Abstract
Over the last several decades, numerous modifications and advancements have been made to design the optimal corneal biomatrix for corneal epithelial cell (CECs) or limbal epithelial stem cell (LESC) carriers. However, researchers have yet to discover the ideal optimization strategies for corneal biomatrix design and its effects on cultured CECs or LESCs. This review discusses and summarizes recent optimization strategies for developing an ideal collagen biomatrix and its interactions with CECs and LESCs. Using PRISMA guidelines, articles published from June 2012 to June 2022 were systematically searched using Web of Science (WoS), Scopus, PubMed, Wiley, and EBSCOhost databases. The literature search identified 444 potential relevant published articles, with 29 relevant articles selected based on inclusion and exclusion criteria following screening and appraising processes. Physicochemical and biocompatibility (in vitro and in vivo) characterization methods are highlighted, which are inconsistent throughout various studies. Despite the variability in the methodology approach, it is postulated that the modification of the collagen biomatrix improves its mechanical and biocompatibility properties toward CECs and LESCs. All findings are discussed in this review, which provides a general view of recent trends in this field.
1. Introduction
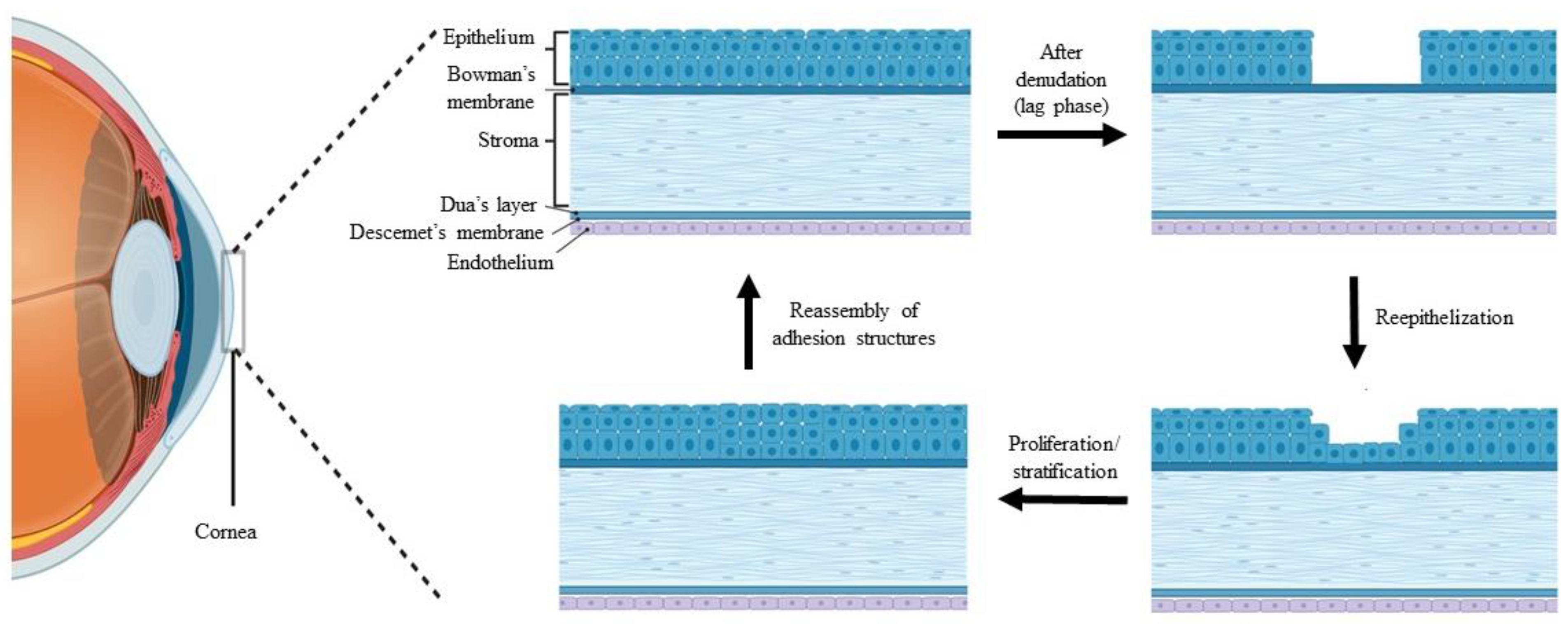
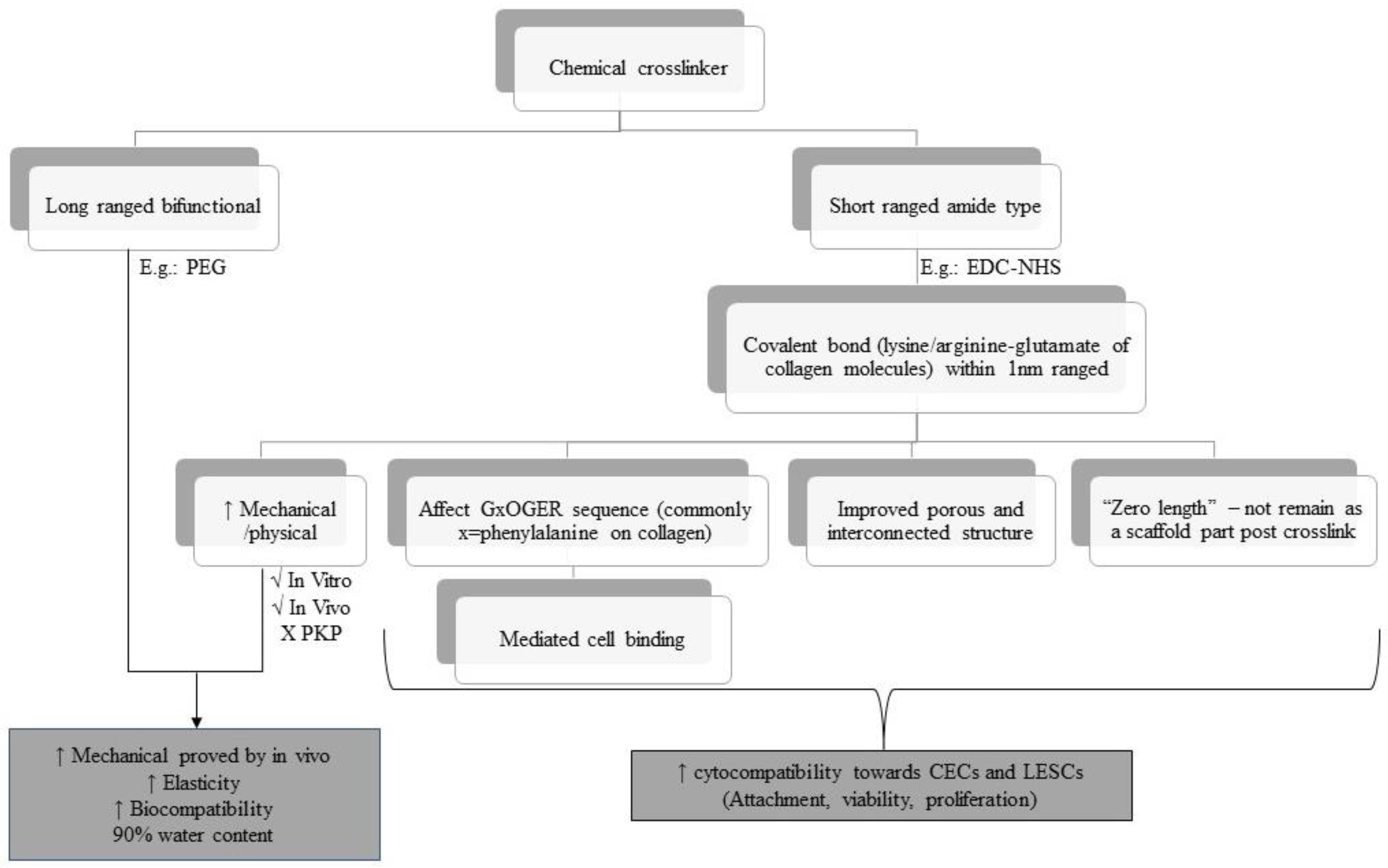
The cornea is a transparent window to the eye that maintains the refractive properties of light transmission to the retina [1,2,3]. The cornea is a multilayered component and is enclosed by a non-keratinized stratified epithelium layer, continuously providing a smooth ocular surface [1,2,3,4], as shown in Figure 1. The smoothness and integrity of the corneal epithelium are essential for transparency, providing adequate light refraction, and homeostasis [1,5]. These important roles depend on a balanced corneal epithelial cell (CEC) turnover, as the new CECs originate from the limbal epithelial stem cells (LESCs), which are located at the periphery of the cornea and replace older CECs [1,2,6]. LESCs maintain the self-renewal of CECs by continuous and slow epithelization processes that are involved in the proliferation and differentiation of LESCs into CECs, followed by centripetal migration of CECs towards the central region of the cornea [1,2,6].
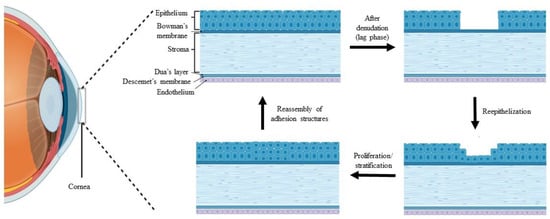
Figure 1.
Stages of the wound healing process after corneal epithelial injury, created by BioRender.
The cornea is also responsible for the frontal barrier that allows the diffusion of oxygen and essential nutrient from the tear film, against invading pathogens, debris, chemical agents, and trauma [7,8,9,10]. Thus, the corneal epithelium is vulnerable to external injury, which compromises its first line defense against corneal damage and can be overcome by a rapid healing process through re-epithelialization activity [7,9,10,11,12,13,14]. The stages of the wound healing process following corneal epithelial injury are illustrated in Figure 1.
However, various potential problems, such as delay in cell migration, epithelial hypertrophy, and recurrent corneal erosion, are prone to occur during the epithelialization process, which eventually leads to the scarring of the stroma, reducing vision quality and corneal damage [11,15,16,17]. This may be due to the dysfunction, destruction, or deficiency of LESCs, which is also known as limbal stem cell deficiency (LSCD). Recent studies also reported that LSCD can induce unstable production of corneal epithelium, followed by corneal ulceration, conjunctival invasion into the cornea, and neovascularization on the corneal surface that leads to inflammation and chronic pain, and thus, ultimately, to vision loss [18,19].
The only recognized treatment strategy for corneal blindness or vision loss is through corneal transplant but it is limited due to corneal shortage [20,21]. Another alternative solution is by a tissue engineering approach which replaces the damaged cornea with a biomaterial-based biomatrix combined with cells to replicate the corneal tissue [22,23,24,25]. Many corneal biomatrices have been developed to replace all or only a part of the cornea, depending on the patient’s requirements [26]. The development of corneal biodegradable biomatrix (specific for the epithelial layer damage treatment) focused on regenerating the damaged epithelial layer either from transplanted CECs or differentiated from transplanted LESCs. Thus, the LESC biomatrix is important in supporting the expansion, stratification, and maintaining LESCs functions [27].
Biological biomatrices, e.g., the human amniotic membrane (HAM) [28,29], fibrin [30,31,32], and feeder layers, such as 3T3 fibroblast [33], are gold standard treatments and widely used as cell carriers as they promote cell expansion. However, as natural carriers, their potential drawbacks such as the tendency to carry infection, not being optically transparent, and inadequate structural compaction and rigidity as a corneal biomatrix, were reported [34,35]. The high economical cost of these natural biomatrix needs to be overcome by discovering new biomaterials for CECs and LESCs [36].
Many studies have focused on biomaterial for CECs and LESCs, including collagen, silk fibroin [37], gelatin [38], chitosan [39], alginate [40], hyaluronic acid [41], and decellularized cornea [27]. Collagen is a well-known biomaterials for the corneal biomatrix which mimics the native corneal structure [4,42,43,44]. Collagen makes up about 70% of the dry–wet weight of the cornea and plays an important role in supporting CECs, LESCs, and corneal fibroblast cell growth [4,6,9,10,23]. Three forms of collagen biomatrix are normally used in tissue engineering research, such as collagen sponge [45,46,47,48], collagen hydrogel [49,50,51], and collagen film [52,53,54].
The important aspect that needs to be considered during the collagen biomatrix development is the collagen biomatrix interaction with CECs and LESCs which is influenced by the physicochemical properties of the biomatrix [26,55]. The source of biomaterial, mechanical strength, biodegradation rate, optical characteristics, and biocompatibility properties need to be tailored during the development of corneal biomatrix [55,56,57]. Considering the aforementioned characterization of the biomatrix, the modification or optimization process is crucial for achieving optimal biomatrix designs [26]. Modifications of collagen biomatrix, such as crosslinking, physical modification, incorporation of other biomolecules or cells into the collagen biomatrix, or incorporation of the collagen into another biomatrix, give different effects in terms of interaction between the biomatrix and cells. Since the main outcome of the produced corneal biomatrix is a prolonged effect after implantation, it is thus important to ensure its ability to regenerate into corneal native tissue.
The mechanical strength of the biomatrix is an important aspect that needs to be optimized as it must resist the high tension during implantation, and in in vivo dynamic environments, such as physiologic intraocular pressure and constant eyelid motion [58], which is closely related to the biocompatibility towards LESCs and CECs [59,60,61]. Modification through the cross-linking process will produce a mechanically strong cross-linked biomatrix [62], which results in multidimensional polymeric chain extension of the biomatrix. Unfortunately, the crosslinker could reduce biomatrix’s transparency and cause cell toxicity that will overshadow their cross-linking potential [51].
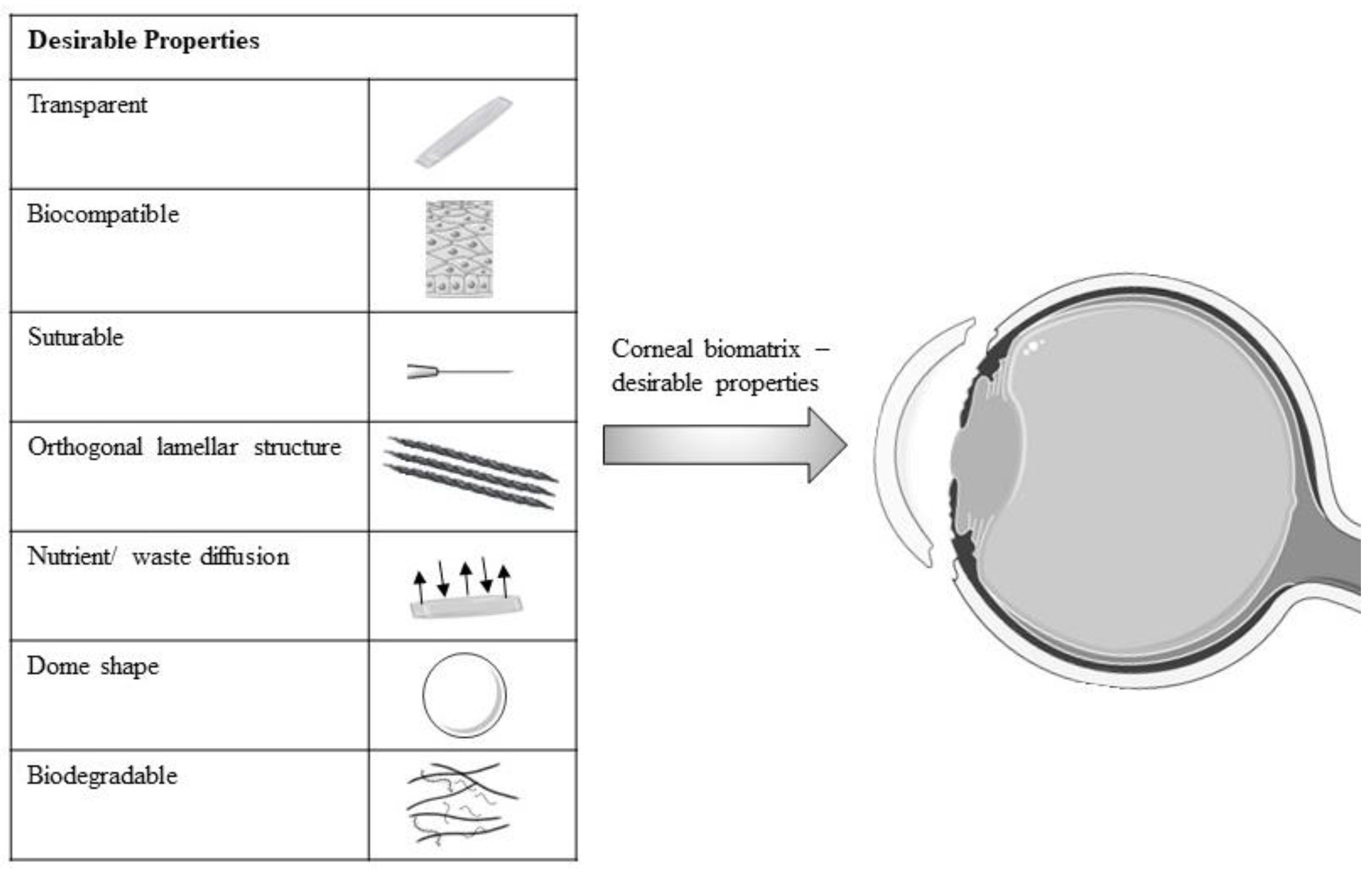
Recently, several studies have reported the improvement of cell–biomatrix interactions via surface alterations of the biomatrix. The biomatrix surface with moderate hydrophilicity, irregular structure and cationic charge was likely to be attached and grown by the LESCs and CECs [63,64]. Some studies have exploited the biocompatibility properties of collagen by incorporating or coating the collagen onto another biomatrix to increase the biomatrix biocompatibility [40,65]. All modified biomatrix with their desirable properties for corneal engineering are shown in Figure 2.
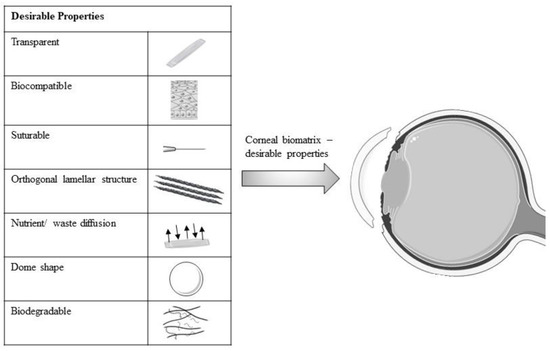
Figure 2.
Schematic diagram of desirable properties of modified biomatrix for corneal engineering. Figure 2 was partly generated using Servier Medical Art, provided by Servier, licensed under a Creative Commons Attribution 3.0 unported license.
A literature search was conducted to identify the recent modifications that have been performed on collagen biomatrix within the last 10 years. This systematic review aims to discover the modification strategies to optimize collagen biomatrices for CEC and LESC carriers in the treatment of LSCD. This review will also provide insight to further explore a better and safer modification of the collagen biomatrix for CECs and LESCs in future studies.
2. Materials and Methods
2.1. Search Strategy
This systematic review was performed to identify the relevant studies of recent modifications on collagen biomatrix and the efficacy of these modifications on the physicochemical properties and biocompatibility of the biomatrix toward CECs or LESCs, both in vitro and in vivo. Briefly, this review was constructed based on PRISMA guidelines to ensure its quality and transparency [66]. Five separate databases including Scopus (Elsevier, Amsterdam, NH, The Netherlands), Web of Science (WoS) (Clarivate Analytics, Philadelphia, PA, USA), PubMed (National Center for Biotechnology Information, NCBI, Bethesda, MD, USA), Wiley (John Wiley and Sons, Inc., Hoboken, NJ, USA), and EBSCOhost (EBSCO Information Services, Ipswich, MA, USA) were systematically searched to discover the studies related to the biomaterial or bioengineering, especially in collagen biomatrix as a biomatrix of CECs and LESCs in corneal therapy.
These databases screened all related published journal articles. This article search was guided by the focus question formulated using the PICO strategy whereby Population (P) was in vitro and in vivo studies on the collagen biomatrix for CECs and LESCs transplantation; Intervention (I) was different modification strategies on the collagen biomatrix; Comparison (C) with other biomaterials was not applicable; and Outcome (O) was physicochemical and cellular characteristics of the biomaterials studied towards CECs or LESCs (in vitro and in vivo).
The combination of three sets of keywords (corneal epithelial cells OR CECs OR corneal epithelium OR limbal epithelium OR limbal epithelial cells OR limbal epithelial stem cells OR LESCs) AND (limbal deficiency OR limbal stem cell deficiency OR LSCD OR corneal limbal stem cell deficiency OR corneal epithelial injury) AND (collagen OR collagen biomatrix OR collagen bio scaffold OR collagen scaffold) were used during the searching process of the relevant articles published.
2.2. Criteria of Selection
Only English articles were included due to limited resources for translation. Studies that provide free full-text articles published within 10 years, with a limit ranging from 2012 to 2022 were considered. Titles and abstracts that have fulfilled the topic requirements were systematically screened. Articles related to humans were included as the relevant basis for the scope of this review. All research articles related to collagen as a component or a part of the biomatrix for CECs and LESCs were also included. All secondary literature and any original article that involved clinical studies were removed. Any studies that focused on the other fields except for physicochemical properties and biocompatibility (in vitro and in vivo) were also omitted.
2.3. Management of Data Extraction Table
Articles were screened and underwent three phases to be selected as part of this systematic review. The first phase involved article title screening that meets the requirement of the topic of interest. The title that did not match the inclusion criteria was removed. The next phase involved the elimination of unrelated articles based on inclusion criteria followed by the removal of all identical articles. The last steps involved omitting the articles that did not meet the inclusion criteria after full-text reading by two independent reviewers. Two reviewers independently assessed the specified inclusion and exclusion criteria of selected published articles to guarantee neutrality in the selection of the final articles.
This was accompanied by a discussion among the reviewers to obtain a consensus on the discrepancies that emerged during the assessment of the articles. Extracted information, as outlined in the data extraction table for in vitro study, are as follows: (1) Author and year published; (2) Type of biomatrix; (3) Modification techniques; (4) Type of cells used; (5) Test and result (physicochemical properties); (6) Test and result (in vitro biocompatibility); and (7) Conclusion. The data extraction table for in vivo study was outlined as follows: (1) Author and year published; (2) Type of biomatrix; (3) Modification techniques; (4) Animal model/injury; (5) Test and result (in vivo); and (6) Conclusion. This review is not suitable to be published in PROSPERO as it included in vitro studies. For quality assessment, this review was carried out systematically, employing the critical appraisal instrument [67]. Each item in the appraisal instrument for each selected study was also discussed by independent reviewers.
3. Results
3.1. Searching Result
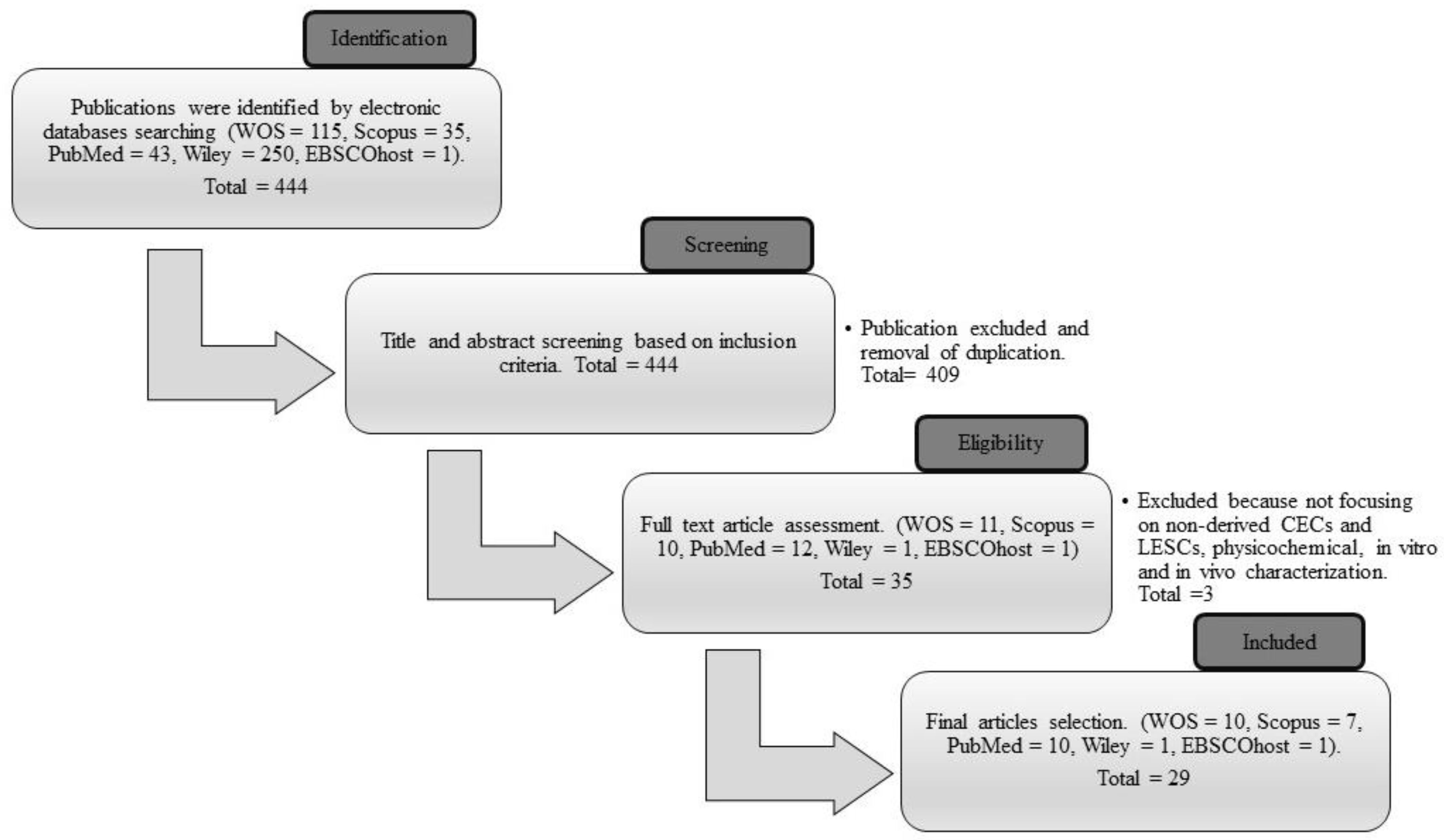
The combination of three sets of keywords during the searching process successfully identified 444 articles as potentially relevant. A total of 409 articles that did not fulfill the inclusion criteria and that were duplicates were removed during the title and abstract screening process. From the remaining 35 articles, the reviewers omitted six more articles that did not meet the requirements of the inclusion criteria. After the selection process, 29 articles were included in the data extraction table, of which ten articles were acquired from WOS, seven articles from Scopus, ten articles from PubMed, one article from Wiley, and one article from EBSCOhost. The article screening and selection process is summarized in Figure 3.
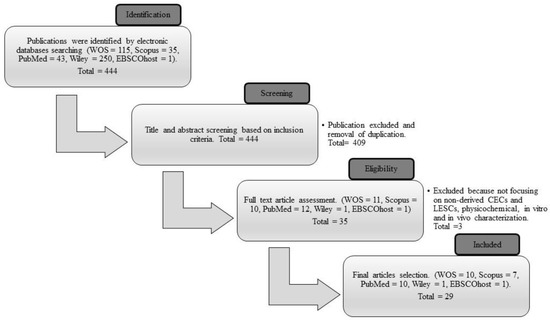
Figure 3.
The process flow of the selection of the final articles from Web of Science (WOS), Scopus, PubMed, Wiley, and EBSCOhost databases.
3.2. Study Characteristics
In this review, the search successfully finalized the studies related to the modification of collagen biomatrix and its effect on CECs or LESCs. To summarize the selected articles from 2012 to 2022, eight studies aimed to develop a new formulation for collagen biomaterials [68,69,70,71,72,73,74,75], whereas twelve studies aimed in improving or characterizing current collagen biomaterials, including the improvement of fabrication methods to produce better collagen biomatrix [27,35,51,58,59,60,61,76,77,78,79,80].
Four studies investigated the interaction of corneal cells on collagen biomaterials with other biological molecules and cells [81,82,83,84]. Meanwhile, four studies exploited the biological benefit function of collagen by the combination of different biomatrices to improve its function as a cell carrier [40,85,86,87]. From these articles, several types of cells were used, such as CECs, LESCs alone or a co-culture of both cells, and the combination of one of these cells with stromal cells (corneal stromal stem cell or limbal fibroblast). Most of these cells were primarily acquired from the cornea of a human cadaveric donor, rabbit, porcine, mini pig (Gottingen), bovine, and mouse. They were freshly obtained from the corneal rim, immortalized, primary or cell line.
All these studies reported various modifications to the collagen biomatrix by extraction of collagen from new sources, physical modification, crosslinkers, or incorporation with other cells and biomolecules. Some researchers also incorporated collagen into another biomatrix. The new source of collagen biomatrix that was obtained is through the decellularization of the bovine eyeballs, porcine conjunctiva and fish scale, production of the synthetic collagen peptide, or modification of collagen’s methacrylate group. The physical modification that performed was compression by using different compressors, embedding the decellularized corneal lenticule (dCL) with compressed collagen, surface patterning or vitrification process.
In the last 10 years, several researchers have used the following crosslinkers: polyethene glycol (PEG), N-hydroxysuccinimide (NHS), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide-N-hydroxysuccinimide (EDC-NHS), 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methyl-morpholinium chloride (DMTMM), methacyloyloxyethyl phosphorylcholine (MPC), or a hybrid crosslinker. Others incorporated different components, such as fibronectin (FN), laminin, stromal cells, ascorbic acid, or stem cell factor (SCF)/C-kit, into the collagen biomatrix. Some studies also incorporated collagen into another biomatrix, such as poly(lactide-co-glycolide) (PLGA) biomatrix, dopamine hydrazone biomatrix-crosslinked hyaluronic acid (HA-DOPA), poly-L/DL-lactic acid (PLA) films, or silk film.
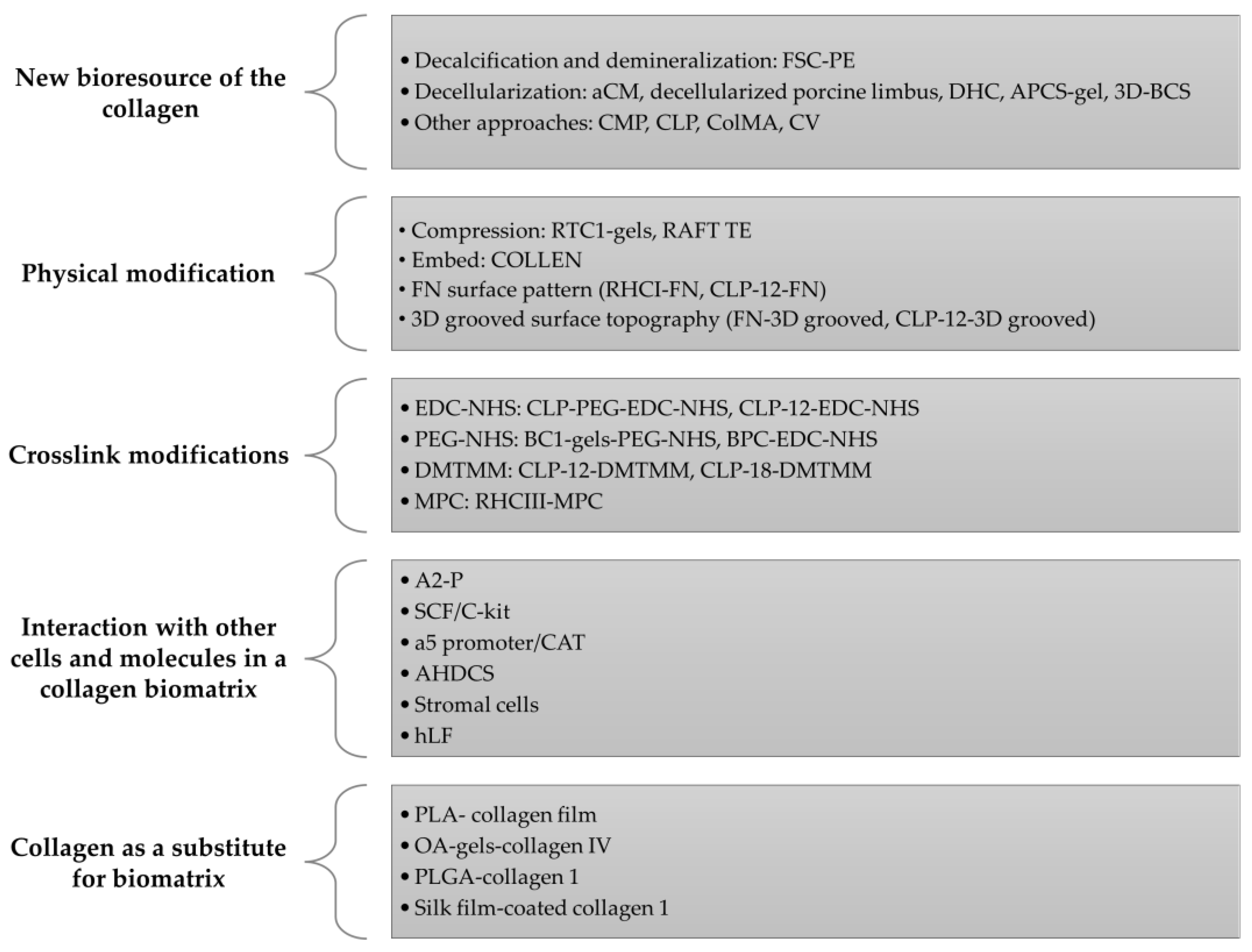
Ten studies were conducted both in vitro and in vivo: two articles involved an in vivo study only, whereas the remainder only involved in vitro study. Ultimately, the selected articles demonstrated optimization of the collagen biomatrix and its effect on the physicochemical and biocompatibility of the biomatrix towards the CECs and LESCs which affect its cellular biocompatibility differently. All articles are summarized in Table 1 for the in vitro study and in Table 2 for the in vivo study. Figure 4 shows an overview of the recent modification performed on the collagen biomatrix in the last 10 years based on the selected articles.

Table 1.
Description of selected in vitro studies on the modified collagen biomatrix.

Table 2.
Description of selected in vivo studies on the modified collagen biomatrix.

Figure 4.
Schematic diagram on the methods or strategies used in the modification of the collagen biomatrix for the last 10 years based on selected articles.
4. Discussion
4.1. New Bioresource and Its Efficacy on CECs and LESCs
It is vital to tailor the collagen source during the development of the biocompatible biomatrix due to the presence of various amino acids in the collagen, depending on the species and tissue sources [1,6,9,10]. This affects the final characteristic, physical properties, and biocompatibility of the biomatrix [90,91]. Decellularization is one of the approaches that is currently being used to produce a new collagen source for the biomatrix.
A previous study by Zhao et al. (2014) decellularized conjunctiva to produce an acellular conjunctiva matrix (aCM) as LESCs carrier [70]. The conjunctiva has a high degree of similarity to the cornea as both are derived from the epidermal ectoderm. Compared to the denuded amniotic membrane (dAM), the aCM biomatrix has better physical characteristics and is biocompatible with CECs. In vivo, the aCM could reconstruct the ocular surface in LSCD rabbits without neovascularization, inflammation, or oedema [70].
A study by Park et al. (2019) also decellularized corneal stromal tissue from bovine eyeballs and produced a three-dimensional (3D) bioprinted decellularized collagen sheet (3D-BDCS), which could re-epithelialize the damaged epithelial layer within a few days [71]. Moreover, decellularized porcine limbus and re-cellularized Statens Seruminstitut Rabbit Cornea (SIRC) limbal epithelial cell line and human adipose-derived mesenchymal stem cells (hADSCs) could produce a biomatrix with a high content of collagen IV, which are able to regenerate the stratified epithelium [75]. This is due to collagen IV affecting the CEC transcriptional factor, which plays a significant role in CEC adhesion and migration properties [92,93].
Fish scale collagen, isolated from the fish scale of fresh fish (L. calcarifer) caught from the catch, is another new collagen bio-source that is rich in collagen I. Following coating with polyethene (FSC-PE), it possesses favorable physical strength, transparency, and biocompatibility with corneal cells [68,94]. FSC-PE allows proper epithelialization due to collagen I up-regulation of the specific gene, which regulates cell viability, attachment, and differentiation. The porous nature of FSC-PE also supported the viability and differentiation of LESCs and enhanced CEC migration and proliferation [35,68,94,95].
Collagen mimetic peptide (CMP) or collagen-like peptide (CLP) is another alternative collagen source for the development of biomatrices [72]. CMP promotes the realignment of damaged collagen, which accelerates wound closure in vivo. This is due to CMP being a short synthetic collagen peptide that is able to intercalate into damaged endogenous collagen I in vivo [96,97]. CMP is also recognized to enhance CEC density and the re-epithelialization process with a better organization of epithelial layers [69,72,77].
Moreover, Qin et al.(2021) produced collagen methacrylate (ColMA) by modifying collagen with a methacrylate group, followed by photo-crosslinking [80]. ColMA is a transparent biomatrix, with high-pressure overload capacity, and is compatible with hCECs. Nanogranules from dislodging ColMA adhere to stromal tissue, promoting re-epithelization, reducing myofibroblast activation, and decreasing scar formation.
During the last 10 years, all new bioresources of collagen, including FSC-PE, aCM, and 3D-BDCS, had the potential to be developed as CEC or LESC carriers. An alternative extracellular matrix (ECM) to the macro-molecule collagen, including CMP and ColMA, is an attractive biomaterial and suitable to be developed as a biocompatible biomatrix for CECs or LESCs.
4.2. Physical Modification of the Biomatrix
Over the past 10 years, several researchers have performed physical modifications to improve the mechanical stability of the collagen biomatrix. The collagen biomatrix was modified through compression technique by Jones et al. (2012) [59] and Xeroudaki et al. (2020) [58]. They found that the compression of collagen hydrogel bioengineered porcine collagen (BPC) crosslinked with EDC-NHS reduced the water content, thus permitting control of the collagen concentration, stiffness, mechanical strength, and surface topography, which contributes to its biocompatibility with corneal cells [49,59,60,61]. The compressed BPC supports the proliferation and maintains the normal morphology of the hCECs (in vitro and in vivo) [58,59].
A study by Gouveia et al. (2019) [60] reported that plastic compression of the collagen hydrogel, i.e., the real architecture for 3D tissue equivalent (RAFT TE), also likely supported the differentiation of the LESCs via mechanotransduction-dependent pathways, whereby Yes-associated protein (YAP) supported the viability of the differentiated CECs. However, RAFT TE promotes the migration of LESCs which maintained a single monolayer, but few stratification cells became round and detached from the basal sheet. These cause RAFT TE to have a low viable differentiated CEC number and flat stretched CEC morphology compared to the uncompressed softer collagen hydrogel [60].
Therefore, LESC maintenance is highly dependent on the softer biomechanical limbus niche region properties which are opposed to the relatively stiff corneal central [98,99,100,101]. This is due to the LESCs niche requiring a specialized microenvironment to maintain the undifferentiated LESCs by slowing their migration rate and preserving their proliferative and stratification capabilities [60,79]. However, these are opposed by studies by Massie et al. [61] and Kureshi et al. (2014) [78]. They showed that the compressed RAFT TE supported the attachment, viability, and proliferation of undifferentiated LESCs in vitro [59,78].
Hong et al. (2018) [27] have conducted another physical modification of collagen bio-composite which embedded the decellularized corneal lenticule (dCL) with compressed collagen as a LESC carrier known as COLLEN. COLLEN takes the biological advantages from compressed collagen and the mechanical properties of the dCL. COLLEN supports the LESC and hCEC attachment, expansion, morphology, and functions, which were similar to the compressed collagen. In vivo, COLLEN-based limbal graft was stably grafted with native limbal region tissues without neovascularization, oedema, conjunctivalization, and inflammation. COLLEN also support multilayered differentiated CECs 2 weeks post-implantation while maintaining the putative stem cell markers on the limbal region compared to central cornea [27].
Another study by Haagdorens et al. (2019) [77] reported that surface modification of the FN pattern on CLP hydrogels influenced the pattern of cell proliferation. Yuncin et al. (2021) [87] stated that parallel ridge on silk film-coated collagen I enhanced CEC growth, spreading and promoting wound recovery. Surface patterning and surface topography, including the curvature of the biomatrix, also affect the adhesion, proliferation, and gene expression of CECs [102].
The vitrification process is another physical modification approach to develop a rigid glassy material collagen vitrigel (CV) biomatrix. It is a thin membrane composed of high-density organized meshwork type I collagen fibrils that has superior mechanical properties, is non-degradable, has a stable water content, and has optically transparent and supporting corneal cells [35,103]. In vivo, CV promotes the regeneration of healthy CECs and LESCs with a low inflammatory response and reduced neovascularization (5 weeks post-surgery) [35].
In conclusion, the compression on the collagen biomatrix was able to increase the physical strength and affected the behavior of cultivated CECs or LESCs. Most of the researchers reported that the compressed collagen biomatrix supported the viability of CECs, but it is not suitable for LESC growth. This is due to the LESCs requiring soft biomechanical region properties of the collagen biomatrix to preserve proliferative and stratification capabilities of the undifferentiated LESCs. However, this was opposite to the result obtained by Kureshi et al. (2015) [83]. Thus, further study is needed to explore the mechanism behind these corneal cells’ behavior. In addition, the surface modification of collagen hydrogel also affects the growth and behavior of cultivated CECs and LESCs. CV and COLLEN are other creative approaches in developing optimal designs of the CEC or LESC biomatrix.
4.3. Crosslinking Effect on the Biocompatibility of the Construct towards CECs/LESCs
Crosslinking is another approach in improving the mechanical stability of the collagen biomatrix. Different cross-linkers have different effects on the mechanical properties in terms of biocompatibility of the biomatrix towards corneal cells, especially CECs and LESCs. Figure 5 shows the overview of the chemical cross-linker used in the last 10 years based on selected articles. Amide-based crosslinkers, such as EDC-NHS, are commonly used with collagen biomatrix as they mimics the lysine-based crosslinker which is naturally present in collagen [104]. EDC-NHS affects the GxOGER sequences of collagen molecules [58], did not remain as a part of the protein structure post-crosslinking [105,106], and improved the porous and interconnected structure of the collagen biomatrix [43], promoting cell attachment, proliferation, and viability of the attached cells.
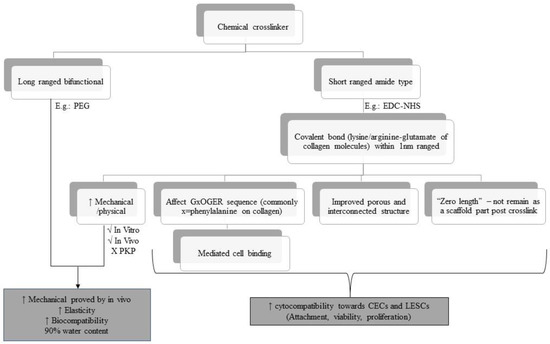
Figure 5.
Overview of the chemical cross-linkers used over the last 10 years based on selected articles.
EDC-NHS enhances the physical properties of the collagen biomatrix, showing sufficient mechanical strength during subcutaneous implantation and implantation in the rabbit cornea, but insufficient strength penetrating keratoplasty (PKP) [58,106,107,108]. This is due to the functional group located on the adjacent collagen microfibril being too far to be bridged by EDC-NHS, as EDC-NHS only can link within 1.0 nm from each other [108]. These drawbacks could be overcome by hybridization of EDC-NHS (amide-type crosslinker) with a bifunctional cross-linker such as PEG, which provides synergistic effects on the physical and biological properties of collagen biomatrix [109,110].
Jangamreddy et al. (2018) [69] and Rafat et al. (2008) [111] managed to conjugate the CLP-EDC-NHS to PEG (four arms or eight arms) to further improve the mechanical strength and promote a stable biomatrix for regeneration. This hybrid crosslinked hydrogel (CLP-PEG-EDC-NHS) enhanced the mechanical strength and elasticity by 100% and 20%, respectively, compared to the non-hybrid biomatrix. The hydrogel comprised over 90% water, compared to the cornea that has 78% water, which contributes to its biocompatibility. It also did not cause any cytotoxicity effects toward CECs as there was minimal CECs death at 48 hours post cell culture and it was able to regenerate the neo cornea with healthy regenerated functional CECs in vivo [69]. Although the tensile strength of CLP-PEG-EDC-NHS was not as high as the control recombinant human collagen type III (RHCIII) conjugated to MPC (RHCIII-MPC) implants, it was more elastic compared to the RHCIII-MPC, which allowed them to withstand the grafting procedure [69,112].
Fernandes-Cunha et al. (2020) [51] found that the concentration and arm number of PEG affect the transparency, directly proportional to the storage modulus and degradation profile of the PEG-crosslinked collagen biomatrix. The highest storage modulus achieved was at 8% (eight arms of PEG) but decreased at 16% PEG content. This may be due to the saturation effect, where beyond a certain concentration of PEG, no further crosslinking was achieved, and any additional PEG reduced the crosslinking density and transparency via inadequate macromolecular mixing, leading to matrix heterogeneity.
In addition, this biomatrix has low cytotoxicity effects, as almost 100% of human immortalized corneal epithelial cells (iCECs) confluent after 2 days post cultivated on all biomatrices. PEG arm concentration and arm number were directly proportional to the iCECs adhesion and proliferation except in 16% of PEG, respectively. The high PEG concentration causes the reduction of biomatrix porosity, which restrains the mobility of the polymer network and thus reduced the proliferation of corneal cells [111]. The presence of iCECs improved the transmittance in the four and six arms by 16% PEG. PEG also affect the alignment of the collagen fiber which affects the iCEC behaviors [112].
Haagdorens et al. (2019) [77] also used an EDC-NHS crosslinker for the biomatrix of human corneal epithelial cells (hCECs) and human limbal epithelial stem cells (hLESCs). They used seven different collagen-derived hydrogels (recombinant human collagen type I (RHCI) and CLP hydrogel) with EDC-NHS or DMTMM cross-linker as a carrier of immortalized human corneal epithelial cells (ihCECs) and primary hLESCs. All these collagen hydrogels met the physical criteria of a good biomatrix for CEC and LESC carriers. RHCI and CLP hydrogel, irrespective of the crosslinker type, except for CLP-12-EDC, is biocompatible towards ihCECs as there was minimal cell death that supported the metabolic activity, attachment, and proliferation of ihCECs and primary hLESCs. However, these biomatrices did not promote LESC differentiation except for CLP [77].
Overall, in the last 10 years, the crosslinker used on the collagen biomatrix as a carrier of CECs or LESCs at its optimal crosslinker concentration was able to improve the mechanical stability of the biomatrix and biocompatibility with CECs or LESCs. The use of a high concentration and arm numbers of the crosslinker leads to the saturation effect which could cause a decrease in the transparency and porosity of the biomatrix and reduce the proliferation of the corneal cells. More novel crosslinkers and techniques need to be explored, either alone or in combination with chemical, enzymatic, or physical crosslinker methods, which may give synergistic effects as a CEC or LESC carrier.
4.4. Interaction of CECs/LESCs with Other Cells and Molecules in a Collagen Biomatrix
Other components, such as FN, laminin, stromal cells, ascorbic acid, and SCF/C-kit, have been incorporated into the biomatrix as they are able to promote re-epithelization [6,10,13,79]. Wilson et al. (2014) [82] investigated the effects of FN on the cultured CECs by seeding the adult porcine CECs on the FN-coating rat tail collagen type 1 hydrogel (RTCI-gel) encapsulated adult human derived corneal stromal (AHDCS) (RTCI-gel-FN-coated–AHDCS). RTCI-gel-FN-coated–AHDCS supported the CEC viability and maintained the normal cobblestone morphology with a tight cell–cell junction. This was due to FN-binding integrin a5b1 promoting the CEC adhesion and migrating to cover the uncovered surface FN [13,113,114,115,116]. It mimicked the physiological cornea, as FN is a temporary ECM that is present in abundance during early corneal wound healing and is progressively replaced by collagen and laminin from the basal membrane as wound healing progresses [10,13,77,117,118,119].
In contrast, CECs appeared to be much smaller, less flattened, and lacking the tight cell–cell junction when cultured on the biomatrix treated with wortmannin (epithelial stroma interaction inhibitor) [82]. These indicated that the mutual interaction of CECs with stromal cells encapsulated in the hydrogel was needed to support the CEC growth and enhance epithelium multilayered organization [82,83,120].
This was also supported by Kureshi et al. (2010) [121], Massie et al. (2015) [84], and Zhang et al. (2015) [122]. Their studies showed that stromal cells/limbal fibroblast cells successfully enhanced the LESC growth on the collagen biomatrix, which mimics in vivo corneal arrangement. It also was due to the LESC differentiation depending on the mediated expression of the Wnt/B catenin of bone morphogenetic protein (proliferative marker) secreted by the stromal cell [60]. Massie et al. (2015) [84] reported that human limbal fibroblast (hLF) (quiescent) was quite safe to be transplanted with hLESCs on the collagen biomatrix. hLF was less activated in terms of stroma and basement membrane remodeling and did not progress toward a scarring-like phenotype compared to diseased fibroblasts (dFib).
Another molecule that is important for corneal wound healing process is ascorbic acid. According to Chen et al. (2017) [81], the presence of L-ascorbic acid 2-phosphate (A2-P), the stable form of derivative ascorbic acid, increased the stemness of mouse corneal epithelial stem/progenitor cells (TKE-2) by regulating the ECM components (collagen). A2-P also enhanced the stemness and proliferation of TKE2. SCF/C-Kit is another biomolecule that is present in normal mouse cornea and plays an important role in promoting corneal wound healing. According to Miyamoto et al. (2012) [89], SCF/C-kit enhanced cell attachment to FN, laminin, and collagen type IV during corneal wound healing via the induction of the avidity and affinity of integrin members in vitro.
In conclusion, corneal stromal/keratocyte cells promoted CEC or LESC growth on the collagen biomatrix. The presence of other components, including FN, ascorbic acid, and SCF/C-kit, enhanced the biocompatibility of the collagen biomatrix towards CECs or LESCs.
4.5. Collagen as a Substitute for Biomatrix
A study by Chakraborty and colleagues [76] reported that a collagen IV-coated surface improved LESC attachment, growth, and proliferation when compared to the untreated plastic surface. Many researchers have investigated the benefits of biocompatibility properties of collagen type IV as the main component of the biomatrix. This includes incorporating or coating collagen IV onto another biomatrix to develop the optimal corneal biomatrix design.
De la Mata et al. (2019) [85] studied the biological properties of collagen IV by incorporating the collagen into a poly-L/DL-lactic acid (PLA) biomatrix. Functionalizing PLA films with collagen IV (70:30) improved LESC attachment, selection, and enrichment and maintained the undifferentiated LESC phenotype with a homogenous polygonal morphology [85]. This finding was supported by Wright et al. (2014) [40], who incorporated collagen IV into an oxidized alginate biomatrix. Incorporating collagen IV further enhanced the CEC viability and provided a niche environment that supports the re-epithelization process and may serve as viable wound healing bandages for the damaged cornea.
Moreover, a study by Kayiran Celebier et al. (2020) [86] exploited the biological benefits of collagen I by incorporating collagen I into poly(lactide-co-glycolide) (PLGA) polymers loaded with naproxen sodium (NS). This incorporation of collagen I did not affect the degradation period or the mechanical strength, but improved hydrophilicity and enhanced the water uptake capacity compared to the plain biomatrix [64,123]. As a result, the PLGA biomatrix coated with collagen I improved CECs attachment, proliferation, and viability [86,124], whereas the incorporation of collagen I coating the silk film also enhanced biomatrix biocompatibility toward CECs [87].
In conclusion, exploitation of the biological benefits of collagen IV or collagen I by incorporating it into another biomatrix can improve the biomatrix biocompatibility toward CECs or LESCs, which has great potential to be further used in in vivo studies and clinically.
4.6. In Vivo Application of a Recently Developed Collagen Biomatrix and Its Efficiency in Corneal Therapy
Recently, many researchers proved that most of the modified collagen scaffolds involved in in vivo study are biocompatible and have the potential to be translated into corneal therapy using a different clinical setting. The main aspect that needs to be considered during the development of the collagen biomatrix for corneal therapy is its biocompatibility with CECs and LESCs [55]. It is closely related to the physicochemical properties of collagen biomatrix and the modification strategies that have been tailored on the collagen biomatrix.
APCs-gel is one of the collagen biomatrices that is produced by the decellurization of the porcine cornea. APCS-gel has potential for clinical use as it maintains the critical characteristics of the native cornea in vivo. APCS-gel promotes faster re-epithelization and this enhances corneal wound healing in vivo [73]. CMP is another alternative to the collagen source which is made up of short synthetic collagen peptides. This CMP is topically applied and able to enhance the closure of the corneal wound by re-alignment of the underlying damaged collagen on the ocular surface in vivo. CMP also promotes re-epithelization by accelerating basal epithelium adherence and promoting CECs density to form organized epithelial layers in the wound area [72].
ColMA hydrogel is a novel therapeutic sutureless wound dressing to repair partial thickness in corneal defects. ColMA hydrogel serves as a physical barrier to prevent bacterial infection and corneal wound dehydration but also produce nanogranules that highly promote re-epithelization. Thus, ColMa hydrogel has great potential to be translated into wound dressing for corneal regenerative therapy [80]. COLLEN-based limbal graft was able to reconstruct the ocular surface of LSCD in a rabbit model. COLLEN can be stably sutured onto the cornea and is highly resistant to biodegradation. It is also biocompatible with ocular transplantation as it supports re-epithelization and limbal reconstruction without inducing neovascularization, stromal oedema, and inflammation. In fact, it also has regenerative properties as it maintains the stemness and normal proliferation activity of LESCs [27].
BCI-gel-PEG-NHS provides an in situ hydrogel over stromal keratectomy injury without the need for any sutures. BCI-gel-PEG-NHS supported the growth, migration, and formation of a multi-layered epithelium surface on the wound area [51]. BPC-EDC-NHS, which is formed by compression, has suitable mechanical properties and proved to be safely integrated into the corneal wound area. BPC-EDC-NHS allowed the migration and population by host cells (CECs and stromal cells) while maintaining corneal transparency and thickness after surgery. Thus, this key point has a great impact in translating the collagen into corneal stromal replacement therapy [58].
In conclusion, most of the in vivo research proves that the current modification strategy is efficient to be translated into corneal therapy. All modifications that have been introduced in in vivo studies enhanced its carrier and regenerative function. However, the underlying mechanisms remain elusive, hence, these modifications still require further investigation to evaluate the potential modified collagen biomatrix as an alternative treatment for corneal repair.
5. Conclusions
In conclusion, various modification strategies have been performed to optimize the collagen biomatrix for CEC and LESC carriers in the treatment of corneal defects. Based on the comparison of all modified collagen, we have identified that different modification strategies provide different effects on the CECs and LESCs. Despite the variability in the methodological approaches, the reviewers suggested that different scaffold modification will eventually contribute to the improvement of scaffold physicochemical and biocompatibility properties towards CEC and LESCs. The presence of the new bioresource of collagen within the last 10 years has the potential to be developed as a biocompatible biomatrix. The physical modification and crosslinking methods improved mechanical strength of the collagen biomatrix and, thus, have a remarkable effect on the CECs and LESCs. The incorporation of the corneal stromal/keratocyte, FN, ascorbic acid, laminin, and SCF/C-kit into the collagen biomatrix enhanced the scaffold biocompatibility towards CECs and LESCs. Some studies also exploited the biological benefit of collagen by incorporating one type of collagen into another biomatrix. These studies reported a great improvement in the biocompatibility of the biomatrix towards CECs and LESCs. This review provides an insight into the current modifications strategies in optimizing the collagen biomatrix for corneal therapy. We believe that a certain scaffold modification is essential in supporting the transition of collagen biomatrix-based therapies into clinical trials.
Author Contributions
Conceptualization, N.A.R., R.C.M., M.B.F., N.A.G. and W.H.W.A.H.; methodology, N.A.R., R.C.M., M.B.F. and W.H.W.A.H.; validation, N.A.R., M.B.F., N.A.G., W.H.W.A.H. and R.C.M.; formal analysis, N.A.R.; investigation, N.A.R., R.C.M., M.B.F., N.A.G. and W.H.W.A.H.; data curation, N.A.R., R.C.M., M.B.F., N.A.G. and W.H.W.A.H.; writing—original draft preparation, N.A.R.; writing—review and editing, R.C.M., M.B.F., N.A.G., M.R.B., N.M.H. and W.H.W.A.H.; visualization, N.A.R., R.C.M., M.B.F., N.A.G. and W.H.W.A.H.; supervision, M.B.F., N.A.G., R.C.M., M.R.B., N.M.H. and W.H.W.A.H.; project administration, W.H.W.A.H.; funding acquisition, W.H.W.A.H. All authors have read and agreed to the published version of the manuscript.
Funding
This study was funded by Fundamental Research Grant Scheme (FRGS) under the Ministry of Higher Education of Malaysia (MOHE), grant number FRGS/1/2020/SKK0/UKM/02/16.
Institutional Review Board Statement
The study was conducted according to the ethical guidelines underpinned by right-based theories, whereby it adheres to the principles of beneficence, nonmaleficence, justice, honesty, and gratitude. The study was approved by the Institutional Review Board (or Ethics Committee) of Universiti Kebangsaan Malaysia (protocol code JEP-2021-383 on 25 August 2022).
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
Acknowledgments
We would like to thank the Ophthalmology department, Pathology department, Anatomy department, Centre of Tissue Engineering and Regenerative Medicine, Institute of Microengineering and Nanoelectronics and Faculty of Medicine UKM for providing resources to complete this review.
Conflicts of Interest
The authors report no conflict of interest in this work.
Abbreviations
| 3D | Three-dimensional |
| 3D-BDCS | 3D bioprinting decellularized collagen sheet |
| A2-P | L-ascorbic acid 2-phosphate |
| aCM | Acellular conjunctiva matrix |
| AHDCS | Adult human derived corneal stromal |
| APCS-gel | Acellular porcine corneal stroma hydrogel |
| BCI-gel-PEG-NHS | Bovine collagen type 1 hydrogel crosslinked to PEG-NHS |
| BMP | Bone morphogenetic protein |
| BPC | Bioengineered porcine collagen |
| CAT | Chloramphenicol acetyltransferase |
| CECs | Corneal epithelial cells |
| CK | Cytokeratin |
| CLP | Collagen-like peptide |
| CLP-12 EDC | Collagen-like peptide type 12 crosslinked with 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride |
| CLP-PEG-EDC-NHS | Collagen-like peptide crosslinked with 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide-N-hydroxy succinimide and conjugated to the polyethene glycol |
| CMP | Collagen mimetic peptides |
| COLLEN | Decellularized corneal lenticule embedded compressed collagen |
| ColMA | Collagen methacrylate |
| CSSCs | Corneal stromal stem cells |
| CV | Collagen vitrigel |
| dAM | Denuded amniotic membrane |
| dCL | Decellularized corneal lenticule |
| dFib | Diseased fibroblasts |
| DHC | Decellularized human corneal tissue remnants |
| DMTMM | 4-(4,6-Dimethoxy-1,3,5-triazin-2-yl)-4-methyl-morpholinium chloride |
| ECM | Extracellular matrix |
| EDC | 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride |
| EDC-NHS | 1–Ethyl–3-(3-dimethylaminopropyl) carbodiimide-N-hydroxysuccinimide |
| FGF | Fibroblast growth factor |
| FN | Fibronectin |
| FSC-PE | Fish scale collagen coated with polyethene |
| FTIR | Fourier-transform infrared spectroscopy |
| H&E | Haematoxylin and eosin stain |
| HA-DOPA | Dopamine hydrazone scaffold-crosslinked hyaluronic acid |
| hADSCs | Human adipose-derived mesenchymal stem cells |
| HAM | Human amniotic membrane |
| hCECs | Human corneal epithelial cells |
| hLESCs | Human limbal epithelial stem cells |
| hLF | Human limbal fibroblast |
| Hyp | Hydroxyproline |
| IF | Immunofluorescence |
| IHC | Immunohistochemistry |
| ihCECs | Immortalized human corneal epithelial cells |
| K | Keratin |
| LEPC | Limbal epithelial progenitor cells |
| LESCs | Limbal epithelial stem cells |
| LM | Limbal melanocytes |
| LMSC | Limbal mesenchymal stromal cells |
| LSCD | Limbal stem cell deficiency |
| MMP | Matrix metalloproteinases |
| MPC | Methacyloyloxyethyl phosphorylcholine |
| MTT | Tetrazolium salt (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NHS | N-hydroxysuccinimide |
| NS | Naproxen sodium |
| OA-gel-CIV | Oxidized alginate hydrogel incorporated with collagen IV |
| OCT | Optical coherence tomography |
| PE | Polyethene |
| PEG | Polyethene glycol |
| PKP | Penetrating keratoplasty |
| PLA | Poly-L/DL- lactic acid |
| PLGA | Poly lactic-co-glycolic acid |
| RAFT TE | Real architecture for 3D tissue equivalent |
| RAFT TE-CI | Real architecture for 3D tissue equivalent treated with collagenase I |
| RAFT TE-NT | Real architecture for 3D tissue equivalent treated without treatment |
| RAFT TE-PBS | Real architecture for 3D tissue equivalent treated with phosphate-buffered saline |
| rCECs | Rabbit corneal epithelial cells |
| RHC | Recombinant human collagen |
| RHCI | Recombinant human collagen type I |
| RHCIII | Recombinant human collagen type III |
| RTCI-gel | Rat tail collagen type I hydrogel |
| RT-PCR | Reverse transcription polymerase chain reaction |
| SCF | Stem cell factor |
| SDS | Sodium dodecyl-sulphate |
| SEM | Scanning electron microscope |
| SIRC | Statens Seruminstitut Rabbit Cornea |
| TEM | Transmission electron microscopy |
| TGF-β1 | Transforming growth factor beta 1 |
| TKE2 | Mouse corneal epithelial stem/progenitor cells |
| TMSCs | Human turbinate-derived mesenchymal stem cells |
| WOS | Web of Science |
| YAP | Yes-associated protein |
References
- DelMonte, D.W.; Kim, T. Anatomy and physiology of the cornea. J. Cataract Refract. Surg. 2011, 37, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Maurice, D.M. The cornea and sclera. In Vegetative Physiology and Biochemistry, 1st ed.; Davson, H., Ed.; Elsevier: Amsterdam, The Netherlands, 1962; Volume 1, pp. 289–368. [Google Scholar] [CrossRef]
- Sridhar, M.S. Anatomy of cornea and ocular surface. Indian J. Ophthalmol. 2018, 66, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, C.E.; Rnjak-Kovacina, J.; Kaplan, D.L. Corneal tissue engineering: Recent advances and future perspectives. Tissue Eng. Part B Rev. 2015, 21, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Espana, E.; Ti, S.; Grueterich, M.; Touhami, A.; Tseng, S. Corneal stromal changes following reconstruction by ex vivo expanded limbal epithelial cells in rabbits with total limbal stem cell deficiency. Br. J. Ophthalmol. 2003, 87, 1509–1514. [Google Scholar] [CrossRef]
- Gipson, I.K.; Stepp, M.A. Anatomy and cell biology of the cornea, superficial limbus, and conjunctiva. In Albert and Jakobiec’s Principles and Practice of Ophthalmology; Springer: Cham, Switzerland, 2022; pp. 3–30. [Google Scholar] [CrossRef]
- Lu, L.; Reinach, P.S.; Kao, W.W. Corneal epithelial wound healing. Exp. Biol. Med. 2001, 226, 653–664. [Google Scholar] [CrossRef]
- Krachmer, J.; Mannis, M.; Holland, E. Cornea, Volume Two: Surgery of the Cornea and Conjuctiva, 2nd ed.; Elsevier Mosby: Maryland Heights, MO, USA, 2005; pp. 997–1011. [Google Scholar] [CrossRef]
- Bergmanson, J.P. Anatomy and physiology of the cornea and related structures. In Contact Lens, 6th ed.; Anthony, J.P., Lynne, S., Eds.; Elsevier Mosby: Maryland Height, MO, USA, 2019; pp. 33–43. [Google Scholar] [CrossRef]
- Downie, L.E.; Bandlitz, S.; Bergmanson, J.P.; Craig, J.P.; Dutta, D.; Maldonado-Codina, C.; Ngo, W.; Siddireddy, J.S.; Wolffsohn, J.S. BCLA CLEAR-Anatomy and physiology of the anterior eye. Contact Lens Anterior Eye 2021, 44, 132–156. [Google Scholar] [CrossRef]
- Fagerholm, P. Wound healing after photorefractive keratectomy. J. Cataract Refract. Surg. 2000, 26, 432–447. [Google Scholar] [CrossRef]
- Kuo, I.C. Corneal wound healing. Curr. Opin. Ophthalmol. 2004, 15, 311–315. [Google Scholar] [CrossRef]
- Liu, C.-Y.; Kao, W.W. Corneal Epithelial Wound Healing. Prog. Mol. Biol. Transl. Sci. 2015, 134, 61–71. [Google Scholar] [CrossRef]
- Park, M.; Richardson, A.; Pandzic, E.; Lobo, E.P.; Whan, R.; Watson, S.L.; Lyons, J.G.; Wakefield, D.; Di Girolamo, N. Visualizing the Contribution of Keratin-14(+) Limbal Epithelial Precursors in Corneal Wound Healing. Stem Cell Rep. 2019, 12, 14–28. [Google Scholar] [CrossRef]
- Choi, J.S.; Joo, C.K. Wakayama symposium: New therapies for modulation of epithelialization in corneal wound healing. Ocul. Surf. 2013, 11, 16–18. [Google Scholar] [CrossRef]
- Notara, M.; Lentzsch, A.; Coroneo, M.; Cursiefen, C. The role of limbal epithelial stem cells in regulating corneal (lymph) angiogenic privilege and the micromilieu of the limbal niche following UV exposure. Stem Cells Int. 2018, 2018, 8620172. [Google Scholar] [CrossRef]
- Bonnet, C.; González, S.; Roberts, J.S.; Robertson, S.Y.; Ruiz, M.; Zheng, J.; Deng, S.X. Human limbal epithelial stem cell regulation, bioengineering and function. Prog. Retin. Eye Res. 2021, 85, 100956. [Google Scholar] [CrossRef]
- Le, Q.; Xu, J.; Deng, S.X. The diagnosis of limbal stem cell deficiency. Ocul. Surf. 2018, 16, 58–69. [Google Scholar] [CrossRef]
- Dua, H.S.; Joseph, A.; Shanmuganathan, V.; Jones, R. Stem cell differentiation and the effects of deficiency. Eye 2003, 17, 877–885. [Google Scholar] [CrossRef]
- Vieira-Potter, V.J.; Karamichos, D.; Lee, D.J. Ocular complications of diabetes and therapeutic approaches. BioMed Res. Int. 2016, 2016, 3801570. [Google Scholar] [CrossRef]
- Córdoba, A.; Mejía, L.F.; Mannis, M.J.; Navas, A.; Madrigal-Bustamante, J.A.; Graue-Hernandez, E.O. Current global bioethical dilemmas in corneal transplantation. Cornea 2020, 39, 529–533. [Google Scholar] [CrossRef]
- Behlau, I.; Martin, K.V.; Martin, J.N.; Naumova, E.N.; Cadorette, J.J.; Sforza, J.T.; Pineda, R.; Dohlman, C.H. Infectious endophthalmitis in Boston keratoprosthesis: Incidence and prevention. Acta Ophthalmol. 2014, 92, 546–555. [Google Scholar] [CrossRef]
- Duan, X.; Sheardown, H. Dendrimer crosslinked collagen as a corneal tissue engineering scaffold: Mechanical properties and corneal epithelial cell interactions. Biomaterials 2006, 27, 4608–4617. [Google Scholar] [CrossRef]
- Zhang, B.; Xue, Q.; Hu, H.-Y.; Yu, M.-F.; Gao, L.; Luo, Y.-C.; Li, Y.; Li, J.-T.; Ma, L.; Yao, Y.-F. Integrated 3D bioprinting-based geometry-control strategy for fabricating corneal substitutes. J. Zhejiang Univ.-Sci. B 2019, 20, 945–959. [Google Scholar] [CrossRef]
- Jameson, J.F.; Pacheco, M.O.; Nguyen, H.H.; Phelps, E.A.; Stoppel, W.L. Recent Advances in Natural Materials for Corneal Tissue Engineering. Bioengineering 2021, 8, 161. [Google Scholar] [CrossRef] [PubMed]
- Ahearne, M.; Fernandez-Perez, J.; Masterton, S.; Madden, P.W.; Bhattacharjee, P. Designing scaffolds for corneal regeneration. Adv. Funct. Mater. 2020, 30, 1908996. [Google Scholar] [CrossRef]
- Hong, H.; Huh, M.-I.; Park, S.M.; Lee, K.-P.; Kim, H.K.; Kim, D.S. Decellularized corneal lenticule embedded compressed collagen: Toward a suturable collagenous construct for limbal reconstruction. Biofabrication 2018, 10, 045001. [Google Scholar] [CrossRef] [PubMed]
- Man, R.C.; Yong, T.K.; Hwei, N.M.; Halim, W.H.W.A.; Zahidin, A.Z.M.; Ramli, R.; Saim, A.B.; Idrus, R.B.H. Corneal regeneration by induced human buccal mucosa cultivated on an amniotic membrane following alkaline injury. Mol. Vis. 2017, 23, 810. [Google Scholar] [PubMed]
- Rohaina, C.M.; Then, K.Y.; Ng, A.M.H.; Halim, W.H.W.A.; Zahidin, A.Z.M.; Saim, A.; Idrus, R.B. Reconstruction of limbal stem cell deficient corneal surface with induced human bone marrow mesenchymal stem cells on amniotic membrane. Transl. Res. 2014, 163, 200–210. [Google Scholar] [CrossRef]
- Rama, P.; Bonini, S.; Lambiase, A.; Golisano, O.; Paterna, P.; De Luca, M.; Pellegrini, G. Autologous fibrin-cultured limbal stem cells permanently restore the corneal surface of patients with total limbal stem cell deficiency1. Transplantation 2001, 72, 1478–1485. [Google Scholar] [CrossRef]
- Rama, P.; Matuska, S.; Paganoni, G.; Spinelli, A.; De Luca, M.; Pellegrini, G. Limbal stem-cell therapy and long-term corneal regeneration. N. Engl. J. Med. 2010, 363, 147–155. [Google Scholar] [CrossRef]
- Alaminos, M.; Sánchez-Quevedo, M.D.C.; Munoz-Ávila, J.I.; Serrano, D.; Medialdea, S.; Carreras, I.; Campos, A. Construction of a complete rabbit cornea substitute using a fibrin-agarose scaffold. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3311–3317. [Google Scholar] [CrossRef]
- Pellegrini, G.; Traverso, C.E.; Franzi, A.T.; Zingirian, M.; Cancedda, R.; De Luca, M. Long-term restoration of damaged corneal surfaces with autologous cultivated corneal epithelium. Lancet 1997, 349, 990–993. [Google Scholar] [CrossRef]
- Behaegel, J.; Ní Dhubhghaill, S.; Koppen, C.; Zakaria, N. Safety of cultivated limbal epithelial stem cell transplantation for human corneal regeneration. Stem Cells Int. 2017, 2017, 6978253. [Google Scholar] [CrossRef]
- Chae, J.J.; McIntosh, A.W.; Espinoza, F.A.; Mulreany, D.G.; Ng, S.; Takezawa, T.; Trexler, M.M.; Schein, O.D.; Chuck, R.S.; Elisseeff, J.H. Regeneration of corneal epithelium utilizing a collagen vitrigel membrane in rabbit models for corneal stromal wound and limbal stem cell deficiency. Acta Ophthalmol. 2015, 93, 57–66. [Google Scholar] [CrossRef]
- Zhu, X.; Beuerman, R.W.; Chan-Park, M.; Cheng, Z.; Ang, L.P.; Tan, D.T. Enhancement of the mechanical and biological properties of a biomembrane for tissue engineering the ocular surface. Ann.-Acad. Med. Singap. 2006, 35, 210–214. [Google Scholar] [CrossRef]
- Li, Y.; Yang, Y.; Yang, L.; Zeng, Y.; Gao, X.; Xu, H. Poly (ethylene glycol)-modified silk fibroin membrane as a carrier for limbal epithelial stem cell transplantation in a rabbit LSCD model. Stem Cell Res. Ther. 2017, 8, 256. [Google Scholar] [CrossRef]
- Dang, X.; Li, Y.; Yang, M. Biodegradable waterborne polyurethane grafted with gelatin hydrolysate via solvent-free copolymerization for potential porous scaffold material. J. Mech. Behav. Biomed. Mater. 2019, 92, 79–89. [Google Scholar] [CrossRef] [PubMed]
- de la Mata, A.; Nieto-Miguel, T.; López-Paniagua, M.; Galindo, S.; Aguilar, M.R.; Garcia-Fernandez, L.; Gonzalo, S.; Vázquez, B.; Román, J.S.; Corrales, R.M. Chitosan–gelatin biopolymers as carrier substrata for limbal epithelial stem cells. J. Mater. Sci. Mater. Med. 2013, 24, 2819–2829. [Google Scholar] [CrossRef]
- Wright, B.; De Bank, P.A.; Luetchford, K.A.; Acosta, F.R.; Connon, C.J. Oxidized alginate hydrogels as niche environments for corneal epithelial cells. J. Biomed. Mater. Res. Part A 2014, 102, 3393–3400. [Google Scholar] [CrossRef]
- Chen, D.; Qu, Y.; Hua, X.; Zhang, L.; Liu, Z.; Pflugfelder, S.; Li, D. A hyaluronan hydrogel scaffold-based xeno-free culture system for ex vivo expansion of human corneal epithelial stem cells. Eye 2017, 31, 962–971. [Google Scholar] [CrossRef]
- Islam, M.M.; AbuSamra, D.B.; Chivu, A.; Argüeso, P.; Dohlman, C.H.; Patra, H.K.; Chodosh, J.; González-Andrades, M. Optimization of collagen chemical crosslinking to restore biocompatibility of tissue-engineered scaffolds. Pharmaceutics 2021, 13, 832. [Google Scholar] [CrossRef]
- Goodarzi, H.; Jadidi, K.; Pourmotabed, S.; Sharifi, E.; Aghamollaei, H. Preparation and in vitro characterization of cross-linked collagen–gelatin hydrogel using EDC/NHS for corneal tissue engineering applications. Int. J. Biol. Macromol. 2019, 126, 620–632. [Google Scholar] [CrossRef]
- Palchesko, R.N.; Carrasquilla, S.D.; Feinberg, A.W. Natural biomaterials for corneal tissue engineering, repair, and regeneration. Adv. Healthc. Mater. 2018, 7, 1701434. [Google Scholar] [CrossRef]
- Crabb, R.A.; Chau, E.P.; Decoteau, D.M.; Hubel, A. Microstructural characteristics of extracellular matrix produced by stromal fibroblasts. Ann. Biomed. Eng. 2006, 34, 1615–1627. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.M.; Chivu, A.; AbuSamra, D.B.; Saha, A.; Chowdhuri, S.; Pramanik, B.; Dohlman, C.H.; Das, D.; Argüeso, P.; Rajaiya, J. Crosslinker-free collagen gelation for corneal regeneration. Sci. Rep. 2022, 12, 9180. [Google Scholar] [CrossRef] [PubMed]
- Varghese, T.; Jacob, J. Fish Collagen as a Potential Biocompatible Composite with Biomedical Applications. J. Sci. Technol. Manag. 2019, 12, 47–51. [Google Scholar] [CrossRef]
- Xu, S.; Zhou, S.; Mao, B.; Chen, J.; Zhang, Z. Cornea-stroma-mimicking films derived from cellulose nanocrystal templating fibrous collagen as therapeutic contact lenses. ACS Sustain. Chem. Eng. 2019, 7, 12248–12260. [Google Scholar] [CrossRef]
- Cheema, U.; Brown, R.A. Rapid fabrication of living tissue models by collagen plastic compression: Understanding three-dimensional cell matrix repair in vitro. Adv. Wound Care 2013, 2, 176–184. [Google Scholar] [CrossRef]
- Chen, F.; Le, P.; Fernandes-Cunha, G.M.; Heilshorn, S.C.; Myung, D. Bio-orthogonally crosslinked hyaluronate-collagen hydrogel for suture-free corneal defect repair. Biomaterials 2020, 255, 120176. [Google Scholar] [CrossRef]
- Fernandes-Cunha, G.M.; Chen, K.M.; Chen, F.; Le, P.; Han, J.H.; Mahajan, L.A.; Lee, H.J.; Na, K.S.; Myung, D. In situ-forming collagen hydrogel crosslinked via multi-functional PEG as a matrix therapy for corneal defects. Sci. Rep. 2020, 10, 16671. [Google Scholar] [CrossRef]
- Kishore, V.; Iyer, R.; Frandsen, A.; Nguyen, T.-U. In vitro characterization of electrochemically compacted collagen matrices for corneal applications. Biomed. Mater. 2016, 11, 055008. [Google Scholar] [CrossRef]
- Ji, P.; Zhang, C.; Kong, Y.; Liu, H.; Guo, J.; Shi, L.; Yang, H.; Gu, Z.; Liu, Y. Collagen Film with Bionic Layered Structure and High Light Transmittance for Personalized Corneal Repair Fabricated by Controlled Solvent Evaporation Technique. J. Funct. Biomater. 2022, 13, 52. [Google Scholar] [CrossRef]
- Qin, L.; Gao, H.; Xiong, S.; Jia, Y.; Ren, L. Preparation of collagen/cellulose nanocrystals composite films and their potential applications in corneal repair. J. Mater. Sci. Mater. Med. 2020, 31, 55. [Google Scholar] [CrossRef]
- Hancox, Z.; Keshel, S.H.; Yousaf, S.; Saeinasab, M.; Shahbazi, M.-A.; Sefat, F. The progress in corneal translational medicine. Biomater. Sci. 2020, 8, 6469–6504. [Google Scholar] [CrossRef]
- Huang, G.; Ji, S.; Luo, P.; Liu, H.; Zhu, S.; Wang, G.; Zhou, P.; Xiao, S.; Xia, Z. Accelerated expansion of epidermal keratinocyte and improved dermal reconstruction achieved by engineered amniotic membrane. Cell Transplant. 2013, 22, 1831–1844. [Google Scholar] [CrossRef]
- Dinescu, S.; Albu Kaya, M.; Chitoiu, L.; Ignat, S.; Kaya, D.A.; Costache, M. Collagen-based hydrogels and their applications for tissue engineering and regenerative medicine. In Cellulose-Based Superabsorbent Hydrogels; Polymers and Polymeric Composites: A Reference, Series; Mondal, M., Ed.; Springer International Publishing: Edinburg, Scotland, 2019; pp. 1643–1664. [Google Scholar] [CrossRef]
- Xeroudaki, M.; Thangavelu, M.; Lennikov, A.; Ratnayake, A.; Bisevac, J.; Petrovski, G.; Fagerholm, P.; Rafat, M.; Lagali, N. A porous collagen-based hydrogel and implantation method for corneal stromal regeneration and sustained local drug delivery. Sci. Rep. 2020, 10, 16936. [Google Scholar] [CrossRef]
- Jones, R.R.; Hamley, I.W.; Connon, C.J. Ex vivo expansion of limbal stem cells is affected by substrate properties. Stem Cell Res. 2012, 8, 403–409. [Google Scholar] [CrossRef]
- Gouveia, R.M.; Vajda, F.; Wibowo, J.A.; Figueiredo, F.; Connon, C.J. YAP, ΔNp63, and β-catenin signaling pathways are involved in the modulation of corneal epithelial stem cell phenotype induced by substrate stiffness. Cells 2019, 8, 347. [Google Scholar] [CrossRef]
- Massie, I.; Kureshi, A.K.; Schrader, S.; Shortt, A.J.; Daniels, J.T. Optimization of optical and mechanical properties of real architecture for 3-dimensional tissue equivalents: Towards treatment of limbal epithelial stem cell deficiency. Acta Biomater. 2015, 24, 241–250. [Google Scholar] [CrossRef]
- Then, K.Y.; Azlina, M.; Ropilah, A.; Ruszymah, B.; Rohaina, C.; Ng, M. The use of bone marrow derived mesenchymal stem cell for cornea regeneration in rabbit model. Asian J. Ophthalmol. 2017, 15, 224–233. [Google Scholar] [CrossRef]
- Hallab, N.J.; Bundy, K.J.; O’Connor, K.; Moses, R.L.; Jacobs, J.J. Evaluation of metallic and polymeric biomaterial surface energy and surface roughness characteristics for directed cell adhesion. Tissue Eng. 2001, 7, 55–71. [Google Scholar] [CrossRef]
- Wang, X.; Majumdar, S.; Ma, G.; Sohn, J.; Yiu, S.C.; Stark, W.; Al-Qarni, A.; Edward, D.P.; Elisseeff, J.H. Chondroitin sulfate–based biocompatible crosslinker restores corneal mechanics and collagen alignment. Investig. Ophthalmol. Vis. Sci. 2017, 58, 3887–3895. [Google Scholar] [CrossRef]
- Kang, K.B.; Lawrence, B.D.; Gao, X.R.; Luo, Y.; Zhou, Q.; Liu, A.; Guaiquil, V.H.; Rosenblatt, M.I. Micro-and nanoscale topographies on silk regulate gene expression of human corneal epithelial cells. Investig. Ophthalmol. Vis. Sci. 2017, 58, 6388–6398. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 105906. [Google Scholar] [CrossRef]
- Aromataris, E.; Fernandez, R.; Godfrey, C.M.; Holly, C.; Khalil, H.; Tungpunkom, P. Summarizing systematic reviews: Methodological development, conduct and reporting of an umbrella review approach. JBI Evid. Implement. 2015, 13, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, S.; Sekar, S.; Katheem, M.F.; Krishnakumar, S.; Sastry, T.P. Fish scale collagen—A novel material for corneal tissue engineering. Artif. Organs 2012, 36, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Jangamreddy, J.R.; Haagdorens, M.K.; Islam, M.M.; Lewis, P.; Samanta, A.; Fagerholm, P.; Liszka, A.; Ljunggren, M.K.; Buznyk, O.; Alarcon, E.I. Short peptide analogs as alternatives to collagen in pro-regenerative corneal implants. Acta Biomater. 2018, 69, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Qu, M.; Wang, Y.; Wang, Z.; Shi, W. Xenogeneic acellular conjunctiva matrix as a scaffold of tissue-engineered corneal epithelium. PLoS ONE 2014, 9, e111846. [Google Scholar] [CrossRef]
- Park, J.; Lee, K.P.; Kim, H.; Park, S.; Wijesinghe, R.E.; Lee, J.; Han, S.; Lee, S.; Kim, P.; Cho, D.W. Biocompatibility evaluation of bioprinted decellularized collagen sheet implanted in vivo cornea using swept-source optical coherence tomography. J. Biophotonics 2019, 12, 201900098. [Google Scholar] [CrossRef]
- Baratta, R.O.; Del Buono, B.J.; Schlumpf, E.; Ceresa, B.P.; Calkins, D.J. Collagen Mimetic Peptides Promote Corneal Epithelial Cell Regeneration. Front. Pharmacol. 2021, 12, 705623. [Google Scholar] [CrossRef]
- Zhou, Q.; Guaiquil, V.H.; Wong, M.; Escobar, A.; Ivakhnitskaia, E.; Yazdanpanah, G.; Jing, H.; Sun, M.; Sarkar, J.; Luo, Y. Hydrogels derived from acellular porcine corneal stroma enhance corneal wound healing. Acta Biomater. 2021, 134, 177–189. [Google Scholar] [CrossRef]
- Polisetti, N.; Roschinski, B.; Schlötzer-Schrehardt, U.; Maier, P.; Schlunck, G.; Reinhard, T. A decellularized human limbal scaffold for limbal stem cell niche reconstruction. Int. J. Mol. Sci. 2021, 22, 10067. [Google Scholar] [CrossRef]
- Sánchez-Porras, D.; Caro-Magdaleno, M.; González-Gallardo, C.; García-García, Ó.D.; Garzón, I.; Carriel, V.; Campos, F.; Alaminos, M. Generation of a biomimetic substitute of the corneal limbus using decellularized scaffolds. Pharmaceutics 2021, 13, 1718. [Google Scholar] [CrossRef]
- Chakraborty, A.; Dutta, J.; Das, S.; Datta, H. Comparison of ex vivo cultivated human limbal epithelial stem cell viability and proliferation on different substrates. Int. Ophthalmol. 2013, 33, 665–670. [Google Scholar] [CrossRef]
- Haagdorens, M.; Cėpla, V.; Melsbach, E.; Koivusalo, L.; Skottman, H.; Griffith, M.; Valiokas, R.; Zakaria, N.; Pintelon, I.; Tassignon, M.-J. In vitro cultivation of limbal epithelial stem cells on surface-modified crosslinked collagen scaffolds. Stem Cells Int. 2019, 2019, 7867613. [Google Scholar] [CrossRef]
- Kureshi, A.K.; Drake, R.A.; Daniels, J.T. Challenges in the development of a reference standard and potency assay for the clinical production of RAFT tissue equivalents for the cornea. Regen. Med. 2014, 9, 167–177. [Google Scholar] [CrossRef]
- Lake, J.; Zaniolo, K.; Gingras, M.-È.; Couture, C.; Salesse, C.; Guérin, S.L. Functional Impact of Collagens on the Activity Directed by the Promoter of the α5 Integrin Subunit Gene in Corneal Epithelial Cells. Investig. Ophthalmol. Vis. Sci. 2015, 56, 6217–6232. [Google Scholar] [CrossRef]
- Zhang, Q.; Tang, Q.; Yang, Y.; Yi, J.; Wei, W.; Hong, Y.; Zhang, X.; Zhou, F.; Yao, X.; Ouyang, H. Wound dressing gel with resisted bacterial penetration and enhanced re-epithelization for corneal epithelial-stromal regeneration. Appl. Mater. Today 2021, 24, 101119. [Google Scholar] [CrossRef]
- Chen, J.; Lan, J.; Liu, D.; Backman, L.J.; Zhang, W.; Zhou, Q.; Danielson, P. Ascorbic acid promotes the stemness of corneal epithelial stem/progenitor cells and accelerates epithelial wound healing in the cornea. Stem Cells Transl. Med. 2017, 6, 1356–1365. [Google Scholar] [CrossRef]
- Wilson, S.L.; Yang, Y.; El Haj, A.J. Corneal stromal cell plasticity: In vitro regulation of cell phenotype through cell-cell interactions in a three-dimensional model. Tissue Eng. Part A 2014, 20, 225–238. [Google Scholar] [CrossRef]
- Kureshi, A.K.; Dziasko, M.; Funderburgh, J.L.; Daniels, J.T. Human corneal stromal stem cells support limbal epithelial cells cultured on RAFT tissue equivalents. Sci. Rep. 2015, 5, 16186. [Google Scholar] [CrossRef] [PubMed]
- Massie, I.; Dale, S.B.; Daniels, J.T. Limbal fibroblasts maintain normal phenotype in 3D RAFT tissue equivalents suggesting potential for safe clinical use in treatment of ocular surface failure. Tissue Eng. Part C Methods 2015, 21, 576–584. [Google Scholar] [CrossRef] [PubMed]
- de la Mata, A.; Mateos-Timoneda, M.A.; Nieto-Miguel, T.; Galindo, S.; López-Paniagua, M.; Planell, J.A.; Engel, E.; Calonge, M. Poly-l/dl-lactic acid films functionalized with collagen IV as carrier substrata for corneal epithelial stem cells. Colloids Surf. B Biointerfaces 2019, 177, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Çelebier, S.K.; Pehlivan, S.B.; Demirbilek, M.; Akıncı, M.; Vural, İ.; Akdağ, Y.; Yürüker, S.; Ünlü, N. Development of an anti-inflammatory drug-incorporated biomimetic scaffold for corneal tissue engineering. J. Ocul. Pharmacol. Ther. 2020, 36, 433–446. [Google Scholar] [CrossRef]
- Luo, Y.; Kang, K.B.; Sartaj, R.; Sun, M.G.; Zhou, Q.; Guaiquil, V.H.; Rosenblatt, M.I. Silk films with nanotopography and extracellular proteins enhance corneal epithelial wound healing. Sci. Rep. 2021, 11, 8168. [Google Scholar] [CrossRef]
- Polisetti, N.; Schmid, A.; Schlötzer-Schrehardt, U.; Maier, P.; Lang, S.J.; Steinberg, T.; Schlunck, G.; Reinhard, T. A decellularized human corneal scaffold for anterior corneal surface reconstruction. Sci. Rep. 2021, 11, 2992. [Google Scholar] [CrossRef]
- Miyamoto, K.; Kobayashi, T.; Hayashi, Y.; Zhang, Y.; Hara, Y.; Higashine, M.; Shiraishi, A.; Ohashi, Y. Involvement of stem cell factor and c-kit in corneal wound healing in mice. Mol. Vis. 2012, 18, 1505–1515. [Google Scholar]
- Antoine, E.E.; Vlachos, P.P.; Rylander, M.N. Review of collagen I hydrogels for bioengineered tissue microenvironments: Characterization of mechanics, structure, and transport. Tissue Eng. Part B Rev. 2014, 20, 683–696. [Google Scholar] [CrossRef]
- Davison-Kotler, E.; Marshall, W.S.; García-Gareta, E. Sources of collagen for biomaterials in skin wound healing. Bioengineering 2019, 6, 56. [Google Scholar] [CrossRef]
- Dyrlund, T.F.; Poulsen, E.T.; Scavenius, C.; Nikolajsen, C.L.; Thøgersen, I.B.; Vorum, H.; Enghild, J.J. Human cornea proteome: Identification and quantitation of the proteins of the three main layers including epithelium, stroma, and endothelium. J. Proteome Res. 2012, 11, 4231–4239. [Google Scholar] [CrossRef]
- Montanino, A.; Gizzi, A.; Vasta, M.; Angelillo, M.; Pandolfi, A. Modeling the biomechanics of the human cornea accounting for local variations of the collagen fibril architecture. ZAMM-J. Appl. Math. Mech. 2018, 98, 2122–2134. [Google Scholar] [CrossRef]
- Qin, D.; Bi, S.; You, X.; Wang, M.; Cong, X.; Yuan, C.; Yu, M.; Cheng, X.; Chen, X.-G. Development and application of fish scale wastes as versatile natural biomaterials. Chem. Eng. J. 2022, 428, 131102. [Google Scholar] [CrossRef]
- Lee, E.; Lee, W.-H.; Kaetzel, C.S.; Parry, G.; Bissell, M.J. Interaction of mouse mammary epithelial cells with collagen substrata: Regulation of casein gene expression and secretion. Proc. Natl. Acad. Sci. USA 1985, 82, 1419–1423. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Raines, R.T. Collagen-based biomaterials for wound healing. Biopolymers 2014, 101, 821–833. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, S.; Murphy, C.J.; McAnulty, J.F.; Raines, R.T. Peptides that anneal to natural collagen in vitro and ex vivo. Org. Biomol. Chem. 2012, 10, 5892–5897. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Jones, R.R.; Mi, S.; Foster, J.; Alcock, S.G.; Hamley, I.W.; Connon, C.J. The mechanical properties of amniotic membrane influence its effect as a biomaterial for ocular surface repair. Soft Matter 2012, 8, 8379–8387. [Google Scholar] [CrossRef]
- Foster, J.W.; Jones, R.R.; Bippes, C.A.; Gouveia, R.M.; Connon, C.J. Differential nuclear expression of Yap in basal epithelial cells across the cornea and substrates of differing stiffness. Exp. Eye Res. 2014, 127, 37–41. [Google Scholar] [CrossRef]
- Moers, K.; Steinberg, T.; Schlunck, G.; Reinhard, T.; Tomakidi, P.; Eberwein, P. Substrate elasticity as biomechanical modulator of tissue homeostatic parameters in corneal keratinocytes. Exp. Cell Res. 2013, 319, 1889–1901. [Google Scholar] [CrossRef]
- Yazdanpanah, G.; Haq, Z.; Kang, K.; Jabbehdari, S.; Rosenblatt, M.I.; Djalilian, A.R. Strategies for reconstructing the limbal stem cell niche. Ocul. Surf. 2019, 17, 230–240. [Google Scholar] [CrossRef]
- Gouveia, R.M.; Koudouna, E.; Jester, J.; Figueiredo, F.; Connon, C.J. Template curvature influences cell alignment to create improved human corneal tissue equivalents. Adv. Biosyst. 2017, 1, 1700135. [Google Scholar] [CrossRef]
- Calderón-Colón, X.; Xia, Z.; Breidenich, J.L.; Mulreany, D.G.; Guo, Q.; Uy, O.M.; Tiffany, J.E.; Freund, D.E.; McCally, R.L.; Schein, O.D. Structure and properties of collagen vitrigel membranes for ocular repair and regeneration applications. Biomaterials 2012, 33, 8286–8295. [Google Scholar] [CrossRef]
- Mouw, J.K.; Ou, G.; Weaver, V.M. Extracellular matrix assembly: A multiscale deconstruction. Nat. Rev. Mol. Cell Biol. 2014, 15, 771–785. [Google Scholar] [CrossRef]
- Hermanson, G.T. Bioconjugate Techniques, 3rd ed.; Academic Press: Cambridge, MA, USA, 2013; pp. 259–273. [Google Scholar]
- Wissink, M.; Beernink, R.; Pieper, J.; Poot, A.; Engbers, G.; Beugeling, T.; Van Aken, W.; Feijen, J. Immobilization of heparin to EDC/NHS-crosslinked collagen. Characterization and in vitro evaluation. Biomaterials 2001, 22, 151–163. [Google Scholar] [CrossRef]
- Nair, M.; Best, S.M.; Cameron, R.E. Crosslinking collagen constructs: Achieving cellular selectivity through modifications of physical and chemical properties. Appl. Sci. 2020, 10, 6911. [Google Scholar] [CrossRef]
- Zeeman, R.; Dijkstra, P.J.; van Wachem, P.B.; van Luyn, M.J.; Hendriks, M.; Cahalan, P.T.; Feijen, J. Successive epoxy and carbodiimide cross-linking of dermal sheep collagen. Biomaterials 1999, 20, 921–931. [Google Scholar] [CrossRef]
- Lee, H.J.; Lee, J.-S.; Chansakul, T.; Yu, C.; Elisseeff, J.H.; Seungju, M.Y. Collagen mimetic peptide-conjugated photopolymerizable PEG hydrogel. Biomaterials 2006, 27, 5268–5276. [Google Scholar] [CrossRef]
- Doillon, C.J.; Côté, M.-F.; Pietrucha, K.; Laroche, G.; Gaudreault, R.C. Porosity and biological properties of polyethylene glycol-conjugated collagen materials. J. Biomater. Sci. Polym. Ed. 1995, 6, 715–728. [Google Scholar] [CrossRef]
- Rafat, M.; Li, F.; Fagerholm, P.; Lagali, N.S.; Watsky, M.A.; Munger, R.; Matsuura, T.; Griffith, M. PEG-stabilized carbodiimide crosslinked collagen–chitosan hydrogels for corneal tissue engineering. Biomaterials 2008, 29, 3960–3972. [Google Scholar] [CrossRef]
- Lin, S.; Gu, L. Influence of crosslink density and stiffness on mechanical properties of type I collagen gel. Materials 2015, 8, 551–560. [Google Scholar] [CrossRef]
- Kang, S.J.; Kim, E.K.; Kim, H.B. Expression and distribution of extracellular matrices during corneal wound healing after keratomileusis in rabbits. Ophthalmologica 1999, 213, 20–24. [Google Scholar] [CrossRef]
- Nishida, T.; Nakamura, M.; Mishima, H.; Otori, T. Differential modes of action of fibronectin and epidermal growth factor on rabbit corneal epithelial migration. J. Cell. Physiol. 1990, 145, 549–554. [Google Scholar] [CrossRef]
- Murakami, J.; Nishida, T.; Otori, T. Coordinated appearance of β1 integrins and fibronectin during corneal wound healing. J. Lab. Clin. Med. 1992, 120, 86–93. [Google Scholar]
- Zheng, M.; Tian, C.; Fan, T.; Xu, B. Fibronectin regulates the self-renewal of rabbit limbal epithelial stem cells by stimulating the Wnt11/Fzd7/ROCK non-canonical Wnt pathway. Exp. Eye Res. 2019, 185, 107681. [Google Scholar] [CrossRef]
- Martin, G.R.; Timpl, R. Laminin and other basement membrane components. Annu. Rev. Cell Biol. 1987, 3, 57–85. [Google Scholar] [CrossRef] [PubMed]
- Nakayasu, K.; Tanaka, M.; Konomi, H.; Hayashi, T. Distribution of types I, II, III, IV and V collagen in normal and keratoconus corneas. Ophthalmic Res. 1986, 18, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, D.R.; Trüeb, B.; Winterhalter, K.H.; Witmer, R.; Fischer, R.W. Type VI collagen is a major component of the human cornea. FEBS Lett. 1986, 197, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Koulikovska, M.; Rafat, M.; Petrovski, G.; Veréb, Z.; Akhtar, S.; Fagerholm, P.; Lagali, N. Enhanced regeneration of corneal tissue via a bioengineered collagen construct implanted by a nondisruptive surgical technique. Tissue Eng. Part A 2015, 21, 1116–1130. [Google Scholar] [CrossRef]
- Kureshi, A.; Cheema, U.; Alekseeva, T.; Cambrey, A.; Brown, R. Alignment hierarchies: Engineering architecture from the nanometre to the micrometre scale. J. R. Soc. Interface 2010, 7, 707–716. [Google Scholar] [CrossRef]
- Zhang, Y.; Yeh, L.-K.; Zhang, S.; Call, M.; Yuan, Y.; Yasunaga, M.; Kao, W.W.-Y.; Liu, C.-Y. Wnt/β-catenin signaling modulates corneal epithelium stratification via inhibition of Bmp4 during mouse development. Development 2015, 142, 3383–3393. [Google Scholar] [CrossRef]
- Li, X.-K.; Cai, S.-X.; Liu, B.; Xu, Z.-L.; Dai, X.-Z.; Ma, K.-W.; Li, S.-Q.; Yang, L.; Sung, K.P.; Fu, X.-B. Characteristics of PLGA–gelatin complex as potential artificial nerve scaffold. Colloids Surf. B Biointerfaces 2007, 57, 198–203. [Google Scholar] [CrossRef]
- Campos, Y.; Almirall, A.; Fuentes, G.; Bloem, H.L.; Kaijzel, E.L.; Cruz, L.J. Tissue engineering: An alternative to repair cartilage. Tissue Eng. Part B Rev. 2019, 25, 357–373. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).