printing technology for CT phantom coronary arteries with high geometrical accuracy for biomedical imaging applications. Bioprinting 2020, 18, e00074. [Google Scholar] [CrossRef]
- Jardini, A.L.; Larosa, M.A.; Filho, R.M.; de Carvalho Zavaglia, C.A.; Bernardes, L.F.; Lambert, C.S.; Calderoni, D.R.; Kharmandayan, P. Cranial reconstruction: 3D biomodel and custom-built implant created using additive manufacturing. J. Cranio-Maxillofac. Surg. 2014, 42, 1877–1884. [Google Scholar] [CrossRef]
- Mobbs, R.J.; Coughlan, M.; Thompson, R.; Sutterlin, C.E.; Phan, K. The utility of 3D printing for surgical planning and patient-specific implant design for complex spinal pathologies: Case report. J. Neurosurg. Spine 2017, 26, 513–518. [Google Scholar] [CrossRef]
- Putra, N.E.; Mirzaali, M.J.; Apachitei, I.; Zhou, J.; Zadpoor, A.A. Multi-material additive manufacturing technologies for Ti-, Mg-, and Fe-based biomaterials for bone substitution. Acta Biomater. 2020, 109, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Chua, K.; Khan, I.; Malhotra, R.; Zhu, D. Additive manufacturing and 3D printing of metallic biomaterials. Eng. Regen. 2021, 2, 288–299. [Google Scholar] [CrossRef]
- Bose, S.; Vahabzadeh, S.; Bandyopadhyay, A. Bone tissue engineering using 3D printing. Mater. Today 2013, 16, 496–504. [Google Scholar] [CrossRef]
- Campoli, G.; Borleffs, M.S.; Yavari, S.A.; Wauthle, R.; Weinans, H.; Zadpoor, A.A. Mechanical properties of open-cell metallic biomaterials manufactured using additive manufacturing. Mater. Design. 2013, 49, 957–965. [Google Scholar] [CrossRef]
- Yavari, S.A.; Wauthlé, R.; van der Stok, J.; Riemslag, A.C.; Janssen, M.; Mulier, M.; Kruth, J.-P.; Schrooten, J.; Weinans, H.; Zadpoor, A.A. Fatigue behavior of porous biomaterials manufactured using selective laser melting. Mater. Sci. Eng. C 2013, 33, 4849–4858. [Google Scholar] [CrossRef]
- van Bael, S.; Chai, Y.C.; Truscello, S.; Moesen, M.; Kerckhofs, G.; Van Oosterwyck, H.; Kruth, J.-P.; Schrooten, J. The effect of pore geometry on the in vitro biological behavior of human periosteum-derived cells seeded on selective laser-melted Ti6Al4V bone scaffolds. Acta Biomater. 2012, 8, 2824–2834. [Google Scholar] [CrossRef]
- Fukuda, A.; Takemoto, M.; Saito, T.; Fujibayashi, S.; Neo, M.; Pattanayak, D.K.; Matsushita, T.; Sasaki, K.; Nishida, N.; Kokubo, T.; et al. Osteoinduction of porous Ti implants with a channel structure fabricated by selective laser melting. Acta Biomater. 2011, 7, 2327–2336. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, N.; Fujibayashi, S.; Takemoto, M.; Sasaki, K.; Otsuki, B.; Nakamura, T.; Matsushita, T.; Kokubo, T.; Matsuda, S. Effect of pore size on bone ingrowth into porous titanium implants fabricated by additive manufacturing: An in vivo experiment. Mater. Sci. Eng. C 2016, 59, 690–701. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, J.; Pavanram, P.; Leeflang, M.; Fockaert, L.; Pouran, B.; Tümer, N.; Schröder, K.-U.; Mol, J.M.C.; Weinans, H.; et al. Additively manufactured biodegradable porous magnesium. Acta Biomater. 2018, 67, 378–392. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jahr, H.; Lietaert, K.; Pavanram, P.; Yilmaz, A.; Fockaert, L.I.; Leeflang, M.A.; Pouran, B.; Gonzalez-Garcia, Y.; Weinans, H.; et al. Additively manufactured biodegradable porous iron. Acta Biomater. 2018, 77, 380–393. [Google Scholar] [CrossRef]
- Shah, F.A.; Snis, A.; Matic, A.; Thomsen, P.; Palmquist, A. 3D printed Ti6Al4V implant surface promotes bone maturation and retains a higher density of less aged osteocytes at the bone-implant interface. Acta Biomater. 2016, 30, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.A.; Omar, O.; Suska, F.; Snis, A.; Matic, A.; Emanuelsson, L.; Norlindh, B.; Lausmaa, J.; Thomsen, P.; Palmquist, A. Long-term osseointegration of 3D printed CoCr constructs with an interconnected open-pore architecture prepared by electron beam melting. Acta Biomater. 2016, 36, 296–309. [Google Scholar] [CrossRef]
- Ngo, T.D.; Kashani, A.; Imbalzano, G.; Nguyen, K.T.Q.; Hui, D. Additive manufacturing (3D printing): A review of materials, methods, applications and challenges. Compos. Part B Eng. 2018, 143, 172–196. [Google Scholar] [CrossRef]
- Bhushan, J.; Grover, V. Additive manufacturing: Current concepts, methods, and applications in oral health care. In Biomanufacturing; Springer International Publishing: Cham, Switzerland, 2019; pp. 103–122. [Google Scholar] [CrossRef]
- Mohan, D.; Teong, Z.K.; Bakir, A.N.; Sajab, M.S.; Kaco, H. Extending cellulose-based polymers application in additive manufacturing technology: A review of recent approaches. Polymers 2020, 12, 1876. [Google Scholar] [CrossRef]
- Lewis, J.A. Direct ink writing of 3D functional materials. Adv. Funct. Mater. 2006, 16, 2193–2204. [Google Scholar] [CrossRef]
- Bikas, H.; Stavropoulos, P.; Chryssolouris, G. Additive manufacturing methods and modeling approaches: A critical review. Int. J. Adv. Manuf. Technol. 2016, 83, 389–405. [Google Scholar] [CrossRef]
- Park, S.; Shou, W.; Makatura, L.; Matusik, W.; Fu, K.K. 3D printing of polymer composites: Materials, processes, and applications. Matter 2022, 5, 43–76. [Google Scholar] [CrossRef]
- Rajpurkar, P.; Chen, E.; Banerjee, O.; Topol, E.J. AI in health and medicine. Nat. Med. 2022, 28, 31–38. [Google Scholar] [CrossRef]
- Zwawi, M. Recent advances in bio-medical implants; mechanical properties, surface modifications and applications. Eng. Res. Express 2022, 4, 032003. [Google Scholar] [CrossRef]
- Jafari, M.; Paknejad, Z.; Rad, M.R.; Motamedian, S.R.; Eghbal, M.J.; Nadjmi, N.; Khojasteh, A. Polymeric scaffolds in tissue engineering: A literature review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 431–459. [Google Scholar] [CrossRef] [PubMed]



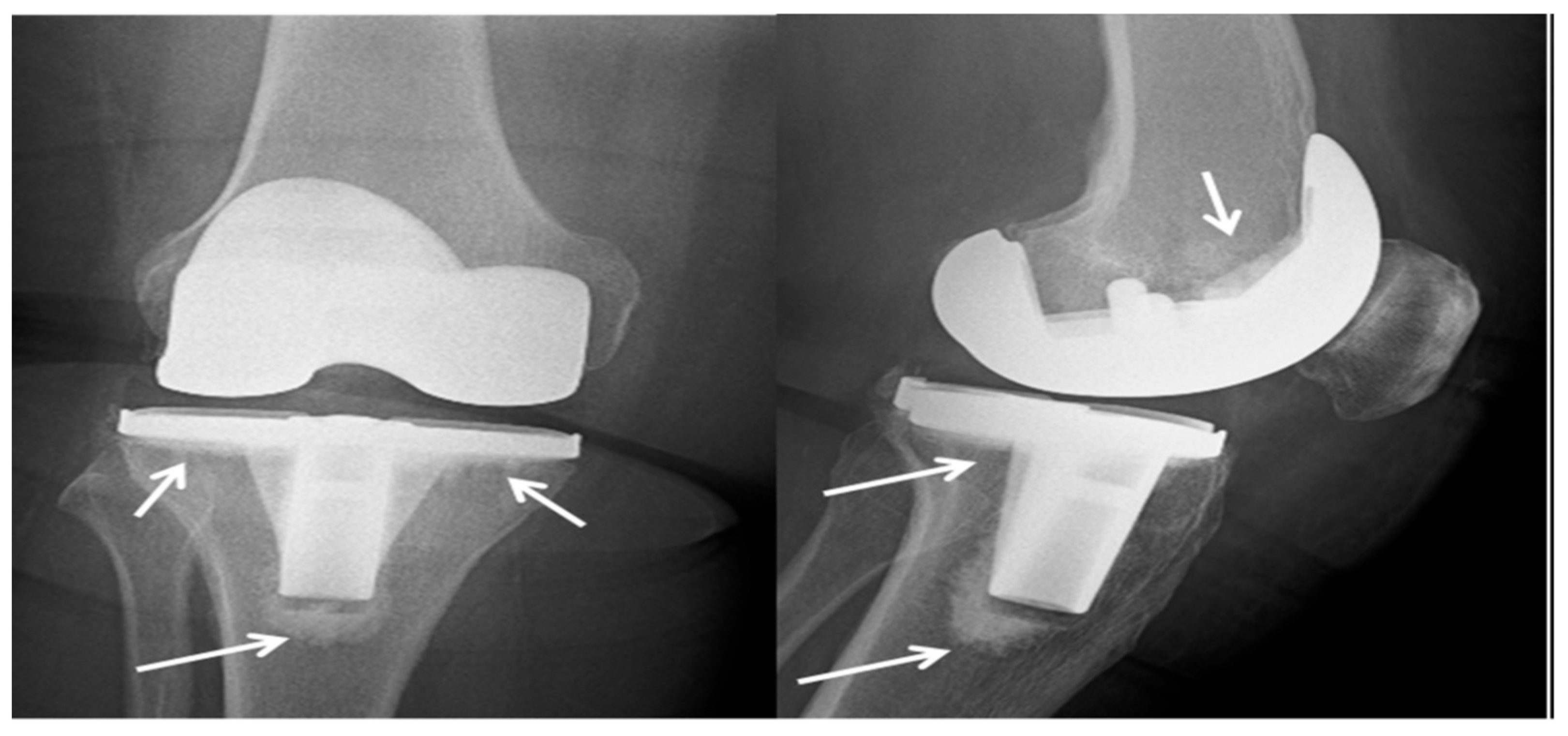
| Fracture Site | Internal Fixators | |
|---|---|---|
| Head | Fracture of the skull | Plates, wires, and pins |
| Craniofacial fracture | Plates, wires, and pins | |
| Trunk | Fracture of the clavicle | Plates and intramedullary nails |
| Fracture of the scapular | Plates and screws | |
| Fracture of the pelvis | External fixators, screws, and plates | |
| Spinal fracture | Fixation implant contains plates, pedicle screws, and rods | |
| Upper limb fracture | Fracture of the humerus | Plates and screws for open reduction and an intramedullary nail for closed reduction |
| Fracture of the radius or ulnar | Plates and screws for open reduction and an intramedullary nail for closed reduction | |
| Fracture of the phalanges and metacarpal fracture | External fixators for close reduction and intramedullary nails, plates, and screws for open reduction | |
| Lower limb fracture | Femoral fracture | Plates and screws for open reduction and an intramedullary nail for closed reduction |
| Tibial and fibular fracture | Plates and screws for open reduction and an intramedullary nail for closed reduction | |
| Fracture of the metatarsus | Plates and screws for open reduction and an intramedullary nail for closed reduction | |
| Calcaneal fracture | Wires and screws for close reduction |
| Materials | Density (g/cm3) | Yield Strength (Mpa) | Tensile Strength (Mpa) | Elongation at Break (%) | Elastic Modulus (Gpa) | Biocompatibility | Corrosion-Resistance | Refs. | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Metal | Non-biodegradable | 316 L steel | 7.9 | 290 | 579 | 40 | 193 | Poor | Reasonable | [50,51,52,53] |
| Ti-6Al-4V | 4.43 | 850–900 | 960–970 | 14 | 110 | Fair | Excellent | |||
| CoCr20Ni15Mo7 | 7.8 | 240–450 | 450–960 | 50 | 195–230 | Poor | Excellent | |||
| Biodegradable | Pure Mg | 1.74–2 | 65–100 | 90–190 | 2–10 | 41–45 | Excellent | Poor | [50,53] | |
| Fe20Mn alloy | 7.73 | 420 | 700 | 8 | 207 | |||||
| Zn-Al-Cu (Zn based alloy) | 5.79 | 171 | 210 | 1 | 90 | |||||
| Polymer | Non-biodegradable | UHMWPE | 0.931–0.949 | 21.4–27.6 | 38.6–48.3 | 3.5–5.25 | 0.894–0.963 | Good | Excellent | [54,55] |
| PMMA | 1.18 | - | 72 | 5 | 310 | Good | Excellent | [56,57,58,59,60] | ||
| PEEK | 1.23–1.32 | 87–95.2 | 70.3–103 | 0.3–1.5 | 3.76–3.95 | Good | Excellent | [61,62,63,64] | ||
| Biodegradable | PLA | 1.21–1.25 | 60 | 21–60 | 6 | 0.35–3.5 | Excellent | poor | [2,65] | |
| PLGA | 1.30–1.34 | 3.8–26.6 | 13.9–16.7 | 5.7 | - | Excellent | Poor | [2,50] | ||
| PLC | 1.11–1.14 | 8.37–14.6 | 20.7–42 | 22.8–28.3 | 0.21–0.44 | Excellent | Poor | |||
| Metal and Alloys | Particular Alloys | Major Applications | Advantages | Disadvantages |
|---|---|---|---|---|
| Stainless steel | 316 L Stainless steel | Surgical implements, stents, fracture fixation | High wear resistance | The modulus is increased compared to bone allergy due to Co, Cr, and Ni |
| Titanium-based alloys | CP–Ti | Dental implants, fracture fixation, bone and joint replacement, pacemaker encapsulation | Low density, excellent biocompatibility, high corrosion resistance, low Young’s modulus | Weak tribological characteristics, the toxic impact of V and Al with long-term use |
| Ti–Al–Nb | ||||
| Ti–6Al–4V | ||||
| Ti–13Nb–13Zr | ||||
| Ti– Mo–Zr–Fe | ||||
| Co and chromium alloys | Co–Cr–Mo | Dental implants and restorations, heart valves, joint and bone replacement | Excellent wear resistance | The modulus is increased compared to bone allergy due to Co, Cr, and Ni |
| Cr–Ni– Cr–Mo | ||||
| Others | Ni–Ti | Orthodontic wires, fracture fixation plates, stents | Low Young’s modulus | Allergy due to Ni |
| Platinum Pt–Ir | Electrodes | Excellent corrosion resistance under maximum voltage potential and charge transfer conditions | - | |
| Hg–Ag–Sn amalgam | Dental restorations | Easily moldable in situ into a desired shape that is resistant to corrosion in the oral environment | Concerns related to Hg toxicity |
| Material | Young’s Modulus (GPa) | 0.2% Offset Yield Point (MPa) | Compressive Strength at 20% Strain (MPa) |
|---|---|---|---|
| Cast Fe | 203 | 157 | 498 |
| Cast Mg | 30–40 | 20–30 | 100–180 |
| Cast Zn | 100 | 95 | 200 |
| Cast Fe–35Mn | - | 240 | 440 |
| Human cortical bone | 1–35 | 1–20 | 103–140 |
| Material | Elastic Modulus (GPa) | Tensile Strength (MPa) |
|---|---|---|
| Trabecular bone | 0.02–0.05 | 1–5 |
| Cortical bone trabecular | 3–30 | 50–151 |
| UHMWPE | 0.9–2.7 | 50–151 |
| PMMA | 1.88–3.3 | 68 |
| PEEK | 3.5–4.0 | 118 |
| Co–Cr–Mo alloy | 210–232 | 1173 |
| Ti–6Al–4V alloy | 116 | 1018 |
| Product Name | Manufacturer | Polymer Composition | Degradation Time |
|---|---|---|---|
| Biofix® SR-PGA | Bionx Implants, Tampere, Finland | SR-PGA | 6 weeks |
| Biofix® SR-PLLA | Bionx Implants, Tampere, Finland | SR-PLLA | 5–7 years |
| Resomer® LR708 | Evonik Industries, Darmstadt, Germany | PLLA (70%) + PDLLA (30%) | 2–3 years |
| MacroPore® | MacroPore Biosurgery Inc., San Diego, CA, USA | PLLA (70%) + PDLLA (30%) | 2–3 years |
| Macrosorb® | MacroPore Biosurgery Inc., San Diego, CA, USA | PLLA (70%) + PDLLA (30%) | 2–3 years |
| Biosorb FX® | Linvatec Biomaterials Ltd., Tampere, Finland | PLLA (70%) + PDLLA (30%) | 2–3 years |
| Resorb X® | KLS Martin Group, Tuttlingen, Germany | PLLA (50%) + PDLLA (50%) | 12–30 months |
| PolyMax® | Synthes, Oberdorf, Switzerland | PLLA (70%) + PDLLA (30%) | 2 years |
| PolyMax® RAPID | Synthes, Oberdorf, Switzerland | PLLA (85%) + PGA (15%) | 12 months |
| Rapidsorb® | DePuy Synthes, West Chester, PA, USA | PLLA (85%) + PGA (15%) | 12 months |
| Lactosorb® | Lorenz, Jacksonville, FL, USA | PLLA (82%) + PGA (18%) | 12 months |
| Delta® | Stryker Leibinger Corp., Kalamazoo, MI, USA | PLLA (85%), PGA (10%), PDLA (5%) | 8–13 months |
| Inion CPS® | Inion Inc., Tampere, Finland | PLLA, PGA, TMC–proportion varies | 2–4 years |
| Inion CPS® baby | Inion Inc., Tampere, Finland | PLLA, PGA, TMC–proportion varies | 2–3 years |
| Osteotrans-MX® | TEIJIN Medical Corp., Osaka, Japan | PLLA (60–70 wt%), u-HA (30–40 wt%) | 4.5–5.5 years |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Shalawi, F.D.; Mohamed Ariff, A.H.; Jung, D.-W.; Mohd Ariffin, M.K.A.; Seng Kim, C.L.; Brabazon, D.; Al-Osaimi, M.O. Biomaterials as Implants in the Orthopedic Field for Regenerative Medicine: Metal versus Synthetic Polymers. Polymers 2023, 15, 2601. https://doi.org/10.3390/polym15122601
Al-Shalawi FD, Mohamed Ariff AH, Jung D-W, Mohd Ariffin MKA, Seng Kim CL, Brabazon D, Al-Osaimi MO. Biomaterials as Implants in the Orthopedic Field for Regenerative Medicine: Metal versus Synthetic Polymers. Polymers. 2023; 15(12):2601. https://doi.org/10.3390/polym15122601
Chicago/Turabian StyleAl-Shalawi, Faisal Dakhelallah, Azmah Hanim Mohamed Ariff, Dong-Won Jung, Mohd Khairol Anuar Mohd Ariffin, Collin Looi Seng Kim, Dermot Brabazon, and Maha Obaid Al-Osaimi. 2023. "Biomaterials as Implants in the Orthopedic Field for Regenerative Medicine: Metal versus Synthetic Polymers" Polymers 15, no. 12: 2601. https://doi.org/10.3390/polym15122601
APA StyleAl-Shalawi, F. D., Mohamed Ariff, A. H., Jung, D.-W., Mohd Ariffin, M. K. A., Seng Kim, C. L., Brabazon, D., & Al-Osaimi, M. O. (2023). Biomaterials as Implants in the Orthopedic Field for Regenerative Medicine: Metal versus Synthetic Polymers. Polymers, 15(12), 2601. https://doi.org/10.3390/polym15122601








